Abstract
BACKGROUND AND OBJECTIVE:
EBV-associated gastric cancer (EBVaGC) is a distinct subtype of GC, and EBV plays an important role in tumor progress. The standard method to identify EBV-positive tumor is determined by in situ hybridization for EBV-encoded EBERs in tumor tissues. The present study aims to detect the serological expression of EBV-related antibodies and ET-1 axis to provide a noninvasive method for diagnosis of EBVaGC.
METHODS:
The content of EBV-related antibodies and ET-1 axis in preoperative peripheral blood of GC was performed by Chemiluminescence and ELISA assay. The EBV DNA copy number was measured by qRT-PCR.
RESULTS:
The results showed that the levels of anti-EBV early antigen (EA) IgG, viral capsid antigen (VCA) IgA, nuclear antigen (NA) IgG, and EBV DNA copy number were significantly higher in EBVaGC. The ET-1 axis level was much lower in EBVaGC than EBVnGC.
CONCLUSIONS:
The combined detection of specific anti-EBV antibodies and ET-1 axis might provide new molecular markers for the identification of EBVaGC.
Introduction
In 1993, Tokunaga et al. defined EBV-encoded small RNAs (EBERs)-positive gastric cancer as EBV-associated gastric carcinoma (EBVaGC), and the remaining gastric cancer types were named EBV-negative gastric carcinoma (EBVnGC) [1]. As well as Helicobacter pylori, EBV has been accepted as an important infective agent causing GC. EBV primarily infects cells in two ways, which are lytic infection and latent infection, and expresses different sets of genes in different processes. In latent state, EBV expresses only 6 nuclear antigens (EBNA1, EBNA2, EBNA3a, EBNA3b, EBNA3c, LP), 3 latent membrane proteins (LMP1, LMP2A, LMP2B) and two small RNAs (EBER1 and EBER2) [2]. The expression of these EBV products can stimulate the body to produce related antibodies to exert humoral immunity, such as anti-EBV early antigen (EA)-IgA, EA-IgG, viral capsid antigen (VCA)-IgM, VCA-IgG, EBV nuclear antigen (NA)-IgG, etc. [3].
EBV-positive tumor is determined by in situ hybridization for EBV-encoded EBERs, the sensitive and specific marker for viral presence. However, in situ hybridization needs to obtain tumor tissue samples, and it is not routinely detected in pathologic examination. Thus, in recent years, serological detection of EBV-related antigens and antibodies has gradually become the main reference for early diagnosis of nasopharyngeal carcinoma (NPC). Serological profiles of EBV have been evidenced in clinical practice to be indicative of tumor burden, treatment response, and prognosis of NPC patients [4, 5]. Some studies have also shown that the content of EBV DNA in the plasma is closely related to the patient’s condition [6, 7, 8]. Unlike other EBV-associated malignancies, EBVaGC is a distinct subtype of GC that has unique clinicopathological features, such as overall lower mortality, more frequently occurs in males than females, greater intra- or peritumoral immune cell infiltration than EBV-negative GC [9, 10]. GC has potential precursor lesions that have been identified and can be evaluated in relation to EBV serological markers. In a longitudinal Chinese study of gastric lesions, persons with elevated baseline VCA-IgG and EBNA IgG titers had a higher likelihood of progressing to more severe gastric dysplasia 2 years later, particularly subjects with baseline diagnoses of intestinal metaplasia [11]. Their study suggested a significant association between anti-EBV antibodies and precancerous gastric lesions, and a possible role for EBV reactivation at an early phase of gastric carcinogenesis. However, serological profiles of EBV in gastric cancer are still unknown.
The endothelin 1 (ET-1) gene encodes a 212-amino acid precursor, which is converted by protease to form an intermediate peptide consisting of 38 amino acids, big-ET-1. Big-ET-1 is further cleaved into ET-1 by ET-converting enzymes (ECE-1 or ECE-2), which exerts biological effects by binding to ETR in an autocrine or paracrine manner [12]. Previous study showed that ET-1 was significantly overexpressed in gastric cancer when compared with adjacent normal [13]. In our previous study, it was found that the expression of ET-1 protein in EBVaGC cell lines was much lower than that of in EBVnGC cell lines (unpublished data). In this manuscript, we aim to investigate whether the content of ET-1 and its receptors in peripheral blood of EBVaGC and EBVnGC cases exhibit similar expression patterns of GC cell lines. Furthermore, we intend to account for authentic tumor EBV status by comparing differences in the associations between the humoral immune response to EBV and the levels of ET-1 axis, which may help to elucidate how this ubiquitous virus contributes to EBV-associated tumors. To our knowledge, this is the first serological study regarding to the correlations among ET-1, ETR and EBV infection in gastric cancer.
Material and methods
Samples
All gastric carcinoma cases (28 EBVaGC and 39 EBVnGC) were collected from Qingdao University Affiliated Hospital and Qingdao Municipal Hospital from October 2017 to August 2019, and then were diagnosed by pathological biopsy. EBV-associated gastric carcinoma cases were identified by in situ hybridization for EBV-encoded EBER1. In addition, we collected preoperative blood samples from these patients to reduce the impact of surgery. Of the 67 samples included 28 EBVaGC (mean age: 62.00
Comparison of clinical pathological features between EBVaGC and EBVnGC
Comparison of clinical pathological features between EBVaGC and EBVnGC
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma;
The contents of EA-IgG (#130215001M, Snibe), EA-IgA (#130215002M, Snibe), VCA-IgG (#130215003M, Snibe), VCA-IgM (#130215004M, Snibe), VCA-IgA (#130215005M, Snibe), and NA-IgG (#130215006M, Snibe) in the serum were measured by Chemiluminescence assay on an Automatic chemiluminescence detector Maglumi 2000 (Snibe, Shenzhen, China). According to manufacturer’s instructions, the reference values of EA-IgG, EA-IgA, VCA-IgG, VCA-IgM, VCA-IgA, and NA-IgG are 2 AU/ml, 3 AU/ml, 4 AU/ml, 3 AU/ml, 2 AU/ml, and 2 AU/ml, respectively. A sample exceeding the reference value is defined as positive case.
ELISA assay
We detected the concentrations of ET-1, ETAR and ETBR in the serum by ELISA assay (Keshun Bio, Shanghai, China). All experimental operations were carried out in accordance with the instruction, including the standard dilution, samples’ addition, incubation, washing, etc. After adding the termination solution, detect the absorbance value at 450 nm wavelength, then calculate the concentration of the samples (Bio-Tek ELx808, USA). Because hemolysis can affect the accuracy of the test results and cause false positive errors, the samples included in the study are non-hemolytic specimens that met the test requirements.
Detection of EBV-DNA copy number
EBV nucleic acid amplification fluorescence quantification kits were purchased from Daangene Company (#DA-B065, Guangzhou, China). First, according to the manufacturer’s instructions, the peripheral blood DNA was extracted with the corresponding reagent in the kit, and then the copy number of EBV DNA was detected on Light Cycler instruments according to the procedure (Roche, Switzerland). The conditions were as follows: 95
Statistical analysis
The chi-squared (
Results
Higher levels of EBV-related antibodies in EBVaGC than EBVnGC samples
To determine whether different concentrations of EBV-related antibodies are present in the serum of GC cases, we analyzed 28 cases of EBVaGC and 39 cases of EBVnGC using Chemiluminescence assay. The results showed that EA-IgA, VCA-IgM, and VCA-IgG were expressed at comparable levels between EBVaGC and EBVnGC, while EA-IgG, VCA-IgA and NA-IgG were at significant higher levels in EBVaGC than EBVnGC (Table 2). Then, we further analyzed the distribution of anti-EBV related antibodies in EBVaGC and EBVnGC (Table 3). Anti-EA-IgG, EA-IgA, VCA-IgA, VCA-IgM, VCA-IgG and EBNA-1-IgG antibodies in GC were 16.42%, 2.99%, 22.39%, 8.96%, 100% and 89.55%, respectively. These data showed that the positive rate of EA-IgG and VCA-IgA in EBVaGC was significantly higher than that in EBVnGC (EA-IgG: 32.1% vs 5.1%; VCA-IgA: 35.7% vs 14.71%). Interestingly, the distribution of NA-IgG showed no significant difference between EBVaGC and EBVnGC, indicating that NA-IgG had only increased titer in EBVaGC without a change in positive rate. Thus, higher EBV-related antibodies in EBVaGC, especially EA-IgG and VCA-IgA, are positively correlated with the development of EBVaGC.
EBV DNA copy number in GC samples
Expression of anti-EBV related antibodies in EBVaGC and EBVnGC
Expression of anti-EBV related antibodies in EBVaGC and EBVnGC
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma; EA, early antigen; VCA, viral capsid antigen; NA, nuclear antigen.
The distribution of anti-EBV related antibodies in EBVaGC, EBVnGC and GC
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma; EA, early antigen; VCA, viral capsid antigen; NA, nuclear antigen.
To see whether there is a relationship between the EBV DNA copy number and EBVaGC occurrence, we examined EBV DNA copy number in EBVaGC and EBVnGC. Due to that DNA copy number of all samples was low (less than 5
Comparison of the number of cases in which DNA copy number can be detected in EBVaGC and EBVnGC
Comparison of the number of cases in which DNA copy number can be detected in EBVaGC and EBVnGC
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma.
Plasma levels of ET-1, ETAR, ETBR in patients with EBVaGC and EBVnGC
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma.
The levels of ET-1, ETAR, and ETBR in the serum of GC cases were determined by ELISA. As shown in Table 5, the expression levels of ET-1, ETAR, and ETBR in EBVaGC were significantly lower than that of EBVnGC (
Correlations between plasma concentration of ET-1, ETAR, ETBR and clinicopathological parameters
Correlations between plasma concentration of ET-1, ETAR, ETBR and clinicopathological parameters
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma.
Correlation analysis among ET-1, ETAR, and ETBR in GC samples. (a) A linear correlation between ET-1 and ETAR. 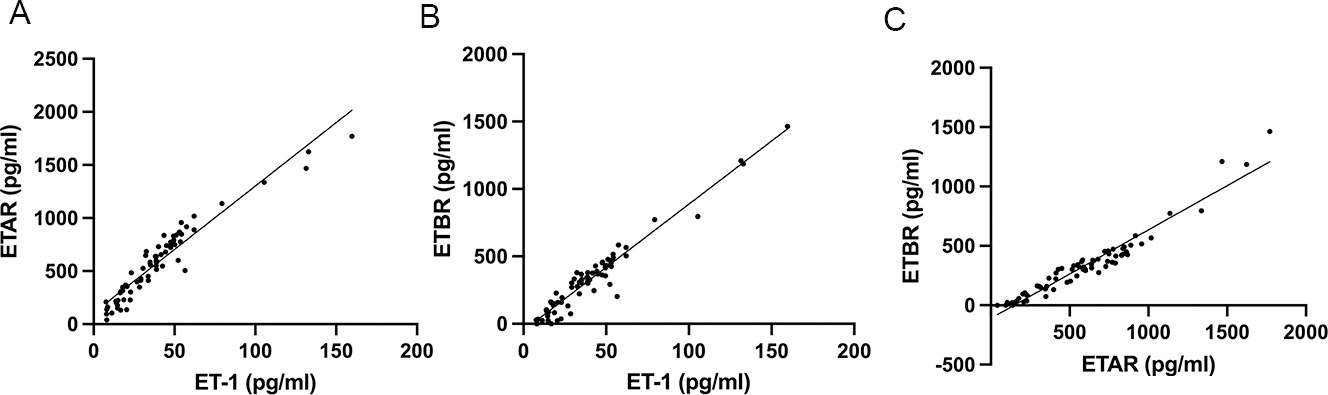
Previous studies have shown that the expression of ET-1 and ETAR is associated with clinical pathological parameters [16, 17]. To test the possibility that ET-1 axis is associated with the unique clinical pathology of EBVaGC, we analyzed the correlation between plasma levels of ET-1 axis and clinicopathological parameters (Table 6). These data suggested that the plasma concentration of ET-1 axis is unrelated with differentiation, lymph node metastasis, tumor classification, gender, years and invasion depth (
To clarify the relationship between ET-1 axis and EBV-related antibodies in EBVaGC, we further analyzed the relationship between the concentration of ET-1 axis content and antibodies. The data showed that the plasma levels of ET-1, ETAR and ETBR are positively correlated with VCA-IgM (
Expression of ET-1 axis was negatively correlated with EBV DNA copy number
We then analyzed the relationship between the concentration of ET-1 and ETR with the copy number of EBV DNA in the serum of 11 patients with positive EBV DNA copy number (Fig. 2). Interestingly, we find that although the difference in copy number and content of ET-1 or ETR is relatively large among individuals, the copy number of EBV DNA has a negative relationship with ET-1 content. In specimens of patients P15 and P36, the copy number of EBV DNA is the highest, while ET-1 or ETR content is the lowest. Conversely, the P2 patient’s specimen had the lowest copy number of EBV DNA and the highest ET-1 or ETR content. Correlation detection of copy number and ET-1 or ETR content in 11 samples is performed using Spearman test (copy vs ET-1:
Discussion
Correlation between ET-1 and anti-EBV-related antibody concentrations in plasma
Correlation between ET-1 and anti-EBV-related antibody concentrations in plasma
EBVaGC, EBV-associated gastric carcinoma; EBVnGC, EBV-negative gastric carcinoma; EA, early antigen; VCA, viral capsid antigen; NA, nuclear antigen.
Relationship between the concentrations of ET-1 axis with EBV DNA copy number in 11 patients with positive EBV DNA copy number. (a) Related analysis of ET-1 and EBV DNA copy number. (b) Related analysis of ETAR and EBV DNA copy number. (c) Related analysis of ETBR and EBV DNA copy number. P2, P15, P16, three cases of GC samples. It indicates that copy number of EBV DNA has a negative relationship with ET-1 content.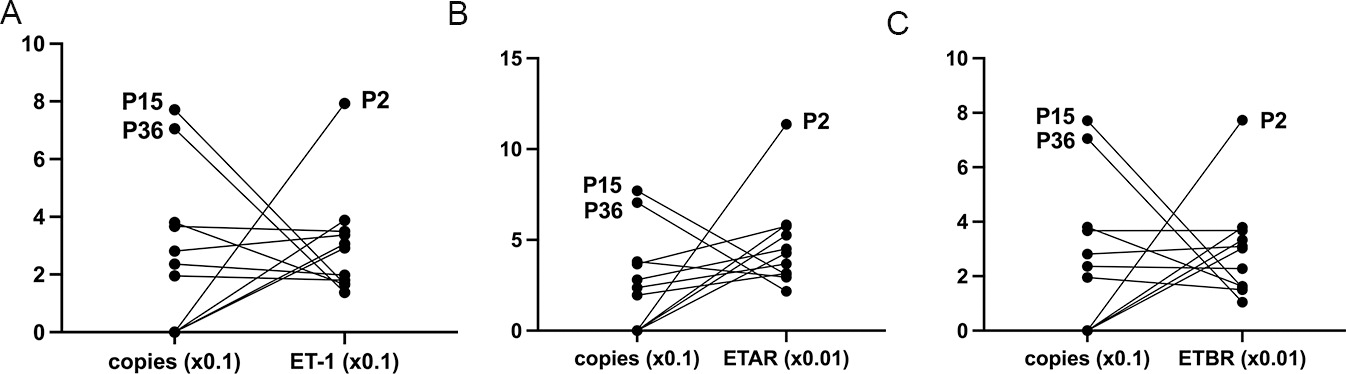
More than 95% of the world’s population have been infected with EBV at the age of 3 to 5 years and have been infected with latent infection for the lifetime [18]. Under certain circumstances, latent infected viruses will be activated and converted to proliferative infections, which can lead to diseases associated with abnormal B lymphocytes and certain epithelial cell tumors. The antibody spectrum produced by EBV infection has clinical significance for diagnosis and disease identification [19, 20], but their performance in GC remains controversial. Several prospective cohort studies [21, 22, 23] and a case-control study [24] evaluated the relationship between GC and EBV serological markers. Two of these studies [21, 22] found no statistically significant increase in GC risk in subjects with elevated anti-VCA or anti-EA antibody titers. The other two studies [3, 23] making the important distinction between EBV-positive and EBV-negative GC tumors, found that seropositivity rates and titers for anti-EBV antibodies were significantly higher in EBV-positive cases than in EBV-negative cases. In this study, it was showed that EA-IgG, EA-IgA, VCA-IgA and VCA-IgM antibodies were lower in gastric cancer patients (2.99%
ET-1, ETAR and ETBR are abnormally expressed in many diseases, such as malignant tumors and cardiovascular diseases. Also, ET-1 and its receptors are involved in the pathogenesis of some viruses, such as HCV [28], HPV [29], HIV [30], and so on. We previously detected the expression of ET-1 by RT-qPCR and Western blot in EBVaGC and EBVnGC cell lines and found that the expression level of ET-1 in EBVaGC was significantly lower than that in EBVnGC. EBV could inhibit the expression of ET-1 through its encoded products (unpublished data). In this report, we further analyzed the expression level of ET-1 in clinical samples. These results were consistent with previous findings. Additionally, there was a negative correlation between ET-1 axis and the copy number of EBV DNA, indicating that ET-1 axis expression is associated with EBV infection both in vivo and in vitro.
ET-1 axis can augment the response of immune cells in the local microenvironment. Particularly, ET-1-driven autocrine or paracrine loops could modulate dendritic cell function, such as increase in cell survival, cytokine production and T cell activation [31]. As the most efficient antigen-presenting cells, dendritic cells produce ET-1 and functional ETAR and ETBR, which are both highly expressed during dendritic cell maturation. Therefore, autocrine or paracrine circuits involving ET-1, ETAR and ETBR seem to regulate the maturation and function of immune system. Herein, low level of ET-1 and ETR in the serum of EBVaGC cases might be related to EBV evading immune surveillance in the body to achieve lifelong latent infection.
In terms of diagnosis, clinical EBV antibody profiles are diverse and inconsistent with EBV-DNA assays. For example, some patients with chronic active EBV infection may have a low EBV antibody upregulation when they have low immunoglobulins or severe splenomegaly, but qRT-PCR assay found a significant increase in viral copy number, suggesting that sometimes EBV antibodies are low, but cross-reactivity due to other factors cannot be ruled out [32, 33]. Here, we found that levels of ET-1 and ETR in the plasma were positively correlated with VCA IgM content but not with other antibodies. This may be due to the fact that VCA IgM is produced in the early stage of acute infection [34], when ET-1 and ETR genes are up-regulated and stimulate the body’s anti-infective immune response. As mentioned above, EBVaGC can only be determined by the presence of virus found directly within tumor cells. For clinical diagnosis, the approaches basing on tumor tissues have more sensitivity and specificity, such as EBERs-ISH and next-generation. While, our study sought to identify noninvasive or minimally invasive markers through the examination of special EBV antibody titers. Based on our research, combined EBV antibody titers and ET-1 axis may provide a useful biomarker for pre-diagnostic screening of EBVaGC.
In conclusion, this study reports the relationship between ET-1/ETR and EBV-related antibodies or EBV DNA copy number for the first time. Patients with EBVaGC have elevated antibodies to EBV and lower expression of ET-1 axis. Due to the small number of cases, further verification with more samples is required. Although EBERs-ISH has more sensitivity and specificity, EBV status is not routinely assessed in clinical pathologic practice. Sometimes, tumor tissue is unavailable, EBV serology has actual utility for next research. Given the limited options for identifying EBVaGC, multiple markers in combination could be useful in the future. These data can help better understand the unique pathogenesis and clinical features of EBVaGC and provides a new molecular marker for the identification of EBV-positive gastric cancer and -negative gastric cancer.
Abbreviations
EBV: Epstein-Barr virus; EBVaGC: EBV-associated gastric cancer; EBVnGC: EBV-negative gastric cancer; EA: early antigen; VCA: viral capsid antigen; NA: nuclear antigen; NPC: nasopharyngeal carcinoma; ET-1: endothelin-1.
Funding
This work was supported by Natural Science Foundation of Shandong Province [ZR2020MH302; ZR2020 MC020]; China postdoctoral Science Foundation [2020 M682126].
Competing interests
The authors have declared that no competing interest exists.
Author contributions
Conception: Bing Luo.
Interpretation or analysis of data: Yan Zhang, Qianqian Zhang.
Preparation of the manuscript: Qianqian Zhang, Hua Xiao, Weiwen Wang.
Revision for important intellectual content: Lin Xu, Yan Zhang.
Supervision: Bing Luo.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-220001.
sj-docx-1-cbm-10.3233_CBM-220001.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-220001.docx
