Abstract
Background:
Esophagogastric junction adenocarcinoma (EJA) is one of the most common malignant tumors of digestive tract with high mortality worldwide. Given a lack of early diagnosis biomarkers, the prognosis of EJA is poor. Non-invasive biomarkers for early-stage EJA are urgently required.
Objective:
We aimed at evaluating the early diagnostic value of serum interleukin-8 (IL-8) level in EJA patients.
Methods:
The IL-8 mRNA expression data were analyzed based on the stomach cardia adenocarcinoma samples of The Cancer Genome Atlas (TCGA) database. Enzyme-linked immunosorbent assay (ELISA) was used to measure the concentration of serum IL-8 in 95 EJA patients and 95 normal controls enrolled from 2 different cancer hospitals. The diagnostic accuracy of serum IL-8 was evaluated by applying Mann-Whitney U test and receiver operating characteristic (ROC) curve.
Results:
The mRNA expression levels and serum levels of IL-8 in EJA group were significantly higher than those in the normal group (all P < 0.001). The areas under the ROC curve (AUC) were 0.661 (95% CI, 0.583-0.740) and 0.745 (95% CI, 0.606-0.885), with the sensitivities of 43.2% (95% CI, 33.2%-53.7%) and 66.7% (95% CI, 46.0%-82.8%) and the specificities of 87.4% (95% CI, 78.6%-93.1%) in EJA group and early-EJA group, respectively, when the optimal cutoff value was 109.086 pg/mL. The clinical data analysis showed there were significant correlations between patient genders, depth of invasion, lymph node metastasis, TNM stage and the serum level of IL-8 (all P < 0.05).
Conclusions:
Serum IL-8 represents a potential diagnostic biomarker to identify early-stage EJA.
Introduction
Esophagogastric junction adenocarcinoma (EJA) is defined as the adenocarcinoma with an epicenter at the junction between the distal esophagus and proximal stomach histopathologically. 1,2 As one of the most common highly lethal and malignant tumors, the incidence of EJA has been reported to alarmingly increase worldwide recently. 3,4 However, controversy still exists on the aspects of pathogenesis, epidemiology, pathology, clinical treatment and prognosis, due to a number of unique biological characteristics of EJA. 5 Without the specific clinical symptoms and the effective methods for early diagnosis, majority of patients with EJA are at an advanced stage when diagnoses are confirmed, meaning that the tumor has probably extensively invaded and distantly metastasized, and leaded to poor prognosis and low survival rate. 6 Hence, the key to improving prognosis and increasing the survival rate is to identify convenient and effective methods to increase the early diagnosis rate of EJA.
Current methods for EJA diagnosis are mainly histopathological biopsy after endoscopy technologies, but issues such as invasive injury, low efficiency and high cost still limit their applications in early detection. Serum tumor biomarkers have several advantages, compared with conventional endoscopy methods, of less invasiveness, lower cost and higher efficiency for clinical utility. 6,7 For example, alpha-fetoprotein has been widely used on clinical detection for hepatocellular carcinoma. 8 A screening for prostate cancer mainly relies on prostate-specific antigen (PSA) or the inactive proenzyme form of PSA (Pro-PSA). 9 Postprandial serum carbohydrate antigen 125 (CA 125) used for supporting vector machine-based algorithm could distinguish ovarian cancer from benign diseases with sensitivity of 91.7% and specificity of 99.2%. 10 Nevertheless, there remains the deficiency of effective biomarkers for the early diagnosis of EJA.
In our previous studies, serum cytokine antibody microarrays of 20 esophageal squamous cell carcinoma (ESCC) patients and 20 normal controls were constructed to search for candidate cytokine biomarkers, and the results showed that interleukin-8 (IL-8) might be a possible molecule for early detection of ESCC. 11 IL-8, a member of the chemokine family, can attract neutrophils infiltrating via acting on C-X-C chemokine receptor type 1/2. 12 IL-8 contributes to tumor angiogenesis, and promotes the immunosuppressive nature in tumor microenvironment through the chemotaxis of neutrophils and myeloid-derived suppressor cells. 13,14 Large quantities of IL-8, secreted by tumor cells, contribute to the potential of progression and metastasis of tumor cells. 14 Serum IL-8 level have been demonstrated to be linked with multiple types of cancers, especially in tumor prediction and oncogenesis. 15 -18
Interestingly, the incidence patterns of ESCC and EJA were similar in same geographic areas in China, which suggested that there were many similar pathogenic factors and mechanisms that lie behind both cancers. 19,20 In brief, IL-8 also has the potential to be used as diagnostic biomarkers in early-stage EJA. Indeed, Deans et al reported that IL-8 is highly expressed both at mRNA and protein levels in EJA specimens. 21 So far as we know, there are few studies that have explored the association between serum IL-8 level and the early diagnosis of EJA in depth. Therefore, this study was designed to probe the early diagnostic efficiency of serum IL-8 in EJA.
Material and Methods
Samples
Between January 2017 and September 2018, sera of 95 EJA patients and 95 normal controls were collected from the Cancer Hospital of Shantou University College of Medicine and Sun Yat-Sen University Cancer Center (Table 1). All patients had gastroscopy and were pathologically diagnosed as EJA without any history of tumor, and sera were collected before the patients received any treatment. Normal controls have been certified free from any tumor-related diseases. EJA stages Ⅰ and Ⅱ were defined as early-stage EJA according to AJCC Cancer Staging Manual eighth.
Participant Information and Clinicopathological Characteristics.

Abbreviation: EJA, esophagogastric junction adenocarcinoma.
a 8 are histologically uncertain.
Peripheral blood samples from patients and normal controls were stored at room temperature (18-25°C) for 30 minutes, and subsequently centrifugated at 1250 G for 5 minutes and stored at −80°C. All the participants signed informed consent and voluntarily joined the study before their sera taken. This study was approved by the institutional ethics review committee at the Cancer Hospital of Shantou University College of Medicine (IRB number: 2016002). This work was in conformity with the principles of the Helsinki Declaration.
Analysis of Gene Expression in the Cancer Genome Atlas (TCGA) Database
Stomach adenocarcinoma (STAD) RNA-seq data (HTSeq counts) were downloaded from the TCGA website (https://tcga-data.nci.nih.gov/tcga/). The data of 18 normal solid tissues, 73 cardia adenocarcinoma tissues and 228 non-cardia adenocarcinoma tissues were used to present the mRNA expression level of IL-8. P < 0.05 (2-sided) was supposed to be statistically significant.
Enzyme-Linked Immunosorbent Assay (ELISA)
Serum concentrations of IL-8 were measured in duplicate at the Cancer Hospital of Shantou University College of Medicine using commercial human IL-8 ELISA kit (ELH-IL8, cat. No. 0802190143, RayBiotech Inc., Georgia, USA) in the same batch. All well-training operators were blinded to the characteristics of samples from EJA patients and normal controls. Briefly, the IL-8 standard was diluted to 8 groups of standard solution at concentrations of 0.0, 0.8, 2.5, 7.4, 22.2, 66.7, 200.0, and 600.0 pg/mL. Serum samples were diluted at a ratio of 1:3 in accordance with our preliminary experiment after warming to room temperature. 100 µL standards and samples were added to 96-well ELISA plates, and were incubated at 37°C for 2.5 hours. After the plates were washed with wash buffer, 100 µL prepared detection antibody solution was added to each well, the plates were incubated at 37°C for 1 hour. Next, the plates were washed, 100 µL prepared horserumdish peroxidase (HRP)-streptavidin solution was successively added, with incubation at 37°C for 1 hour. The plates were washed again. Finally, 100 µL staining agent was added to each well. After 30 minutes of incubation in darkness at 37°C, 50 µL stop solution was added.
Within 5 minutes after completion, the optical density (OD) value of each well was measured at 450 nm using a microplate reader (Multiskan ELX800, BioTek Instruments Inc., Vermont, USA). The average OD values were calculated after the difference of the duplicate wells was smaller than 10%. If it was higher than the maximum OD value of the standard, a 1:7 dilution experiment was performed. Ultimately, Sigma Plot 10.0 (Systat Software Inc., California, USA) was used to obtain the corresponding serum IL-8 concentration.
Statistical Analysis
The experimental data were collated and analyzed using Microsoft Excel 365 (Microsoft Corp., California, USA), SPSS 24.0 (IBM Corp., Armonk, USA), Sigma Plot 10.0, GraphPad Prism 8.0 (GraphPad Software Inc., California, USA) and R 3.6.1 (The R Foundation, open source project). Mann-Whiney U test was used to compare the serum IL-8 level between 2 groups. The receiver operating characteristic (ROC) curves were presented to evaluate the diagnostic efficacy of IL-8 according to the area under ROC curve (AUC) with the 95% confidence interval (CI). The optimum diagnostic cut-off value was calculated using the MinValueSp method of the OptimalCutpoints package (CRAN, Tsinghua Mirror, Beijing, China) in R. The basic principle of this method was to set the minimum cut-off value for maximizing specificity and sensitivity under the premise of specificity greater than 85%. Based on the same cut-off value, the diagnostic value of IL-8 in early-stage EJA was assessed. The positive predictive value (PPV), negative predictive value (NPV), positive likelihood ratio (PLR), and negative likelihood ratio (NLR) were also presented for the improvement of clinical diagnosis interpretation. The relationship between the positive rate of IL-8 and Clinical variables in EJA group was analyzed by Chi-squared test. In all the statistical analysis, P value (2-sided) lower than 0.05 was considered statistically significant.
Results
IL-8 Expression Level in STAD and Normal Tissue in Silico
To identify the source of IL-8 secretion, we downloaded and analyzed the RNA sequencing data of STAD. As we predicted, the results revealed that IL-8 expression increased in cardia adenocarcinoma tissue and non-cardia adenocarcinoma tissue when compared with normal stomach tissue (all P < 0.001, Figure 1). However, the difference of IL-8 expression was not significant between cardia adenocarcinoma and non-cardia adenocarcinoma (P = 0.157, Figure 1).

IL-8 expression level in STAD and normal tissue from TCGA database. Box plot illustrates the visualization of interquartile range including minimum, 25th percentile, median, 75th percentile and maximum values of IL-8 in cardia adenocarcinoma tissue and non-cardia adenocarcinoma tissue of STAD, and normal tissue. T test shows significant difference when the 2 groups are compared with the normal group respectively. The difference of IL-8 expression was not significant between cardia adenocarcinoma and non-cardia adenocarcinoma. IL-8, interleukin-8; STAD, stomach adenocarcinoma. (***Mean P < 0.001).
IL-8 Expression Level in EJA Group and Normal Control
To assess whether IL-8 could be presented as a feasible serum biomarker for the early diagnosis of EJA, we performed ELISA assay to explore in serum of 95 patients and 96 normal controls. The expression level of IL-8 was measured by ELISA. The average serum IL-8 concentration of EJA patients (n = 95, 390.76 ± 188.42 pg/mL) and early-stage EJA patients (n = 27, 788.42 ± 522.99 pg/mL) were significantly higher than those of the control group (n = 95, 52.69 ± 11.41 pg/mL) (Table 2, Mann-Whitney U test, P < 0.001). There was no significant difference between early-stage EJA group and EJA group (Table 2, Mann-Whitney U test, P > 0.05). The distribution of serum IL-8 concentration was positively skewed both in EJA group and normal control. Compared with the samples of normal controls, the samples of EJA group were more densely distributed in higher IL-8 concentrations (Figure 2A). The median serum IL-8 concentration in EJA group, early-stage EJA group and normal control was 84.06 pg/mL, 174.74 pg/mL and 30.56 pg/mL, respectively (Figure 2B and C).
Comparison of Serum IL-8 Level Between 3 Groups.
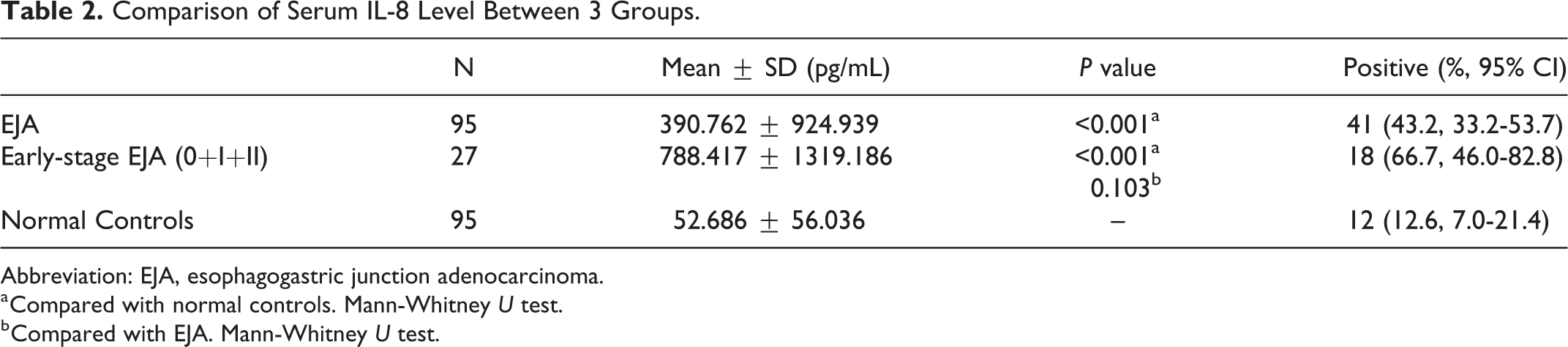
Abbreviation: EJA, esophagogastric junction adenocarcinoma.
a Compared with normal controls. Mann-Whitney U test.
b Compared with EJA. Mann-Whitney U test.

Serum IL-8 level in EJA and normal controls. (A) The EJA group is in red and the normal controls group is in blue. The sample counts on higher concentration in EJA group are more than those of normal controls. (B) Box plot elucidates median level and interquartile ranges, and the whiskers show minimum and maximum value of serum IL-8 in EJA patients and normal controls. (C) Scatter plots show the serum IL-8 level from EJA patients and normal controls. Black horizontal lines are mean, and error bars are SEs. Mann-Whitney U test was executed to verify the statistics differences. IL-8, interleukin-8; EJA, esophagogastric junction adenocarcinoma. (***Mean P < 0.001).
Diagnostic Value of IL-8 in EJA and Early-Stage EJA
To evaluate the diagnostic value of serum IL-8, we drawn the ROC curve based on IL-8 concentration in the serum of the EJA group and early-stage EJA group (Figure 3). The optimum diagnostic cut-off value was 109.086 pg/mL. The AUC was 0.661 (95% CI, 0.583-0.740) and 0.745 (95% CI, 0.606-0.885), with the sensitivity of 43.2% (95% CI, 33.2%-53.7%) and 66.7% (95% CI, 46.0%-82.8%) respectively and the same specificity of 87.4% (95% CI, 78.6%-93.1%) versus the normal group. The rest of the diagnostic efficacy parameters were shown in Table 3.

ROC curve analysis of serum IL-8 in EJA and early-stage EJA. ROC curve of EJA groups and normal controls group is in red and blue, respectively. The area under the black diagonal line is 0.5 for reference. EJA, esophagogastric junction adenocarcinoma; ROC curve, receiver operating characteristic curve.
Evaluation of the Detection Value of IL-8 in the Diagnosis of EJA.a

Abbreviations: EJA, esophagogastric junction adenocarcinoma; NC, normal controls; AUC, area under the ROC curve; SEN, sensitivity; SPE, specificity; PPV, positive predictive value; NPV, negative predictive value; PLR, positive likelihood ratio; NLR, negative likelihood ratio.
a 95% CI were given in brackets for each group.
Relationships Between IL-8 Positive Rate and Clinical Characteristics
To further explore the relationships between the positive rate and the clinical characteristics of the EJA group, chi-squared test showed the correlations between positive rate and patient gender, depth of invasion, lymph node metastasis, and TNM stage, respectively (Table 4, P < 0.05).
Correlation Between IL-8 and Clinical Data in EJA Patients.

Abbreviation: EJA, esophagogastric junction adenocarcinoma.
a 3 are uncertain of depth of tumor invasion.
b 3 are uncertain of regional lymph nodes.
c 26 are histologically uncertain.
Discussion
The incidence of EJA has rapidly increased in Eastern countries in the last few decades. 22 The low early-stage diagnosis rate of EJA remains low 5-year survival rate mainly because of the lack of effective means for early detection. Serum tumor biomarkers serving as a non-invasive test, are more convenient and precise and have fewer secondary actions of infection and hemorrhage, have long been used as feasible screening methods in multiple tumors, which benefits the further diagnosis of cancer and the generation of treatment plans. Hence, it is essential to search for serum EJA biomarkers. To our knowledge, this study is the first to assess early diagnostic value to IL-8 in EJA with polycentric hospitals.
Our previous study documented that IL-8 could be a useful cytokine marker in ESCC. 11 Chen et al and Tran et al reported that the high incidence area of ESCC and EJA was in a high degree of overlap in regional distribution. 19,20 There are possibly like to be similar etiology and pathogenesis pattern between ESCC and EJA though it remains poorly understood. Known as Barrett’s esophagus, the metaplasia of the simple columnar epithelium is the precancerous lesion to the development of EJA. 23 Isomoto et al showed high level of IL-8 expression was associated with intraepithelial neutrophils which were drawn to chemotactic influences of presumably main IL-8 secretion from the epithelium under inflammation in Barrett’s esophagus tissue. 24 Unsurprisingly, increasing IL-8 mRNA and protein levels were observed in several lines of research covering esophageal adenocarcinoma and EJA. O’Riordan et al reported significantly increased IL-8 expression in adenocarcinoma tissue, with stepwise increase in the expression of IL-8 from normal through Barrett’s epithelium to adenocarcinoma by ELISA. 25 Confirmed by immunohistochemistry, Jenkins et al illustrated the increased IL-8 protein levels in both Barrett’s tissues and adenocarcinoma. 26 Moreover, Deans et al reported mRNA for IL-8 was detected in EJA tumor tissue at significantly elevated concentrations. 21 Similar results were obtained by the analysis of TCGA-STAD data in this article. Above all, it demonstrated that IL-8 might be a promoting early detection biomarker in EJA.
In this study, we explore the feasibility of IL-8 as a marker for a better early diagnosis of EJA. The significantly higher serum IL-8 levels in both EJA group and early-stage EJA group and the ROC curve analysis results show a degree of diagnostic accuracy in early detection of EJA. As 2 major serum tumor markers for auxiliary diagnosis in gastro-intestinal cancers, carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA19-9) had not achieved a magnificent effect. A research containing 211 EJA patients reported that the positive ratio of CEA and CA19-9 was 20.3% and 12.9%, which was similar to the results of research conducted by Scarpa et al. 27,28 Although the result in this study is superior to that of the tumor markers currently used in clinical practice, the sensitivity of IL-8 seem to be not enough to serve as an independent early diagnosis biomarker, as the same holds true to its specificity, so multiple-marker panels included IL-8 should be considered to develop. Nevertheless, endoscopy combined with biopsy is needed to carry out accurate diagnosis. The positive rate of serum IL-8 level has certain correlations to gender, depth of invasion, lymph node metastasis and TNM stage (all P < 0.05). Possible explanation about gender difference might be the different incidence in male and female. 29 The fact that advanced-stage patients have occupied relatively large proportion, probably increases the potential biases. However, a growing body of studies demonstrated IL-8 are overexpressed in various tumors and are correlated with tumor stages and grades. 30
IL-8 is a chemokine promoting inflammation via its receptors, CXCR1 and CXCR2 which exist not only on neutrophils and endothelial cells, but also on cancer cells and tumor-associated macrophages. When infected, neutrophils trend to the focus of infection by CXCL8-CXCR1/2 axis. Neutrophils rupturing by oxidation and degranulation reduce inflammation and bacteria amount. For the signaling cascades affected by different cytokines, up to 100-fold up-regulation of CXCL8 expression is stimulated. 30 However, more tumor microenvironment responses contribute to the interactions between IL-8 and cancers. Besides EJA, IL-8 is overexpressed in esophageal cancer, breast cancer, colon cancer and other solid tumors. 12,30 It employs tumor cells autocrine and paracrine to increase its amount, and combines with CXCR1/2 in the tumor microenvironment to implement effects. The important effects of increasing cell proliferation (stem cell as well), motion, migration and invasion in addition to angiogenic response are mainly stimulated by tumor-associated macrophages. 30 To wit, although IL-8 have been implicated in both cancer and inflammation, the secretory cell and signal pathways are disparate. The careful interpretation of IL-8 level is required to conclude the correlation with particular condition’s diagnosis or prognosis. 31 Consequently, although IL-8 has the potential of as a promoting marker for numerous types of cancers or other clinical issues, further studies are needed to address this.
Some improvements are scheduled to uncover expected serum autoantibody for useful marker panels in early detection. Several kinds of serum tumor biomarkers have gradually been exposed, including circulating DNAs, micro RNA, long noncoding RNAs and metabolites by genome and proteome technology. 32 -35 The feasibility of these type of methods were found in the experimental research, but few of them could be applied to the clinical practice. 36 In the field of EJA, exploration of effective panels is a longstanding issue. The detection rate (AUC of 0.730, sensitivity of 64%, specificity of 87%) of tumor-associated autoantibodies (TAAs) panel reported by Zhou et al was slightly inferior to that of IL-8. 37 Similarly, in Xu et al’s panel (6 TAAs of p53, NY-ESO-1, PRDX6, MMP-7, Hsp70, and Bmi-1), the AUC, sensitivity and specificity in early-stage EJA reported in their study were 0.785, 50% and 90.5%. 38 Although the detection differences might be caused by the source and size of samples, IL-8 is worthy to be further studied. Further validation studies are needed with a larger sample of early-stage EJA patients from different institutions. Considering the flow of diagnosis, a cut-off point of early diagnosis biomarkers should be established, rising the specificity at the expense of sensitivity. The specificity of 87.4%, at this sensitivity of 66.7%, reveals that IL-8 is potential to be a screening test for early EJA. In the future researches, we will explore biomarkers with high sensitivity and specificity to optimize EJA diagnosis biomarkers panels with IL-8.
There are still some limitations in our study. One limitation of this study is the sample size. The other is lack of independent external validation cohort to verify the results. Therefore, an optimized serum biomarker panel (IL-8 included) in the larger early-stage patient cohorts should be established to further explore the efficacy of early diagnosis in EJA.
Conclusion
In conclusion, this study is the first to investigate the efficacy of serum IL-8 in the early diagnosis of EJA, and preliminarily identifies the high expression of serum IL-8 as a potential molecular marker for early diagnosis of EJA.
Supplemental Material
Supplemental Material, sj-docx-1-ccx-10.1177_10732748211004883 - The Early Diagnostic Value of Serum Interleukin-8 in Esophagogastric Junction Adenocarcinoma
Supplemental Material, sj-docx-1-ccx-10.1177_10732748211004883 for The Early Diagnostic Value of Serum Interleukin-8 in Esophagogastric Junction Adenocarcinoma by Zheng Li, Haijie Xu, Jiaming Yu, Cantong Liu, Chunwen Zheng, Ruijie Zeng, Liyan Xu, Enmin Li, Yuhui Peng and Yiwei Xu in Cancer Control
Footnotes
Abbreviations
AUC, areas under the ROC curve; CA 125, carbohydrate antigen 125; CA19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; EJA, esophagogastric junction adenocarcinoma; ELISA, enzyme-linked immunosorbent assay; ESCC, esophageal squamous cell carcinoma; HRP, horserumdish peroxidase; IL-8, interleukin-8; NC, normal controls; NLR, negative likelihood ratio; NPV, negative predictive value; OD, optical density; PLR, positive likelihood ratio; PPV, positive predictive value; Pro-PSA, proenzyme form of PSA; PSA, prostate-specific antigen; ROC, receiver operating characteristic; SEN, sensitivity; SPE, specificity; STAD, stomach adenocarcinoma; TAAs, tumor-associated autoantibodies; TCGA, The Cancer Genome Atlas.
Authors’ Note
Zheng Li, Haijie Xu, Jiaming Yu contributed equally to this study. This study was approved by the institutional ethics review committee at the Cancer Hospital of Shantou University College of Medicine (IRB number: 2016002). This work was in conformity with the principles of the Helsinki Declaration. All the participants signed informed consent and voluntarily joined the study before their serum taken.
Acknowledgment
The authors thank the patients who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China [grant number #2018YFC1313101]; the National Natural Science Foundation of China [grant number #81972801]; the Natural Science Foundation of Guangdong Province [grant number #2018A030307079, #2019A1515011873]; Student Innovation and Entrepreneurship Training Program of Shantou University College of Medicine [grant number #201910560130]; the Medical Project of Science and Technology Planning of Shantou [grant number #200605115266724]; and 2020 Li Ka Shing Foundation Cross-Disciplinary Research Grant [grant number #2020LKSFG01B].
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
