Abstract
BACKGROUND:
Differential diagnosis between malignant pleural effusion (MPE) and benign pleural effusion (BPE) remains a clinical challenge.
OBJECTIVE:
The aim of the study is to assess the efficacy of the serum and pleural fluid (PF) miRNA panels in distinguishing MPE from BPE.
METHODS:
Fourteen candidate miRNAs which were shown aberrant expression in lung cancer based on previous studies were tested by quantitative real-time PCR (qRT-PCR) in 20 MPE patients and 20 BPE patients. Significantly aberrantly expressed miRNAs were further assessed by qRT-PCR in all patients enrolled in this study. A receiver operating characteristic (ROC) curve was constructed, and the area under the ROC curve (AUC) was calculated to evaluated the diagnostic performance of the miRNAs.
RESULTS:
miR-21, miR-29c and miR-182 were found to be significantly aberrantly expressed in the serum and PF of MPE patients. The AUCs for the combination of miR-21, miR-29c and miR-182 in serum and PF were 0.832 and 0.89 respectively in distinguishing MPE from infection-associated PE including tuberculous pleurisy and parapneumonia PE, and 0.866 and 0.919 respectively for differentiating MPE from heart failure-associated PE, which were superior to AUC of each individual miRNAs.
CONCLUSIONS:
miR-21, miR-29c and miR-182 in serum and PF could be useful biomarkers for diagnosis of MPE.
Patient demographics and etiology of pleural effusion (PE)
Patient demographics and etiology of pleural effusion (PE)
Pleural effusion (PE) are common complications in patients with advanced tumor, infection or inflammation [1]. Cancer is one of the main causes of PE, and
microRNAs (miRs) represent a class of naturally occurring non-coding RNA molecules with regularly 19–25 nucleotides. The role of miRNA deregulation in carcinogenesis, cancer progression, and metastasis has been well established. miRNA signature may be used to identify specific type of cancer and thus are considered a promising tool for patient diagnosis, prognosis and clinical management [10]. miRNAs may be extracted from all biological samples, including tissue and liquid specimens, which include blood, saliva, sputum, urine, and bronchial lavage and serous fluids [11, 12]. In recent years, miRNA analyses conducted in surgical specimens, plasma, serum, sputum, and urine from patients with cancer have demonstrated its potential role in diagnosis and prognosis. However, the expression of miRNA in PE specimens has been less investigated [13, 14]. In the present study, we assessed the utility of miRNAs in serum and PF samples as biomarkers of differential diagnosis of MPE and BPE. Because most of MPE originate from lung cancer, in this study, 14 miRNAs proven to has aberrant expression in the tissue and blood specimens of lung cancer patients in previous studies were chosen as candidate diagnostic biomarkers (Supplementary Table 1 and Supplementary References). Then these miRNAs were examined by quantitative real-time PCR (qRT-PCR) in serum and pleural fluid (PF) specimens from 20 patients with MPE caused by lung cancer and 20 patients with benign pleural effusion (BPE). Significantly aberrantly expressed miRNAs were further analyzed for their diagnostic value in distinguishing MPE from BPE.
Material and methods
Study subjects, specimens, and study design
We prospectively enrolled 235 PE patients who were administrated to the Affiliated Hospital of Jiangsu University in China from March 2015 to December 2018. All patients with PE had a definite etiology documented by examination of medical imaging, PF biochemistry, microbiology, cytology, pleural biopsy, or endoscopic biopsy. The demographics and etiology of PE patients are summarized in Table 1. Etiology of MPE included lung adenocarcinoma (LAD), lung squamous cell carcinoma (LSCC), breast cancer, colorectal and ovarian cancer, etc. BPE consisted of tuberculous pleurisy (TBP), parapeumonia PE and heart failure-associated PE (HFAPE). In order to evaluate precisely diagnostic value of serum and PE miRNAs for MPE and BPE, the PE diagnosis protocol follow the recommendations set out in the guidelines published by different medical societies [15, 16, 17]. The diagnosis of MPE was based on the presence of cancer cells or tissue in either PF or biopsy pleural specimen, or if patients had disseminated malignancy and there was no alternative explanation for the PE [15, 16]. TBP was defined by the presence of pleural caseating granulomas, or the detection of Mycobacterium tuberculosis in patients sputum, PF or pleural biopsy specimens, or a high levels of PF adenosine deaminase (ADA) was determined (
PF samples were collected by thoracocentesis or during pleural biopsy before start any treatment. Peripheral blood sample were collected from PE patients and healthy controls. The time interval between collection of PF and blood samples in PE patients was 0–2 days in order to prevent possible miRNA data inconsistency between PF and matched serum samples caused by longer sampling interval. All samples were transported to the laboratory within 30 min of collection. Then the PF were centrifuged at 3000 g for 20 min and supernatants were transferred to new microfuge tubes (500 ul in each tube). Blood samples were centrifuged at 5000 g for 30 min to completely remove any cell debris and serum was transferred to new microfuge tubes. PF supernatants and serum were frozen instantly and stored at
In the initial phase of this study, we selected 14 candidate miRNAs which have been reported to be aberrantly expressed in the tissue or blood samples of lung cancer patients (Supplementary Table 1 and Supplementary References). The PF and serum levels of each of these miRNAs were tested by quantitative real-time PCR (qRT-PCR) in 20 patients with MPE caused by lung cancer (15 LAD and 5 LSCC) and 20 patients with BPE (10 TBP, 5 parapneumonia-PE and 5 HFAPE). When comparing MPE patients with BPE patients, only those miRNAs with a mean fold change
RNA extract and quantitative RT-PCR
Total RNA was extracted from each pleural fluid supernatant and serum sample using miRVana PARISKit (Ambion, TX) according to the manufacturer’s protocol. RNA concentration was quantified using Nano Drop 1000 Spectrophotometer (NanoDrop Technologies, MA), total RNA concentration
Statistical analysis
Results were expressed as mean
Scatter plots showing the expression levels (log2 values) of miR-21, miR-29c and miR-182 in serum (A–C) and pleural samples (D–F), as well as the values of their pleural fluid/serum ratio (G–H) in different types of cancer, different groups of benign pleural effusion and healthy control. ADC, lung adenocarcinoma; SCC, lung squamous cell carcinoma; SCLC, small cell lung cancer; BC, breast cancer; TB, tuberculous pleurisy; HF, heart failure; HC, healthy control. Other cancer included colorectal, ovarian, gastric, renal, and pancreatic cancer.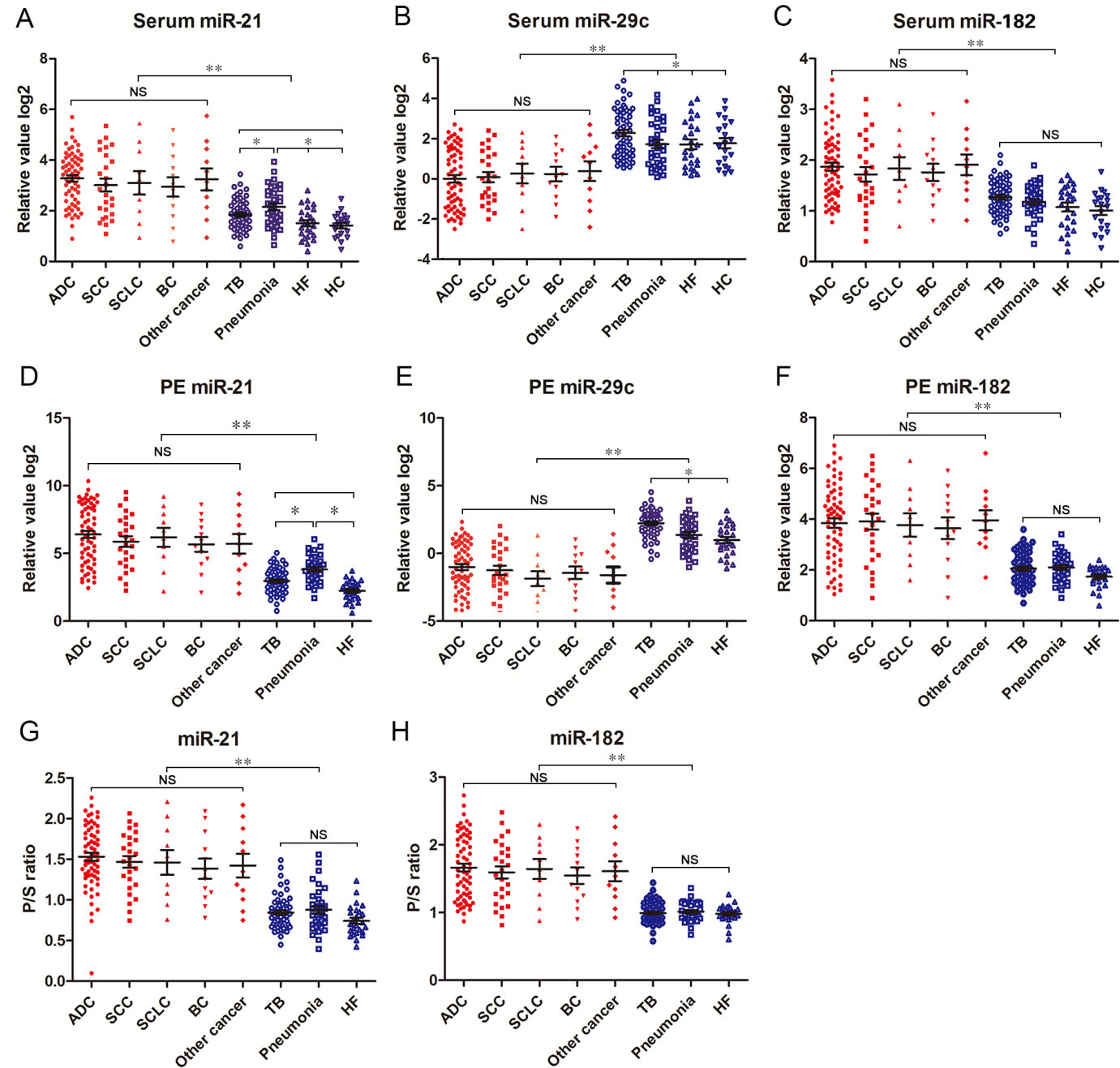
Serum levels of miR-21, miR-29c and miR-182, and their ROC curves. (A–C) The diagram at the top of each panel show serum levels of the 3 miRNAs in MPE and different groups of BPE. The diagram at the bottom of each panel show the ROC curves of miR-21, miR-29c and miR-182 in distinguishing MPE from IAPE, HF and healthy controls, respectively. MPE, malignant pleural effusion; IAPE, infection associated pleural effusion; HF, heart failure; HC, healthy control; ROC, receiver-operating characteristic.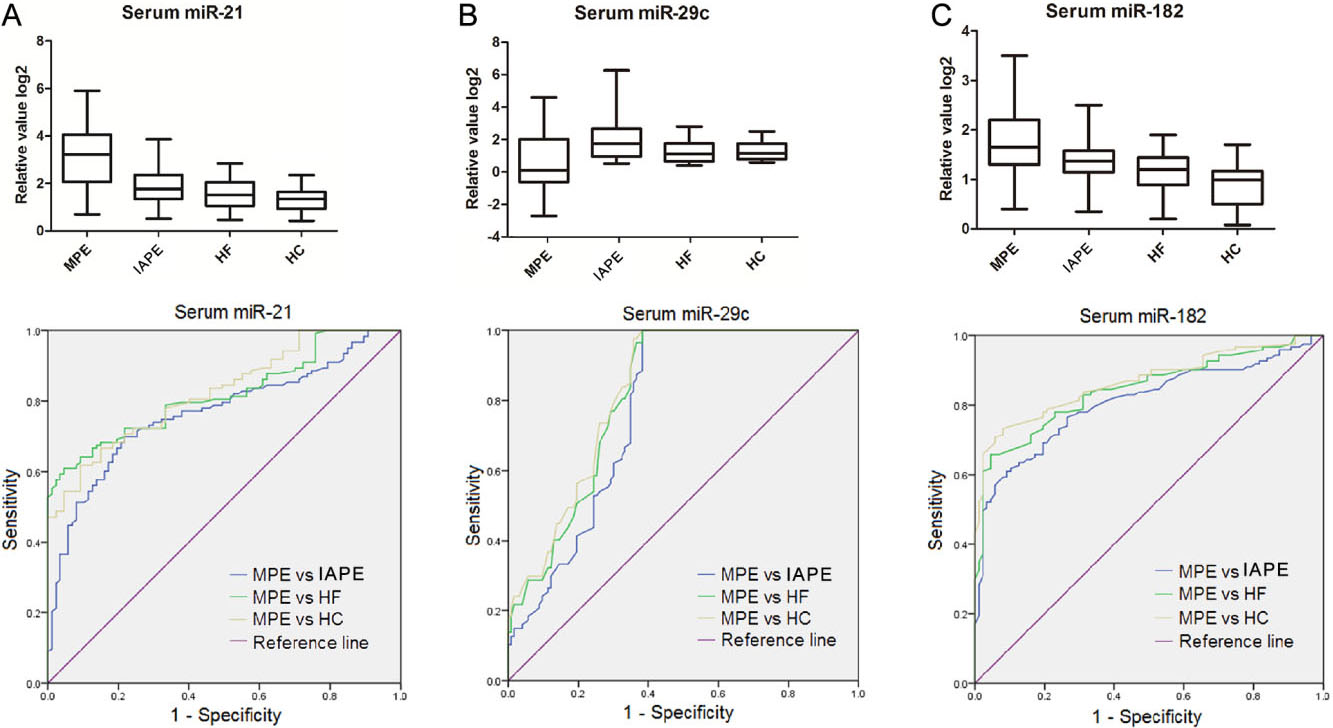
Expression levels of candidate miRNAs in serum and PF samples of MPE patients and BPE patients
Results showed that the levels of miR-21 and miR-182 expression in serum and PF samples were significantly up-regulated in all subgroup MPE patients as compared with all BPE patients (Fig. 1A, C, D and F). No difference was found in the serum or PF miR-21 and miR-182 expressions between lung cancer patients and other cancer patients. Serum and PF miR-29c levels were found to be markedly down-regulated in the MPE patients in comparison with the BPE patients (Fig. 1B and E). Likewise, there was no significant difference in the serum and PF miR-29c levels between MPE caused by lung cancer and MPE caused by other cancer.
Interestingly, the serum and PF miR-21 levels were elevated in patients with pneumonia compared with patients with TB and heart failure, but they were still significantly lower in patients with pneumonia than in patients with MPE caused by various cancer (Fig. 1A and D). In addition, miR-29c levels in serum and PF were markedly higher in patient with TBP than in patients with pneumonia and heart failure. Nevertheless, serum and PF miR-29c levels in all subgroup BPE patients were markedly higher than those in all MPE patients (Fig. 1B and E). In addition, we noted that the levels of miR-21 and miR-182 in the patients with MPE were higher in PF than in serum. Hence PF/serum (P/S) ratios of the 2 miRNAs were evaluated. Because the levels of serum and PF miR-29c have declined below zero in varying degrees, their P/S ratio can not be evaluated. We found that the P/S ratios of miR-21 and miR-182 were all markedly elevated in the MPE patients in compared to the BPE patients (Fig. 1G–H).
Diagnostic performance of miR-21, miR-29c and miR-182 for distinguishing MPE from BPE
To assess the diagnostic performance of the 3 miRNAs for discriminating MPE from BPE, the AUC of each miRNA in serum and PF was calculated by the ROC analysis. TBP and parapneumonia-PE were combined into one group which defined as infection-associated PE (IAPE). The individual AUC values of the serum miR-21, miR-29c and miR-182 ranges between 0.767–0.808 in distinguishing MPE from IAPE. These AUC values went up to 0.819–0.845 for discriminating MPE from HFAPE, and 0.825–0.868 for discriminating MPE from healthy controls (Fig. 2A–C, Tables 2 and 3, and Supplementary Table 2). When the optimal cut-off value of each miRNA was determined based on ROC curve analyses, the 3 serum miRNAs yielded sensitivity of 66.7–80.5%, specificity of
Diagnostic performance of three miRNAs in serum and pleural fluid and their combined use in distinguishing between MPE and IAPE
Diagnostic performance of three miRNAs in serum and pleural fluid and their combined use in distinguishing between MPE and IAPE
MPE, malignant pleural effusion; IAPE, infection-associated pleural effusion; PF, pleural fluid; P/S, pleural fluid/serum.
Diagnostic performance of three miRNAs in serum and pleural fluid and their combined use in distinguishing between MPE and HFAPE
MPE, malignant pleural effusion; HFAPE, heart failure-associated pleural effusion; PF, pleural fluid; P/S, pleural fluid/serum.
Then, we evaluated the diagnostic performance of the 3 PF miRNAs in distinguishing MPE from BPE. The ROC analyses revealed that the AUC values of the 3 PF miRNAs were 0.805–0.844 (MPE vs IAPE), and 0.824–0.878 (MPE vs HFAPE). The miR-182 showed better diagnostic performance than PF miR-29c and miR-21 (Fig. 3A–C and Tables 2 and 3), corresponding sensitivity, specificity and accuracy are shown in Tables 2 and 3. We observed that the AUC values of the 3 PF miRNAs were higher than those of the 3 serum miRNAs in discriminating MPE from IAPE or HFAPE.
Pleural effusion levels of miR-21, miR-29c and miR-182, and their POC curves. (A–C) The diagram at the top of each panel show pleural fluid levels of the 3 miRNAs in MPE and different groups of BPE. The diagram at the bottom of each panel show the ROC curves of miR-21, miR-29c and miR-182 in distinguishing MPE from IAPE and HF, respectively. PE, pleural effusion; MPE, malignant pleural effusion; IAPE, infection associated pleural effusion; HF, heart failure; ROC, receiver-operating characteristic.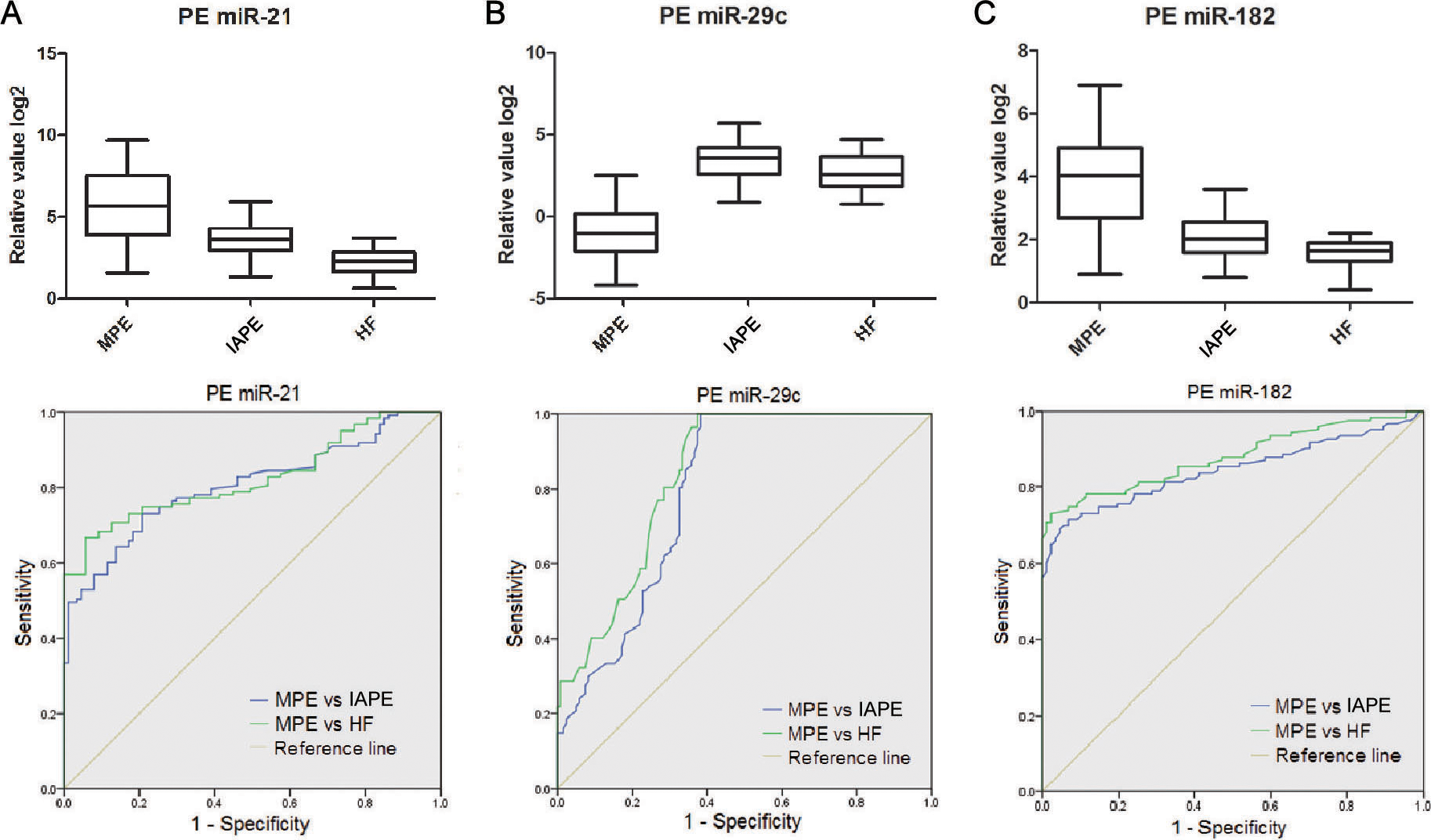
Finally, the capacity of P/S ratios of miR-21 and miR-182 to discriminate MPE from BPE was examined with ROC analyses. As shown in Fig. 4A and B and Tables 2 and 3, the AUCs of the P/S ratios of the 2 miRNAs were 0.856 and 0.868 in differentiating MPE from IAPE, and 0.895 and 0.916 in discriminating MPE from HFAPE. The sensitivity, specificity and accuracy of the P/S ratios of the 2 miRNA in diagnosing MPE were listed in Tables 2 and 3, which were better than those of the 2 serum and PF miRNAs. These results suggest that serum and PF miR-21, miR-29c and miR-182, as well as the P/S ratios of miR-21 and miR-182 may be useful tools for diagnosing MPE.
Pleural fluid/serum ratios of miR-21 and miR-182, and their POC curves. (A–C) The diagram at the top of each panel show pleural fluid/serum ratios of the 2 miRNAs in MPE and different group of BPE. The diagram at the bottom of each panel show the ROC curves of pleural fluid/serum ratio of miR-21 and miR-182 in differentiating MPE from IAPE and HF, respectively. MPE, malignant pleural effusion; IAPE, infection associated pleural effusion; HF, heart failure; P/S, pleural fluid/serum; ROC, receiver-operating characteristic.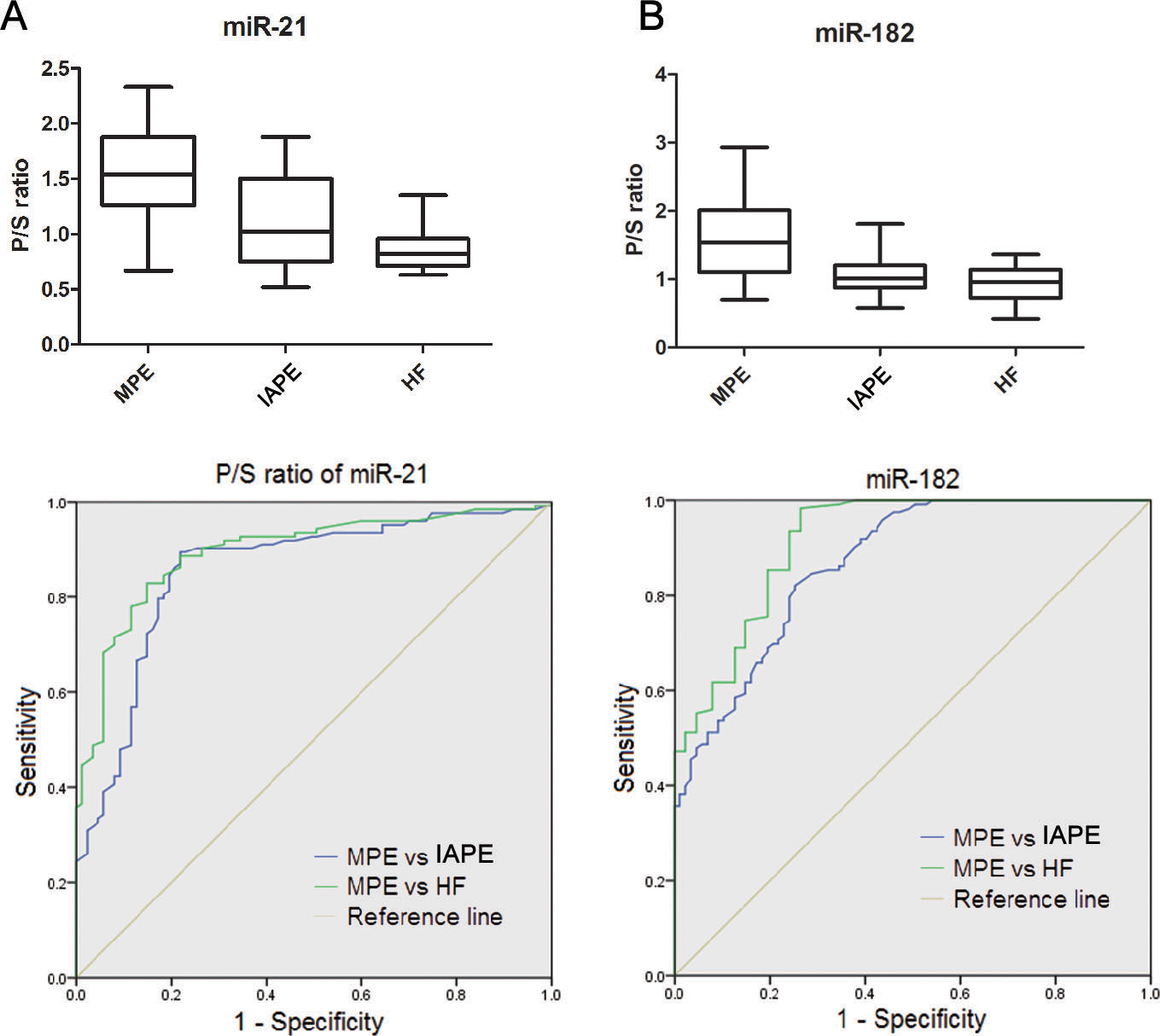
To assess if an optimal miRNA panel, instead of the single individual miRNA, could be applied to the diagnosis of MPE with higher sensitivity and accuracy, we used a 3-miRNA signature by combining the 3 miRNAs using the logistic regression analysis to construct the diagnostic model of the 3 miRNA. Results showed that the AUCs for the combination of the 3 miRNA in serum and PF were 0.832 and 0.89 respectively, and the AUC for the combination of P/S ratios of miR-21 and miR-182 was 0.933, for distinguishing MPE from IAPE, which were considerably higher than AUC value of each individual miRNAs (Table 2 and Fig. 5A).
ROC curve analyses of the 3-miRNA panel (combination of miR-21, miR-29c and miR-182) in serum, pleural fluid and their pleural fluid/serum ratio. (A) Distinguishing MPE patients from IAPE patients. (B) Distinguishing MPE patients from HF patients. (C) Distinguishing MPE patients from healthy control. MPE, malignant pleural effusion; IAPE, infection associated pleural effusion; HF, heart failure; P/S, pleural fluid/serum; ROC, receiver-operating characteristic.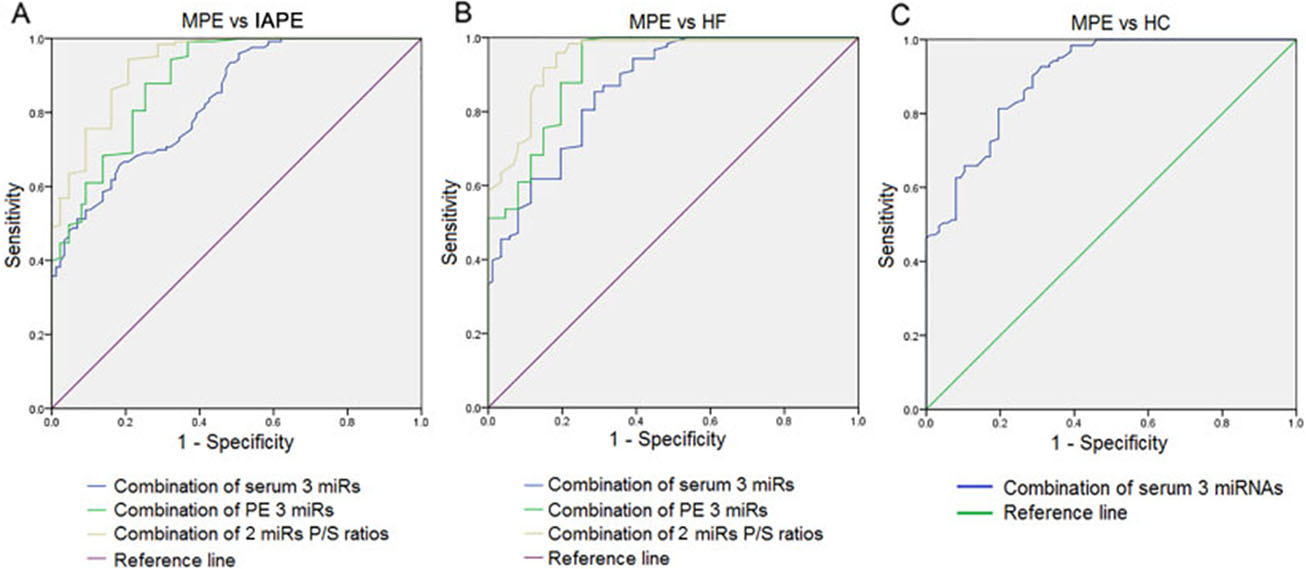
By using the logistic regression model again, we found that the 3-miRNA signature set diagnostic performance equal well in differentiating MPE from HFAPE, the corresponding AUCs were 0.866 and 0.919 respectively in serum and PF samples. In addition, combining miR-21 and miR-182 P/S ratios achieved an AUC of 0.945 (Table 3 and Fig. 5B), which were also substantially higher than the AUC value of each individual miRNA. When we applied this logistic regression model to analyze the 3 miRNAs combination discriminating between MPE and healthy control only in serum, we obtained an AUC of 0.898 (Fig. 5C and Supplementary Table 2). The sensitivity, specificity and accuracy in discriminating MPE from IAPE, HFAPE, or healthy control by combining the 3 miRNAs of serum and PF, and combining miR-21 and miR-182 P/S ratios were shown in Tables 2 and 3 and Supplementary Table 2, all of them were superior to those of each individual miRNA.
Considering that PF cytology is one of the gold standard for diagnosis of MPE, it is worthwhile to assess the potential clinical applications of the 3 PF miRNAs when combined with PF cytological examination. The sensitivity of initial cytological examination for MPE diagnosis in the present study was 41.5% indicating that only of 51/123 MPE patients were initially examined as positive cytology in PF samples. When cytology in combination with PF miR-21, miR-29c, or miR-182, the sensitivity increased from 41.5% for cytology alone to 80.4%, 79.7% and 82.9%, respectively (Supplementary Table 3). Combination of cytology and the 3-miRNA signature further raised the sensitivity to 91.1% (112/123). This results indicate that 61 of 72 MPE samples missed by initially cytological examination were rescued by the 3-miRNA signature in PF.
Although the diagnosis of MPE is critical, making correct diagnosis is still challenging. Cytological examination of pleural fluid obtained through thoracetesis is a standard and non-invasive method for the diagnosis of MPE. However, false-negative results of cytological examination in PF are serious problem. Being able to establish a diagnosis of MPE in a non-invasive way through tumor markers is of great investigational interest. Consequently, there have been a number of studies on the utility tumor marker measurement in PE [8, 9, 20, 21, 22].
In the present study, we found that a 3-miRNA panel consisting of miR-21, miR-29c and miR-182 can efficiently differentiating MPE patients from BPE patients. We demonstrate that the combination of the 3 miRNAs in either serum or PF exhibited better diagnostic performance in compared to each individual miRNA, as evidenced by increased AUC values, sensitivity and accuracy (Tables 2 and 3). Furthermore, compared to serum and PF samples, the combination of the P/S ratios of miR-21 and miR-182 showed the best diagnostic performance in differentiating MPE from BPE. Meantime, our results indicate that the 3 PF miRNA contributed the additional information to PF cytology alone. Frequently, the 3 miRNAs were positive in the PF of patients exhibiting malignant cytology. However, a striking finding was the demonstration of the 3 miRNAs positivity in 61 of 72 initially cytological negative MPE.
In the present study, we noted that the levels of serum and PF miR-21 in patients with parapneumonia PE were markedly elevated than those in patients with BPE caused by TBP or heart failure. Nevertheless, the levels of serum and PF miR-21 in MPE patients were dramatically higher than those in all BPE patients including parapneumonia PE cases. Abd-El-Fattah et al reported that the levels of serum miR-21, miR-155 and miR-197 in pneumonia patients were markedly higher than healthy controls, and could be used to distinguish pneumonia patients from healthy controls but the serum miR-21 and miR-155 levels in pneumonia patients were still lower than those in lung cancer patients [23], which are similar to our results.
Also we found that the levels of serum and PF miR-29c in patients with TBP were not only significantly elevated than those in patients with MPE, but also much higher than those of patients with parapneumonia PE and heart failure. A study showed that six serum miRNAs (miR-378, miR-483-5p, miR-22, miR-29c, miR-101, and miR-3206) could serve as distinct biosignature of pulmonary tuberculosis compared to healthy controls, and groups of pneumonia, lung cancer, and chronic obstructive pulmonary disease (COPD) patients [24]. Among six miRNAs, serum miR-29c was up-regulated in the serum of the pulmonary tuberculosis patients than those of the healthy control, pneumonia, lung cancer and COPD patients [24]. These results resemble our finding.
miR-21 functions as an oncogene in several cancer. In non-small cell lung cancer (NSCLC), it was stated that NF-
miR-29c plays the role as tumor suppressor in several kinds of tumor. miR-29c was shown to inhibit cell proliferation, migration and invasion by targeting VEGFA in LAD. Its expression down-regulation in tumor tissue was significantly associated with unfavorable prognosis in LAD [32]. In LSCC, the miRNA-29 family including miR-29c was significantly reduced in tumor tissue compared to normal tissue, loss of miR-29c enhanced cancer cell migration and invasion through direct regulation of oncogenic LOXL2 [33]. It was reported that the expression of miR-29c was decreased with the progression of breast cancers and was closely associated with an overall survival of patients. Overexpression of miR-29c inhibited the migration, colony formation and growth in breast cancer cells by regulating the TIMP3/STAT1/FOXD1 pathway [34]. miR-29c was found to be down-regulated in gastric cancer tissue and cell lines, and miR-29c overexpression suppressed cell proliferation, promoted cell apoptosis and arrested cell cycle at G1/G0 phage by targeting NASP [35]. Although many studies showed that miR-29c functioned as a tumor suppressor in various type of cancer and its expression was down-regulated in tumor tissue or serum of cancer patients [33, 34, 35, 36], some researchers reported that the levels of miR-29c expression were up-regulated in NSCLC tissue and/or serum samples compared to normal lung tissue or healthy control serum, and it was a member of panel of miRNAs proposed as biomarker for NSCLC early detection. For example, a panel of miRNAs (miR-29c, miR-93, and miR-429) detected in serum samples that includes also miR-29c, could serve as a potential tool for early diagnosis, as their elevated levels could discriminate between NSCLC patients and healthy controls [37]. In another study, along with serum miR-1466, miR-205, and miR-306, serum miR-29c was up-regulated and present as a member of the 4 miRNAs signature with high potential for early diagnosis of NSCLC [38]. The reason for the discrepancies between these studies are not clear, which may be explained by variance in the methods applied for miRNA analysis, the criteria for analyzed miRNA of interest, the ethnicity and/or clinicpathological features of recruited patients. It is necessary to further study miR-29c expression trend in various specimens (tissue, serum, plasma and PF) of different types of cancer patients with reasonable stratification of other relevant features.
Some studies signified that overexpression of miR-182 showed increased tumor cell growth and proliferation, highly invasive features, and tumor progression through repression of FBXW 7 and FBXW 11 in NSCLC [39], PECK in breast cancer [40], and the ST6GALNACa and PI3K/AKT pathway in colorectal cancer [41]. Some researchers demonstrated that circulating miR-182 can stand as a biomarker for tumor diagnosis, including NSCLC and breast cancer [42, 43]. A meta-analysis has revealed that the expressions of miR-183 family including miR-182 were up-regulated in many type of cancer, in which NSCLC was ranked in third place [44]. Thus it is generally believed that miR-182 acts as a oncogene in the biological process of cancer. However, other studies showed different conclusions. Zhang et al reported that overexpression of miR-182 inhibits the epithelial to mesenchymal transition and metastasis via inactivation of Met/AKT/Snail in NSCLC cells [45]. Sun et al reported that the miR-182 induced cervical cancer cell apoptosis by suppressing DNMT32 expression [46]. The reason of these conflicting results is unknown, but it may be related to the limited sample size and study population, or different design and experimental methods. This is also a subject worthy of in-depth study.
Conclusion
Our results show that the 3-miRNA panel is more reliable diagnostic signature than each individual miRNA in differentiating MPE from BPE. Overall, the combination of the 3 PF miRNAs are superior to the combination of the 3 serum miRNAs in diagnosing MPE. Although the positive of the 3 miRNA in serum or PF would not be used to confirm the diagnosis of MPE, the molecular analyses can serve as an important adjunct to cytological examination in MPE diagnosis.
Author contributions
Conception: Li-Rong Zhu, Rong-Xia Yuan and Jian Li.
Interpretation or analysis of data: Li-Rong Zhu, Rong-Xia Yuan, Xian-Bin Xia, Yi Wang, Yu-Min Zhu and Ling Fi.
Preparation of the manuscript: Li-Rong Zhu, Rong-Xia Yuan and Jian Li.
Revision for important intellectual content: Jian Li.
Supervision: Yi Wang.
Supplementary data
The supplementary files are available to download from
sj-docx-1-cbm-10.3233_CBM-210090.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-210090.docx
Footnotes
Acknowledgments
This work was supported by The Social Development Foundation of Zhenjiang (SH2013076, SH2014076).
Conflict of interest
The authors declare that they have no conflict of interest.
