Abstract
BACKGROUND:
The prognostic role of annexin A5 (ANXA5) in stomach adenocarcinoma (STAD) has not been studied, and its relationship with immune infiltration is still unclear.
OBJECTIVE:
This investigation aimed at exploring the role of ANXA5 in STAD using an integrated bioinformatics analysis.
METHODS:
The expression of ANXA5 in STAD and the correlations between the effect of ANXA5 and survival of STAD patients were investigated using database. The clusterProfiler package in R software was used to perform enrichment analysis on the top 100 co-expressed genes of ANXA5 from the COXPRESdb online database. Correlations between ANXA5 and immune cell infiltrates were analyzed using the TIMER database.
RESULTS:
In STAD, ANXA5 expression was significantly upregulated and increased ANXA5 expression was significantly correlated with poor overall survival (
CONCLUSIONS:
Together with our findings, ANXA5 could serve as a potential biomarker to assess prognosis and immune infiltration level in STAD.
Introduction
Stomach adenocarcinoma (STAD) is one of the most common malignant tumors and the fourth leading cause of cancer-related deaths in the world [1]. STAD has become a major global health concern due to its high aggressiveness, rapid progression and poor prognosis [2, 3]. Although there has been some improvements in STAD prevention, diagnostic screening, surgical resection and adjuvant chemotherapy, the clinical effects of treatments still need to be improved.
The treatment strategy represented by programmed cell death protein I (PD-1)/programmed cell death ligand I (PD-L1) has been used in cancer for the recent years [4]. Immunotherapy has been successful, and has shown considerable effects in cancers such as malignant melanoma, non-small cell lung cancer and metastatic urothelium [5, 6]. However, progress in immunotherapy for STAD has been slow compared to the aforementioned cancers. Therefore, identifying new immune-related therapeutic targets and elucidating the immunophenotype of STAD-immune interaction is fundamental for STAD immunotherapy.
Annexin A5 (ANXA5) is a member of the annexin family of proteins. It has a conserved C-terminus and a unique N-terminus and its functions include membrane fusion, ion channels and signal transduction [7]. Recent studies have confirmed that ANXA5 is an essential tumor marker that is not only abnormally expressed in a variety of tumor tissues but also linked to tumor differentiation [8, 9]. According to previous studies, ANXA5 is significantly increased in breast, cervical and colon cancers [10, 11], but decreased in lung cancer [12]. However, the prognostic role of ANXA5 in STAD has not been reported and its relationship with immune infiltration is still unclear.
In this study, databases were analyzed with the aim to explore the expression of ANXA5 in STAD patients, its relationship with STAD prognosis, putative pathogenic mechanism and the relationship between ANXA5 and tumor infiltrating immune cells and immunological markers.
Materials and methods
Data acquisition
ANXA5 gene expression profiles and paired clinical information of STAD patient datasets, which included 408 tumor tissues and 36 normal tissues were downloaded from The Cancer Genome Atlas (TCGA). Cases with insufficient or missing data on age, gender, overall survival time, local invasion, lymph node metastasis, and distant metastasis were excluded in the subsequent analyses. Finally, 352 cases with eligible clinical information were subjected to Cox regression analysis in order to investigate the predictive factors of prognosis. To study the effects of ANXA5 expression on immune microenvironment, 375 tumor tissues with eligible expression levels of the ANXA5 gene and immune cell-related genes were retained to perform Tumor IMmune Estimation Resource (TIMER) analysis.
Survival and expression analysis by GEPIA
The Gene Expression Profiling Interactive Analysis (GEPIA) (
Immunohistochemistry
The protein expression levels of ANXA5 in normal and STAD tissues were assessed using the Human Protein Atlas (HPA) database, which comprises immunohistochemistry-based expression of the most common cancers. The protein expression levels were divided into four levels based on staining intensity and staining quantity.
Survival outcome and expression difference of ANXA5 in STAD (*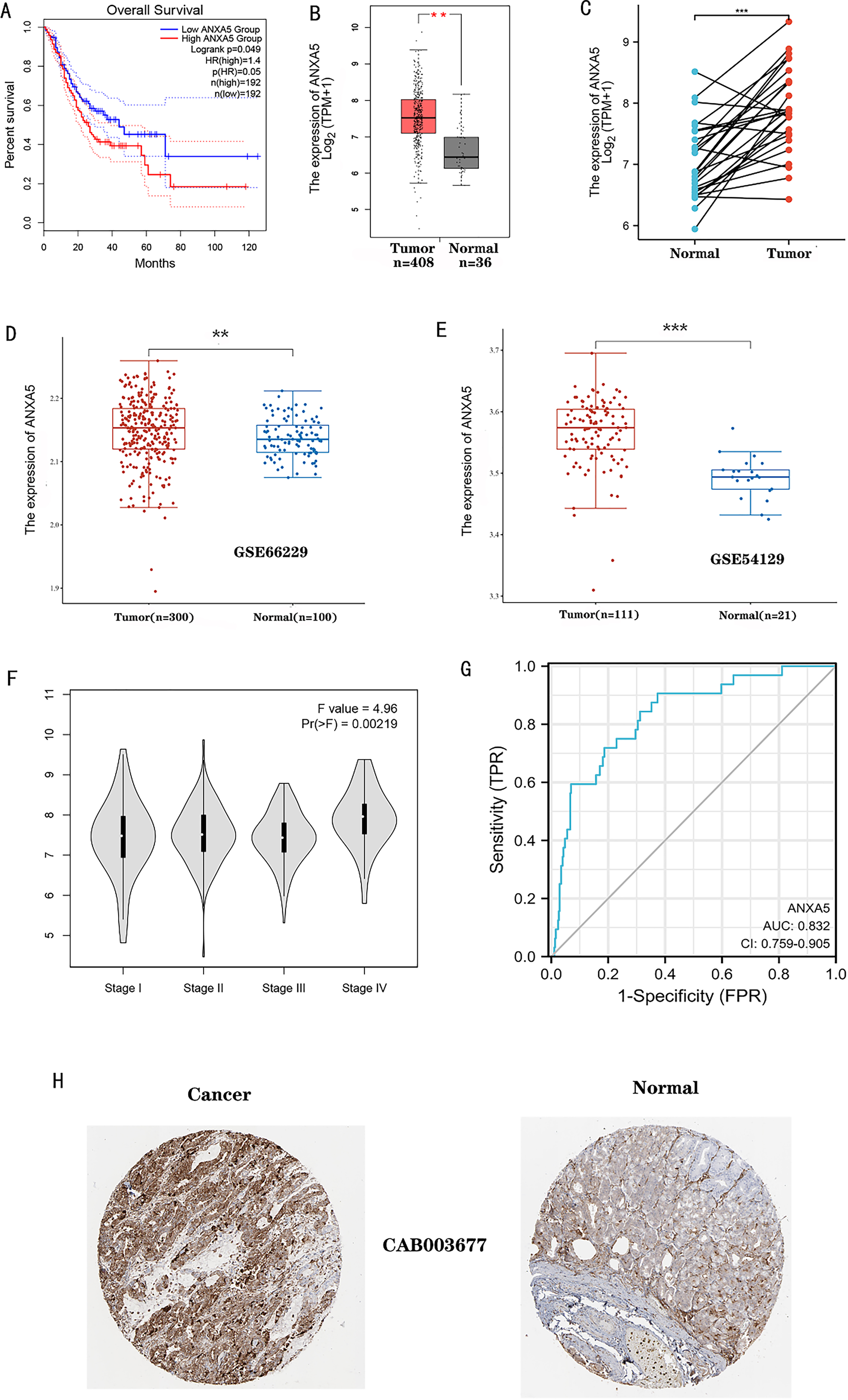
Multivariate analysis with the use of a Cox proportional hazards model for overall survival
To make reliable immune infiltration estimations, we utilized the immunedeconv, an R package that integrates TIMER and has been systematically benchmarked. TIMER is a comprehensive tool and routinely used for analyzing cancer-related immune infiltrates [14]. The abundance of six immune infiltrates (B cells, CD4
Analyses of genes co-expressed with ANXA5
The top 100 co-expressed genes of ANXA5 were estimated using the COXPRESdb online database (
Gene set enrichment analysis (GSEA)
To explore the potential pathogenic biological processes of ANXA5, we conducted gene set enrichment analysis (GSEA) for TCGA-STAD data using GSEA v4.3.0 software. The file c2.cp.kegg.v7.4.symbols.gmt was selected for further analysis. The number of permutations was 1000, and the cut-offs for significant enrichment were NES (normalized enrichment score)
Statistical methods
Statistical analyses on data acquired from TCGA were conducted by R- 4.0.3. The specificity and sensitivity of ANXA5 for the diagnosis of gastric cancer was evaluated using a receiver operating characteristic (ROC) curve. The correlations between clinical characteristics and ANXA5 expression were analyzed using the logistic regression. The Cox regression analysis was used to identify overall survival-related clinical characteristics in TCGA patients. In this study, a
Association between ANXA5 expression and clinicopathologic variables
Association between ANXA5 expression and clinicopathologic variables
Multivariate Cox analysis of ANXA5 expression and other clinicopathological factors. The up-regulated ANXA5 expression, age 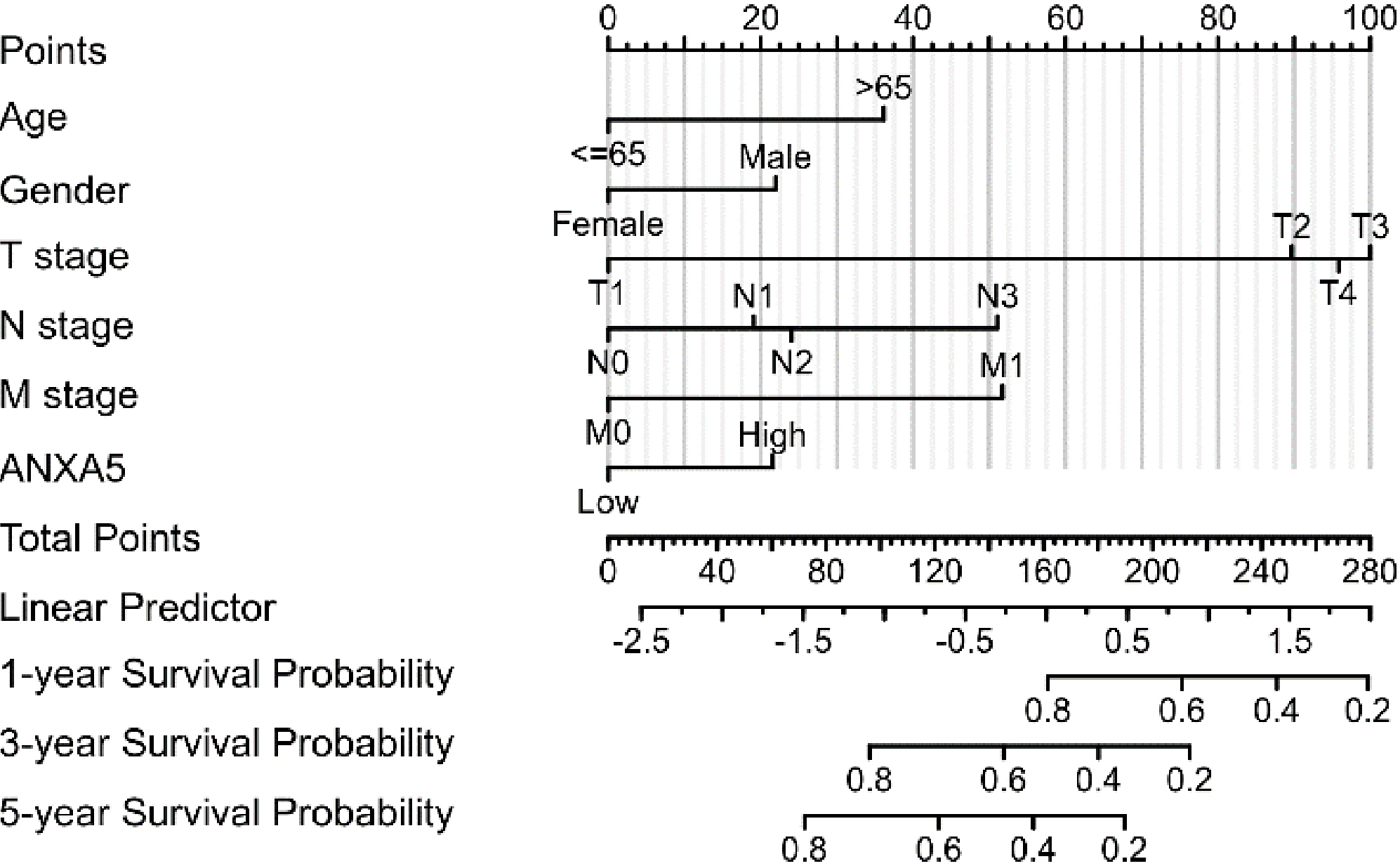
Relationship between ANXA5 expression and tumor-infiltrating immune cells. A. Immune cell score heat map, where different colors represent the expression trend in different samples (**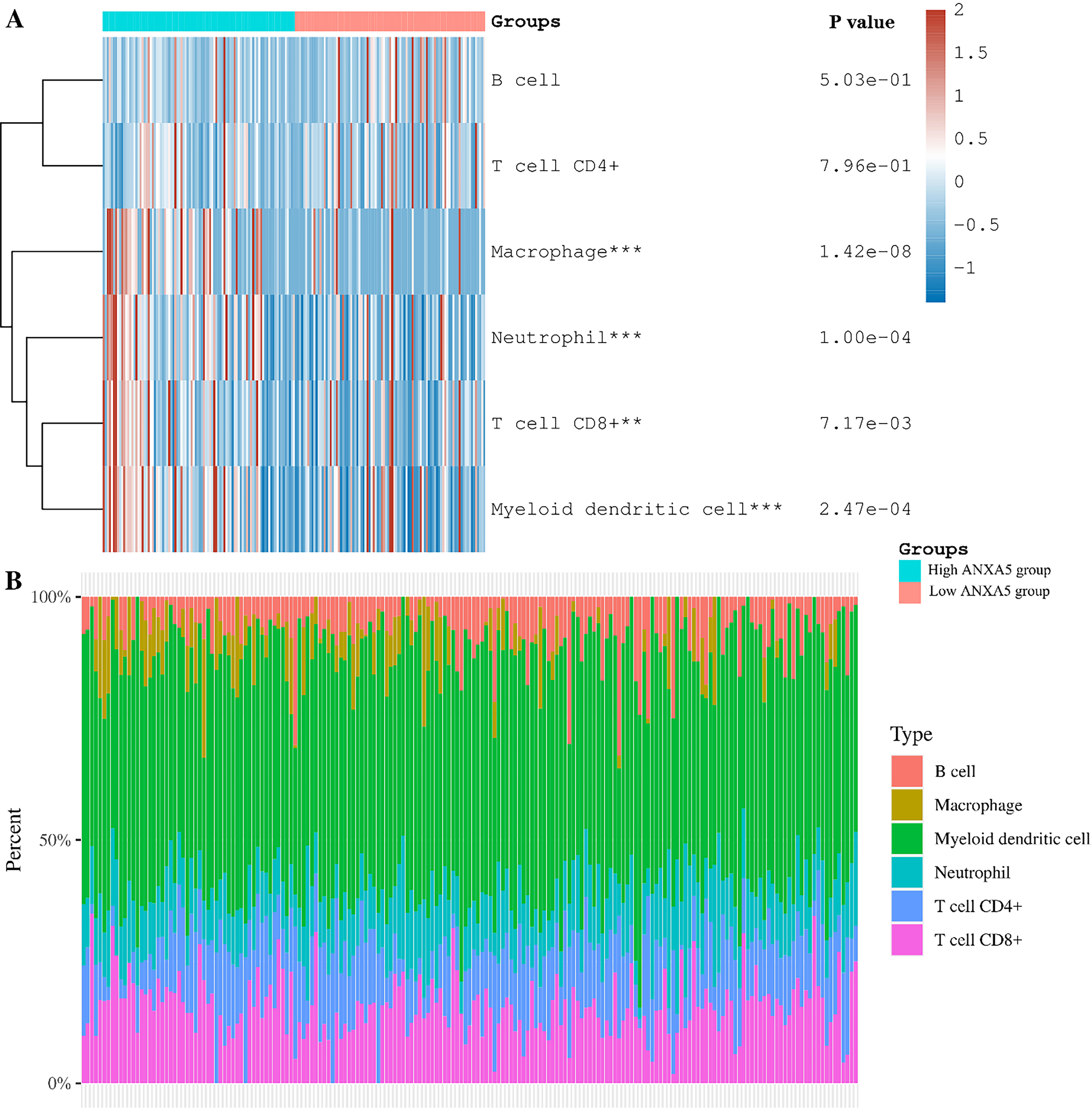
Survival outcomes and multivariate analysis
384 cases with eligible OS information and ANXA5 gene expression download from TCGA database were divided into high expression group (
Association between ANXA5 expression and clinicopathologic variables
We investigated the underlying mechanism ofANXA5 expression in cancer and its correlation with certain clinical aspects in STAD patients. Cases of STAD with eligible clinical information were analyzed using R version 4.0.3. Univariate analysis using logistic regression, and ANXA5 expression as a categorical dependent variable (based on median expression value), revealed that elevated ANXA5 expression correlated significantly with pathological stage (stage I vs. stage IV,
Relationship between ANXA5 expression and tumor-infiltrating immune cells
The results of TIMER are exhibited in Fig. 3: In the high ANXA5 expression group, CD8
Correlation analysis between ANXA5 expression and possible genes markers of tumor infiltrating immune cells via “correlation” module of GEPIA
Correlation analysis between ANXA5 expression and possible genes markers of tumor infiltrating immune cells via “correlation” module of GEPIA
The top 100 co-expressed genes of ANXA5 were selected to conduct the enrichment analysis. The terms extracellular matrix (ECM) organization, extracellular structure organization, and urogenital system development were significantly enriched in the GO biological process (BP) analysis (Fig. 4 and Table 4). The terms collagen-containing ECM, ECM component, and collagen trimer were significantly enriched in the GO term cellular component (CC) analysis (Fig. 4 and Table 4). According to the molecular function (MF) analysis, the terms ECM structural constituent, collagen binding, and ECM structural constituent conferring tensile strength were highly enriched (Fig. 4 and Table 4). The KEGG pathway analysis indicated that focal adhesion, amoebiasis, protein digestion, and absorption were significantly enriched (Fig. 4 and Table 4).
Enrichment analysis of co-expressed genes with ANXA5 in STAD
Enrichment analysis of co-expressed genes with ANXA5 in STAD
The top 100 co-expressed genes of ANXA5 were selected to conduct the enrichment analysis and the results showed that the co-expressed genes were involved in extracellular matrix-related processes.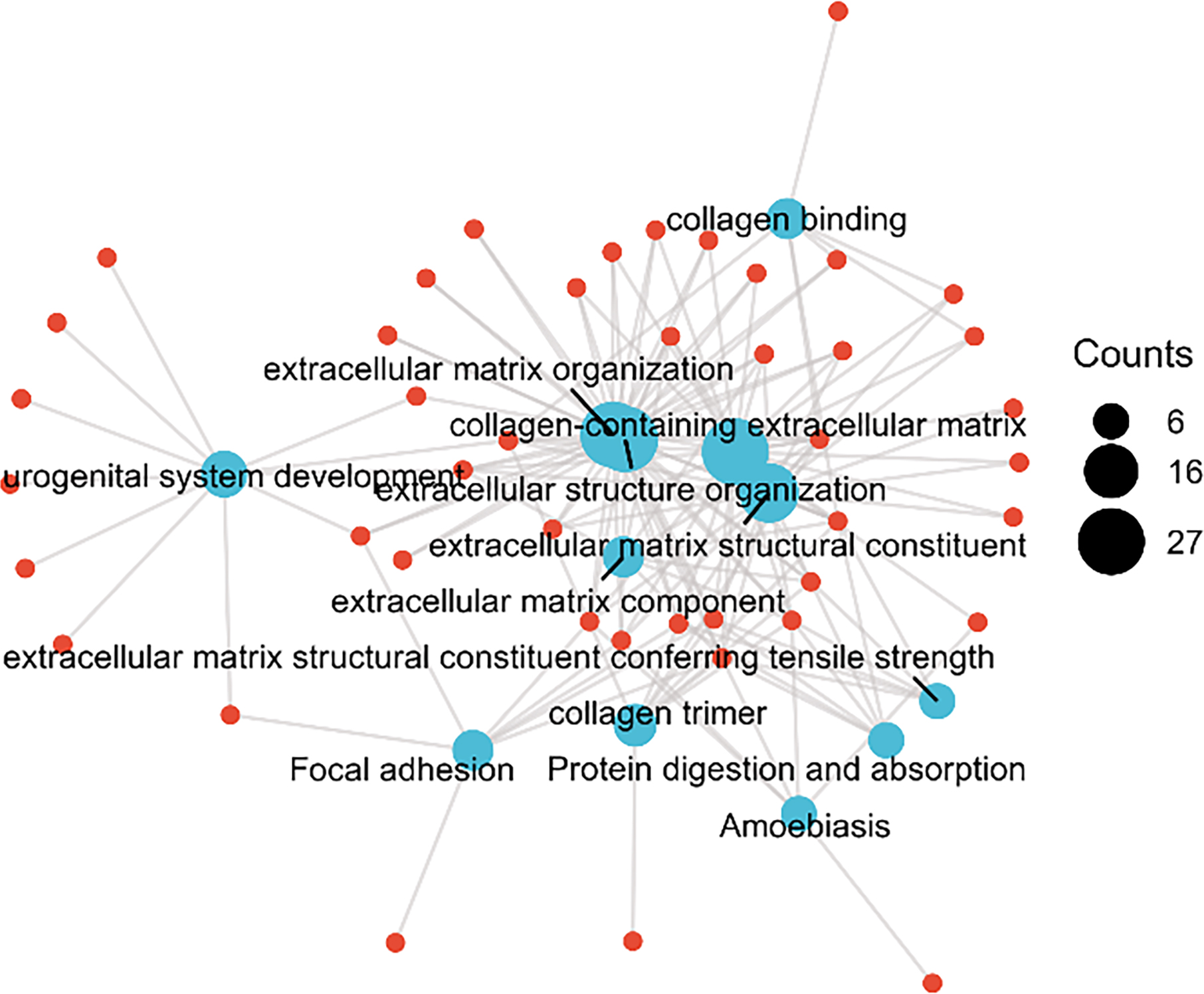
GSEA were conducted between high and low ANXA5 expression groups from TCGA-STAD dataset to analyze the possible biological pathways regulated by ANXA5 in STAD. A total of 81 pathways were significantly enriched in the ANXA5 high expression group. The results showed that the top 9 setsize terms included neutrophil degranulation (Fig. 5A), olfactory transduction (Fig. 5B) and singaline pathway (Fig. 5C), secreted factors (Fig. 5D), ECM organization (Fig. 5E), core matrisome (Fig. 5F), cytokine receptor interaction (Fig. 5G), platelet activation signaling and aggregation (Fig. 5H) and ECM regulators (Fig. 5I) were significantly enriched.
GSEA analysis of ANXA5 collected through TCGA-STAD dataset. NES: normalized enrichment score; FDR: false discovery rate.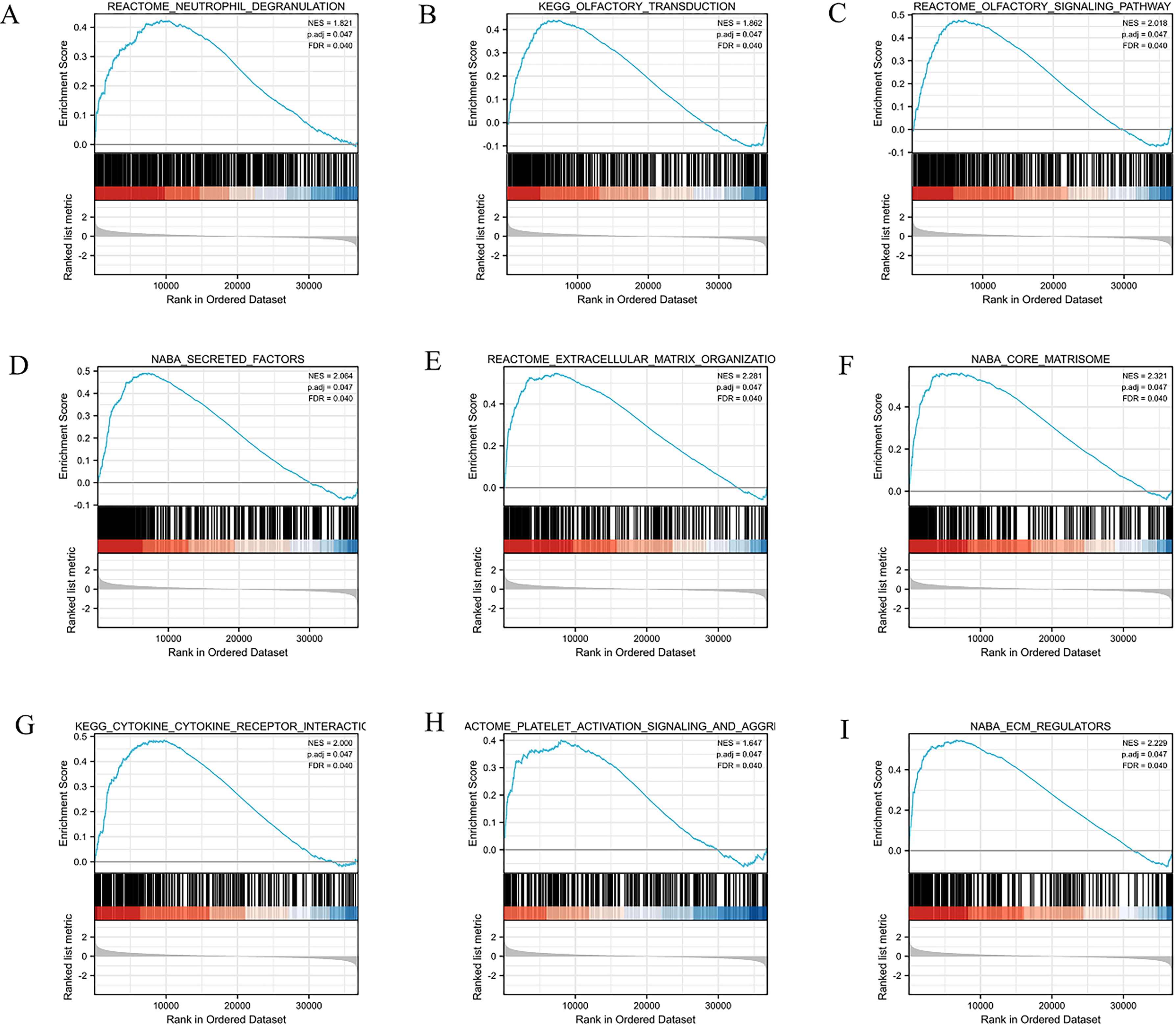
In this study, we found the correlation of ANXA5 expression with prognosis of STAD patients by using GEPIA. In multivariate analysis, upregulated ANXA5 expression was an independent predictive factors of poor prognosis. We also observed that ANXA5 expression differs in normal and tumor tissues of STAD through the analysis of multiple databases. To further study the underlying mechanisms of ANXA5 in STAD, we downloaded datasets from TCGA and enrichment analysis was performed. The results showed that the co-expressed genes were involved in ECM-related processes. We further analyzed the correlations between ANXA5 and immune cell infiltrates by using the TIMER database and found that ANXA5 expression was significantly correlated with various infiltrating immune cells in STAD.
ANXA5 is a member of the annexin family of proteins. Its gene, which is located on human chromosome 4q26-q28, encodes a non-glycosylated single-chain protein with 319 amino acids [17]. Annexin A5 has a molecular weight of 35.7 kD [18]. Accumulating evidence confirm that its abnormal expression is closely related to the biological behavior of a variety of malignant tumors, and that it plays multiple roles in proliferation of tumor cells, apoptosis, migration, and invasion. Ding et al. [19] found that inhibiting the expression of ANXA5 can significantly reduce the proliferation, migration, and invasion of human cholangiocarcinoma QBC-939 cells while increasing their apoptosis. However, Li et al. [11] demonstrated that overexpression of ANXA5 may inhibit the proliferation and metastasis of human cervical cancer cells. Although ANXA5 has both pro-tumor and anti-tumor effects, there are few studies in STAD, which piqued our interest in ANXA5 research.
Previous findings revealed that ANXA5 mRNA expression varies among cancers, suggesting that ANXA5 functions as both an oncogene and a tumor suppressor in different cancers [10, 11, 12, 19]. The role of ANXA5 in gastric cancer is still unclear. Recently, Wang et al. [20] detected the role of ANXA5 in gastric cancer cell lines MGC-803 and MKN-45. It was found that when the ANXA5 gene was knocked down, cell proliferation and metastasis were promoted, while apoptosis was inhibited. However, our investigation based on large transcriptome sequencing data from TACG, GEO and immunohistochemistry showed that ANXA5 was overexpressed in STAD relative to normal tissue. Logistic regression results revealed that ANXA5 expression significantly correlated with the pathological stage and tumor status. The ROC curve also demonstrated that ANXA5 is capable of predicting STAD. Furthermore, we found that increased ANXA5 expression was significantly associated with poor survival in STAD, and served as an independent risk factor for STAD. Altogether, these results suggested that ANXA5 may have a role in the STAD carcinogenic process and can be used as a prognostic marker.
Tumor-infiltrating immune cells are essential components of tumor stroma, and they play a role in tumor progression and response to cancer therapy [21, 22]. A noteworthy finding of our investigation was that there is a strongly positive relationship between the level of ANXA5 expression and invasion of CD8
To explore the potential biological processes of ANXA5, we conducted GO and KEGG analyses of the co-expressed genes, as well as GSEA analysis for ANXA5. The enrichment analysis of the top 100 co-expressed genes showed that ECM-related processes were significantly enriched. Moreover, the GSEA revealed that the terms neutrophil degranulation, secreted factors, ECM organization, core matrisome, cytokine receptor interaction and ECM regulators were significantly enriched in ANXA5 high expression phenotype. Relevant research has confirmed the ECM components and secreted factors contribute to tumor cell proliferation, migration and invasion in various cancers [26, 27]. The top set-size term of GSEA in our investigation was neutrophil degranulation. Neutrophil was originally considered responders to infection. However, it has been increasingly recognized as important players in cancer development. In addition, a growing body of evidence shows that neutrophil degranulation has been associated with tumor progression [28, 29]. Therefore, ANXA5 might participate in a variety of biological pathways in the STAD carcinogenic process and closely linked to immune infiltration.
The limitation of this study is that our findings were mainly based on bioinformatics analysis of multiple datasets and may not fully revealed the underlying biological mechanism of ANXA5 in STAD. Subsequent investigation for mechanism should combine in vivo and in vitro methods in the future. However, it is undeniable that the results of our investigation have demonstrated that increased ANXA5 expression correlates with poor prognosis and immune infiltration in STAD. Our work highlights the critical function of ANXA5 in immune infiltration, with a deeper understanding of its function, which has the potential to be used as a STAD prognostic biomarker and a potential target for immunotherapy.
Footnotes
Acknowledgments
This research is supported by the Clinical Medicine Special Project of Nantong University (No. 2019JQ015).
Author contributions
Conception: Su Zhaoran and Li Guangyao.
Interpretation or analysis of data: Su Zhaoran and Shu Kuanshan.
Preparation of the manuscript: Su Zhaoran.
Revision for important intellectual content: Li Guangyao.
Supervision: Li Guangyao.
