Abstract
BACKGROUND:
More novel biomarkers need to be discovered to improve the therapeutic efficiency of non-small cell lung cancer (NSCLC). lncRNA ELFN1-AS1 (ELFN1-AS1) was proved to play crucial roles in numerous diseases, its intention in NSCLC remains unclear.
OBJECTIVE:
This study aimed to investigate the function of ELFN1-AS1 and its potential mechanism in NSCLC development.
METHODS:
A total of 117 NSCLC patients were recruited and provided paired NSCLC tissues and normal tissues. The expression of ELFN1-AS1 was analyzed by PCR. The biological function of ELFN1-AS1 was estimated by CCK8 and Transwell assay. Additionally, the potential mechanism underlying the function of ELFN1-AS1 was explored by the dual-luciferase reporter assay and western blotting.
RESULTS:
The significant upregulation of ELFN1-AS1 was found in NSCLC tissues and cells, which was closely associated with the TNM stage, lymph node metastasis status, and overall survival of patients. The knockdown of ELFN1-AS1 was found to inhibit the cellular processes and EMT of NSCLC. Moreover, ELFN1-AS1 was found to serve as a sponge to binding with miR-497, and CCNE1 was demonstrated to be the downstream target of miR-497, which was speculated as the potential mechanism underlying the function of ELFN1-AS1.
CONCLUSIONS:
ELFN1-AS1 acts as an independent prognostic biomarker and tumor promoter of NSCLC by sponging miR-497/CCNE1 axis.
Introduction
Non-small cell lung cancer (NSCLC), including the subtypes of squamous cell carcinoma, adenocarcinoma, and large cell carcinoma, is one of the major pathological classifications of lung cancer, accounting for a large percentage of all cancer death causes [1]. Due to the lack of representative indicators, most NSCLC patients are always diagnosed at an advanced stage, and therefore results in the poor treatment efficiency and shorten survival [2]. On the other hand, metastasis is closely associated with disease development, therefore, monitoring tumor progression is also a critical means to improve the therapy of NSCLC. With the development of science and technologies, great progress has been made in the early detection and therapeutic strategies of NSCLC, but the clinical prognosis of patients is still unoptimistic.
Long non-coding RNAs (lncRNAs) are a family of RNAs in 200 nucleotides length that comprise 9277 manually annotated lncRNA genes and produce 14880 transcripts [3, 4]. The function of lncRNAs has attracted special attention in recent genetic studies. lncRNAs were known to participate in cell differentiation, growth, and migration, which makes them serve as novel master regulators in the initiation, progression, and therapeutic response of various malignancies [5]. In human cancers, a number of lncRNAs were considered as important regulators of tumor progression, which were involved in tumor proliferation, invasiveness, and stem cell reprogramming [6]. These functional lncRNAs always possess abnormal expression levels. These kinds of lncRNAs have also been identified in NSCLC, such as lncRNA TUC338, lncRNAMALAT1, and LINC PINT that regulated the development and associated with prognosis of patients [7, 8, 9]. LncRNA ELFN1-AS1 (ELFN1-AS1) was found to be dysregulated in NSCLC, while it was also revealed to be dysregulated in other cancers and play important roles, such as esophageal cancer, ovarian cancer, and colorectal cancer [10, 11, 12, 13]. However, whether ELFN1-AS1 was involved in the development of NSCLC and the mechanism underlying its potential functional role remains unclear.
The expression of ELFN1-AS1 was investigated in NSCLC and its clinical significance was also estimated. Meanwhile, the biological function of ELFN1-AS1 was also studied in cell growth, migration, and invasion of NSCLC.
Materials and methods
Clinical samples
A total of 117 paired NSCLC tissues and adjacent normal tissues were collected from patients diagnosed with NSCLC and received surgical resection at China-Japan Union Hospital of Jilin University from January 2013 to December 2015. The collected tissues were frozen in liquid nitrogen and stored at
Cell culture and cell transfection
A549, H1299, HCC827, HCC2279, and BEAS-2B cells were purchased from ATCC. All cells were supplied with the RPMI-1640 medium (Invitrogen, USA) containing 10% FBS (Invitrogen, USA) and 100
Cells were transfected with si-ELFN1-AS1 or corresponding negative controls with the help of Lipofectamine 2000 reagent (Invitrogen, USA) according to the instructions. Transfection efficacy was verified by real-time qPCR after 48 h of cell transfection.
Real-time qPCR
The TRIzol (Invitrogen, USA) was used to lysed collected tissues and cultured cells. The lysis was mixed with chloroform for 30 s followed by centrifuging. The aqueous phase was replaced by an equal volume of pre-cold isopropanol. Then, the precipitate was washed with 75% ethanol and was air dried.
The extracted RNA was reverse transcribed to cDNA with the ReverTra Ace qPCR RT kit (Toyobo, Japan). Real-time qPCR was performed with SYBR Green PCR Kit (Promega, USA) and the StepOnePlus real-time PCR system (Applied Biosystem, USA). The relative expression of ELFN1-AS1, miR-497, and CCNE1 was calculated by the 2
CCK8 assay
Cells were seeded into 96-well plates and supplied with the RPMI-1640 medium. After incubating for 0, 24, 48, and 72 h, the CCK8 (Dojindo, Japan) was added to each well and the absorbance (450 nm) was detected by a microplate reader.
Transwell assay
Cells were seeded into the Transwell chambers with or without the pre-coating of Matrigel (for cell invasion assay). The top chamber was filled with RPMI-1640 medium without FBS, while the completed culture medium was added to the bottom chamber as a chemoattractant. The Transwell chambers were cultured at 37
Dual-luciferase reporter assay
The wild or mutated binding sites of ELFN1-AS1 (Fig. 5A) or CCNE1 (Fig. 7A) with miR-497 were inserted into the pGL3 luciferase reporter vectors (Promega, USA) to synthesize the ELFN1-AS1 wild (ELFN1-AS1 WT), ELFN1-AS1 mutant (ELFN1-AS1 MT), CCNE1 wild, and CCNE1 mutant vectors. The pGL3 reporter luciferase vectors were co-transfected with miR-497 mimic or miR-497 inhibitor into HCC827 cells for 48 h. The relative luciferase activity was estimated with the Dual-Luciferase Reporter Assay System (Promega, USA) according to the instructions.
Western blotting
Proteins were extracted from HCC827 cells and evaluated the concentration with the BCA kit (Thermo Fisher, USA). The isolated protein was mixed with loading buffer and boiled followed by electrophoresing on the 10% SDS-polyacrylamide gel for 1.5 h and transferring to a PVDF membrane for 2 h. Then the membrane was blocked with 5% skim milk at room temperature for 2 h followed by the incubation with antibodies (anti-E-cadherin, anti-Vimentin, anti-N-cadherin, and anti-GAPDH) at 4
Statistical analysis
All data were represented as mean value
Results
ELFN1-AS1 was upregulated in NSCLC
The expression of ELFN1-AS1 in NSCLC tissues was significantly higher than that in matched normal tissues (
ELFN1-AS1 was associated with the development of NSCLC and the prognosis of patients
Among the clinicopathological features of patients, the TNM stage (
Association between ELFN1-AS1 and the clinicopathological features of NSCLC patients
Association between ELFN1-AS1 and the clinicopathological features of NSCLC patients
LNM: lymph node metastasis;
The expression levels of ELFN1-AS1 in NSCLC tissues and cells. A. ELFN1-AS1 was significantly upregulated in NSCLC tissues compared with matched normal tissues. B. The expression of ELFN1-AS1 was raised in NSCLC cells, A549, H1299, HCC827, and HCC2279. 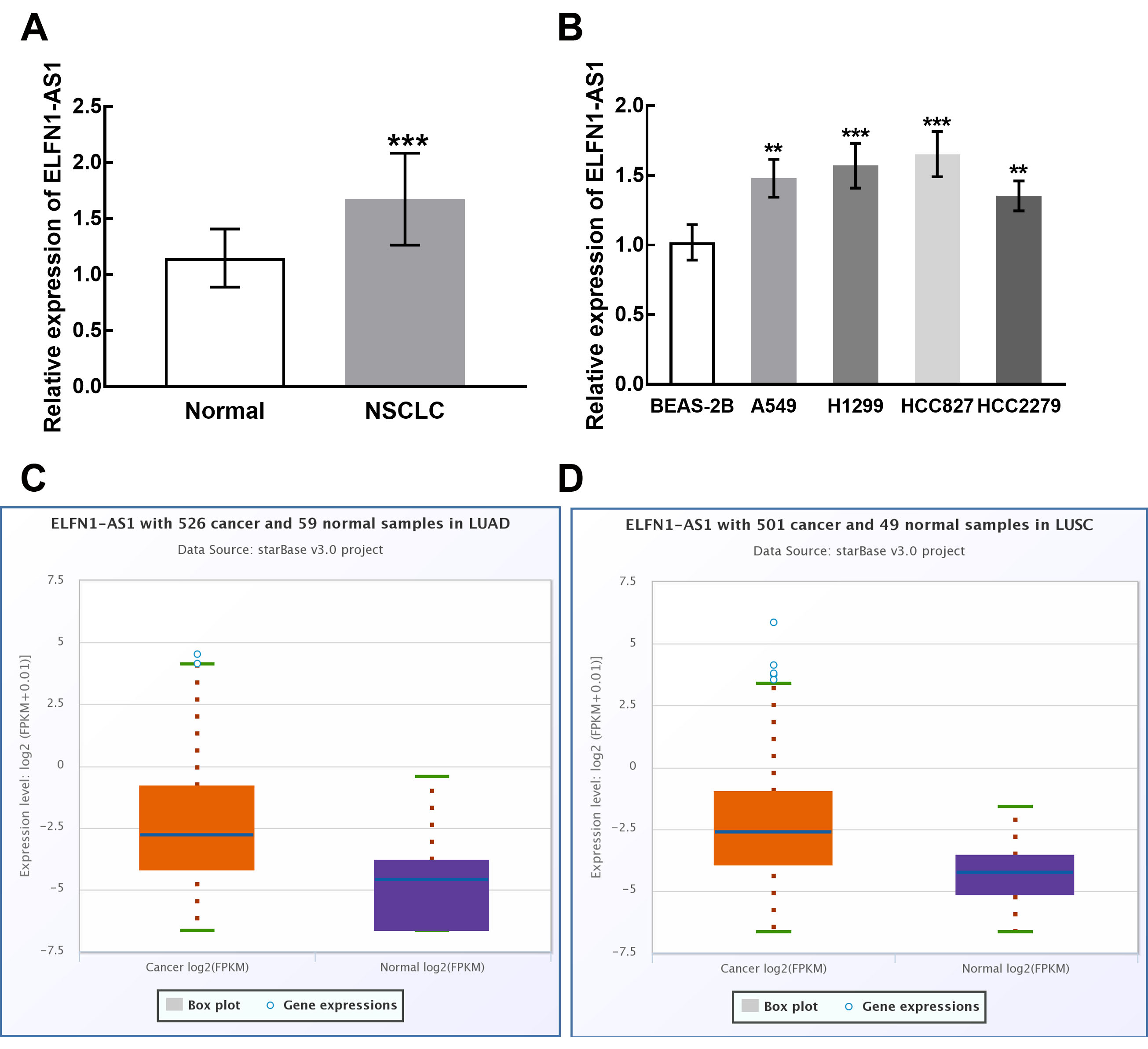
On the other hand, patients that possessed high ELFN1-AS1 expression had a shorter survival time than that of patients who possessed low ELFN1-AS1 expression (Log-rank
Cox regression analysis evaluated the prognostic significance of patients’ clinicopathological features
LNM: lymph node metastasis;
Association of ELFN1-AS1 expression with the survival of NSCLC patients. Patients in the high expression group possessed a shorter survival time than that of patients in the low expression group. The difference was significant. Log-rank 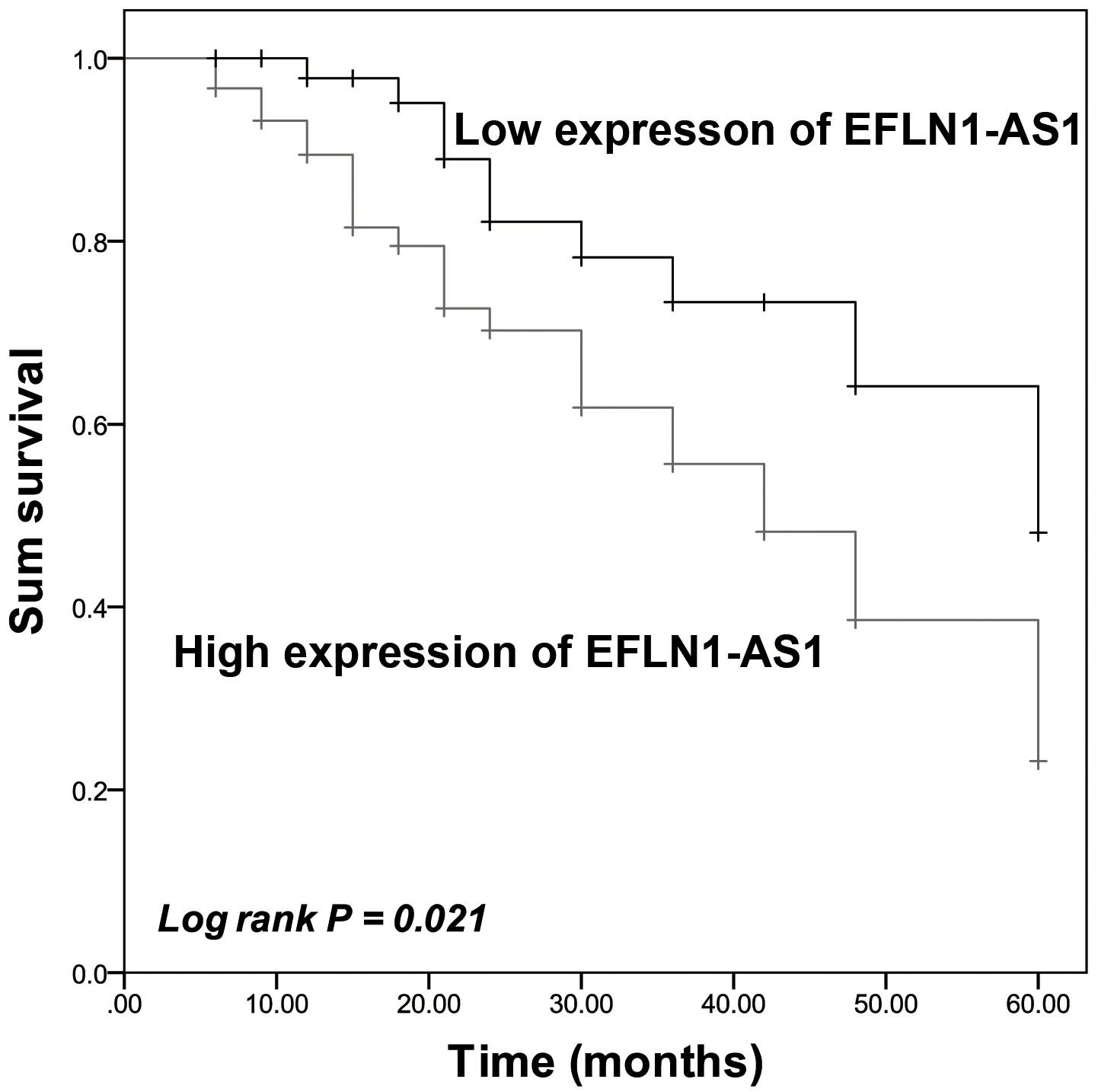
The transfection of si-ELFN1-AS1 significantly suppressed the expression of ELFN1-AS1 in H1299 and HCC827 cells (
The expression levels of ELFN1-AS1 after cell transfection in H1299 and HCC827 cells. A. The expression of ELFN1-AS1 was significantly suppressed by the transfection of siRNA-ELFN1-AS1 in H1299 cells. B. The expression of ELFN1-AS1 was significantly suppressed by the transfection of siRNA-ELFN1-AS1 in HCC827 cells. 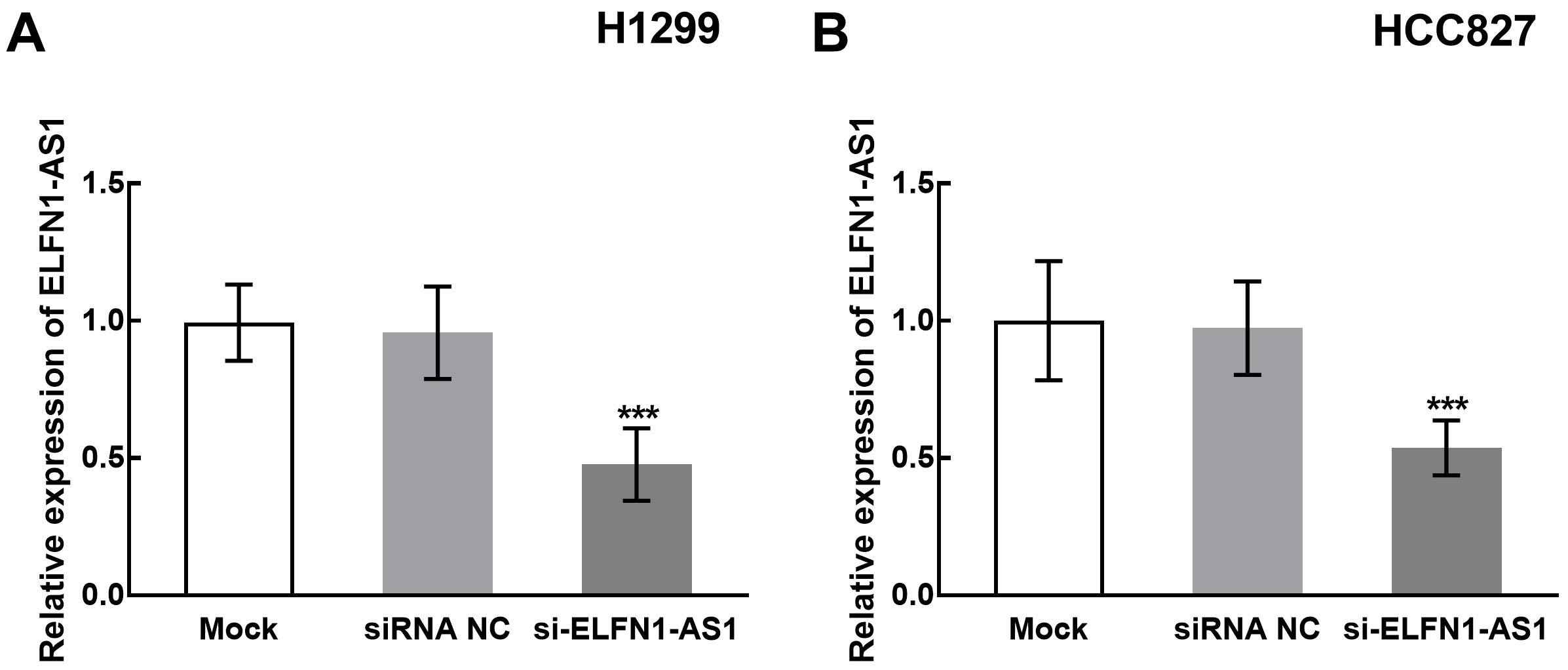
Effect of ELFN1-AS1 on the proliferation, migration, and invasion of NSCLC cells. A. The knockdown of ELFN1-AS1 significantly inhibited the proliferation of H1299 and HCC827 cells. B. The migration of H1299 and HCC827 cells was significantly suppressed by the knockdown of ELFN1-AS1. C. The invasion of H1299 and HCC827 cells was significantly suppressed by the knockdown of ELFN1-AS1. 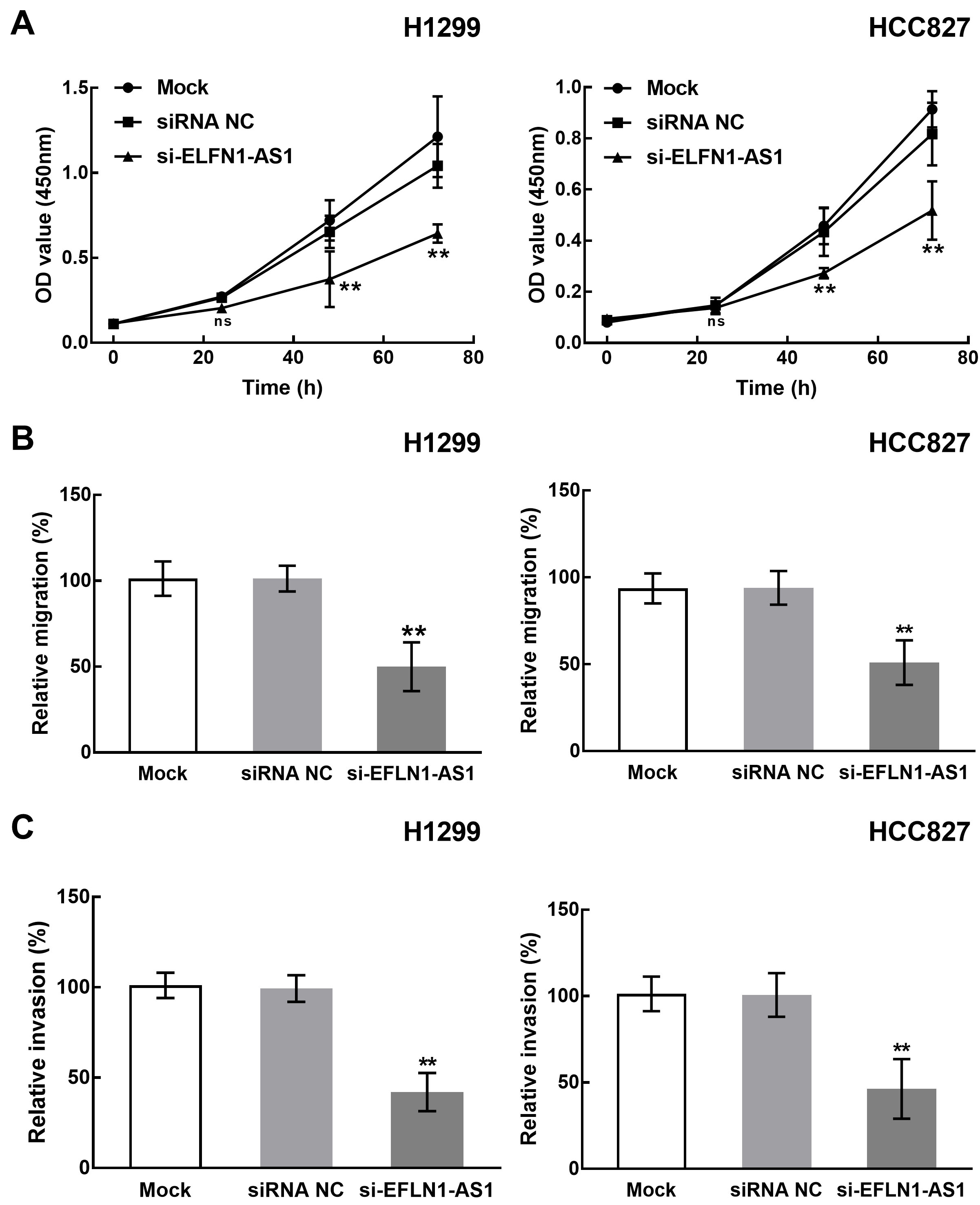
The interaction between ELFN1-AS1 and miR-497 in HCC827 cell. A. The overexpression of miR-497 significantly inhibited the luciferase activity of ELFN1-AS1, whereas the silencing of miR-497 significantly enhanced the luciferase activity of ELFN1-AS1. B. The knockdown of ELFN1-AS1 by siRNA transfection was reversed by the downregulation of miR-497. C. The knockdown of ELFN1-AS1 significantly promoted the expression level of miR-497. D. ELFN1-AS1 silencing significantly suppressed the expression of Vimentin and N-cadherin and enhanced E-cadherin, which was alleviated by miR-497 silencing. 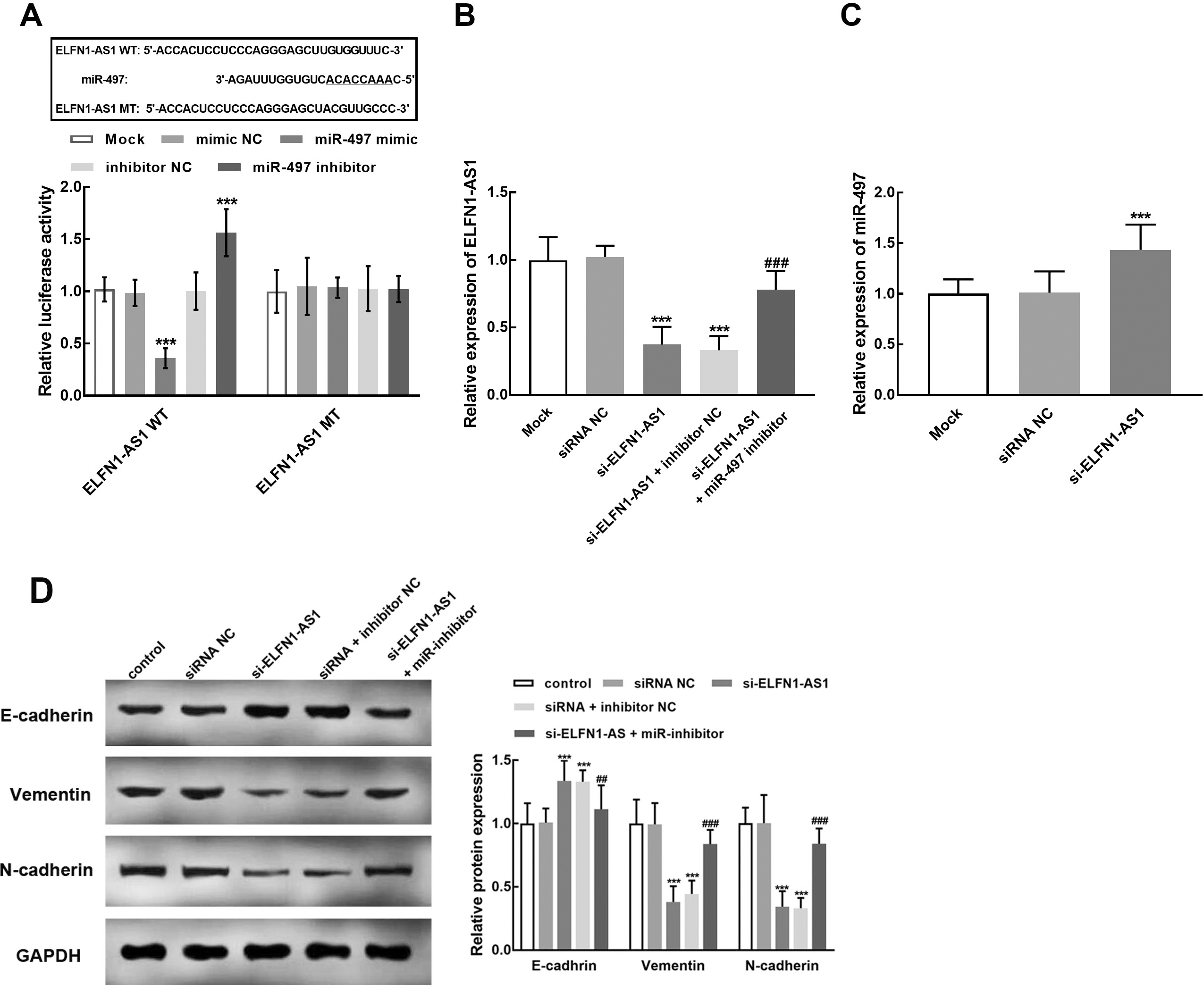
The binding region between miR-497 and ELFN1-AS1 was predicted and constructed into the luciferase reporter vectors (Fig. 5A). The overexpression of miR-479 significantly suppressed the relative luciferase activity of ELFN1-AS1, while the silencing of miR-479 dramatically enhanced the luciferase activity of ELFN1-AS1 (
Meanwhile, the reversed effect of miR-497 was also observed in the regulatory effect of ELFN1-AS1 in HCC827 cells. Specifically, the knockdown of miR-497 significantly attenuated the inhibitory effect of ELFN1-AS1 silencing on the proliferation (Fig. 6A), migration (Fig. 6B), and invasion (Fig. 6C) of HCC827 cells (
The role of miR-497 during the regulatory effect of ELFN1-AS1 in NSCLC. The knockdown of miR-497 significantly reversed the inhibitory effect of ELFN1-AS1 downregulation on the proliferation (A), migration (B), and invasion (C) of NSCLC cells. 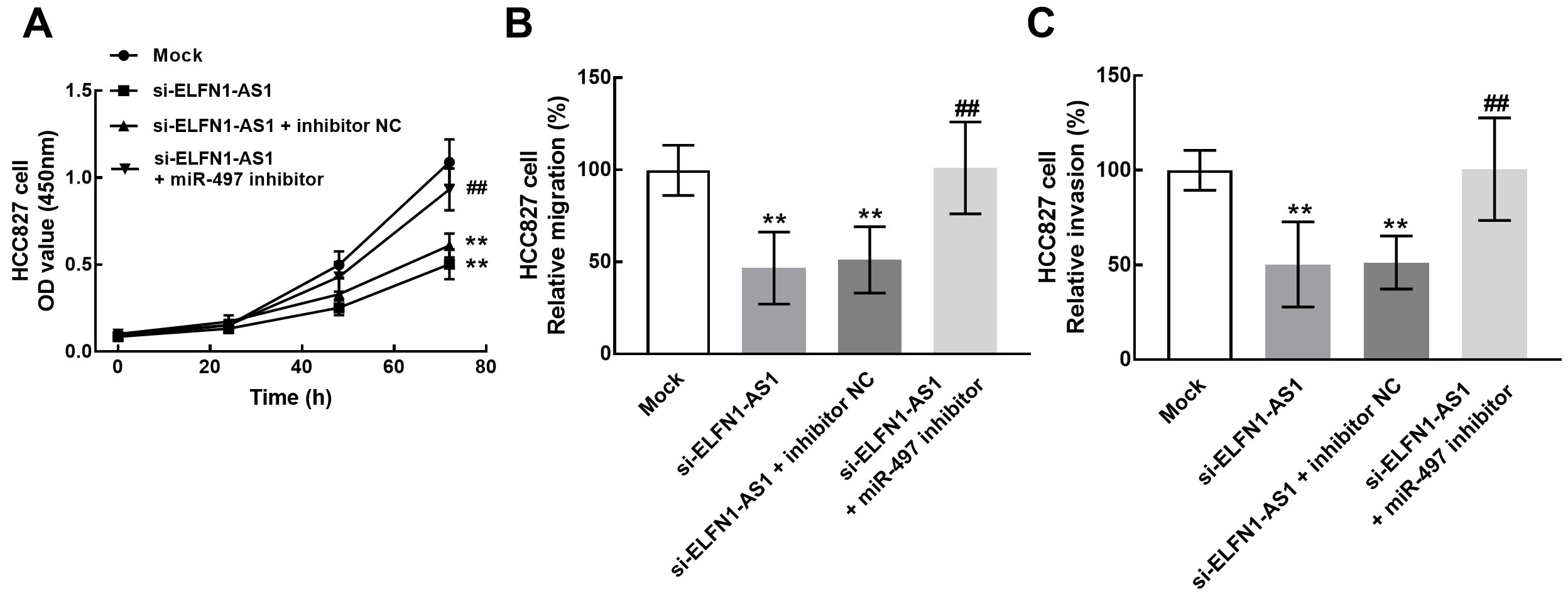
The role of CCNE1 during the regulatory effect of ELFN1-AS1 and miR-497 in NSCLC. A. miR-497 negatively regulated the luciferase activity of CCNE1 WT, while the CCNE1 MT was not affected. B. The knockdown of ELFN1-AS1 dramatically suppressed the expression of CCNE1, which was reversed by the silencing of miR-497. 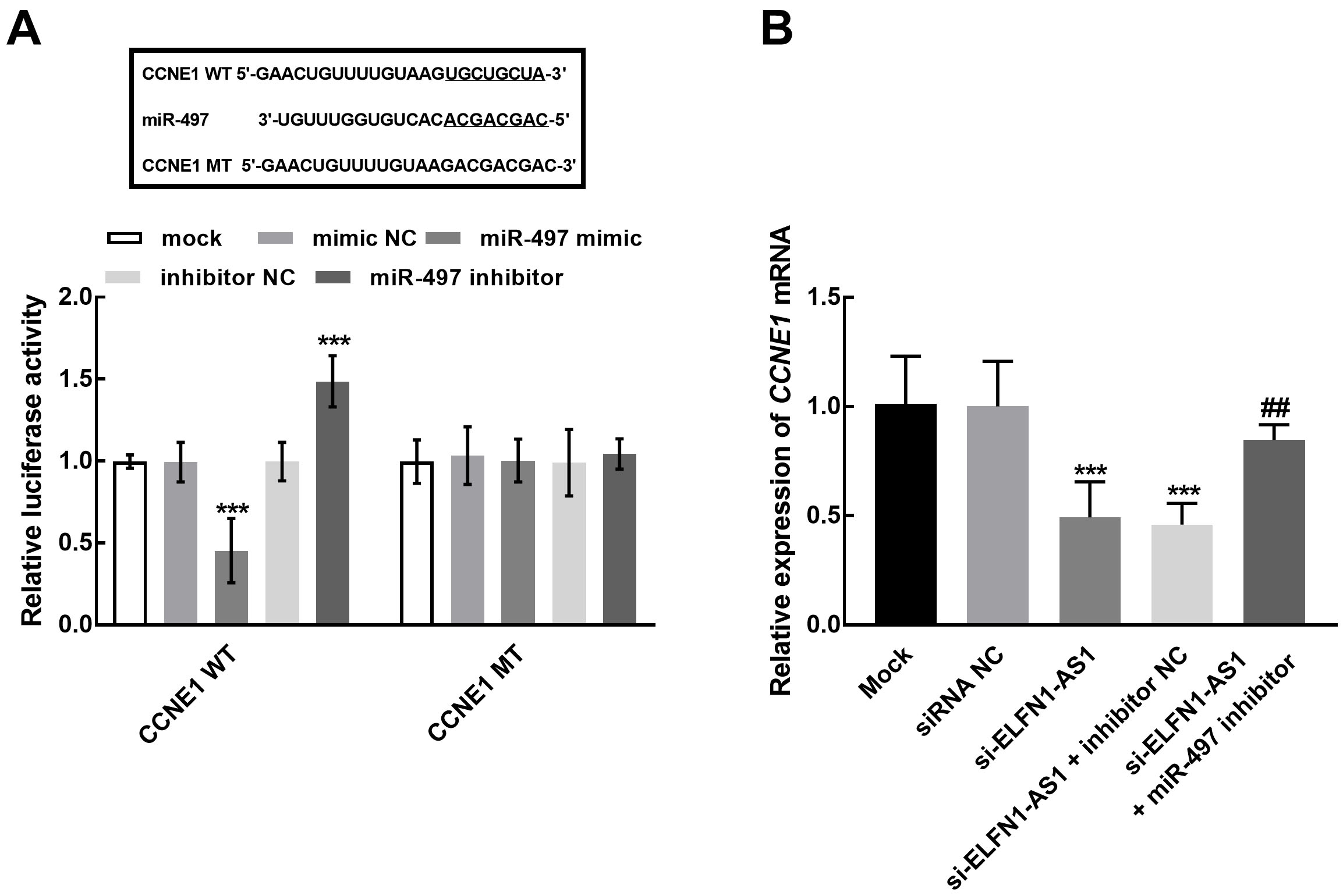
Recently, the abnormally expressed lncRNAs have attracted special attention in the studies on cancer occurrence and development [14, 15, 16, 17]. In NSCLC, several lncRNAs have been identified as functional molecules that play roles in the tumor progression and act as diagnostic and prognostic indicators [18, 19]. Here, the function of ELFN1-AS1 in NSCLC was investigated with a series of in vivo and in vitro experiments. The significantly raised expression of ELFN1-AS1 was found in NSCLC. The upregulation of ELFN1-AS1 was related to the positive lymph node metastasis status and the advanced TNM stage, which are critical indicators of disease progression. Therefore, ELFN1-AS1 was speculated to be involved in the development of NSCLC. Previously, the clinical significance of lncRNAs has been reported. For instance, Lin et al. filtered seven dysregulated lncRNAs and demonstrated the significant association with the overall survival of NSCLC patients [20]. Circulating lncRNA GAS5 and SOX2OT were identified as biomarkers for the diagnosis and prognosis of NSCLC with high sensitivity and specificity [21]. The significance of ELFN1-AS1 in the prognosis of NSCLC patients was further estimated. The expression of ELFN1-AS1 was revealed to be negatively correlated with the survival of NSCLC patients. ELFN1-AS1 was also identified as an independent prognostic indicator together with the TNM stage of patients.
For the biological function of ELFN1-AS1, the expression levels of ELFN1-AS1 in NSCLC cells were downregulated by the transfection of siRNA-ELFN1-AS. Previous studies reported that ELFN1-AS1 could drive colon cancer cell growth and cell invasion through regulating the corresponding molecular axis and umbilical cord mesenchymal stem cells [22, 23]. In esophageal cancer, ELFN1-AS was also found to exert promoted effects on tumor progression through regulating GFPT1 and sponging miR-183-3p [12]. The knockdown of ELFN1-AS1 showed significant inhibitory effects on cellular processes of NSCLC, indicating the tumor promoter role of ELFN1-AS1 in NSCLC, which is consistent with previous reports.
Additionally, the mechanism underlying the function of ELFN1-AS was also explored. The interplay between ELFN1-AS1 and miRNAs has been widely studied [24]. ELFN1-AS1 has been demonstrated to interact with miR-497 in ovarian cancer, and miR-497 also serve as a tumor regulator in various human cancers, including NSCLC. It has been reported that miR-497 inhibited the malignant phenotype and could inhibit the tumorigenesis of lung cancer [25, 26]. Herein, in NSCLC cells, ELFN1-AS1 was disclosed to binding with miR-497 as a role of sponge, which provides direct evidence for the potential mechanism underlying ELFN1-AS1’s functional role. Meanwhile, miR-497 was also found to mediate the inhibitory effect of ELFN1-AS1 on the major cellular processes of NSCLC, including cell proliferation, migration, and invasion. On the other hand, EMT is a process that involved a number of distinct mesenchymal markers, such as N-cadherin, Vimentin, and E-cadherin, and has been demonstrated to be closely related to cancer progression [27]. The enhancement of EMT by ELFN1-AS1 was observed in the present study, which was mediated by miR-497. Hence, ELFN1-AS1 was speculated to promoted EMT through sponging miR-497 and therefore promoted the progression of NSCLC. Moreover, the downstream target of miR-497 is also a vital mechanism that participate the function of ELFN1-AS1 in NSCLC progression. Previously, the functional role of miR-497 was demonstrated to result from targeting various tumor-related genes, such as SOX5, YAP1, FGFR1, and KDR [26, 28, 29, 30]. CCNE1 has been widely demonstrated to play roles in various tumor progression and mediate the function of numerous molecules. For instance, in triple-negative breast cancer, CCNE1 was disclosed to mediate the promoter role of circAGFG1 and served as a potential biomarker [31]. The interaction between miR-497 and CCNE1 has been revealed in lung cancer, which significantly retard the growth of lung cancer cells [32]. Herein, miR-497 was found to negatively regulate CCNE1, while ELFN1-AS1 showed a positive effect on the expression of CCNE1. Therefore, regulating CCNE1 was speculated to be the underlying mechanism of ELFN1-AS1/miR-497 axis in NSCLC development.
In conclusion, ELFN1-AS1 was significantly upregulated in NSCLC, which was associated with the disease development of NSCLC. ELFN1-AS1 served as an independent prognostic indicator to predict the survival of NSCLC patients. Moreover, ELFN1-AS was identified as a tumor promoter that promoted cell proliferation, migration, invasion, and EMT through sponging miR-497/CCNE1 axis.
Footnotes
Acknowledgments
None.
Author contributions
Conception: Shuai Miao.
Interpretation or analysis of data: Bin Yang.
Preparation of the manuscript: Bin Yang.
Revision for important intellectual content: Shuai Miao.
Supervision: Shuai Miao.
