Abstract
OBJECTIVE:
MicroRNA-629 (miR-629) has been found to play an important role in the pathogenesis of human cancers. However, the function of miR-629 is still unknown in non-small-cell lung cancer (NSCLC). The purpose of this study is to preliminarily elucidate the regulatory mechanism of miR-629 in NSCLC.
MATERIALS AND METHODS:
The mRNA and protein expression was measured by real-time quantitative polymerase chain reaction (RT-qPCR) and western blot analysis. The function of miR-629 was investigated by MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) and Transwell assays. The relationship between miR-629 and FOXO1 was confirmed by dual luciferase assay.
RESULTS:
MiR-629 was upregulated in NSCLC tissues and cells. High expression of miR-629 predicted poor prognosis in patients with NSCLC. Moreover, miR-629 promoted cell proliferation, migration and invasion in NSCLC cells. In addition, FOXO1 was confirmed as a direct target of miR-629 in NSCLC. Furthermore, knockdown of FOXO1 also promoted proliferation, migration and invasion of NSCLC cells. More importantly, overexpression of FOXO1 weakened the carcinogenesis of miR-629 in NSCLC. Besides that, miR-629 promoted EMT and activated the PI3K/AKT pathway in NSCLC.
CONCLUSIONS:
MiR-629 promotes the progression of NSCLC by targeting FOXO1 and regulating EMT/PI3K/AKT pathway.
Introduction
With the increase of the elderly population, the acceleration of industrialization and the aggravation of environmental pollution, the incidence and mortality of lung cancer have been rising in the past few decades [1]. Lung cancer has become a leading cause of cancer death in China [2]. Therefore, the prevention and early detection of lung cancer have become research focus in China. Lung cancer is mainly divided into non-small cell lung cancer (NSCLC) and small cell lung cancer. Moreover, NSCLC cells grow and differentiate more slowly than small cell cancer cells. Currently, surgical resection adjuvant radiotherapy and chemotherapy are the primary treatment for patients with NSCLC [3]. It has been reported that the survival rate is closely associated with clinical stage in NSCLC patients. In addition, many patients with NSCLC have poor prognosis due to delayed diagnosis [4]. Therefore, early diagnosis and reasonable treatment are very important to improve the survival rate of patients with NSCLC.
Previous studies have shown that microRNAs (miRNAs) are involved in tumorigenesis by binding to the 3’-UTR of target genes [5]. In addition, abnormal expression of miRNAs has been identified in various cancers that regulate tumorigenesis. In particular, many miRNAs have been reported to act as onco-miR or inhibitory miRNAs in NSCLC. For example, miR-92b was upregulated in NSCLC and promoted NSCLC progression by targeting RECK [6]. In contrast, miR-194 was found to be downregulated and suppress cell metastasis in NSCLC by regulating BMP1 and p27 expression [7]. Recently, the specific functions of miR-629 have caught our attention. It has been reported that miR-629 was upregulated and acted as an oncogene in gastric cancer [8]. Furthermore, miR-629 has been found to promote cell metastasis in ccRCC by targeting TRIM33 [9]. More importantly, it has been shown that the miR-629/NBS1 axis caused an increased risk of lung cancer in the Southern and Eastern Chinese population [10]. However, few reports have reported the biological effect of miR-629 in NSCLC, particularly on cell proliferation, migration and invasion.
In addition, previous studies have shown that forkhead box O1 (FOXO1) is an important regulator in the pathogenesis of human cancer [11]. For example, FOXO1 has been identified as a tumor suppressor in gastric cancer [12] and breast cancer [13]. In addition, FOXO1 has been reported to be regulated by several miRNAs in different cancers, such as miR-9 [14], miR-425 [15] and miR-544 [16]. Besides, FOXO1 has been found to be involved in the PI3K/AKT pathway via upstream activation of AKT [17]. Moreover, miRNA-regulated epithelial-mesenchymal transition (EMT)/PI3K/AKT pathway has been reported in NSCLC [18]. For example, miR-26a has been found to promote metastasis of lung cancer cells via the EMT/AKT pathway [19]. However, the interaction between miR-629 and FOXO1 remains unclear in the progression of NSCLC.
In this study, the functions of miR-629 and FOXO1 were investigated in NSCLC. At the same time, the effects of miR-629 and FOXO1 on the EMT/PI3K/AKT pathway were also explored in NSCLC. Further, the relationship between miR-629 and FOXO1 in NSCLC was confirmed. These findings will provide a useful therapeutic target for the treatment of NSCLC.
Patients and methods
Clinical tissues
NSCLC tissues and adjacent normal lung tissues were obtained from 74 patients in Shanghai Chest Hospital affiliated to Shanghai Jiaotong University. Written informed consents were obtained from all patients. All NSCLC patients did not receive any treatment prior to surgery. Theses tissues were frozen in liquid nitrogen and then stored in a
Cell lines culture
The human normal bronchial epithelium cell line BEAS-2B (BNCC338205) and H1299 (BNCC100859), A549 (BNCC100258), H460 (BNCC102112) NSCLC cell lines were purchased from Cell Bank of the Chinese Academy of Sciences (Shanghai, China). These cell lines were then inoculated in dulbecco’s modified eagle medium (DMEM) medium (Gibco, Rockville, MD, USA) with 10% fetal bovine serum (FBS) (Gibco, Rockville, MD, USA) and cultured in an atmosphere with 5% CO
Cell transfection
MiR-629 mimics or inhibitor and negative control (NC) were obtained from GenePharma (Shanghai, China). FOXO1 siRNA and vector were also synthesized by GenePharma. MiR-629 mimics: 5’-UAUGGC ACUGGUAGAAUUCACU-3’, miR-629 inhibitor: 5’-AGUGAAUUCUACCAGUGCCAU C-3’, and negative control (NC): 5’-UUGUACUACACAAAAGUAAUG-3’. They were then transfected into A549 and H1299 cells using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA), respectively, based on the manufactures’ protocols.
Real-time quantitative polymerase chain reaction (RT-qPCR) analysis
Total RNA in NSCLC tissues and cells was extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The cDNA was synthesized using the PrimeScript
MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide) assay
First, 6
Transwell assay
Transwell chambers (8-
Western blot analysis
Transfected A549 and H1299 cells were lysed using radioimmunoprecipitation assay (RIPA) lysis buffer (Beyotime, Shanghai, China). The protein concentration was detected using a bicinchoninic acid (BCA) Protein Assay kit (Pierce, Rockford, IL, USA) according to the manufacturer’s instruction. Then, 30
Bioinformatics analysis
Bioinformatics prediction TargetScan version 7.1 online software (
Dual luciferase assay
The 3’-UTR of wild type or mutant FOXO1 was inserted into the pcDNA3.1 plasmid vector (Promega, Madison, WI, USA). Next, the above vector and miR-629 mimics were transfected into A549 and H1299 cells. Forty-eight hours after transfection, luciferase activity was measured by a dual luciferase assay system (Promega, Madison, WI, USA).
MiR-629 was upregulated in NSCLC tissues. (A) The expression of miR-629 was detected in NSCLC tissues and normal tissues. (B) High miR-629 expression was correlated with shorter overall survival in NSCLC patients. 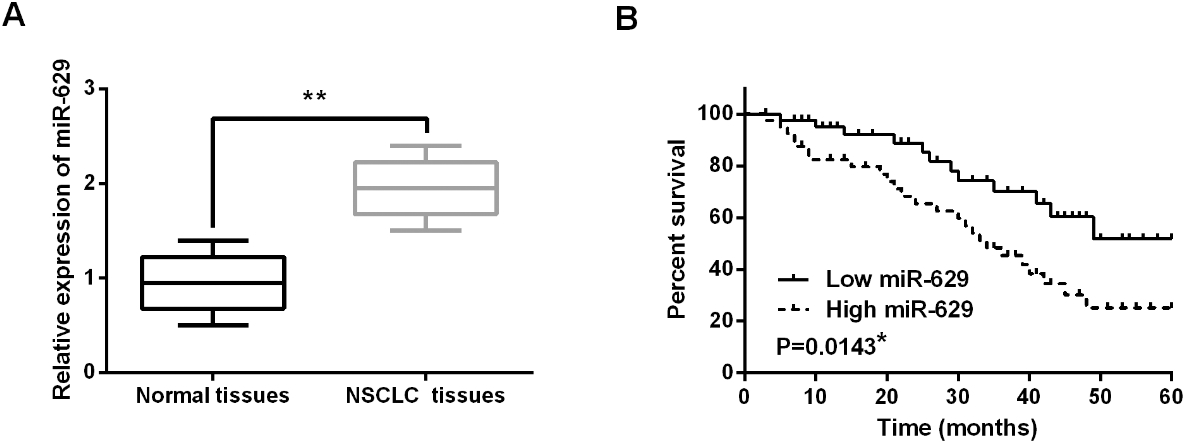
Data are shown as mean
Results
MiR-629 was upregulated in NSCLC tissues
The expression of miR-629 was detected in NSCLC tissues by RT-qPCR assay. The expression of miR-629 was found to be increased in NSCLC tissues compared to normal tissues (Fig. 1A). Next, we found that the abnormal expression of miR-629 was associated with lymph nodes metastasis (
Relationship between miR-629 expression and clinic-pathological characteristics in NSCLC patients
Relationship between miR-629 expression and clinic-pathological characteristics in NSCLC patients
Statistical analyses were performed by the
Next, the expression of miR-629 was detected in H1299, A549, H460 and BEAS-2B cell lines. MiR-629 was found to be upregulated in H1299, A549 and H460 cell lines compared to BEAS-2B cells (Fig. 2A). The function of miR-629 in NSCLC was investigated in A549 and H1299 cells with miR-629 mimics or inhibitors. RT-qPCR assay showed that miR-629 mimics increased the expression of miR-629, while miR-629 inhibitor decreased its expression in A549 and H1299 cells (Fig. 2B). Functionally, overexpression of miR-629 promoted cell proliferation, while downregulation of miR-629 suppressed cell proliferation in A549 and H1299 cells (Fig. 2C and D). Similarly, upregulation of miR-629 was also found to promote cell migration, when downregulation of miR-629 inhibited cell migration in A549 and H1299 cells (Fig. 2E). In addition, the same effect of miR-629 on cell invasion was also identified in A549 and H1299 cells (Fig. 2F). Briefly, these results reveal that miR-629 acts as a tumor promoter in NSCLC by promoting cell proliferation, migration and invasion.
MiR-629 promoted proliferation, migration and invasion of NSCLC cells. (A) MiR-629 expression was examined in H1299, A549, H460 NSCLC cells and human normal bronchial epithelium cell line BEAS-2B. (B) The expression of miR-629 was examined in A549 and H1299 cells with miR-629 mimics or inhibitor. (C) and (D) Cell proliferation was measured in A549 and H1299 cells containing miR-629 mimics or inhibitor. (E) and (F) Cell migration and invasion were detected in A549 and H1299 cells with miR-629 mimics or inhibitor (magnification: 200 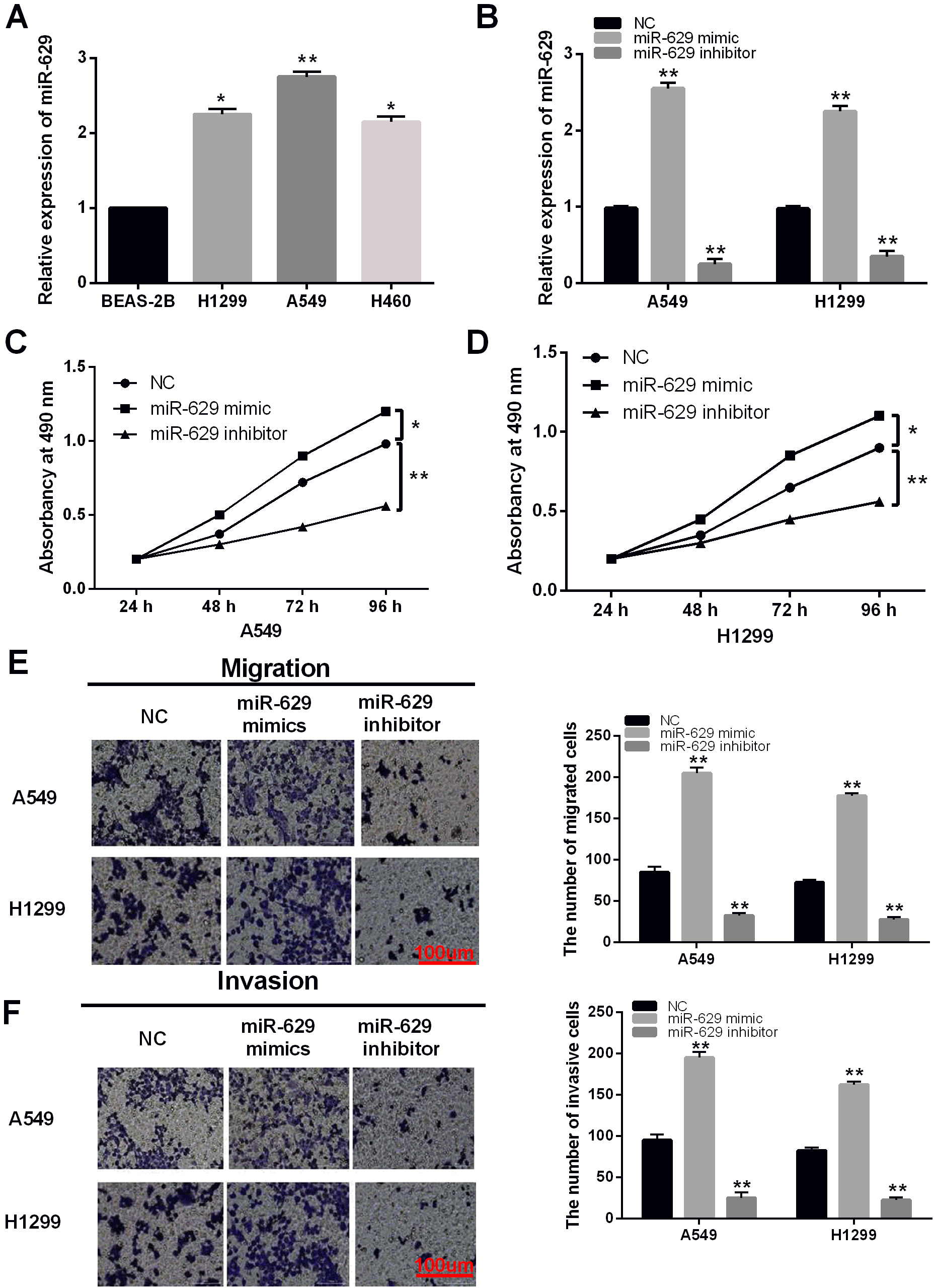
FOXO1 is a direct target of miR-629 in NSCLC. (A) FOXO1 has a binding site with miR-629. (B) Luciferase reporter assay. (C) MiR-629 was negatively correlated with FOXO1 expression in NSCLC tissues. (D) and (E) The expression of FOXO1 were observed in A549 and H1299 cells containing miR-629 mimics or inhibitor. 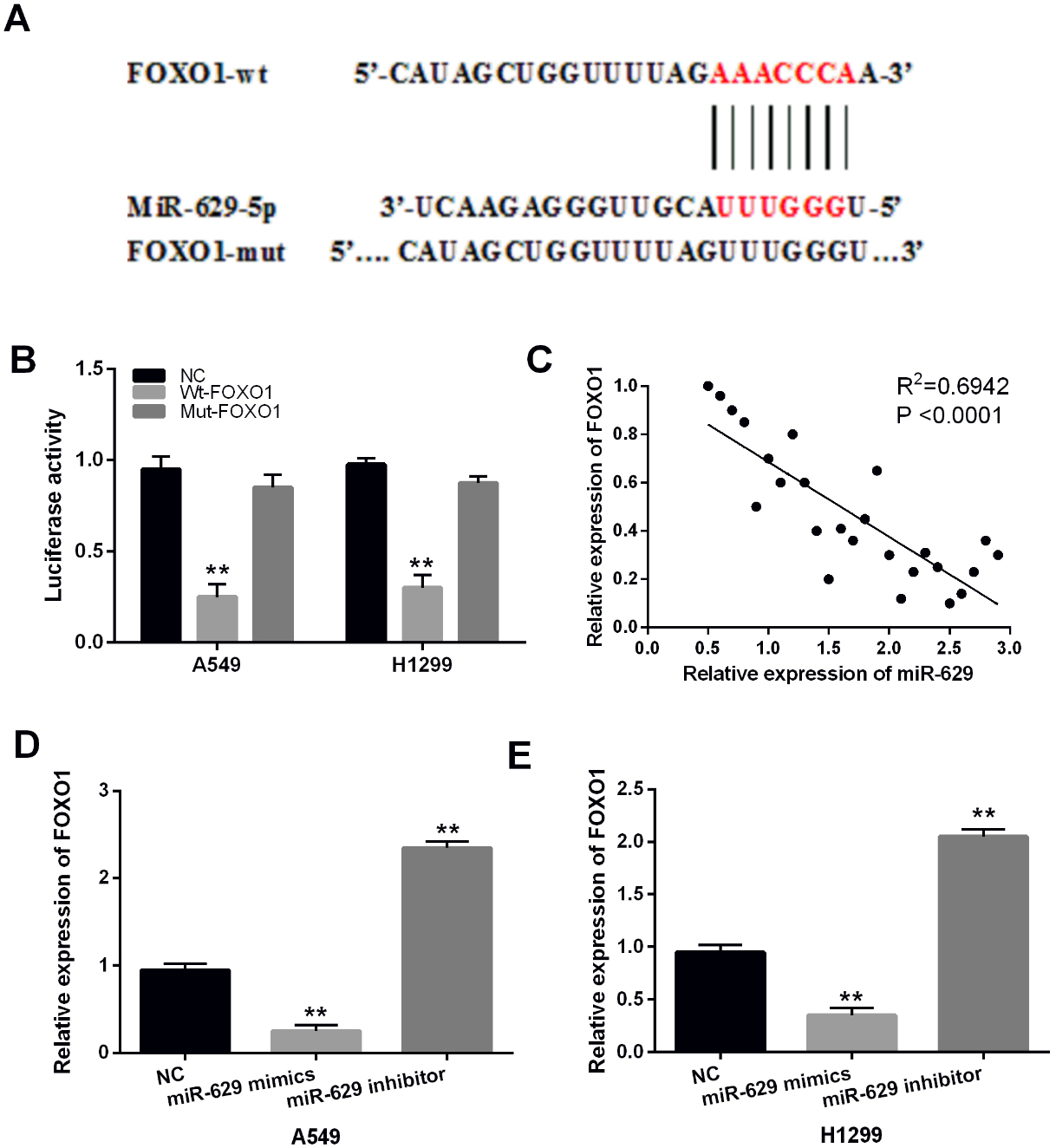
In addition, TargetScan database (
FOXO1 was downregulated in NSCLC
Next, the alternation of FOXO1 expression was examined in NSCLC tissues and cell lines. We found that FOXO1 was downregulated in NSCLC tissues compared to adjacent normal tissues (Fig. 4A). In addition, downregulation of FOXO1 was also detected in H1299, A549 and H460 cell lines compared to BEAS-2B cells (Fig. 4B). Besides that, low FOXO1 expression was found to be associated with shorter overall survival in patients with NSCLC (
FOXO1 was downregulated in NSCLC tissues and cells. (A) The expression of FOXO1 was measured in NSCLC tissues and normal tissues. (B) The expression of FOXO1 was observed in H1299, A549, H460 NSCLC cells and human normal bronchial epithelium cell line BEAS-2B. (C) High expression of FOXO1 was related to longer overall survival in NSCLC patients. 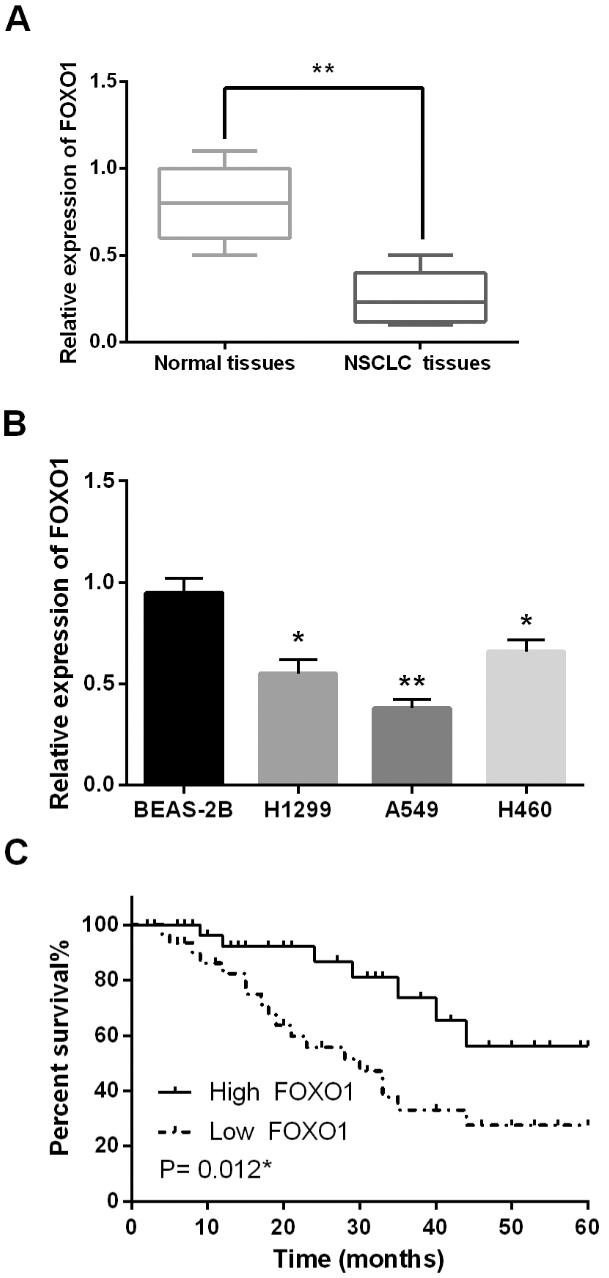
Then, the function of FOXO1 and its interaction with miR-629 in NSCLC were investigated in A549 and H1299 cells with FOXO1 siRNA or miR-629 mimics and FOXO1 vector. FOXO1 siRNA was found to reduce its expression in A549 and H1299 cells. In addition, we found that the decreased expression of FOXO1 induced by miR-629 mimics was restored by FOXO1 vector in A549 and H1299 cells (Fig. 5A and B). Functionally, knockdown of FOXO1 was found to promote cell proliferation in A549 and H1299 cells. Moreover, the promotion of cell proliferation mediated by miR-629 mimics was hampered by upregulation of FOXO1 in A549 and H1299 cells (Fig. 5C and D). In addition, knockdown of FOXO1 also promoted cell migration and invasion in A549 and H1299 cells. Upregulation of FOXO1 attenuated the promotion of cell migration and invasion induced by miR-629 mimics (Fig. 5E and F). In conclusion, FOXO1 functions as a tumor suppressor in NSCLC. And miR-629 promoted the development of NSCLC by downregulating FOXO1.
MiR-629 promoted the development of NSCLC through targeting FOXO1. (A) and (B) The expression of FOXO1 was measured in A549 and H1299 cells with FOXO1 siRNA, miR-629 mimics, or FOXO1 vector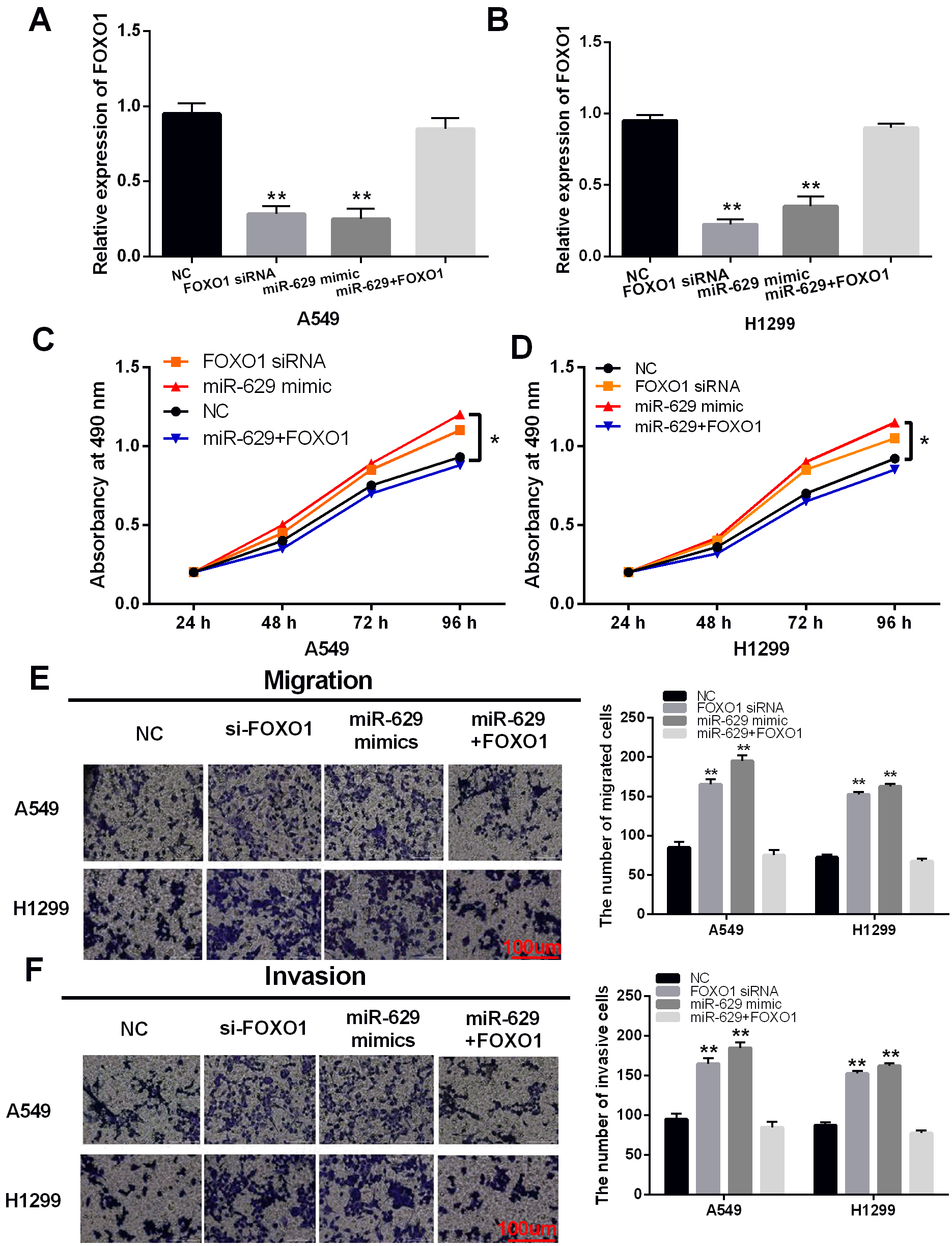
Finally, the effects of miR-629 and FOXO1 on EMT and PI3K/AKT pathway were investigated in NSCLC cells. We found that overexpression of miR-629 and knockdown of FOXO1 promoted the expression of N-cadherin and Vimentin and inhibited E-cadherin expression in A549 and H1299 cells (Fig. 6A). In contrast, downregulation of miR-629 and upregulation of FOXO1 had opposite effects on these markers (Fig. 6B). In addition, overexpression of miR-629 and knockdown of FOXO1 were found to promote p-AKT expression (Fig. 6C), while downregulation of miR-629 and upregulation of FOXO1 inhibited p-AKT expression in A549 and H1299 cells (Fig. 6D). However, the expression of AKT was not affected by miR-629 and FOXO1 in A549 and H1299 cells. These results imply that miR-629 and FOXO1 are involved in the EMT/PI3K/AKT pathway to regulate tumorigenesis of NSCLC.
MiR-629/FOXO1 was involved in EMT/PI3K/AKT pathway in NSCLC. (A) and (B) The protein expression of E-cadherin, N-cadherin, Vimentin, AKT and p-AKT was measured in A549 cells containing miR-629 mimics or inhibitor and FOXO1 siRNA or vector. (C) and (D) The protein expression of E-cadherin, N-cadherin, Vimentin, AKT and p-AKT in H1299 cells containing miR-629 mimics or inhibitor and FOXO1 siRNA or vector.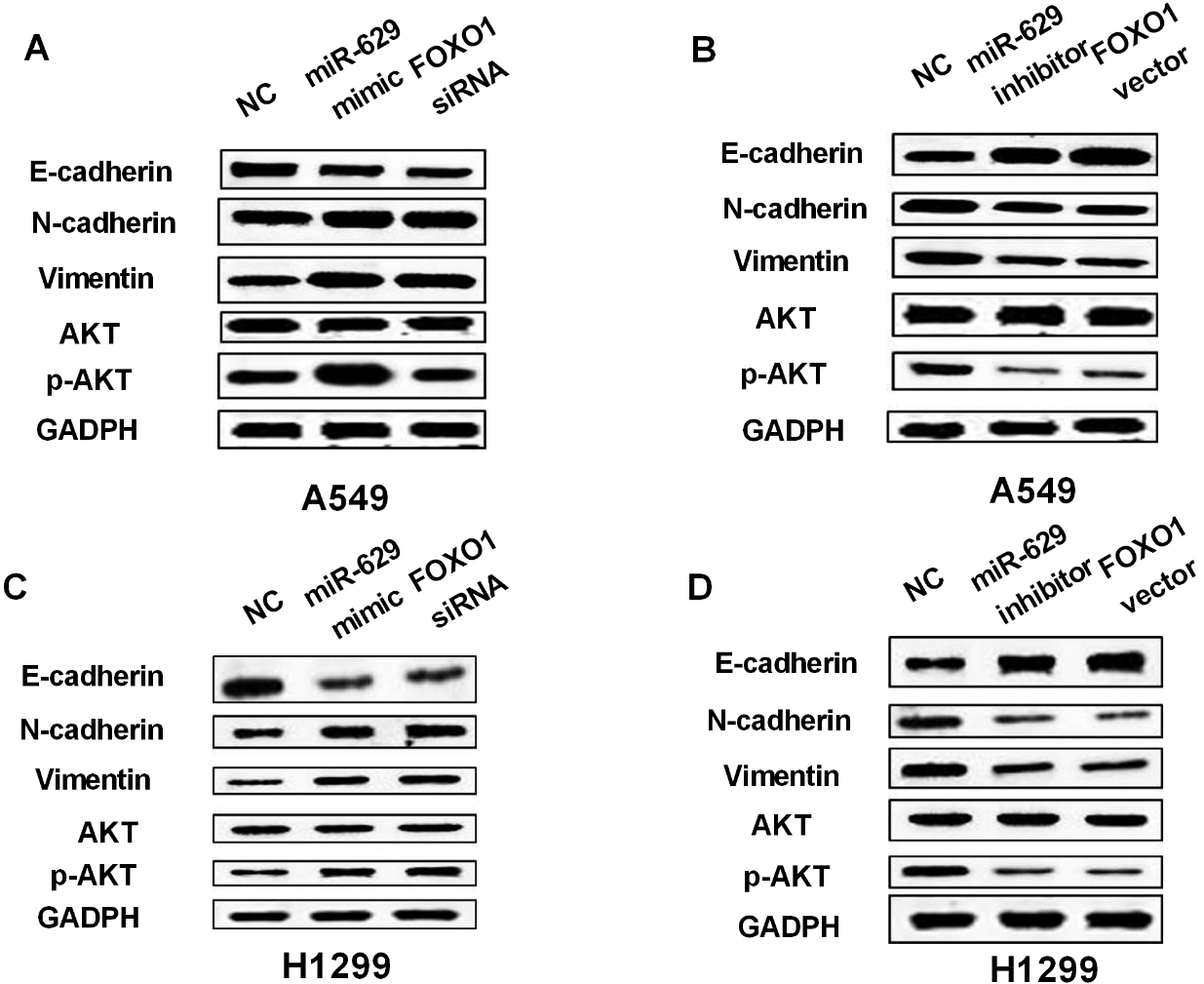
Recently, specific functions of miRNAs have been identified in the pathogenesis of human cancers. Previous studies have shown that multiple miRNAs are involved in the development of NSCLC, such as miR-150 [21], miR-195 [22] and miR-210 [23]. In this study, upregulation of miR-629 was identified in NSCLC and predicted poor prognosis in patients with NSCLC. Similarly, renal cell carcinoma patients with high miR-629 expression were also found to have poor prognosis [9]. In addition, we found that overexpression of miR-629 promoted cell proliferation, migration and invasion in NSCLC. These findings suggest that miR-629 serves as a tumor promoter in NSCLC.
In addition, miR-629 has been reported as a novel biomarker and potential therapeutic target for triple-negative breast cancer lung metastasis [24]. Consistent with our findings, upregulation of miR-629 has been reported to promote ovarian cancer cell migration and invasion and inhibited apoptosis [25]. It has been proposed that miR-629 promoted proliferation and metastasis of human pancreatic cancer cells by targeting FOXO3 [26]. In addition, we found that miR-629 promoted EMT in NSCLC, which has not been reported in previous studies. EMT is an important malignant behavior that affects the metastasis of NSCLC cells. For example, miR-186 has been shown to inhibit migration of NSCLC cells by affecting the EMT process [27]. It was also found that downregulation of miR-638 promoted cell invasion in NSCLC by inducing EMT [28]. However, the cell morphology induced by EMT has not been testified in this study due to the limitation of our lab conditions. In addition, previous studies have shown that miR-629 plays an important role in human cancers by regulating target genes. For example, miR-629 has been reported to promote the development of cervical cancer by targeting RSU1 [29]. Here, FOXO1 was confirmed as a direct target of miR-629 and was negatively correlated with miR-629 expression in NSCLC.
FOXO1, as a target gene, has been reported to be negatively correlated with several miRNAs, including miR-135a [30], miR-182 [31] and miR-222 [32]. In this study, downregulation of FOXO1 was found in NSCLC tissues and cells. Furthermore, knockdown of FOXO1 promoted the progression of NSCLC. In particular, miR-629 was found to promote the development of NSCLC by regulating FOXO1 expression. Similarly, miR-183 was found to promote tumor growth of NSCLC by inhibiting FOXO1 expression [33]. These findings confirm that FOXO1 acts as a tumor suppressor in the pathogenesis of NSCLC. Besides that, we found that miR-629/FOXO1 axis was involved in EMT/PI3K/AKT pathway in NSCLC. Moreover, TargetScan showed that AKT3, a member of PI3K/AKT pathway, is a target of miR-629. However, the effect of miR-629 on AKT3 has not been investigated in this study due to the absence of AKT3 antibody. In addition, we also found that the expression of total AKT was not affected by miR-629. The reason caused that result may be the unstable binding of AKT3 to miR-629. Similarly, miR-132 has been reported to play a carcinogenic role in laryngeal squamous cell carcinoma by targeting FOXO1 and activating the PI3K/AKT pathway [34]. This study also reveals that miR-629 functions as a tumor promoter in NSCLC by targeting FOXO1 and activating the PI3K/AKT pathway.
Conclusions
In conclusion, upregulation of miR-629 and downregulation of FOXO1 were detected in NSCLC tissues and cells. And upregulation of miR-629 and downregulation of FOXO1 predicted poor prognosis in patients with NSCLC. More importantly, miR-629 promotes the progression of NSCLC by targeting FOXO1 and activating the EMT/PI3K/AKT pathway. These findings will provide new avenues for the diagnosis and treatment of NSCLC.
Footnotes
Conflict of interest
The authors declared no conflict of interest.
