Abstract
BACKGROUND:
PPM1G, a member of the serine/threonine protease family, dephosphorylates various proteins and may be involved in cancer development. The role and mechanism of PPM1G in HCC still needs to be verified.
OBJECTIVE:
This study aims to explore the role of PPM1G in the occurrence, development and prognosis of HCC.
METHODS:
Using bioinformatics (UALCAN, cBioPortal, Linkedomics, STRING and GSEA) to analyze the expression of PPM1G mRNA in HCC, its clinical relevance and possible involved signaling pathways. The expression of PPM1G protein was determined by immunohistochemistry in 311 cases of HCC to evaluate the association between PPM1G and clinical features and prognosis.
RESULTS:
The expression of PPM1G was significantly upregulated in HCC (
CONCLUSION:
PPM1G showed an essential function involving in tumor-related pathways in HCC, providing a biological basis for targeted treatment of HCC clinically.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the fourth cause of cancer-related death worldwide [1], which has a high-grade malignancy with poor prognosis, and presents a serious health problem [2]. Therefore, it is necessary to identify biomarkers for early diagnosis and evaluation of prognosis in HCC. These biomarkers would help us to gain a better understanding of the pathogenesis of HCC and find potential therapeutic targets [3].
PPM1G belongs to the PPM family of proteins and is a type 2C phosphatase with a unique acidic domain. The phosphatase activity of human PPM1G depends on Mn
In light of the involvement of PPM1G in cell cycle arrest and DNA damage response, the possibility of PPM1G related to tumorigenesis was noticed. Xu et al. [9] demonstrated that activity of PPM1G is modulated by PI-3K/AKT signaling pathway, which shifts the balance between the hyper- and hypo-phosphorylated 4E-BP1 and regulates the expression of Id1 at the translational level, thus promoting the occurrence and development of glioma. Nevertheless, Phannasi et al. [10] found that PPM1G is upregulated during the suppression of Moringa oleifera pod (bMO) on azoxymethane (AOM)/dextran sodium sulfate (DSS)-induced mouse colon carcinogenesis, which suggested that PPM1G may act as a tumor suppressor. The above reports showed that PPM1G has tissue-specific function in carcinogenesis and development. However, the role of PPM1G in the tumorigenesis and development of HCC remains to be determined.
In the present study, we first used bioinformatic analysis to detect the mRNA level of PPM1G in HCC, and then performed immunohistochemistry to analyze the correlation between the expression of PPM1G and clinicopathological features by our cohort. Gene ontology (GO) enrichment analysis, the Kyoto Encyclopedia of Gene and Genome (KEGG) pathway analysis, and protein-protein interaction (PPI) network were used to explore the molecular mechanism of PPM1G in HCC.
Materials and methods
UALCAN
UALCAN (
cBioPortal
cBioPortal (
Linkedomics
The LinkedOmics (
STRING
In order to obtain the protein interaction network of PPM1G in human cells, the STRING (
mRNA level of PPM1G in liver hepatocellular carcinoma and its relationship with clinicopathological features (UALCAN). (A) Bar graph showing relative expression of PPM1G mRNA in normal tissue and HCC samples. (B) Bar graph showing relative expression of PPM1G mRNA in normal tissue or HCC samples corresponding to tumor grade 1, 2, 3, and 4 (1 represents highly differentiated, 2 represents moderately differentiated, 3 represents poorly differentiated, 4 represents undifferentiated). (C) Bar graph showing relative expression of PPM1G mRNA in normal tissue and HCC samples corresponding to tumor TNM (T: Tumor; N: Lymph node metastasis; M: Distant metastasis) stage 1, 2, 3, and 4. (D) Bar graph showing relative expression of PPM1G mRNA in normal tissue or HCC samples with metastasis. 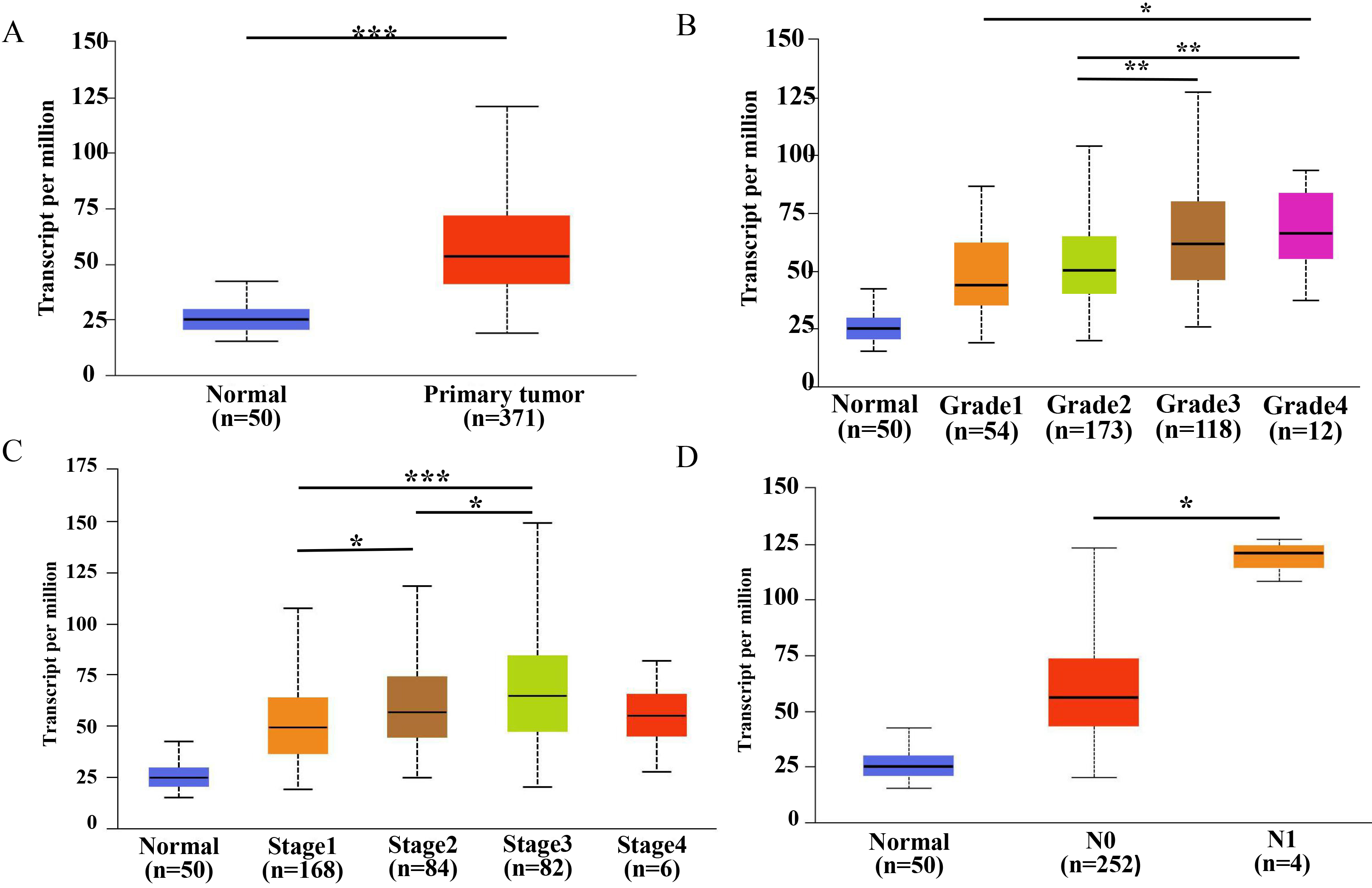
The gene expression matrix of 374 HCC patients downloaded from TCGA (
Immunohistochemistry staining
The tissue microassay (TMA) was purchased from Shanghai Biochip Co., Ltd. The TMA included 311 samples of cancer tissues obtained from HCC patients between 2008 and 2015 and 176 corresponding normal adjacent tissues. Paraffin slices were placed in an oven at 60–65
Immunohistochemistry staining was evaluated according to previous study [11]. The staining score was based on four intensity grades (0: negative, 1: weakly positive, 2: moderately positive, 3: strongly positive) and the fraction of positive cells (0:
Expression of PPM1G in hepatocellular carcinoma tissues (
311)
Expression of PPM1G in hepatocellular carcinoma tissues (
SPSS 26.0 statistical software was used for data analysis. Categorical variables were represented as frequency, and the relationship between PPM1G expression and clinicopathological features of HCC was determined using the
Results
PPM1G mRNA in HCC and normal tissue
Based on the data in UALCAN, the level of PPM1G mRNA was significantly higher in HCC than in normal liver tissue (
Univariate and multivariate Cox regression survival analysis of clinicopathological parameters and PPM1G expression in hepatocellular carcinoma patients
Univariate and multivariate Cox regression survival analysis of clinicopathological parameters and PPM1G expression in hepatocellular carcinoma patients
To further verify the expression of PPM1G, we performed IHC staining of TMA containing 311 HCC samples, and found that PPM1G was mainly localized in the nuclei (Fig. 2). Consistent with the results obtained by UALCAN data, PPM1G was upregulated in HCC tissues. The high expression of PPM1G in HCC was found in 64.63% (201/311) of cases, which was significantly higher than that in adjacent noncancerous tissue (25.57%, 45/176;
Representative cases of high and low PPM1G expression detected by immunohistochemistry. (A) Cancer tissue, metastasis, high expression. (B) Cancer tissue, no metastasis, low expression. (C) Paracancerous tissue, low expression. Yellow or brown represents the positive staining of PPM1G; blue refers to the nucleus stained by hematoxylin.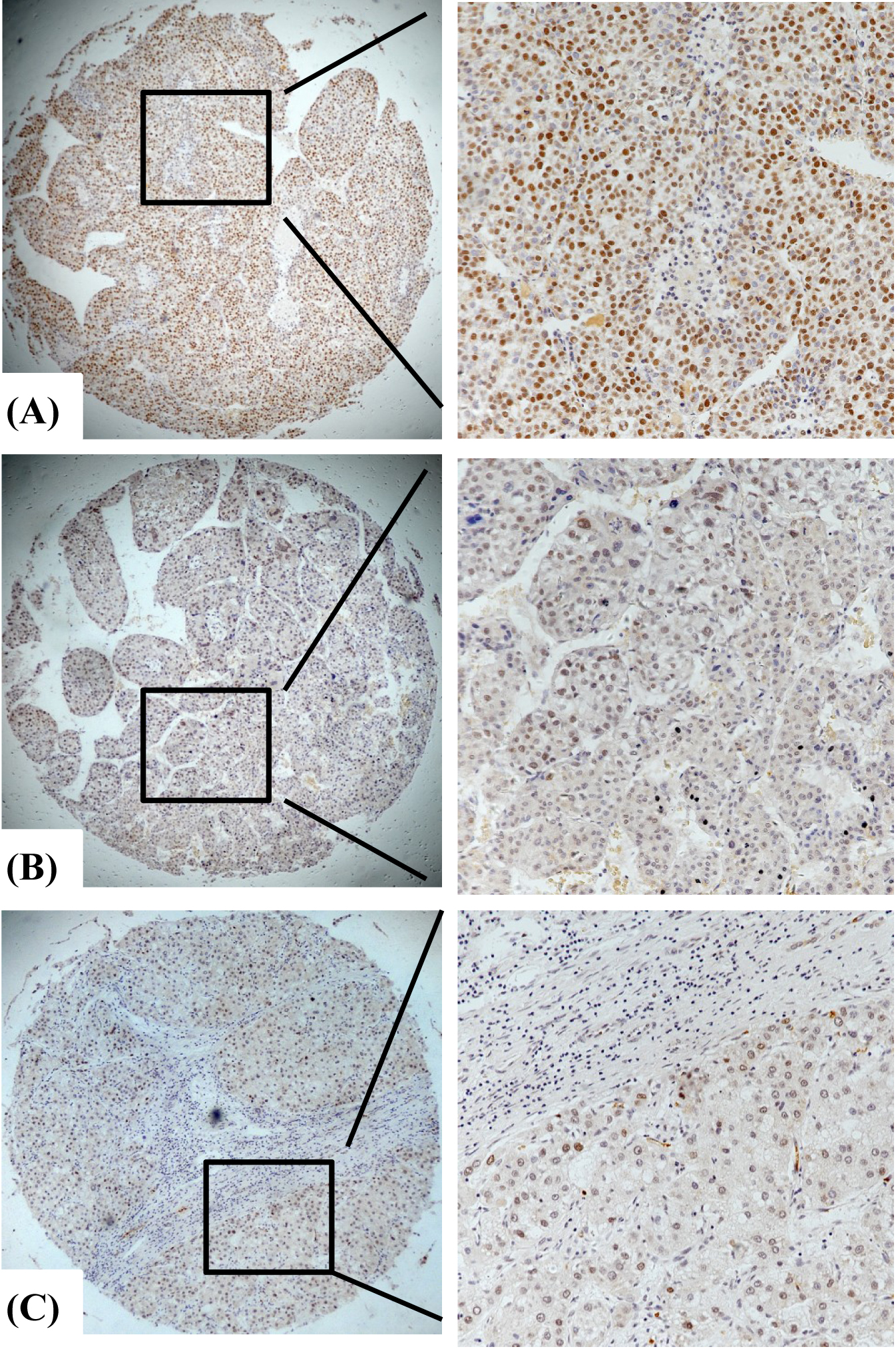
The survival curves of HCC patients with different levels of PPM1G expression based on databases and immunohistochemistry. (A) UALCAN, (B) GEPIA, (C) Kmplot, (D) immunohistochemistry.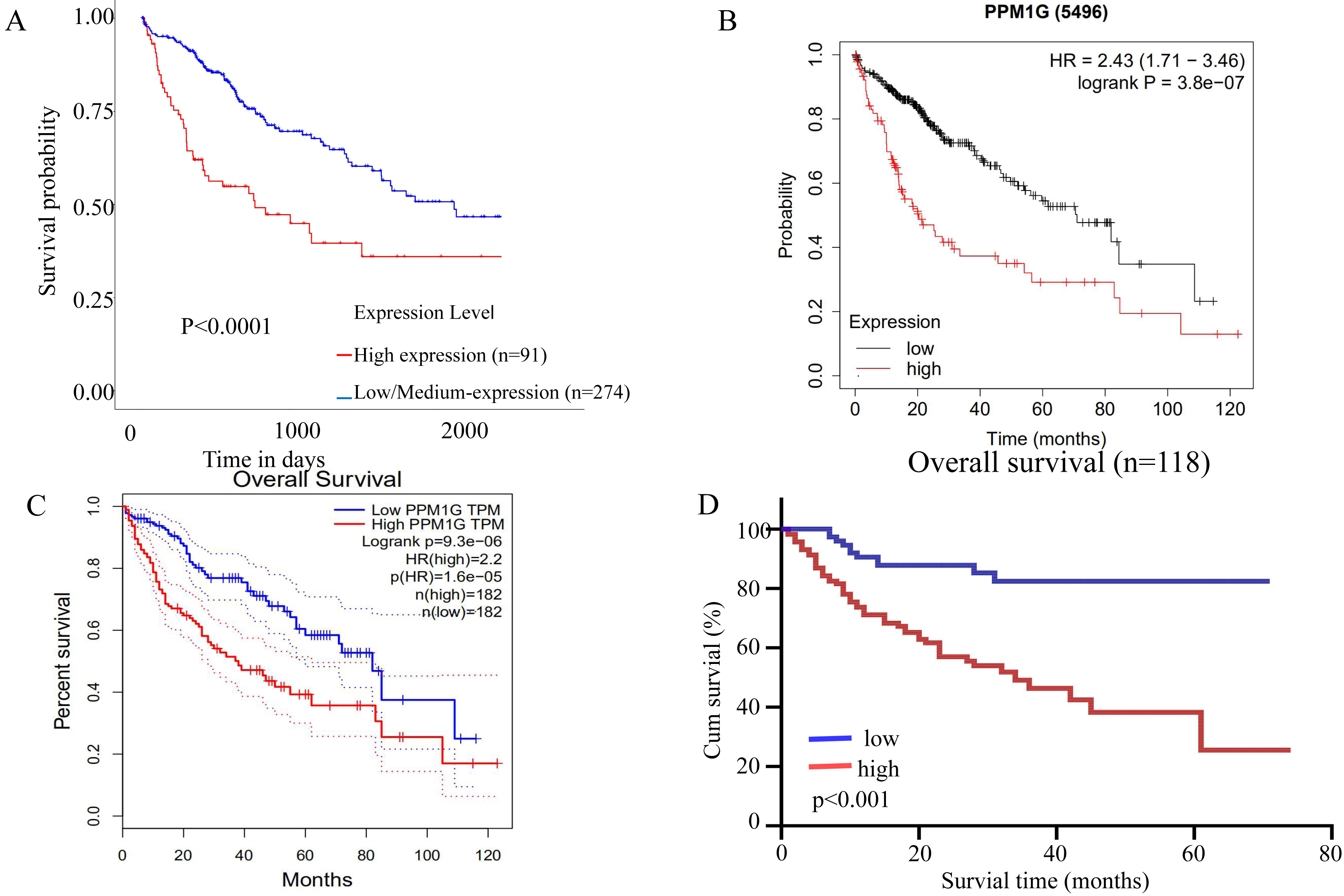
We selected several databases to analyze the impact of the mRNA level of the PPM1G gene on the survival of HCC patients. The results showed that the overall survival of patients with low mRNA level of PPM1G was significantly longer than those with high mRNA level of PPM1G (
Survival analysis performed by the Kaplan-Meier method using our present 311 cases of HCC patients also documented that the survival time (38
Cox univariate regression analysis showed that the protein level of PPM1G (
Analysis of genetic mutations and DNA methylation level in PPM1G (A) Genetic mutations of PPM1G in HCC (cBioPortal). (B) Patients with PPM1G mutations have poor survival (cBioPortal). (C) The methylation level of PPM1G promoter in HCC (UALCAN). (D) Correlation between the expression level of PPM1G mRNA and methylation level of PPM1G promoter (cBioPortal).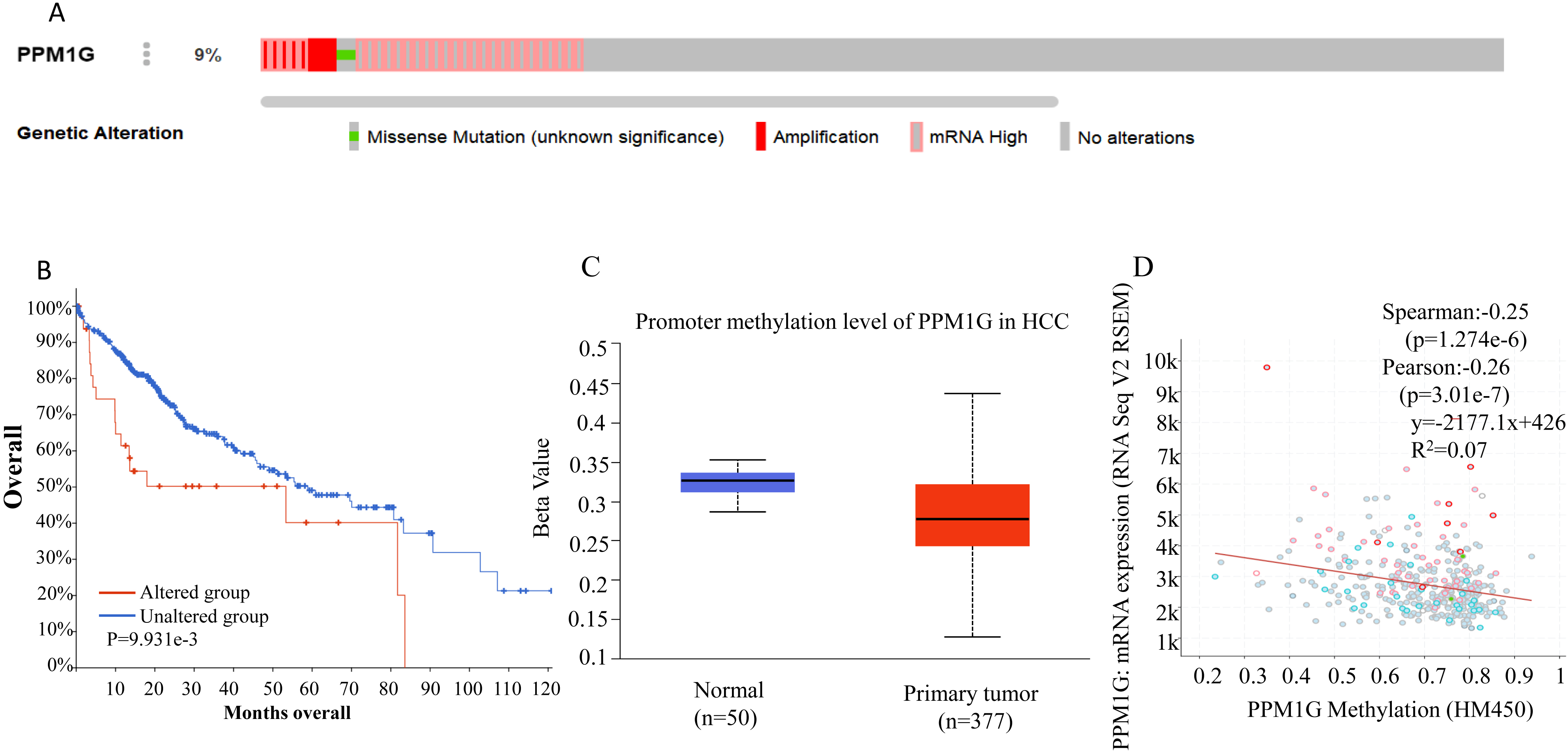
Correlation between differentially expressed genes and PPM1G level in HCC (LinkedOmics). (A) The Pearson’s test was used to analyze correlations between the level of PPM1G and genes differentially expressed in HCC. (B) and (C) Heat maps showing the top 50 genes negatively and positively correlated with PPM1G in HCC.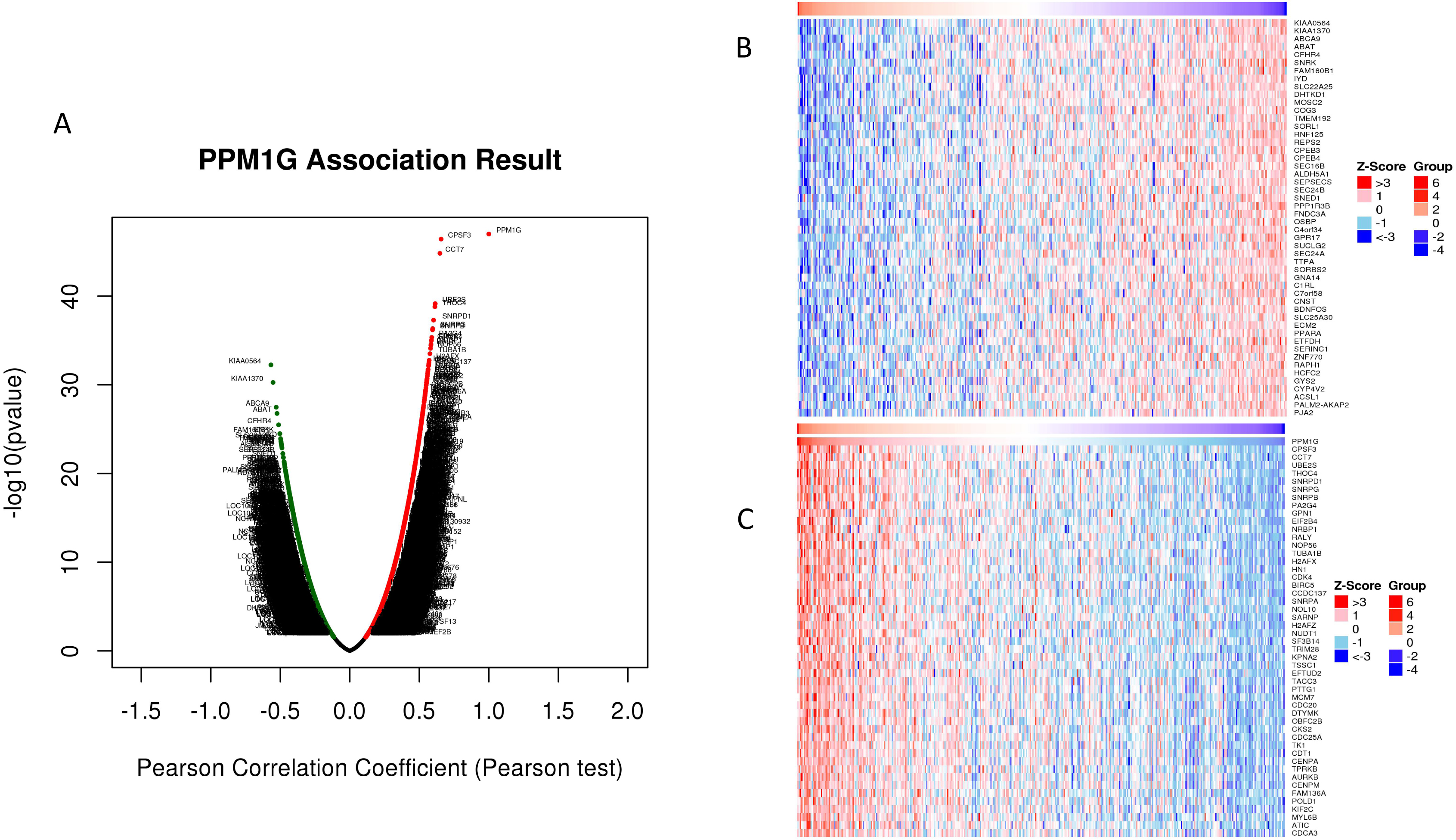
The PPM1G gene was altered in 9% (34/372) HCC patients (Fig. 4A). The most frequent alteration type was amplification, identified in 6 cases (2%). Importantly, patients with genetic alterations of PPM1G had lower overall survival than those without alterations (Fig. 4B).
Methylation within the promoter region of the PPM1G gene was significantly lower in HCC than in normal liver tissue (
Identification and functional analysis of genes co-expressed with PPM1G in HCC
Genes co-expressed with PPM1G were analyzed by LinkedOmics using mRNA sequencing data of 371 HCC patients from the TCGA. All genes associated with PPM1G are shown by the volcano plot in Fig. 5A, while Fig. 5B and C depict 50 significant genes that are positively or negatively correlated with PPM1G.
Co-expressed genes (correlation coefficient
Significantly enriched GO annotations and KEGG pathways of PPM1G in HCC. (A) Cellular component, (B) Molecular function, (C) Biological process, (D) KEGG enrichment.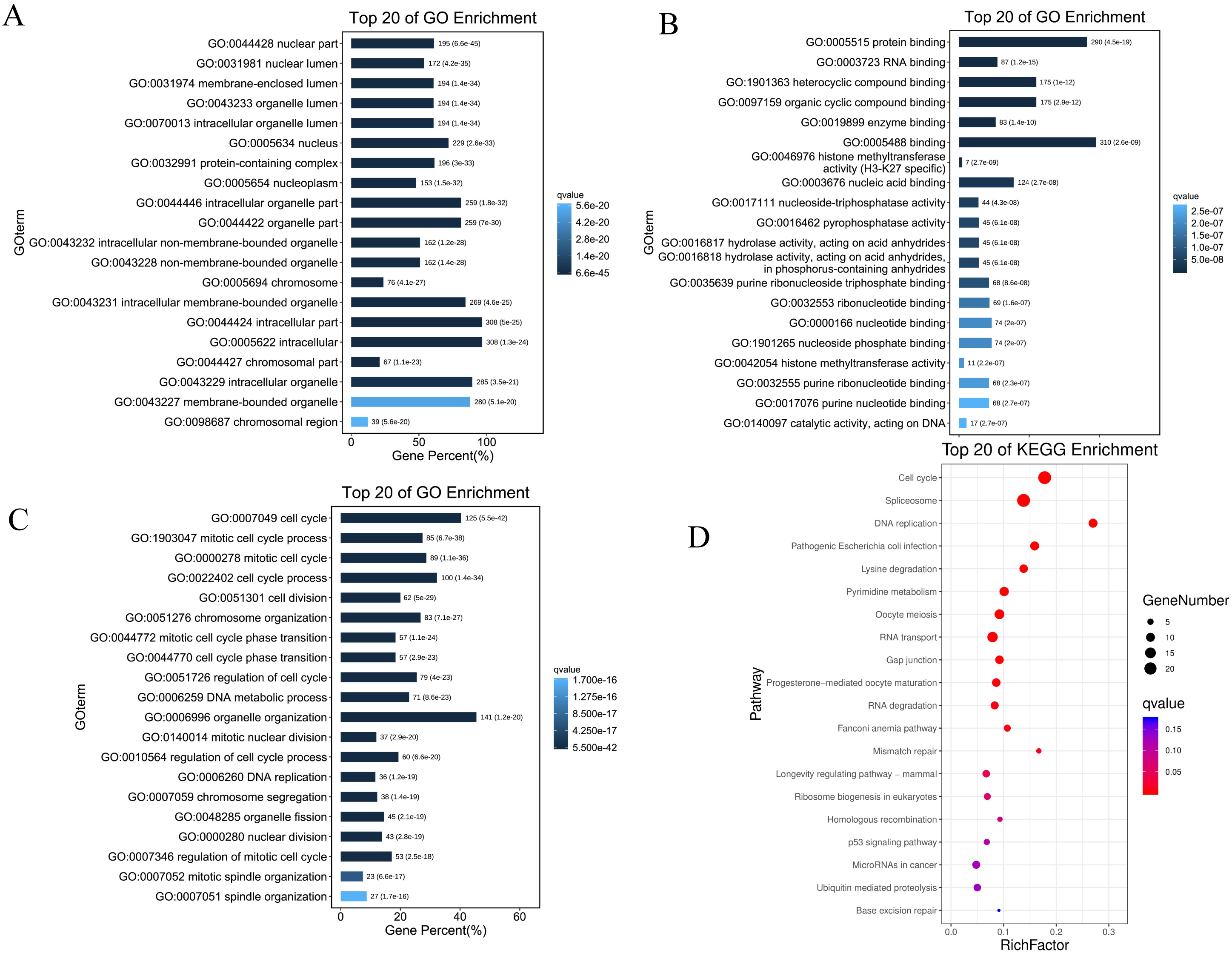
Ten of these pathways were selected based on FDR
GSEA was used to analyze the pathways promoted by the high expression of PPM1G in HCC.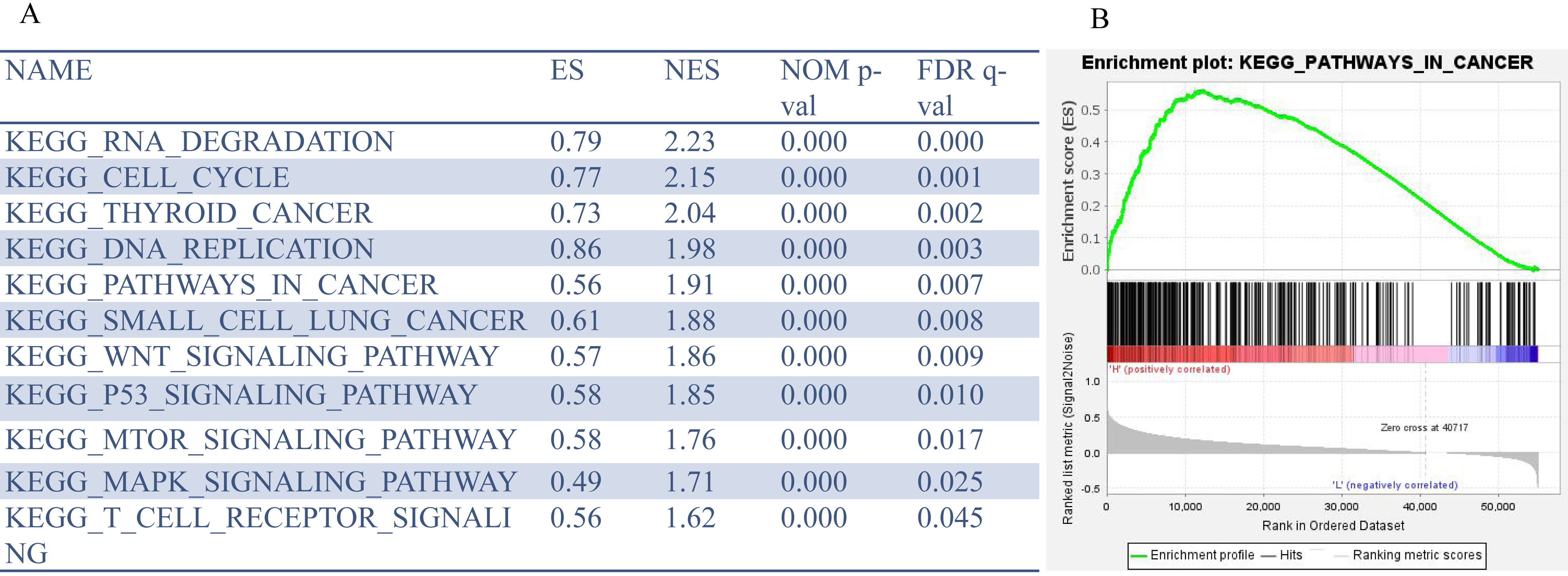
The protein interaction network of PPM1G. (A) The protein-protein interaction of PPM1G in human cells. (B) Pearson’s correlation analysis between PPM1G and USP7. (C) The network diagram of the protein interaction of hub genes. The genes in the red, orange, and yellow circles are the hub genes, while the blue circle indicates other genes associated with the hub genes.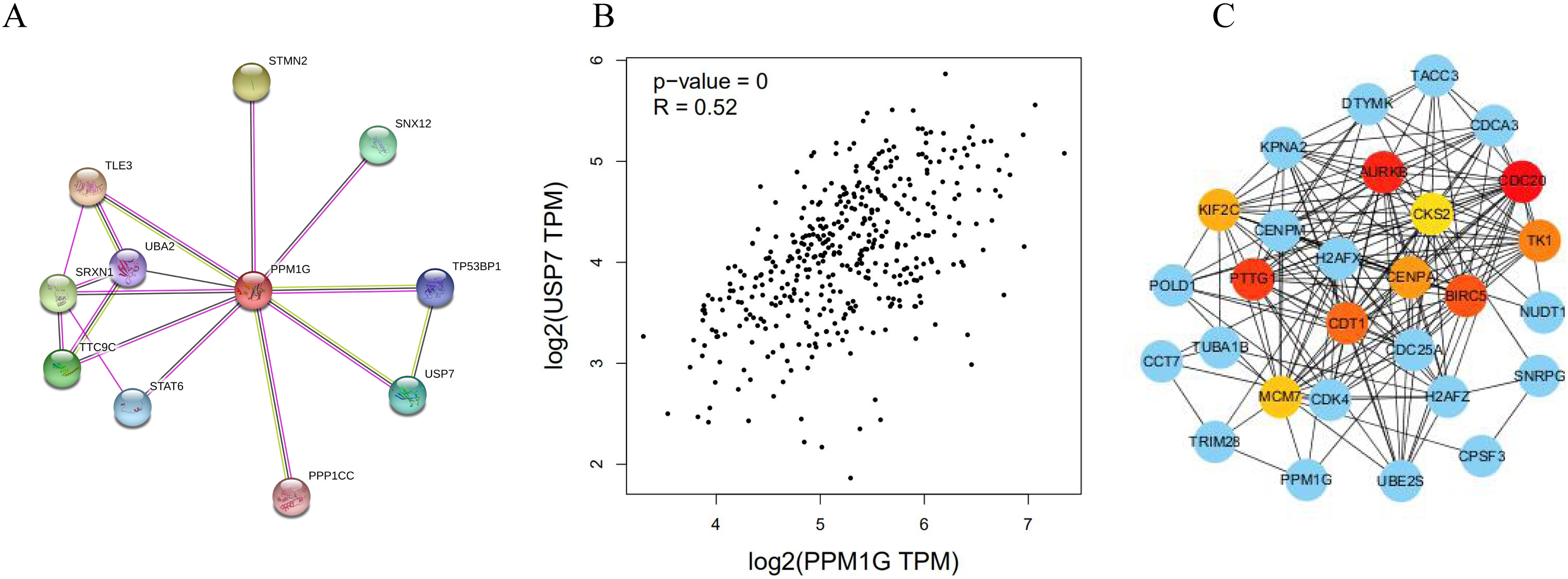
The protein network interacting with PPM1G analyzed by STRING showed that USP7 was one of the interacted proteins with PPM1G (Fig. 8A). So we analyzed the correlation between USP7 and PPM1G by GEPIA in HCC, and we found that USP7 correlated with PPM1G significantly (
Phosphorylation and dephosphorylation are key processes involved in the regulation of protein expression, which may play as an on-off role in cellular biological functions. Most studies on PPM1G (PP2C
Next, immunohistochemistry was employed to determin the protein level of PPM1G using TMA containing 311 HCC samples and 176 adjacent noncancerous samples. The results demonstrated that PPM1G protein level was upregulated in HCC, and its high expression was positively correlated with tumor grade, metastasis, HBV infection and vascular invasion. PP2C isoforms have been implicated in signaling networks controlling cell differentiation, proliferation, growth, survival, and metabolism [12]. Previous studies [13] have also demonstrated that PPM1G has anti-apoptotic activity during neural development in mice. Once stimulated with the pro-inflammatory cytokine tumor necrosis factor-
Recent study [17] has also found PPM1G showed good performance in predicting overall survival for HCC patients by internal and external validation. Consistent with the previous study, we also showed that the high expression of PPM1G was positively correlated with a poor prognosis of HCC patients and was an independent prognostic factor by Cox multivariate regression analysis. Thus, PPM1G may associate with the occurrence, development, and prognosis of HCC.
Study found that these point mutants of PPM1G showed reduced binding to
In summary, high expression of PPM1G is associated with a worse pathological stage of HCC, which might play an important role in the progression of HCC. Additionally, PPM1G may be used as prognostic and survival indicator. However, role and molecular mechanism of PPM1G still needs to be further studied by experiments in vitro and in vivo.
Authors’ contributions
Conception: XMF.
Interpretation or analysis of data: QL.
Preparation of the manuscript: DLX.
Revision for important intellectual content: HW and WLJ.
Supervision: YYM.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-203248.
sj-xlsx-1-cbm-10.3233_CBM-203248.xlsx - Supplemental material
Supplemental material, sj-xlsx-1-cbm-10.3233_CBM-203248.xlsx
Footnotes
Acknowledgments
We thank the Department of Key Laboratory of Gastroenterology of Zhejiang Province for helping us to use the equipment. This work was supported by the project of science and technology of Zhejiang province (grant no. GF19H160085).
