Abstract
BACKGROUND:
Esophageal squamous cell carcinoma (ESCC) has poor prognosis mainly due to the difficulty of making early diagnosis. Therefore, novel biomarkers are critically needed.
OBJECTIVE:
We aimed to investigate the diagnostic value of serum interleukin-8 (IL-8) in ESCC.
METHODS:
Data mining of TCGA was used to analyze expression level of IL-8 mRNA in esophageal carcinoma. Serum levels of IL-8 were measured in 103 ESCC patients and 86 normal controls by ELISA. Receiver operating characteristic (ROC) curve was used to evaluate its diagnostic accuracy.
RESULTS:
IL-8 mRNA expression level and serum IL-8 concentration were both statistically higher in patients than normal controls (
CONCLUSIONS:
Serum IL-8 was highly expressed in ESCC patients and may be a potential marker for early diagnosis of ESCC.
Background
Esophageal cancer (EC) is the eighth common malignant disease worldwide, and the incidence of which in China ranks the fifth in the world [1, 2]. As one of the subtypes of EC, esophageal squamous cell carcinoma (ESCC), accounts for more than 80% of the world’s total EC death [3]. The 5-year survival rate of early EC is up to 90%, while the 5-year survival rate of advanced EC after surgical treatment is less than 20% [4]. However, due to the lack of typical symptoms, it is hard for patients to be diagnosed as ESCC in the early stage [5]. Most of them have progressed to advanced stage when diagnosed [6]. Therefore, it is of certain significance to perform ESCC screening, especially in high incidence areas.
Endoscopy combined with histopathological examination is the gold standard for the diagnosis of ESCC [7]. However, in addition to technical limitations, its efficacy is affected by other factors, for example, the compliance of population [8]. The relevant cost-benefit model also demonstrated that the disadvantages of conducting endoscopic screening and monitoring outweighed its advantages [9, 10]. This remains a substantial challenge even in a high-resource environment and is not tenable, even in the regions of high incidence [11]. Thus, it is important to establish some additional biological methods for early warning and early diagnosis in high-risk population and to narrow the scope of using endoscopic examination.
Detection of serum tumor markers is inexpensive and could be an accurate supplementary for the early diagnosis of some cancers [12]. As reported, the area under the receiver operating characteristic (ROC) curve (AUC) of insulin-like growth factor-binding protein 3 is 0.826 with a sensitivity of 70% and a specificity of 85.5% in colorectal cancer [13]. In breast cancer, the AUC of the combination of trefoil factor 1, trefoil factor 2 and trefoil factor 3 is up to 0.96 [14]. However, in ESCC, there is still no specific and effective marker. Nowadays, serum markers used for the diagnosis of ESCC clinically include squamous cell cancer antigen (SCC-Ag), carcinoembryonic antigen (CEA), and cytokeratin fragment antigen 21-1, but they are not completely satisfactory in the early diagnosis of ESCC due to their low diagnostic sensitivity [4, 15, 16]. Consequently, novel biomarkers are critically needed to improve the diagnosis of patients with ESCC.
To address this issue, in our previous research, cytokines antibody array was used to screen candidate biomarkers. The detection results demonstrated that serum interleukin-8 (IL-8) was highly expressed in 20 ESCC patients when compared to 20 normal controls [17]. In recent years, inflammation has been accepted as a hallmark in cancer progression, and as an inflammation factor, the role of IL-8 in tumors was gradually revealed. IL-8 is overexpressed in many cancers and is correlated with tumor progression. In ESCC, IL-8 expression has been validated to impair the function of natural killer cells and be positively correlated with tumor progression [18]. However, to our knowledge, few studies evaluated the efficacy of serum IL-8 in the early diagnosis of ESCC. Publications reported by Krzystek-Korpacka et al. and Tong et al., respectively, demonstrated elevated IL-8 levels in ESCC patients [19, 20]. These studies indicate that IL-8 has the potential to be used as a biomarker for diagnosis of ESCC. This study is the continuation of our previous research to further assess the diagnostic efficiency of IL-8 in ESCC.
Method
Samples
A retrospective study was performed on 103 ESCC and 86 normal samples collected from the Cancer Hospital of Shantou University Medical College and Sun Yat-Sen University Cancer Center between January 2017 and January 2019. ESCC serum samples were collected from the patients newly diagnosed as ESCC. None of the patients had received any cancer-related treatment before. The normal controls were collected from physical examination centers in the same hospitals without any cancer-detected evidence. Fasting venous blood was collected from each subject and was allowed to clot at room temperature for 30 minutes. Serum was separated after centrifugation at 2,500 g for 10 minutes, and then stored at
All of the patients in this study were histopathologically diagnosed and staged according to the Eighth Edition of the American Joint Committee on Cancer (AJCC) Cancer Staging Manual. AJCC TNM stage 0
Expression of IL-8 mRNA in esophageal carcinoma and normal samples. (a) Relative expression of IL-8 mRNA in normal and esophageal carcinoma samples. (b) Expression levels of IL-8 mRNA in normal and different stages of esophageal carcinoma samples. The line in the box is the median. The degree of dispersion was shown by interquartile ranges. The Mann-Whitney 
In our previous study, sera of 20 ESCC patients and 20 normal controls were sent to RayBiotech Company for antibody array detection. Briefly, the serum samples were dialyzed and then biotin labeled. Following secondary dialysis, the chip was blocked and incubated with biotin-conjugated anti-cytokines for sample hybridization. Finally, streptavidin-conjugated fluorescent dye (Cy3 equivalent) was added. The laser fluorescence scanning (GenePix 4000B scanner) recorded the signals, and these signals were converted and presented by RayBio
Data mining using UALCAN and UCSC Xena for cancer genomics
The Cancer Genome Atlas (TCGA) is an international database containing a large collection of genomics data from multiple different technical platforms, providing an opportunity for data exploration and analysis. UALCAN (
Enzyme-linked immunosorbent assay (ELISA) for serum IL-8 level detection
Serum level of IL-8 was detected in the light of double-antibody sandwich ELISA kit (ELH-IL8, cat. No. 0802190143, RayBiotech Inc, Georgia, USA). The concentrations of IL-8 standards were diluted into 0.0, 0.8, 2.5, 7.4, 22.2, 66.7, 200.0, 600.0 pg/ml for tracing a standard curve. A preliminary experiment was carried out before the formal experiment, and a dilution ratio of 1:3 for serum was determined to be the preferable ratio. After preparation, 100
Statistical analysis
Microsoft Office 365, SPSS 24.0, Sigma Plot 10.0, GraphPad Prism 8.0 and R programming language 3.6.1 were utilized for data analysis. The Mann-Whitney
Cytokines antibody chip analysis results. Volcano plots of cytokines antibody expression in ESCC and normal controls from the Human Cytokine Array AAH-CYT-G4000. X-axis: log2 ratio of protein expression levels (fold change) between normal controls and ESCC. Y-axis: the 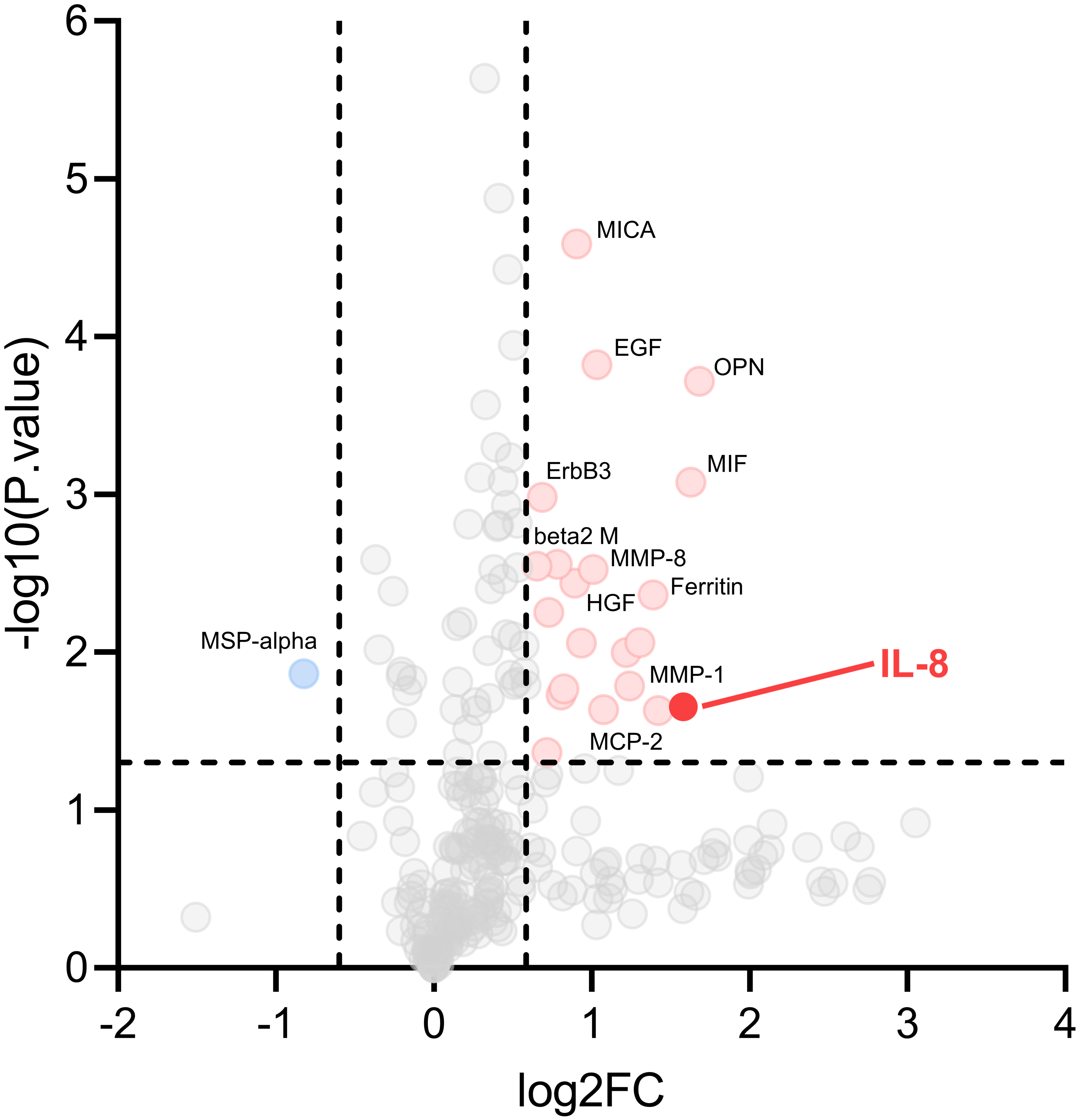
IL-8 gene expression in public available datasets
According to UALCAN, the result of IL-8 gene expression analysis provided information on the relative expression levels of IL-8 mRNA in normal versus cancer samples (11 and 184 samples, respectively). According to the box plot, IL-8 mRNA was expressed higher in esophageal carcinoma samples than that in normal samples (
Participant information and clinical characteristics
Participant information and clinical characteristics
ESCC, esophageal squamous cell carcinoma.
A high-throughput proteomics analysis of ESCC was performed in our previous research to identify potential serum diagnostic biomarkers. According to a standard of
IL-8 concentrations in ESCC patients and normal controls. (a) The diagram showed distribution of serum IL-8 concentration in ESCC patients and normal controls. The lowest concentration was 4.771 pg/mL in normal controls and the highest one was 2650.343 pg/mL in ESCC. Diagrams of ESCC are in yellow and normal controls are in blue. ESCC accounts for more histogram volume on higher concentration compared to normal controls. (b) Box plot illustrated median levels and interquartile ranges in four groups. (c) In scatter plot, the line in the dots is mean with SD. The Mann-Whitney 
ROC curve analysis in the diagnosis of ESCC and early-stage ESCC. The performance of IL-8 in ESCC was showed in ROC curves. ROC curve for two groups versus normal controls group were drawn in different colors. The area under the black line is 0.5, for reference. ROC curve, Receiver operating characteristic curve. ESCC, esophageal squamous cell cancer.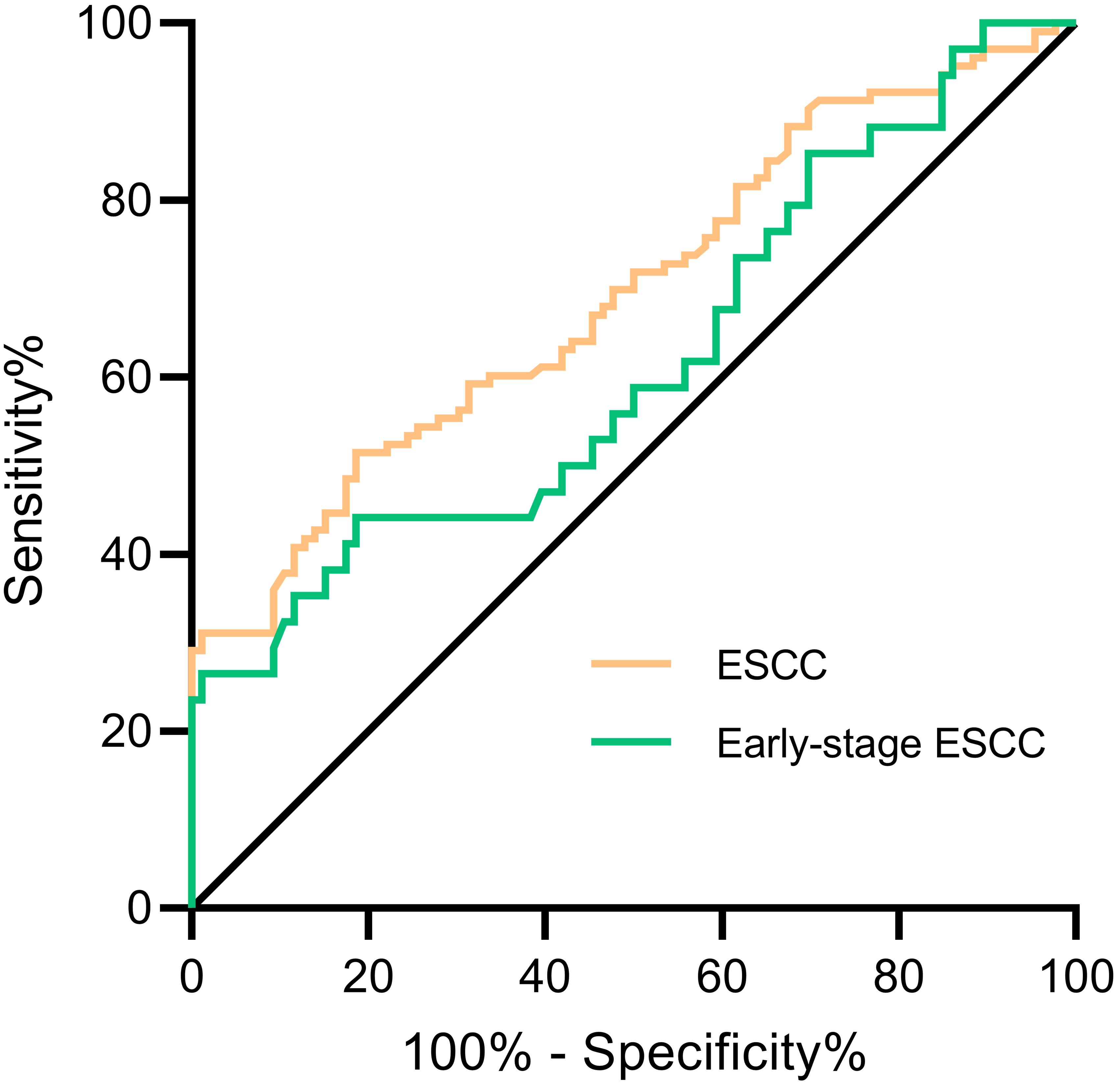
The detail of the recruited participants applied to detect IL-8 serum level was shown in Table 1. Using ELISA, it was obviously shown that the distributions of ESCC group and normal controls were similar, while more IL-8 levels of ESCC group distributed at higher concentration when compared to the normal group (Fig. 3a). For better observation on distribution and degree of dispersion, the levels of serum IL-8 in different groups were shown in box plot and scatter plot (Fig. 3b and c). The median concentration of serum IL-8 in ESCC group was 66.545 pg/mL, and the median concentration of IL-8 in the control group was 25.324 pg/mL. The Mann-Whitney
The ROC curve was drawn according to IL-8 levels in ESCC group and normal control group (Fig. 4). According to the evaluation criteria mentioned in the method, 80.082 pg/mL was indicated as the optimal diagnostic critical value. As shown in Table 2, AUC was 0.694 (95% CI: 0.620–0.768) with specificity of 86.0% and sensitivity of 42.7%. Using the same cut-off, the detection of IL-8 in the early diagnosis of ESCC demonstrated an AUC of 0.618 (95% CI: 0.499–0.737), with sensitivity of 35.3% and specificity of 86.0%.
Evaluation efficiency of IL-8 in the diagnosis of ESCC
Evaluation efficiency of IL-8 in the diagnosis of ESCC
ESCC, esophageal squamous cell carcinoma; CI, exact confidence interval; NC, normal controls; NLR, negative likelihood ratio; NPV, negative predictive value; PLR, positive likelihood ratio; PPV, positive predictive value.
Correlation between IL-8 and clinical indicators in ESCC patients
Kaplan-Meier analysis of the overall survival for IL-8 in esophageal cancer. (a) Patients were divided into two groups according to their median expression level of IL-8 mRNA. The line in red and the line in blue represent high IL-8 expression and low IL-8 expression, respectively. The 
Considering that the incidence of ESCC in different regions would affect their prediction probability, the standardized predicted values should be calculated according to the prevalence rates of 25%, 50% and 75% respectively. At this point, the corresponding positive predictive values of ESCC were 50.6% (95% CI: 0.397–0.614), 75.9% (95% CI: 0.625–0.857), and 89.8% (95% CI: 0.770–0.962) [25]. A standardized predicted value of 89.8% suggested IL-8 had the potential for helping screening ESCC in high incidence areas.
The correlation between IL-8 level and clinical indicators in ESCC patients was assessed and showed in Table 3. The positive rate of serum IL-8 was statistically associated with gender and the degree of differentiation (
Correlation between IL-8 and prognosis
IL-8 expression level and prognostic data of patients with esophageal cancer were obtained from TCGA database. Patients were divided into two groups according to the median expression level of IL-8 mRNA. Kaplan-Meier analysis and log-rank test revealed that there was no statistically significant difference of overall survival time between patients with high IL-8 mRNA expression and low IL-8 mRNA expression (
Discussion
ESCC is a malignant tumor with a low 5-year survival rate for its low detection rate in the early stage. Early screening and treatment are the most feasible means to reduce mortality and improve prognosis. Since there is still a lack of recognized serum tumor markers in ESCC, additional serum markers must be established. Detection of biomarker candidates for early diagnosis has been identified in the field of cancer proteomics study [26]. Antibody array, as a dominant kind of affinity proteomics, is utilized to detect target proteins in a high-throughput platform. Its sensitivity and specificity have been tremendously improved in the past decade with the development of many related technologies [27]. Now it could be helpful in cytokine profiling and provide unique opportunities for biomarker discovery [28].
Through high-throughput proteomics technologies in our previous study, IL-8 was indicated to be a possible detection marker [17]. Data mining and analysis also suggested the importance of IL-8 mRNA in esophageal cancer. Although there was no exact evidence showing the responsible cells for the secretion of IL-8 in ESCC patients, some research indicated that cancer cells might be the main source of IL-8 [29, 30]. To our knowledge, high expression of IL-8 in ESCC tissues has been confirmed by immunohistochemistry before. Reported by Wu et al., the number of IL-8 positive tumor cells was significantly higher in tumor tissues of patients with ESCC when compared with matched adjacent tissues [18]. Moreover, Li et al. also reported significantly higher expression of IL-8 protein in ESCC tissues compared to matched marginal tissues [31]. Actually, IL-8 is secreted by all type of nucleated cells within the body [32]. As reported by Boehlen and Clemetson, IL-8 was mainly produced by monocytes but also by many other cells such as T lymphocytes, neutrophils and platelets [33]. Indeed, high expression level of IL-8 derived from tumor-associated macrophages has been confirmed and reported to promote the migration and invasion of ESCC cell lines in vitro [34]. IL-8 was suggested to function as a significant regulatory factor within the tumor microenvironment for its increased expression in cancer cells and many other cells [35].
In this article, serum IL-8 was further studied to identify if it could serve as an indicator for a better early diagnosis. According to the detection results, serum levels of IL-8 were significantly higher in both ESCC group and early-stage ESCC group than the normal controls. Compared to classical tumor markers, the AUC for IL-8 was similar to AUC for CEA and higher than that for SCC-Ag [36]. It seems that it was not accurate enough for IL-8 to be used as an early diagnostic marker independently due to the insufficient sensitivity, while its specificity indicated that it could be valuable to be considered for the formation of multi-marker panels. The fact that the positive rate of serum IL-8 was correlated with gender, could be explained by the different incidence between males and females [3]. Except for gender, the positive rate was also correlated to the depth of tumor invasion and the degree of differentiation, indicating the relationship between IL-8 and tumor cells’ motility and invasiveness. As we know, smoking and drinking can cause chronic inflammation and are high risk factors for esophageal cancer [37]. In this study, we found that the positive rate of IL-8 expression was correlated with smoking in patients with ESCC. Similarly, Melinceanu et al. also found that heavy smoking was associated with higher levels of IL-8 in patients with squamous cell carcinoma of the larynx [38]. These findings suggested that over-expression of IL-8 in association with smoking might play an important role in the genesis and development of cancer, which is needed to be confirmed in the future study.
A growing body of evidence has confirmed the significance of IL-8 and its high expression in cancer. In physiological conditions, macrophages, endothelial and epithelial cells produce IL-8 in response to injury or infection. One of the functions of IL-8 is to induce the chemotaxis of granulocytes, especially neutrophils. At the same time, neutrophils account for a large proportion of tumor-associated immune cells, therefore they are also called tumor-related neutrophils. IL-8 can enhance their effect and thus plays an active part in the development of tumors [30]. In addition, IL-8 can increase the number of cancer stem cells, strengthen the invasive ability of cancer cells in pancreatic cancer [39] and enhance the proliferation of tumor cells in lung cancer [40]. Similar results were found in breast cancer, colon cancer, ovarian cancer, kidney cancer and melanoma [41]. Although there were many studies showing that the level of serum IL-8 and its receptor would increase in ESCC patients [18, 19, 42, 43], many of them focused on their mechanism [18, 43]. To our knowledge, our present study is the first to evaluate the early diagnostic value of IL-8 in ESCC through such multicenter and large-scale trials.
Currently, it is unrealistic to expect achieving the early diagnosis of ESCC by using a single marker for lacking of definite specific markers [44]. Many potential markers have been found in these years, but still none of them is good enough to be used in early diagnosis of ESCC independently [4, 45]. Considering the heterogeneity of tumors, multi-markers combined detection would be a potential solution for early diagnosis of ESCC. Tong et al. reported that interleukin-6 (IL-6) and IL-8 were significantly upregulated in early-stage ESCC patients according to the results of protein chips, suggesting that the combined detection of IL-6 and IL-8 is beneficial to ESCC diagnosis [20]. Moreover, Chadha et al. reported that the combination of serum IL-8, tumor necrosis factor-
Given the fact that IL-8-G-protein coupled receptor chemokine (C-X-C motif) receptor 1/2 (CXCR1/2) signaling pathway appears to be essential for tumor progression and metastasis, its potential as a prognostic factor has been explored. Although IL-8 was suggested to be a poor prognostic factor in many types of cancers [47, 48, 49], it was demonstrated that serum IL-8 was not significant enough to be a prognostic factor for ESCC [50]. In our research, it was also shown that the expression of IL-8 mRNA and serum IL-8 concentration were not significantly correlated with the patients’ overall survival time. However, contrary results were observed when referring to the prognostic value of IL-8 protein expression in ESCC tissues on survival of patients. Li et al. found that higher IL-8 expression was associated with poorer overall survival [31], while Hosono et al. reported that overall survival of the patients was not significantly different among the negative-, low- and high-expression IL-8 groups [34]. For CXCR-2, Liang et al. indicated CXCR-2 mRNA as a better prognostic factor in esophageal cancer development [42]. Conversely, Łukaszewicz-Zaja̧c et al. reported that serum CXCR-2 was not an independent prognostic factor for the survival of EC patients [51]. Moreover, the immunohistochemical analyses performed by Ogura et al. demonstrated that patients in the IL-8(
There are some limitations in our study. Firstly, patient samples and normal controls were not totally age-matched or gender-matched. It is unavoidable that this selection bias may lead to some errors in our results. Also, due to the limitation of the sample size, we didn’t set a validation cohort to confirm our results. In conclusion, this study preliminarily determined that serum IL-8 was highly expressed in ESCC patients and even in early-stage ESCC. The results showed that it may be promising as a candidate marker to compose a multi-marker panel and to explore the improvement in the early diagnosis of ESCC. Its early diagnostic efficiency still needs more follow-up evaluations.
Footnotes
Acknowledgments
This work was supported by the Natural Science Foundation of China (81972801 and 31600632), the Natural Science Foundation of Guangdong Province (2018A030307079 and 2019A1515011873), Student Innovation and Entrepreneurship Training Program of Shantou University Medical College (201910560130), the Innovative and Strong School Project of Guangdong (2018KTSCX068), 2020 Li Ka Shing Foundation Cross-Disciplinary Research Grant (2020LKSFG01B), the National Key Research and Development Program of China (2018YFC1313101), the Guangdong Esophageal Cancer Institute Science and Technology Program (Q201906) and Grant for Key Disciplinary Project of Clinical Medicine under the Guangdong High-level University Development Program.
Conflict of interest
The authors declare that there is no conflict of interest.
