Abstract
BACKGROUND:
Nuclear pore membrane protein 121 (POM121) is a novel biomarker involved in tumorigenesis and metastasis. However, little is known about the role of POM121 in non-small-cell lung cancer (NSCLC).
OBJECTIVE:
The aim of this study was to detect the expression of POM121 in NSCLC and its relationship with clinicopathologic feature and cell biological behavior, and explore the underlying mechanisms.
METHODS:
The expression of POM121 in NSCLC tissues and para-carcinoma tissues was compared by quantitative real-time PCR and immunohistochemistry analysis. The relationship between POM121 protein and clinicopathological characteristics in NSCLC was investigated. Roles of POM121 in NSCLC cells were investigated by CCK-8 assay, clone formation assay, transwell migration and invasion assay, and in vivo experiments. Variations of signaling pathways were determined by qRT-PCR and Western blot.
RESULTS:
The POM121 expression in NSCLC tissues was significantly higher than that in para-carcinoma tissues, both at the mRNA and protein level. The POM121 expression was related to sex, advanced differentiation, tumor diameter, lymph node metastases, distant metastases, American Joint Committee on Cancer (AJCC) stage, venous invasion, and perineural invasion in NSCLC. Kaplan-Meier analysis indicated that NSCLC patients with high POM121 expression had poor overall survival. Downregulation of POM121 inhibited cell proliferation, clone formation, migration and invasion. TGF-
CONCLUSION:
POM121 plays an oncogenic role in NSCLC through TGF-
Introduction
Lung cancer is the most frequently diagnosed cancer worldwide and the leading cause of cancer death among males [1]. Over half of lung cancer patients are declared to be in advanced stage at initial diagnosis because of the atypical symptoms that present in the early stages [2]. The 5-year survival rate is 26.1% for patients with regional disease and only 3.9% for patients with distant metastases [3]. Non-small-cell lung cancer (NSCLC) accounts for 80%–85% of lung cancers [4]. Thus, it is necessary to identify new sensitive and specific biomarkers of NSCLC.
NPCs serve a vital role in maintaining normal cellular functions through regulating cellular signaling between the cytoplasm and nucleus [5]. Dysregulation of NUPs can trigger abnormal nucleocytoplasmic transport, resulting in mislocalization of transcription factors and tumor suppression proteins [6]. As a member of NUPs, nuclear pore membrane protein 121 (POM121) gene is located on human chromosome 7q 11.23 [7]. POM121 protein is 121 kDa, composed of a single transmembrane domain and two peptide domains extending into the endoplasmic reticulum and the cytoplasm [8, 9]. POM121 and NUP107-160 complex is involved in inner/outer nuclear membrane fusion events, which are required for nuclear envelope formation [10]. POM121 is also essential for nuclear pore complex (NPC) assembly [11, 12]. Replacement of POM121 was connected with defective nuclear transport, aberrant cytoplasmic membrane stacks, and decreased cell viability [13]. Guo et al. reported that POM121 enabled efficient HIV-1 pre-integration complex nuclear import during KPNB1-dependent cargo nuclear transportation [14]. Rodriguez-Bravo et al. demonstrated the important role of POM121 in prostate cancer aggressiveness [15]. but the association between POM121 and NSCLC remains unknown.
To explore the role of POM121 in NSCLC, POM121 expression in NSCLC tissues was detected, and associations between POM121 expression and pathological characteristics were analyzed. The function and mechanism of POM121 in regulating proliferation and metastasis of NSCLC were also explored in vitro and vivo. Combined with the above results, POM121 has the potential to be a therapeutic target for NSCLC patients.
Materials and methods
Tissue samples and clinical data
Formalin-fixed, paraffin-embedded lung tissue samples were collected from 857 patients whichincluded 796 NSCLC tissues, 61 small-cell lung cancer (SCLC) tissues, and 212 normal lung tissues. All samples were obtained from patients who were histologically diagnosed at Bayannur Hospital between 2005 and 2012 and at Nanjing First Hospital from 2008 to 2013. None of the patients enrolled in this study received chemotherapy, immunotherapy, or radiation therapy before surgery or biopsy. The clinicopathological features of tissue donors included age, sex, tumor size, lymph node metastases, distance metastasis, histological type, differentiation grade, histological type, AJCC stage, and venous invasion. OS was defined as the period from initial diagnosis to death (or last follow-up). The project was approved by the Human Ethics Committee of Bayannur Hospital (BSYY2019217), and all patients participating in the study signed informed consent.
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
POM121 mRNA expression was detected in 31 pairs of NSCLC tissues and matched normal lung tissues. Total RNA was extracted from tissues using Trizol reagent (#15596026, ThermoFisher, Waltham, MA, USA) according to the manufacturer’s instructions. DNase was utilized to get rid of all traces of genomic DNA. Extracted RNA was transcribed into cDNA using the SuperScript IV First-Strand Synthesis System (#18091050, ThermoFisher). qRT-PCR was performed using SYBR Select Master Mix (#4472920, ThermoFisher) on an ABI StepOne Plus Real-Time PCR System using the following protocol: 95
Tissue microarrays (TMA) and immunohistochemistry analysis (IHC)
Tissue specimens were shaped in special molds and fixed with 4% paraformaldehyde. After embedding in paraffin, tissue cores were collected to construct tissue microarrays. Blocks were sectioned to a thickness of 4
Cell lines and cell culture
Human NSCLC cell lines A549 and HCC827 were obtained from NHC Key Laboratory of antibody technique (Nanjing Medical University). The medium was RPMI 1640 with 10% fetal bovine serum and 1% double antibody. The cells were cultured in a incubator of 5% CO
Western blot
The total protein was extracted with RIPA lysate (#15596018, Invitrogen, USA) and heated at 95
CCK-8 proliferation experiment
Cells of each group were planted in 96-well plates at a density of
Clone formation assay
The cells were re-inoculated into 6-well plates with 1
Transwell migration and invasion assay
Migration and invasion assays were conducted using transwell chambers without and with Matrigel respectively. Cells (5
Tumor formation experiment in nude mice
A549 cells transfected with shControl and shPOM121 were prepared into cell suspension with a density of 1
POM121 expression in NSCLC tissues. (A) POM121 mRNA levels in the 31 NSCLC tissues (T) were higher than that in matched normal tissues (N). (B) Positive rates of high POM121 in NSCLC tissues and SCLC tissues were higher than that in normal tissues. (C) Representative images of POM121 protein in lung tissues. (a) Non-small-cell squamous carcinoma with high POM121 expression (staining score 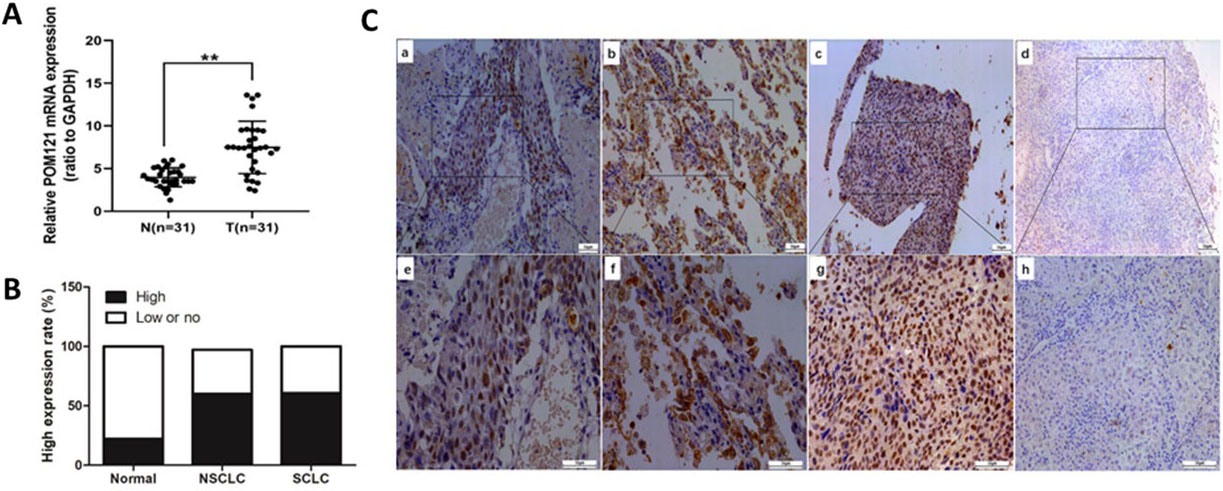
Statistical analyses were performed using SPSS version 18.0 software (SPSS Inc., USA). Measurement data were expressed as mean
Results
POM121 is overexpressed in NSCLC
POM121 mRNA expression levels were detected in 31pairs of NSCLC tissues and matched normal lung tissues by qRT-PCR. POM121 mRNA expression in NSCLC tissues was higher than that in matched normal lung tissues (
To detect POM121 protein expression in lung cancer tissues and normal lung tissues, IHC assays were performed. Results showed that POM121 protein was located in the nuclear membrane and was overexpressed in cancerous tissues (Fig. 1B). High POM121 expression was more prevalent in NSCLC tissues (60.07%, 502/796) than that in normal lung tissues (22.17%, 47/212), consistent with the comparison between SCLC tissues and normal tissues (Fig. 1C).
POM121 expression is connected with clinical characteristics in NSCLC
The correlation between POM121 expression and pathological parameters in NSCLC patients was analyzed. POM121 expression was significantly associated with sex (
Association of POM1121expression with clinicopathological characteristics in NSCLC patients
Association of POM1121expression with clinicopathological characteristics in NSCLC patients
Prognostic factors for NSCLC patients were detected by both univariate and multivariate analyses. POM121 expression was significantly associated with the OS of NSCLC patients in univariate analysis, as was smoking, differentiation, tumor diameter, distant metastasis, and AJCC stage. In multivariate analysis, independent prognostic factors for OS included high POM121 expression (HR
Univariate and multivariate analysis of prognostic factors for overall survival in lung cancer
Univariate and multivariate analysis of prognostic factors for overall survival in lung cancer
HR, Hazard ratio; CI, confdence interval. *
Survival curves of NSCLC patients using the Kaplan-Meier method and the log-rank test. (A) Overall survival curves for patients with low POM121 expression (blue line) and patients with high POM121 expression (green line). (B) Overall survival curves by TNM stage, TNM 0, I and II (blue line), TNM III and IV (green line). (C) Overall survival curves for tumor size, Tis, T1 and T2 (blue line), T3 and T4 (green line). (D) Overall survival curves for patients with well differentiation (blue line), patients with moderate differentiation (green line) and patients with poor differentiation (yellow line). (E) Overall survival curves by lymph node metastases, N0 (blue line), N1 (green line). (F) Overall survival curves by distant metastases, M0 (blue line), M1 (green line).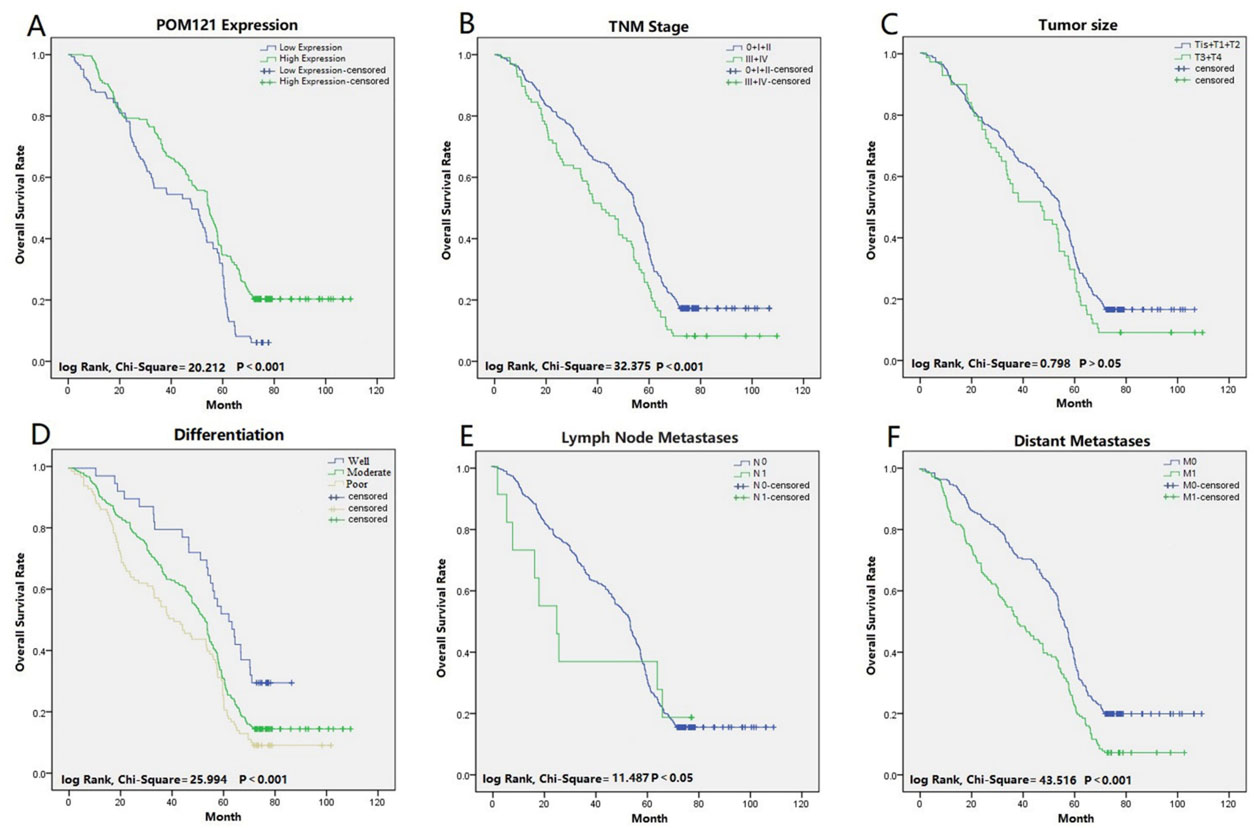
In further investigation of the functions of POM121 on cell biological process, shRNA-POM121 was constructed and transfected into A549 and HCC827 cell lines. POM121 protein expressions in both cell lines were reduced after transfection (Fig. 3A). In contrast to the control group, shPOM121 significantly inhibited the cell proliferation (Fig. 3B), clone formation (Fig. 3C), migration (Fig. 3D) and invasion (Fig. 3E).
POM121 plays an oncogenic role in NSCLC cells. (A) A549 and HCC 827 cells showed a significant decrease in protein level after shPOM121 transfection. (B) POM121 downregulation significantly inhibited the proliferation of both cell lines. (C) A significant decrease in cell anchorage-dependent growth was detected after POM121 knockdown. (D, E) Decreased POM121 expression impaired abilities of migration (D) and invasion (E) of NSCLC cells. (F) Downregulation of POM121 inhibited the tumor growth in the xenograft mouse model.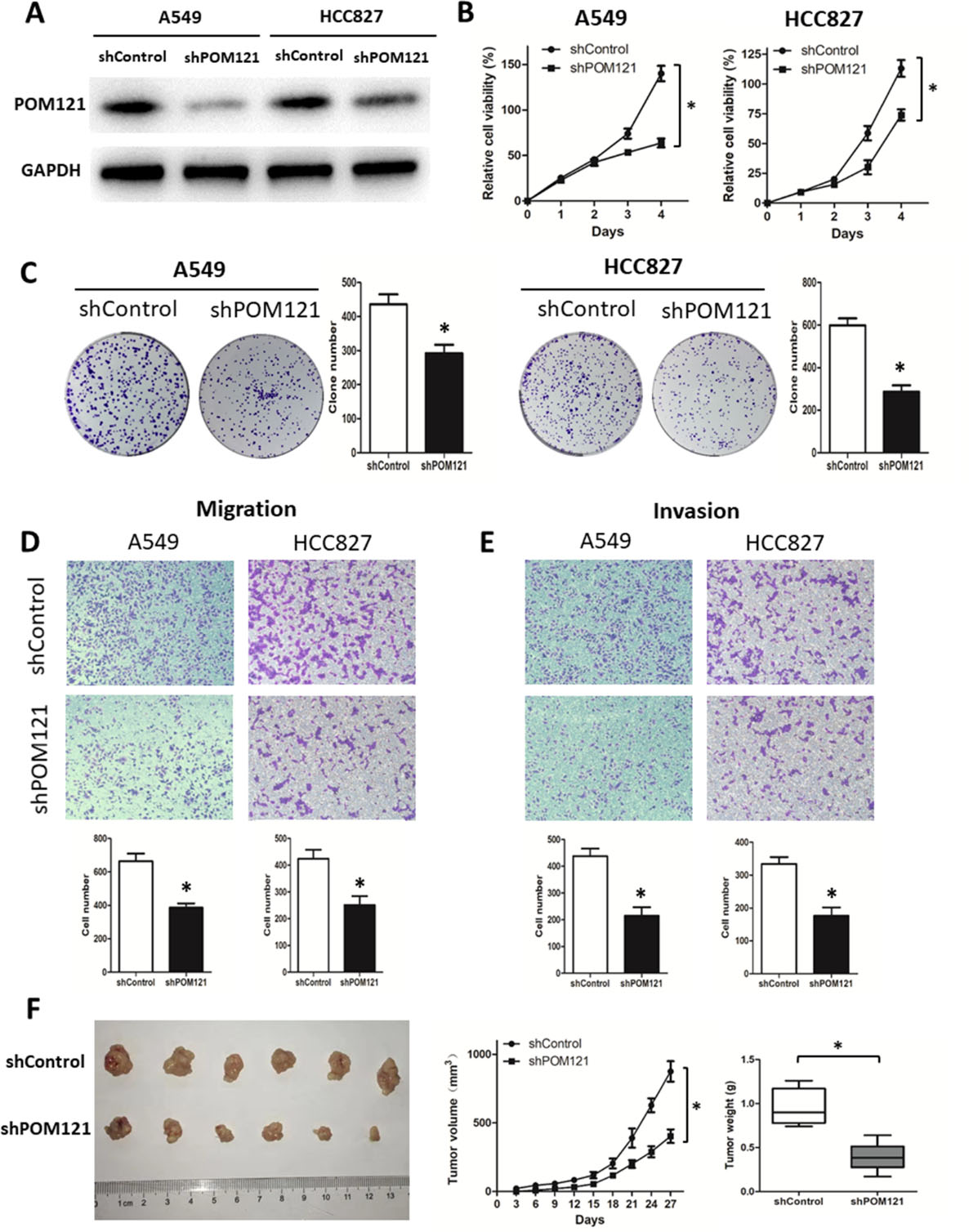
Subcutaneous transplanted models in nude mice were constructed to explore the modulating effect of POM121 in vivo. Downregulation of POM121 reduced tumor growth within 27 days (Fig. 3F). Compared with the control group, the mice in shPOM121 group generated tumors with lessened size and weight (Fig. 3F).
In exploration of the mechanism by which POM121 promotes the malignant biological behaviors in NSCLC, 343 proteins correlated with POM121 were determined in UALCAN dataset (data not shown). Signaling pathway enrichment analysis of these proteins showed that POM121 may affect ubiquitin mediated proteolysis, cell cycle, RNA transport, TGF-
POM121 promotes carcinogenesis through TGF-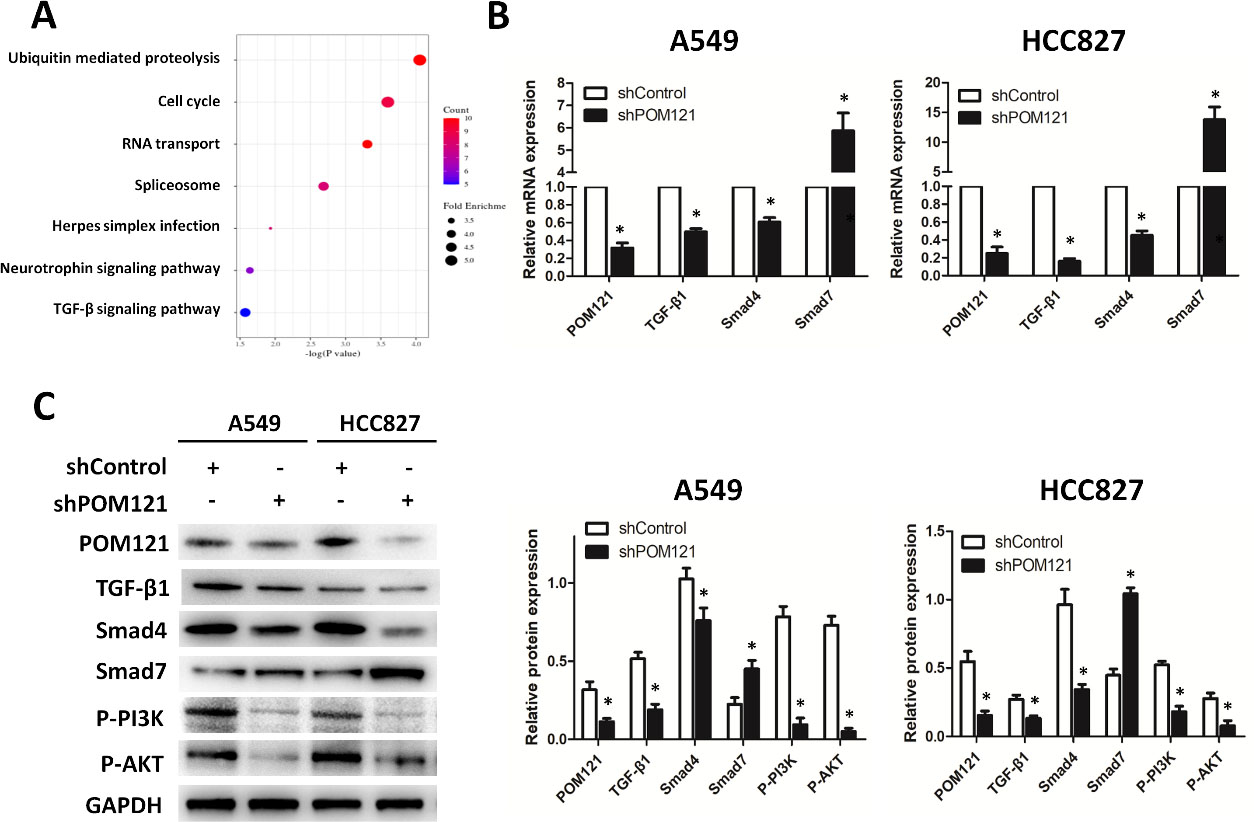
In this study, qRT-PCR and IHC were performed to detect POM121 mRNA and protein levels in NSCLC tissues. POM121 mRNA levels were much higher in carcinomatous tissues than that in matched para-carcinoma tissues. As was the case with mRNA, a larger proportion of high POM121 protein expression in cancerous tissues (60.07%) was confirmed compared with normal lung tissues (22.17%). High expression of POM121 has also been confirmed in colorectal cancer [18], laryngeal cancer [19], and oral squamous cell carcinoma [20]. Our results suggested that POM121 is overexpressed in NSCLC tissues, which is consistent with previous studies.
The relationship between POM121 and clinicopathological features of NSCLC patients was analyzed according to 796 NSCLC tissue samples and their associated clinical data. Increased POM121 expression was found to be significantly associated with advanced differentiation, lymph node metastases, distant metastases, and venous invasion. Multivariate cox regression analysis and Kaplan-Meier survival curves confirmed high POM121 expression as a negative prognostic factor for NSCLC patients. These results suggested that POM121 may play a critical role in the progression and metastasis of NSCLC, and NSCLC patients with high POM121 expression had poor OS rates.
So far, investigations to explore the function of POM121 in human cancers are few. Veronica et al. demonstrated that POM121 promotes tumorigenesis, proliferation, and drug-resistance in prostate cancer cells [15]. In this study, modulating effect of POM121 on NSCLC was detected in vitro and vivo. After POM121 downregulation, both A549 and HCC827 cells presented weakened abilities of cell proliferation, migration and invasion. Subcutaneously implanted tumor model also proved negative effect of shPOM121 on NSCLC cell growth. These results confirmed that POM121 inhibition suppressed proliferation, migration and invasion of NSCLC cells, which means POM121 could contribute to NSCLC proliferation and metastasis.
TGF-
Beside SMAD-mediated signaling, the TGF-
In conclusion, the study confirmed that POM121 knockdown could weaken proliferation, migration and invasion of NSCLC through TGF-
Footnotes
Acknowledgments
We thank H. Nikki March, PhD, from LiwenBianji, Edanz Editing China (
Author contributions
Conception: Lirong Guan, Kun Zhao
Interpretation or analysis of data: Lirong Guan, Lingli Zhang, Tengqi Wang, Lizhou Jia, Ning Zhang
Preparation of the manuscript: Lirong Guan, Lingli Zhang, Ning Zhang, Huishan Yan
Revision for important intellectual content: Tengqi Wang, Lizhou Jia
Supervision: Kun Zhao
