Abstract
Aim of the study. In recent years, the incidence of lung cancer, as well as the mortality rate from this disease, has increased. Moreover, because of acquired drug resistance and adverse side effects, the effectiveness of current therapeutics used for the treatment of lung cancer has decreased significantly. Chinese medicine has been shown to have significant antitumor effects and is increasingly being used for the treatment of cancer. However, as the mechanisms of action for many Chinese medicines are undefined, the application of Chinese medicine for the treatment of cancer is limited. The formula tested has been used clinically by the China National Traditional Chinese Medicine Master, Professor Zhonging Zhou for treatment of cancer. In this article, we examine the efficacy of Ke formula in the treatment of non–small cell lung cancer and elucidate its mechanism of action. Methods. A Balb/c nude mouse xenograft model using A549 cells was previously established. The mice were randomly divided into normal, mock, Ke, cisplatin (DDP), and co-formulated (Ke + DDP) groups. After 15 days of drug administration, the animals were sacrificed, body weight and tumor volume were recorded, and the tumor-inhibiting rate was calculated. A cancer pathway finder polymerase chain reaction array was used to monitor the expression of 88 genes in tumor tissue samples. The potential antiproliferation mechanism was also investigated by Western blot analysis. Results. Ke formula minimized chemotherapy-related weight loss in tumor-bearing mice without exhibiting distinct toxicity. Ke formula also inhibited tumor growth, which was associated with the downregulation of genes in the PI3K/AKT, MAPK, and WNT/β-catenin pathways. The results from Western blot analyses further indicated that Ke blocked the cell cycle progression at the G1/S phase and induced apoptosis mainly via the PI3K/AKT pathway. Conclusion. Ke formula inhibits tumor growth in an A549 xenograft mouse model with no obvious side effects. Moreover, Ke exhibits synergistic antitumor effects when combined with DDP. The mechanism of action of Ke is to induce cell cycle arrest and apoptosis by suppressing the PI3K/AKT pathway. Further research will be required to determine the mechanism of action behind the synergistic effect of Ke and DDP.
Keywords
Introduction
Non–small cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer cases.1,2 Although lung cancer can be treated using various therapies, including chemotherapy and radiotherapy, these treatments often have adverse side effects.3-5 As such, new drugs with fewer negative side effects are needed. A potential strategy for the treatment of NSCLC is to use medicinal plants that are effective against lung cancer but have fewer side effects.
For many years, traditional Chinese medicine (TCM) has been a prime source of anticancer treatments. However, a single herb is seldom used alone. Herbal formulas use synergy and interactions between a myriad phytochemicals present in different herbs to achieve therapeutic efficacy by targeting multiple biological and pathological processes while minimizing adverse side effects.6-9 One such formula, Ke formula, has been used to treat cancer for many years. Indeed, clinical observations from studies performed using Ke formula suggest that this formula has some efficacy as a cancer treatment.10-12 Therefore this formula is of sufficient interest to merit laboratory testing to further investigate its antitumor properties and elucidate its mechanism of action.
In this study, we used polymerase chain reaction (PCR) microarrays to investigate the activity of Ke formula in murine xenograft models to examine gene expression. Based on the microarray data, we performed an in-depth study of potential Ke formula–mediated signaling pathways involved in the regulation of cell growth and apoptosis.
Materials and Methods
Cell Line and Culture Conditions
Human lung carcinoma (NCI-A549) cells obtained from the Cell Line Bank (Shanghai, China) were used for the experiments. The cell line was cultured in RPMI-1640 (Gibco, Carlsbad, CA) with 10% fetal bovine serum (Gibco, Carlsbad, CA), 100 U/mL penicillin, and 100 mg/mL streptomycin (Amresco) in a 5% CO2 cell culture incubator (Forma, Thermo Scientific, Waltham, MA) at 37°C.
Drugs
Ke formula is made up of 13 herbs, Semen Benincasae (15 g), Arisaema Cum Bile (12 g), Houttuynia Cordata (15 g), Rhizoma Fagopyri Dibotryis (15 g), Cremastra Appendiculata (10 g), Herba et Gemma Agrimoniae (15 g), Carthami Flos (10 g), Lignum Sappan (10 g), Rhizoma Pinelliae (10 g), Radix Ranunculi Ternati (15 g), Radix Adenophorae (12 g), Radix Ophiopogonis (12 g), and Rhizoma Polygonati Odorati (12 g), according to the conventional method. The table in the appendix lists the names of these herbs in Chinese, English, and Latin.
The criteria for the quality of the herbs used were in accordance with the 2005 edition of Chinese pharmacopoeia (Chinese Pharmacopoeia Commission, Pharmacopoeia of People’s Republic of China, Beijing: People’s Medical Publishing House; 2005). Before use in experiments, the herbs were tested for heavy metals, microbial contamination, and residual pesticides; all results met the safety standards in China. Laboratory personnel were blinded as to the identity of the herbs. A trained technician prepared the decoction according to a standardized procedure. Each unit of formula yielded 1200 mL of decoction. Cisplatin (cis-diamminedichloroplatinum [DDP]) was purchased from Nanjing Pharmaceutical Factory, Ltd.
Experimental Animals
Balb/c athymic (nude) mice (female, age 6-8 weeks, n = 40) weighing 20 ± 2 g were purchased from the Animal Center in the Academy of Military Medical Sciences (Beijing, China). The animal room was controlled for temperature (22°C ± 2°C), light (12-hour light/dark cycle), and humidity (50% ± 10%). All laboratory feed pellets and bedding were autoclaved.
The study was approved by the Jiangsu Animal Care and Use Committee and followed national and institutional rules regarding animal experiments.
Xenograft Experiments
A nude mouse model of tumor regression was used to evaluate suppression of solid tumor growth in mice treated with different drugs. A total of 1 × 107 A549 cells in 0.2 mL culture medium were subcutaneously injected into the axilla of each mouse. After 7 days of observation, solid tumor masses were excised from the NCI-A549-inoculated mice. When the tumor volume reached approximately 50 mm3, the mice were randomly distributed into 4 groups of 8: mock (saline alone), Ke (1.8 g/kg/d Ke formula), DDP (5 mg/kg/d DDP), and co-formulated (Ke formula + DDP, same dosage as for individual treatment). Ke formula was orally administrated. DDP was peritoneally injected daily for 15 days. Mice with no tumors served as the control group and were orally administered saline. The dosage of Ke formula was converted from the human clinical dosage. The effects of different concentrations of Ke formula on Lewis lung carcinoma cell transplanted tumors and survival conditions of C57/BL6 mice were examined.
Antitumor Activity In Vivo
All animals were monitored for activity, physical condition, bodyweight, and tumor growth. The body weight of each animal was measured once every 2 days. The longest (a) and the shortest (b) tumor diameters (mm) were obtained, and the formula for an ellipsoid sphere (0.52 × a × b2) was used to calculate the tumor volume every 2 days. The tumor weights were also measured on the final day of the experiment after the animals were sacrificed. The antitumor activities of the different treatments were expressed as inhibition rate:
where Wmock and Wtreatment are the tumor weights of the mock and treatment groups, respectively. After the weight was measured, the xenograft tumors were frozen in liquid nitrogen for further experiments.
Polymerase Chain Reaction Microarray Analysis
Total RNA was extracted from experimental tumor samples using Trizol reagent (Invitrogen, Carlsbad, CA). RNA was purified using an RNeasy MinElute Cleanup Kit (Qiagen, Valencia, CA) according to the manufacturer’s protocol. RNA concentration was quantified by UV absorption using a NanoDrop spectrophotometer (Bio Lab, Ltd, Jerusalem, Israel). cDNA was synthesized from total RNA with Super Script.III Reverse Transcriptase (Invitrogen, Carlsbad, CA), and used for real-time PCR with the lung cancer pathway finder PCR array (Cat. No. PA-112, Super Array, Bioscience Corporation, Foster City, CA). The ΔΔCt method was used to analyze the PCR data.
ΔCt was calculated for each pathway-focused gene in each treatment group.
ΔCt (group 1) = average Ct − average of HK genes’ Ct for group 1 array
ΔCt (group 2) = average Ct − average of HK genes’ Ct for group 2 array
ΔΔCt was calculated for each gene across 2 PCR arrays (or groups).
ΔΔCt = ΔCt (group 2) − ΔCt (group 1)
Fold-change was calculated for each gene from groups 1 to 2 as 2-ΔΔCt.
Western Blot Analysis
Cancer tissues were ground in a mortar with liquid nitrogen, lysed in tissue lysis buffer (50 mM Tris (pH 7.4), 150 mM NaCl, 1% Triton X-100, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), and 1 × cocktail containing 0.5 mM Na3VO4, 50 mM NaF, and 5 mM phenylmethyldulfonyl fluoride), then sonicated on ice with an ultrasonic disrupter for 30 minutes. The resulting homogenate was centrifuged at 12000 rpm/min for 30 min. After the supernatant was harvested, equal amounts of proteins were separated by 10% SDS–polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane (Millipore, Billerica, MA). The membrane was probed with antibodies directed against p-AKT (ser473), p-AKT(thr308), AKT, Bax, Bcl-2, CDK4, CyclinD3, E2F1, P21, P27, p-PTEN(ser380), p-GSK3β(ser9) (all from Cell Signaling Technology, Danvers, MA), and β-actin (Santa Cruz Biotechnology, Santa Cruz, CA). The antibodies were diluted in phosphate-buffered saline (pH 7.4) containing 0.05% (v/v) Tween-20 and 5% (w/v) dried milk. The blots were incubated with the appropriate horseradish peroxidase–conjugated secondary antibodies (Cell Signaling Technology, Danvers, MA). Bound antibodies were visualized by Super Signal West Dura Extended Duration Substrate (Thermo Scientific, Waltham, MA) using a Gel Imaging System (ChemDoc XRS System170-8070, Bio-Rad, Hercules, CA).
Identification of Pathways Using the Kyoto Encyclopedia of Genes and Genomes (KEGG) and BioCarta Databases
Gene identification was performed to determine which biochemical pathways were altered by the different treatments. After identifying genes from the PCR microarray data, we investigated whether any of these genes were part of the same pathway. To this end, the KEGG (http://www.genome.ad.jp/kegg/pathway.html) and BioCarta (http://www.biocarta.com/genes/allpathways.asp) biochemical pathway databases were searched using the genes selected from the PCR microarray and Western blot analyses.
Statistical Analysis
The data were analyzed using SPSS 11.5 software and presented as mean ± standard deviation (
Results
Ke Suppresses the Growth of NCI-A549 Cells in Mice
Over the course of treatment, mice in the DDP group exhibited significant weight loss and slowed activity (Figure 1A). In contrast, those in the Ke group maintained stable weight and normal activity, and those in the Ke + DDP group exhibited marked improvement (Figure 1A). While all treatments inhibited the growth of NCI-A549-transplanted solid tumors as compared with the mock group (Figure 1B), Ke significantly inhibited tumor growth as compared with the mock group (significant differences in final tumor weight [P < .05] and volume [P < .01] were observed). After the animals were sacrificed (Figure 1C), the tumor-inhibition rate was 62.8%. After DDP combination, the tumor-inhibition rate was 82.5% (Table 1). These data indicate that Ke formula resulted in minimal chemotherapy-related body weight loss in mice bearing tumors and did not adversely affect activity.
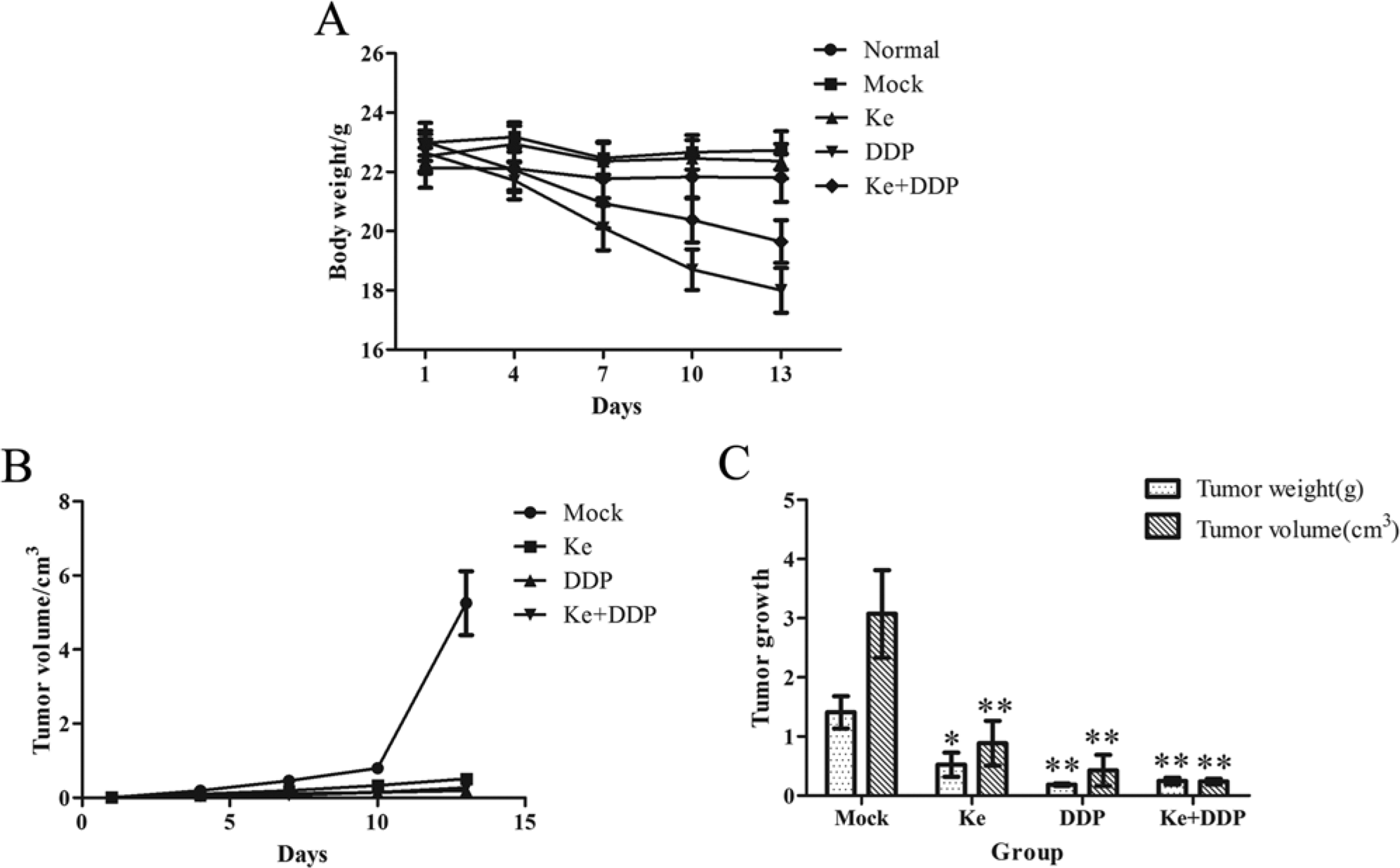
Effects of different drugs on the suppression of NCI-A549 transplanted tumor and survival condition of nude mice. (A) Mice of DDP (Cisplatin) group lost weight significantly, and mice of Ke + DDP group have markedly improved, and all the mice of Ke group keep stable weight with normal activity. (B) All drugs significantly suppress the solid tumor growth compared with the mock group. (C) Ke and DDP significantly inhibited both the volume and weight of tumors, and their combination displays a certain synergy. Values are means of 3 separate experiments ± standard deviation. *P < .05 and **P < .01 by Student’s paired t-test compared with the mock group.
Effects of the Ke on the Tumor Volume and Weight of NCI-A549 Tumor-Bearing Mice.
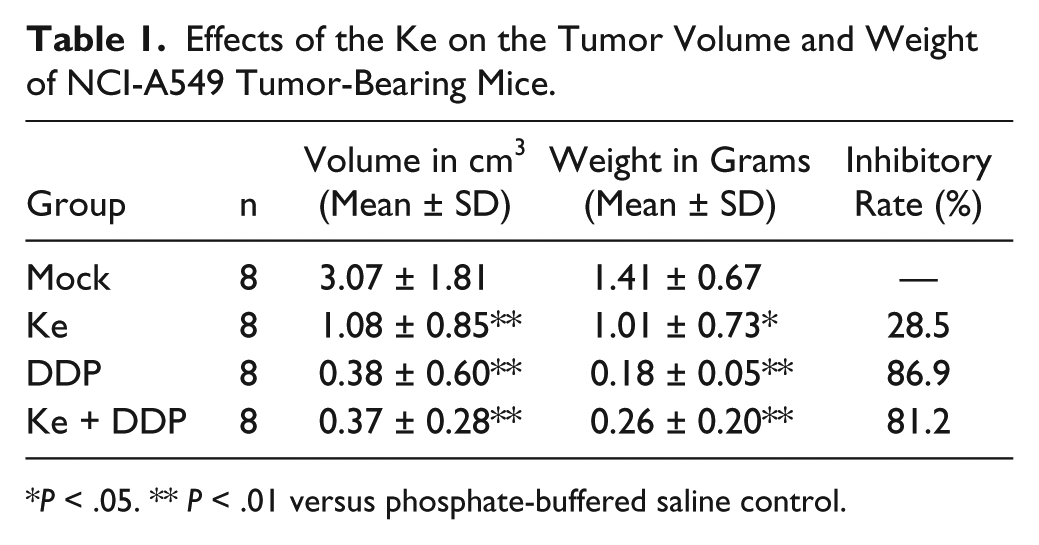
P < .05. ** P < .01 versus phosphate-buffered saline control.
Gene Expression Profiles Vary Among the Different Treatment Groups
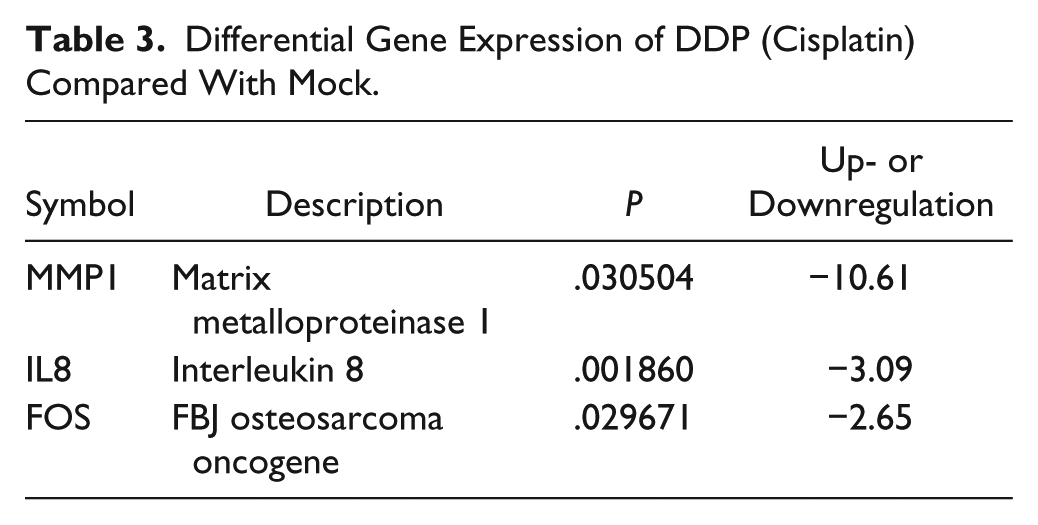
The lung cancer pathway finder PCR array was used to analyze changes in gene expression after the treatment and to elucidate the molecular mechanisms by which Ke inhibits tumor growth. A total of 8, 3, and 12 genes exhibited a 2-fold difference in expression after treatment with the different drugs as compared with the mock group. Of these genes, 1, 0, and 2 were upregulated and 7, 3, and 10 were downregulated in the Ke, DDP, and co-formulated groups, respectively (Figure 2). The genes that were differentially expressed are listed and categorized by their putative functions in Tables 2 to 4. In general, the upregulated genes were involved in the TGFβ signal pathway, while the genes that were downregulated were involved in cell cycle, apoptosis, proliferation, and angiogenesis belong to the PI3K/AKT, MAPK, and WNT/β-catenin pathways.

Putative genes expression in differences compared with mock group after drugs treatment. (A) Differential gene expression of Ke. (B) Differential gene expression of DDP (Cisplatin). (C) Differential gene expression of Ke + DDP.
Differential Gene Expression of Ke Compared With Mock.

Differential Gene Expression of DDP (Cisplatin) Compared With Mock.
Differential Gene Expression of Ke + DDP Compared With Mock.

Ke Induces Cell Cycle Arrest in the G1/S Phase and Activates Apoptotic Proteins
To predict the putative signaling pathway(s) affected by treatment with Ke, we used the KEGG and BioCarta pathway databases to analyze the differentially expressed profiles of drug-induced genes to fit the transduction-signaling map. Two major pathways were downregulated by Ke: the cell cycle regulatory pathway and apoptotic pathway. Our results indicate that Ke treatment decreased the expression levels of E2F1 (2.08-fold) and CDK4 (1.09-fold), as well as Bcl-2 and AKT (2.08- and 1.18-fold, respectively). Combination of Ke with DDP resulted in further downregulation of these genes; E2F1 (2.4-fold), CDK4 (1.25-fold), Bcl-2 (2.72-fold), and AKT (1.60-fold). These data suggest that Ke inhibits tumor growth by suppressing PI3K/AKT activation and that the combination of Ke and DDP has a synergistic additive effect.
Ke Suppresses PI3K/AKT Pathway Activation
To determine whether Ke can suppress PI3K/AKT pathway activation, Western blot analysis was used to examine changes in PI3K/AKT pathway activity after herbal treatment. Western blot analysis confirmed that Ke increased the ratio of Bax/Bcl-2, induced apoptosis (Figure 3A), and decreased the levels of CDK4, CyclinD3, and E2F1. However, in terms of apoptosis and cell cycle regulation, the expression levels of P21 and P27 did not change (Figure 3B). In addition, Ke also downregulated the levels of p-AKT (ser473), p-AKT (thr308), p-GSK3β, and p-PTEN (Figure 3C). In terms of apoptosis and cell cycle, Ke combined with DDP increased the ratio of Bax/Bcl-2, decreased the levels of CDK4 and P27, increased the level of E2F1, and did not affect the levels of CyclinD3 and P21 as compared with the mock group. The levels of p-AKT (ser473), p-AKT (thr308), and p-GSK3β were upregulated, whereas the level of PTEN was not changed in the Ke+DDP group as compared with the mock group. These results indicate that Ke could suppress PI3K/AKT pathway activation, induce apoptosis, arrest the cell cycle in the G1/S phase, and inhibit the growth of NCI-A549 cell-transplanted solid tumors. Further studies will be required to more precisely define the mechanisms behind the synergistic effect of Ke and DDP.

Ke formula induces cell cycle arrest in G1/S phase and activates apoptotic proteins via suppress PI3K/AKT/GSK3β pathway. (A) The expression levels of cell cycle regulatory proteins were analyzed by Western blotting. (B) The expression levels of apoptosis-related proteins were analyzed by Western blotting. (C) The expression levels of phosphorylation of AKT and PTEN were analyzed by Western blotting.
Discussion
Traditional Chinese medicine provides a rich source of potential chemopreventive and therapeutic agents for the treatment of cancer. Rigorous and systematic evaluations are, however, necessary to establish the efficacy of herbal formulas and transform traditional herbal practices into science-based medicines.13-17 In the present article, we evaluated the anticancer activity of Ke formula, which has been shown to be effective in the clinic. The crude formula used in this study followed TCM treatment principles based on syndrome differentiation. TCM theory proposes that lung cancer is mainly related to deficiency of both Qi and Yin, as well as pathological changes of Qi stagnation, blood stasis, and accumulation of phlegm. Therefore, the treatment principle is to resolve phlegm, activate blood and Qi circulation, remove stasis, and nourish Yin. The herbs in Ke formula include herbs of drying dampness and resolving phlegm (Semen Benincasae, Rhizoma Pinelliae, and Arisaema Cum Bile), herbs of relieving masses (Houttuynia Cordata, Rhizoma Fagopyri Dibotryis, Cremastra Appendiculata, and Radix Ranunculi Ternati), herbs of activating blood circulation and removing stasis (Herba et Gemma Agrimoniae, Carthami Flos, and Lignm Sappan); and herbs of invigorating Qi and nourishing Yin (Radix Adenophorae, Rhizoma Polygonati Odorati, and Radix Ophiopogonis). In this study, we demonstrated that Ke can inhibit NCI-A549 lung cancer cell growth in vivo and induce cell cycle G1/S arrest and apoptosis. Moreover, the animal study revealed that Ke significantly inhibited the growth of subcutaneous human NCI-A549 xenograft tumors with no obvious side effects. Data from the microarray experiments demonstrated that Ke can upregulate the expression of TGFβ, which has a dual function in tumors and could negatively regulate the expression of CDK. 18 In general, Ke suppressed the expression of genes belonging to certain pathways, including those involved in MAPK signaling, cell cycle progression, and apoptosis. Western blot analysis demonstrated that the key pathway affected was that regulated by activation of AKT. Indeed, our data suggest that Ke affects cell growth mainly by inhibiting the PI3K/AKT pathway.
The PI3K/AKT pathway plays an important role in survival when cancer cells are exposed to different kinds of apoptotic stimuli, such as oxidative and osmotic stresses, irradiation, matrix adhesion, ischemic shock, and chemotherapeutic drugs.19,20 Once activated, AKT controls cellular functions, such as apoptosis, cell cycle regulation, gene transcription, and protein synthesis, through the phosphorylation of downstream substrates, such as GSK3β and Bad. AKT is overexpressed and constitutively active in many human cancers, including NSCLC.21-23 The activation of AKT can be blocked by PTEN through inhibition of phosphorylation.24,25 In the present study, we evaluated the effect of Ke on the PI3K/AKT pathway. Interestingly, the levels of p-PTEN, p-AKT (ser473), and p-AKT (thr308) decreased after Ke treatment, indicating that Ke may inhibit AKT activation by suppressing PTEN phosphorylation.
Cell cycle regulation is important in growth control, and dysregulation of the cell cycle machinery has been implicated in tumorigenesis.26,27 The CDK4/cyclin D complex promotes the progression through the G1 phase into the S phase by phosphorylating retinoblastoma (Rb) protein and releasing E2F1.28,29 Overexpression of CDK4 has been described in many tumors, including NSCLC. The activity of CDK/cyclin complexes is negatively regulated by binding to cyclin-dependent kinase inhibitors belonging to the INK4 family (P16, P15, P18, and P19) and the Cip/Kip family (P21, P27, and P57).30,31 Among these, P21 induces cell cycle arrest in the G0/G1 phase by inhibiting the activity of the CDK4/cyclin D complex, and seems to be a fundamental partner of P53.32-34 Our results suggest that the cell cycle could be blocked as the levels of CDK4/CyclinD3 and E2F1 were decreased and those of P21 and P27 did not change after Ke treatment. Therefore, Ke could possibly induce G1/S arrest via a P53-independent mechanism.
Through investigation of signaling pathways, we found another gene-regulating cell cycle process to be important in response to Ke treatment. GSK3β, as a downstream target of the PI3K/AKT pathway, regulates cell proliferation and differentiation via phosphorylation of a serine residue 9 by AKT.35,36 GSK3β is a key regulatory enzyme in the WNT/β-catenin transduction pathway. In addition, it could regulate transcription factors such as JUN and MYC as well as phosphorylate the CDK4/cyclin D complex, leading to rapid ubiquitination,37-40 degradation of Rb and E2F1, and induction of G1/S arrest.
Apoptosis, an important regulator in developmental processes, maintenance of homeostasis, and elimination of damaged cells, is the outcome of a complex interaction between pro-apoptotic and anti-apoptotic molecules. Proteins of the Bcl-2 family are key regulators of the apoptotic pathway. The Bcl-2 family can be divided into 2 subfamilies: anti-apoptotic proteins such as Bcl-2 and pro-apoptotic proteins such as Bax. Accumulating evidence indicates that many anticancer agents induce apoptosis by targeting the proteins of the Bcl-2 family. The ratio of Bax/Bcl-2 is a critical determinant of whether cells will undergo apoptosis.41-43 In the present study, Ke increased pro-apoptotic Bax expression and decreased anti-apoptotic Bcl-2 expression, leading to an increased Bax/Bcl-2 ratio, which may be one of the molecular mechanisms by which Ke induces apoptosis.
In conclusion, Ke can induce cell cycle arrest at G1/S phase by disrupting regulatory proteins involved in cell cycle progression and initiate apoptosis and by suppressing the expression of genes of focal adhesion and proliferation through PI3K/AKT pathway inactivation (Figure 4). Conversely, the mechanism behind the synergistic effect of Ke and DDP does not rely on the PI3K/AKT pathway. DDP is a nonspecific cell cycle drug that mainly interferes with DNA replication. Analysis of the microarray data indicates that a close relationship exists between DDP and the MAPK pathway. Taken together, our results indicate that treatment with Ke alone, or in combination with DDP, would provide therapeutic efficacy against NSCLC, without adverse side effects.

Schematic diagram of Ke-regulated genes in different pathways analyzed by Western blotting and KEGG and BioCarta pathway databases. On regulation of the cell cycle, Ke leads to inactivation of AKT, and hence GSK3β inhibits the CDK4/CyclinD3 complex, reduces the p-Rb and E2F1’s expression, and eventually induces G1/S phase arrest, meanwhile, Ke also increase the expression of Bax and decrease the expression of Bcl-2 to promote apoptosis. Ke-induced genes are colored red, and Ke-suppressed genes are colored green, and those colored yellow mean Ke-unregulated genes.
Footnotes
Appendix
Names of Herbs
| Chinese | Pinyin | English | Latin |
|---|---|---|---|
| 冬瓜子 | Dongguazi | Benincasae Semen | Semen Benincasae |
| 法半夏 | Fabanxia | Pinellia Tuber | Rhizoma Pinelliae |
| 胆南星 | Dannanxing | Bile Arisaema | Arisaema Cun Bile |
| 山慈菇 | Shancigu | Pseudobulb of Appendiculate Cremastra | Cremastra Appendiculata |
| 猫爪草 | Maozhaocao | Catclaw Buttercup Root | Radix Ranunculi Ternati |
| 金荞麦 | Jinqiaomai | Wild Buckwheat Rhizome | Rhizoma Fagopyri Dibotryis |
| 鱼腥草 | Yuxingcao | Heartleaf Houttuymia Herb | Houttuynia Cordata |
| 仙鹤草 | Xianhecao | Hairyvein Agrimonia Herb and Bud | Herba et Gemma Agrimoniae |
| 苏木 | Sumu | Sappan Wood | Lignm Sappan |
| 红花 | Honghua | Safflower | Carthami Flos |
| 沙参 | Shashen | Fourleaf ladybell Root | Radix Adenophorae |
| 玉竹 | Yuzhu | Fragrant solomonseal Rhizome | Rhizoma Polygonati Odorati |
| 麦冬 | Maidong | Dwarf Lilyturf Tuber | Radix Ophiopogonis |

Acknowledgements
We thank members of the Fu Lab, Emory University, and Jiangsu Key Laboratory for their assistance in traditional Chinese medicine formula research and the staff of Nanjing University of Chinese Medicine for their assistance and enlightening discussions and Cheryl Meyerkord for editing the text.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the National Science and Technology Pillar Program in the 11th Five-Year Plan of China 2006BAI11B08-01 (to XZ and HF), the Priority Academic Program Development of Jiangsu Higher Education Institutions (to XZ), and the Research and Innovation Program of Postgraduates in Jiangsu Province (to MC) and the U.S. National Institutes of Health grants P01 CA116676 (to HF).

