Abstract
Background
The aim of this study was to investigate the relationship between Nin one binding (NOB1) protein expression and prognosis for resected non-small cell lung cancer (NSCLC).
Methods
A prospective cohort of 70 consecutive patients with resected NSCLC was studied in 2009. Immunohistochemistry was used in the detection of NOB1 protein expression. Prognosis outcomes included overall survival (OS) and progression-free survival (PFS). The log-rank test and Cox hazard model were used to estimate the relationship between NOB1 expression and prognosis.
Results
In the 70 NSCLC tissue specimens, 14 (20%) stained -, 24 (34%) stained +, 21 (30%) stained ++ and 11 (16%) stained +++. The NOB1 high expression rate was 16%. NOB1 expression was significantly different between TMN stage (p=0.024) and lymph node metastasis (p=0.001), as well as histopathological grades (p=0.037). Median OS was 43 months (95% confidence interval [95% CI], 35-51 months), and median PFS was 37 months (95% CI, 25-49 months). OS and PFS were related to TMN stage and lymph node metastasis, as well as NOB1 expression (p<0.05). After adjustment for TMN stage and lymph node metastasis, the hazard ratio (HR) for high NOB1 expression was 1.7 (95% CI, 1.1-3.0, p=0.027) for OS, and 1.8 (95% CI, 1.3-3.7, p=0.031) for PFS.
Conclusions
Our results suggest that enhanced expression of NOB1 is related to poor overall survival and progression-free survival in patients with resected NSCLC.
Introduction
Lung cancer is the most common cancer in the world. Non-small cell lung cancer (NSCLC) is the most common type of lung cancer; about 85% of lung cancers are NSCLC (1, 2). Surgical resection remains the single most consistent and successful option for early-stage lung cancer (1, 2). However, the prognosis is still poor after surgical resection. The 5-year disease-specific mortality in stage I-IIIA subgroups is approximately a 30%, 60% and 75%, respectively (3). It is important to predicate the prognosis in resected NSCLC.
However, accurate prediction of prognosis outcome in NSCLC also remains challenging. Even within the same stage, same performance status, same treatment group and same response to treatment, survival varies from patient to patient (4). Some studies have found that many molecular markers were related to the prognosis for resected NSCLC. These poor prognosis–related molecular markers include epidermal growth factor receptor overexpression (5), excision repair cross-complementing 1 overexpression and xeroderma pigmentosum group G low expression (6), positive expression of CD98 (7) and of carbonic anhydrase XII overexpression (8).
Nin one binding (NOB1) protein is a subunit of the 26S proteasome and plays a crucial role in protease function and RNA metabolism (9). Recently, it has been increasingly recognized that abnormal expression of NOB1 plays a significant role in tumorigenesis (10). In some clinical studies, NOB1 has been confirmed to be related to cancer prognosis. Oehler et al found NOB1 was one of the 6 gene expression profiles which can predict disease and prognosis of chronic myeloid leukemia (11). Zhou et al reported that high levels of NOB1 were associated with unfavorable prognosis of glioma patients (12). Che et al also found in patients with prostate carcinoma that NOB1 expression had a significant positive correlation with patient survival (13).
In NSCLC, our previous study found that NOB1 protein was significantly increased in cancer tissue compared with adjacent tissue and normal lung tissue, and NOB1 expression was significantly associated with NSCLC TNM stage, lymph node metastasis and histopathological grade (14). In this study, we investigated the relationship between NOB1 expression and prognosis for resected NSCLC in the same patient cohort.
Materials and Methods
Study design
We studied a prospective cohort among consecutive patients with resected NSCLC in 2009. Inclusion criteria included patients undergoing resection for NSCLC and with NSCLC diagnosis confirmed by histopathological examination. Exclusion criteria were patients having received preoperative chemotherapy or radiotherapy. The study was approved by the medical ethics committee of our hospital.
Patients and clinicopathological data
Clinicopathological data of the 70 patients included were available from the previous studies of the same cohort (14). The cohort consisted of 39 men and 31 women, ranging in age from 48 to 79 years. Forth-two patients had tumors <3 cm; 28 patients had tumors ≥3 cm. According to the current Union for International Cancer Control (UICC) criteria, 47 patients had stage I or II disease, and 23 patients had stage III disease. There were 29 cases of lymph node metastasis and 41 with no lymph node metastasis; 1 well-differentiated case, 45 moderately differentiated cases and 24 poorly differentiated cases.
Immunohistochemistry and quantitative analysis
We collected tissue blocks of the resected lung cancer tissue of the 70 patients. Immunohistochemistry for NOB1 expression was completed as described previously (14). All staining was analyzed by 2 observers unaware of clinicopathological data.
Quantitative analysis included percentages of NOB1-positive cells and the NOB1 staining intensities. The percentages of positive cells were scored in 4 categories according to staining: 0 for 0%, 1 for 1%-33%, 2 for 34%-66% and 3 for 67%-100%; and the staining intensities were also scored in 4 grades: 0 for no stain, 1 for light yellow, 2 for yellowish brown and 3 for brown. The multiplied values of the percentages and intensity scores were used as the final staining scores: 0 for -, 1-3 for +, 4-6 for ++ and 7-9 for +++. High NOB1 expression was defined as staining score +++.
Follow-up and prognosis outcomes
All patents received follow-up after surgery. The follow-up period was set as 60 months. Data for patients lost to follow-up or for those whose follow-up reached 60 months were censored.
The outcomes included overall survival (OS) and progression-free survival (PFS). OS was defined as the proportion of patients within a group who were expected to be alive after a specified time. OS takes into account death due to any cause – both related and unrelated to the cancer in question. PFS was defined as the proportion of patients among those treated for a cancer whose disease remained stable (without signs of progression) at a specified time after treatment.
Statistical analysis
For comparison of the baseline values between different NOB1 high expression groups, we used the chi-square test and Fischer's exact test. To compare the prognosis outcomes (OS and PFS) between the NOB1 low expression group and NOB1 high expression group, we used a time-to-event strategy with Kaplan-Meier estimates, followed by a log-rank test for univariate analysis. Hazard ratios (HRs), their 95% confidence intervals (95% CI) and p values were calculated by Cox proportional hazard model. We also used the Cox proportional hazard model to adjust the effects by other factors related to OS and PFS for multivariate analyses. All statistical analyses were performed using IBM SPSS software (version 19.0).
Results
NOB1 expression in NSCLC tissues
Immunohistochemistry staining showed NOB1 protein was localized in both the cytoplasm and the nucleus of the malignant cells, but intensely localized in the cytoplasm. In the 70 NSCLC tissue specimens, 14 (20%) stained -, 24 (34%) stained +, 21 (30%) stained ++ and 11 (16%) stained +++. The NOB1 high expression rate was 16%. NOB1 expression in NSCLC tissue by immunohistochemistry is shown in Figure 1.
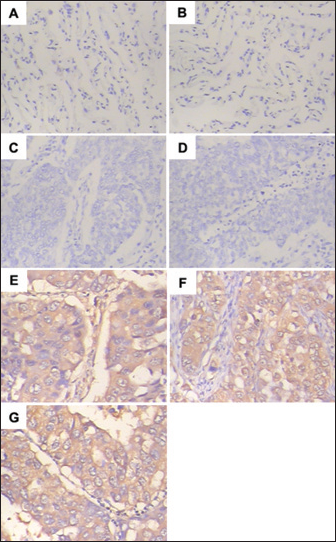
Evaluation of NOB1 expression in non-small cell lung cancer (NSCLC) tissue by immunohistochemistry. A-D) NOB1 negative expression; E-G) NOB1 positive expression (E: staining +; F: staining ++; and G: staining +++).
Relationship between NOB1 expression and clinicopathological features in patients with NSCLC
There were no significant differences between NOB1 expression and sex, age at the time of surgery, smoking status or tumor diameter (p>0.05). However, NOB1 expression showed significant differences for TMN stage (p=0.024) and lymph node metastasis (p=0.001), as well as histopathological grade (p=0.037). The relationships between NOB1 expression and clinicopathological features are summarized in Table I.
Relationships Between Nob1 Expression And Clinicopathological Features Of Study Population
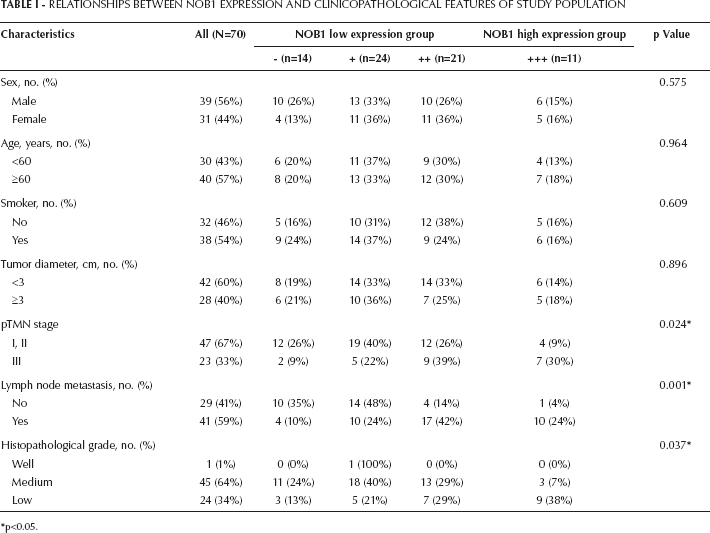
p<0.05.
NOB1 expression and prognosis in patients with NSCLC
Follow-up data were available for all patients. Median OS was 43 months (95% CI, 35-51 months), and median PFS was 37 months (95% CI, 25-49 months).
The log-rank test showed OS and PFS had no relationship with patients‘ sex, age, smoking status, tumor diameter and histopathological grade (p>0.05), but were related with TMN stage and lymph node metastasis, as well as NOB1 expression (p<0.05). Median OS in the NOB1 low expression group was 48 months (95% CI, 34-62 months) and 22 months (95% CI, 0-48 months) in the NOB1 high expression group (Kaplan-Meier curves are shown in Fig. 2A). The same difference was found for median PFS between the NOB1 low expression group and high expression group: 43 months (95% CI, 23-63 months) and 15 months (95% CI, 5-39 months), respectively (Kaplan-Meier curves are shown in Fig. 2B). Univariate log-rank test analysis results for OS and PFS of clinical, histopathological and immunohistochemical parameters are listed in Table II.
Univariate Analysis For Overall Survival And Progression-Free Survival, Of Clinical, Histopathological And Immunohistochemical Parameters
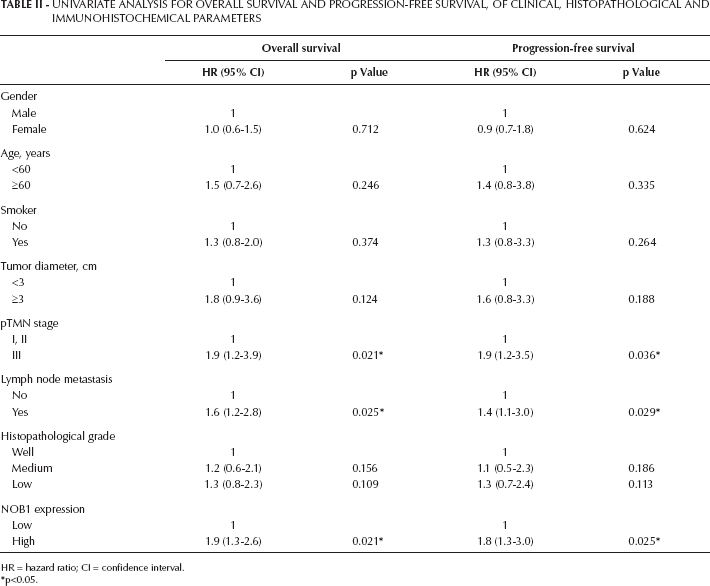
HR = hazard ratio; CI = confidence interval.
p<0.05.
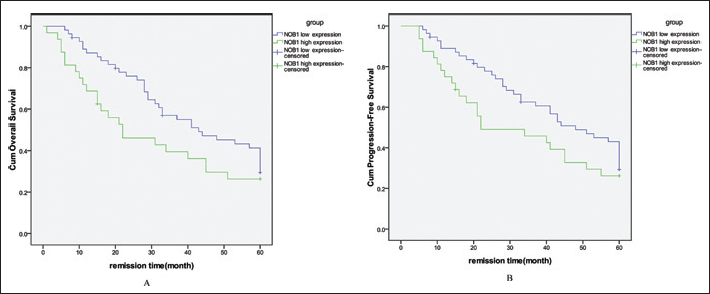
Kaplan-Meier curves for cumulative overall survival (A) and cumulative progression-free survival (B) according to level of NOB1 expression.
Multivariate Cox analysis showed TMN stage, lymph node metastasis and NOB1 expression were independent prognostic factors for OS and PFS in patients with NSCLC. After adjustment for TMN stage and lymph node metastasis, the HR for high NOB1 expression was 1.7 (95% CI, 1.1-3, p=0.027) for OS, and 1.8 (95% CI, 1.3-3.7, p=0.031) for PFS. Multivariate Cox analysis results for OS and PFS of clinical, histopathological and immunohistochemical parameters are shown in the Table III.
Multivariate Analysis For Overall Survival And Progression-Free Survival, Of Clinical, Histopathological And Immunohistochemical Parameters
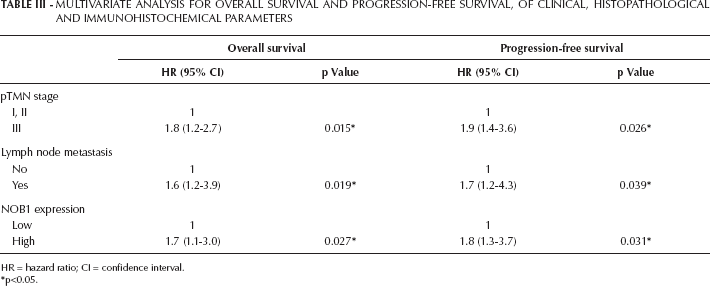
HR = hazard ratio; CI = confidence interval.
p<0.05.
Discussion
NOB1 was first isolated by the yeast 2-hybrid screening method using Rpn12, a subunit of the lid complex of the 19S regulatory particle (15). The nuclear protein NOB1p serves as a chaperone to join the 20S proteasome with the 19S regulatory particle in the nucleus and facilitates the maturation of the 20S proteasome. Therefore, the function of NOB1p is necessary for ubiquitin proteasome pathway-mediated proteolysis for cell cycle progression (16, 17). NOB1 has a critical function of ubiquitin-dependent proteolysis. In our study, we found that high expression levels of NOB1 were independent prognostic factors for OS and PFS in patients with resected NSCLC, with HR=1.7 (95% CI, 1.1-3.0, p=0.027) for OS, and HR=1.8 (95% CI, 1.3-3.7, p=0.031) for PFS. Lin et al found the RNA interference -mediated the downregulation of NOB1 expression markedly reduced the proliferative and colony-formation ability of ovarian cancer cells, and NOB1 shRNA-expressing lentivirus-treated ovarian cancer cells tended to arrest in the G0/G1 phase (18). Chen et al reported NOB1-knockdown markedly inhibited cell growth and caused G2/M-phase arrest in human osteosarcoma cells (19). Liu et al also found that NOB1 silencing arrested the cell cycle in the G0 / G1 phase in 6 different colon cancer cell lines (20). These results suggested that by silencing the NOB1 gene, the functions of the NOB1 protein in cell cycle progression were inhibited and thereby cancer cell proliferation was suppressed.
Our study showed high expression levels of NOB1 were independent prognostic factors for OS and PFS in patients with resected NSCLC. Potti et al identified gene expression profiles for the lung metagene model, and found this lung metagene model predicted prognosis for individual patients significantly better than did clinical prognostic factors across all early stages of NSCLC (21). Wan also identified a 12-gene signature using combinatorial gene selection methods. With this 12-gene risk score algorithm, early-stage NSCLC patients with poor prognosis could be identified (22). However, we found NOB1 was not included in these gene models. We presume that the predictive effect will be increased when NOB1 was added to the gene model. This is the purpose of our future studies.
In conclusion, our results suggest that enhanced expression of NOB1 is related to poor overall survival and progression-free survival in patients with resected NSCLC. Evaluation of NOB1 expression after surgery could contribute to a molecular classification of resected NSCLC.
