Abstract
Introduction:
Dyspnea due to pleural effusions is a common clinical scenario and has many underlying etiologies. Our case of a recurrent pleural effusion was misdiagnosed in part due to the complicated nature of the patient’s medical history. Nonetheless after a bedside thoracic ultrasound examination (TUE), the diagnosis became obvious.
Case Description:
A 63-year-old black female with congestive heart failure, end-stage renal disease, atrial fibrillation as well as a previously treated rare odontogenic carcinoma presented with shortness of breath. She reported having had “fluid drained” from her lungs a few months prior. During our encounter with this patient, a TUE was performed at beside and a large diaphragmatic mass was visualized. A malignant pleural effusion was suspected and an efficient and focused workup was tailored to our patient.
Discussion:
Apart from guiding the thoracentesis, the TUE is not part of the current recognized guidelines when evaluating a pleural effusion. The TUE may be undervalued and play an important impact on diagnosis and management of this condition. Our case demonstrates the utility of a thorough TUE in conjunction with the full clinical picture. A few studies have shown good sensitivities and specificities in the use of the TUE to determine the type and etiology of pleural effusions. This case is a further example of the utility in performing this relatively inexpensive diagnostic test in the evaluation of the patient with a pleural effusion.
Introduction
Dyspnea due to pleural effusions is a common clinical presentation and has many underlying etiologies. Current guidelines as defined by the British Thoracic Society Pleural Disease Guideline 2010 in the diagnosis of a pleural effusion follow an algorithm that includes the history, physical examination, chest X-ray, thoracentesis, computed tomography (CT) scan, and followed with the performance of a Video-Assisted Thoracoscopic Surgery (VATS), thoracotomy, or radiographically guided biopsy depending on the clinical situation. The thoracic ultrasound examination (TUE) as a diagnostic tool is not currently the standard of care although its utility has been demonstrated most notably by Qureshi et al, quoting a 73% sensitivity and 100% specificity in diagnosis of malignancy via TUE. Our case of a recurrent pleural effusion with misdiagnoses due to the complicated nature of patient’s medical history became obvious after a thorough TUE. This is a further example of the utility of the TUE in guiding diagnosis and management through a quick and noninvasive test that already is part of the workup of pleural effusions, especially now that ultrasound-guided thoracentesis has become the standard of care.
Case
A 63-year-old black female presented to our emergency department with the chief complaint of shortness of breath. Her initial chest X-ray demonstrated a unilateral pleural effusion (Figure 1). She reported that she had a pleural effusion that was drained a few months prior. She had seen multiple specialists but couldn’t make it to for follow-up appointment due to the severity of her dyspnea. She reported that this was similar to the previous episodes, describing progressive dyspnea over the past 2 to 3 weeks, orthopnea, and chest tightness. She denied fever, productive cough, or hemoptysis.

Chest X-ray taken in the emergency department prior to hospital admission. Left lower hemithorax opacification with effusion and compressive atelectasis can be seen.
Her medical history was extensive and included congestive heart failure (CHF) ejection fraction of 40% status post automatic implantable cardioverter defibrillator, pulmonary hypertension with right ventricular systolic pressure of 50 to 55 mm Hg, end-stage renal disease (ESRD) requiring hemodialysis, atrial fibrillation (AF) with atrial thrombus requiring chronic anticoagulation with warfarin, coronary artery disease status post coronary stent placement, hypertension, a ghost cell tumor of her right mandible status postresection in 2010, and radiation therapy through 2012 as well as a previous cerebrovascular accident.
Two months prior to presenting to our emergency department, our patient had been admitted for a 6-day hospitalization at an outside facility, during which she had multiple chest X-rays, an echocardiogram, and underwent a thoracentesis. On discharge, she was started on hemodialysis and scheduled to follow-up with her physicians. During the next few weeks after discharge, she continued dialysis and saw a pulmonologist with no change in management as likely it was assumed her effusions were due to CHF and ESRD. Ten days prior to this admission, she again presented to our emergency department with a similar complaint and was told that she “did not have enough fluid to tap” and was sent home.
We decided to admit our patient for the night and in the morning gave her 2 units of fresh frozen plasma due to elevated international normalized ratio for her AF and performed a thoracentesis. Prior to the thoracentesis, a thorough TUE was performed demonstrating a large mass on the diaphragm as well as a mass visible on the pleural surface (Figure 2). We drained approximately 1500 mL of bloody fluid from the left plueral space. At this point, we became extremely concerned for a malignant effusion, sent the pleural fluid for cytology as well as flow cytometry, ordered a CT with contrast, and consulted the thoracic surgery team.
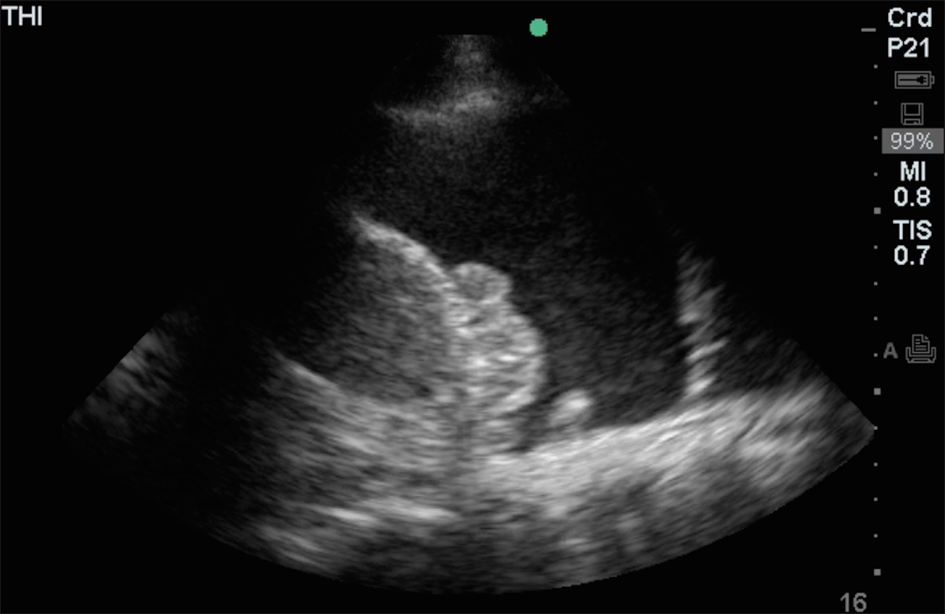
Still image taken from the thoracic ultrasound exam done prior to thoracentesis demonstrating a diaphragmatic mass and pleural mass.
A CT thorax was done without contrast and demonstrated innumerable pleural nodules bilaterally (Figure 3). The pleural fluid cytology and flow cytometry both came back normal. A VATS was performed, and a biopsy of the pleural nodules was consistent with well-differentiated metastatic odontogenic carcinoma similar to histology seen from right mandible in 2009 (Figure 4).

CT scan slice demonstrating one of many pleural nodules seen on CT scan. Throughout the rest of the CT extensive pleural nodular densities throughout both lungs as well as mediastinal lymphadenapathy. The largest nodular density measures 17 x 11 mm.

Histopathology of pleural biopsy, demonstrating highly cellular neoplastic tissue consistent with odontogenic carcinoma.
Discussion
The British Thoracic Society pleural disease guideline 2010 described the algorithm for evaluating a unilateral pleural effusion. First a careful history including drugs and physical examination should be performed. Next the initial diagnostic imaging of choice would be a posteroanterior chest X-ray, which can demonstrate a pleural effusion with as little as 200 mL of fluid and as little as 50 mL of fluid on lateral chest X-ray. Next if diagnosis remains unclear, a pleural aspirate with ultrasound guidance should be performed and sent for at least cytology, protein, low-density lipoprotein, pH, Gram stain, culture, and sensitivity. Next if necessary a contrast-enhanced CT thorax should be performed after which video-assisted thoracoscopic surgery or other tissue biopsy may be considered. Lastly if a diagnosis has not been made, treatable conditions such as pulmonary embolism, tuberculosis, CHF, and lymphoma is reevaluated. 1
The TUE is incorporated as part of this algorithm in the diagnosis of a pleural effusion but only during the thoracentesis. The use of ultrasound during thoracentesis has been shown to decrease iatrogenic pneumothorax, increase yield of pleural fluid, and decrease cost associated with the procedure. 2
Further utility of the TUE has been demonstrated in regard to the diagnostic utility of the image itself. It has been shown that septations within pleural fluid can been seen with a greater sensitivity than CT scanning. 3 Yang et al in a prospective analysis of 320 patients with pleural effusions demonstrated the ability to distinguish exudative from transudative effusions based on TUE alone. In addition, 10 patients demonstrated pleural nodules on examination, all of which ended up having malignant effusions. 4 More recently, the thoracic ultrasound features have been further refined in distinguishing malignant from benign effusions. Qureshi et al demonstrated an overall 95% specificity for a malignant diagnosis where 95% of malignant effusions had parietal pleural thickening >1 cm, 100% had visceral pleural thickening, 95% had diaphragmatic thickening >7 mm, and 100% had diaphragmatic nodules as visualized on ultrasound examination. 5 The overall sensitivity of the ultrasound in the differentiation of malignant from benign effusions was 79% (95% confidence interval [CI] 61%-91%) with a specificity of 100% (95% CI 82%-100%).
Conclusion
This case demonstrates the utility of a thorough TUE in conjunction with the full clinical picture. This patient clearly had multiple reasons that she could be presenting with recurrent pleural effusions including CHF and ESRD. The bloody fluid could additionally have been attributed to the patient’s chronic anticoagulation. On retrospective analysis of records, the original pleural fluid cytology demonstrated “atypical cells,” and her echocardiogram demonstrated a “fibrous effusion.” Nonetheless, without this information, we utilized a bedside TUE to get to the appropriate diagnosis quickly. Our patient was able to leave the hospital with a definitive diagnosis and follow-up with oncology for treatment options.
Further studies regarding the training and who should be performing these studies are required. In addition, not utilizing the full potential of this quick and noninvasive examination is missing an important diagnostic opportunity, especially in the patient with suspected malignant pleural effusion.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
