Abstract
BACKGROUND:
MicroRNAs (miRNAs) have been proven to be critical players in many different types of tumors including esophageal squamous cell carcinoma (ESCC).
OBJECTIVE:
This study aimed at investigating the correlation of miR-138 expression and clinical outcome of patients with ESCC.
METHODS:
A total of 168 serum samples and 128 fresh cancer tissues as well as their corresponding adjacent non-cancerous tissues were collected. Real-time PCR was performed to evaluate the clinical value of miR-138 in ESCC.
RESULTS:
Our results showed that tissue and serum miR-138 levels were both significantly reduced in ESCC compared to their respective controls. Tissue miR-138 levels were highly correlated with serum miR-138 levels. Serum miR-138 differentiated patients with ESCC from healthy controls with high accuracy. In addition, reduced tissue/serum miR-138 levels were correlated with unfavorable clinicopathological parameters including T stage, lymph node metastasis and TNM stage. ESCC patients with lower tissue/serum miR-138 levels had shorter five year overall survival compared with those with higher tissue/serum miR-138 levels. Finally, downregulation of miR-138 was demonstrated to be an independent prognostic risk factor for ESCC.
CONCLUSIONS:
In conclusion, both tissue and serum miR-138 levels are reduced in ESCC, and might be promising prognostic biomarkers for ESCC.

The expression level of tissue and serum miR-138 in ESCC.
Esophageal squamous cell carcinoma (ESCC) is the most predominant histological subtype of esophageal cancer [1]. Although multidisciplinary treatment modalities have been employed, ESCC is still one of the leading causes of cancer related mortality, with a 5-year overall survival rate of 15%–20% [2]. Due to the fact that the prognosis of ESCC remains poor, it is imperative to identify novel molecular biomarkers for predicting survival outcome and effective intervention of ESCC.
MicroRNAs (miRNAs) are short, non-coding RNAs, which bind to the 3’-untranslated region (3’-UTR) of target mRNAs leading to their degradation or translational repression [3]. Individual miRNAs potentially target hundreds or thousands of mRNAs, thus these small molecules has been demonstrated to involve in a wide variety of biological processes such as proliferation, survival, apoptosis and differentiation [4, 5, 6]. Growing number of evidence have indicated that miRNAs can function as either oncogenes or tumor suppressors, depending on the specific tissue microenvironment and the downstream targets [7, 8]. Their important roles in the initiation and progression of ESCC not only help early diagnosis, but also provide prognostic information which might improve the treatment and clinical management. For instance, ectopic expression of miR-92b could significantly suppress the invasion and metastasis capacity of ESCC cells both in vitro and in vivo by targeting ITGAV. In addition, high miR-92b expression in ESCC tissues was associated with favorable clinical outcome, suggesting that miR-92b acted as a tumor suppressor in ESCC [9]. miR-483 was significantly upregulated in ESCC and its overexpression was associated with worse prognosis as well as multidrug resistance [10]. A recent study showed that miR-483 promoted the proliferation, migration and chemo-resistance of ESCC cells by regulating EI24 [11].
Previous studies suggested that miR-138 played an important role in regulating osteogenesis, stem cell differentiation and inflammation processes [12, 13, 14]. More importantly, deregulation of miR-138 has been reported in many types of cancer such as colorectal cancer, ovarian cancer and non-small cell lung cancer [15, 16, 17]. However, the clinical significance of miR-138 in ESCC remains unclear. Therefore, the aim of current study was first to evaluate the expression pattern of tissue and serum miR-138 in patients with ESCC. Then their correlation with clinical outcome was further analyzed.
Materials and methods
Patients and samples
Tissue and serum samples were obtained from 128 patients who underwent ESCC resection at our hospital. ESCC patients who had received adjunct therapies (radiotherapy or chemotherapy or targeted therapy) before surgical resection were excluded. The clinical information for each ESCC patient, including age, gender, location, T stage, clinical stage, lymph node metastasis and differentiation, was documented. The tumor-node-metastasis (TNM) staging system of the American Joint Committee on Cancer 7th edition was used for clinical staging. The follow up time was 5 year. Paired adjacent normal tissues and forty serum samples from healthy volunteers were used as the controls. This study was approved by the Research Ethics Committee of the The First Affiliated Hospital, and College of Clinical Medicine of Henan University of Science and Technology, and conducted in accordance with the ethical guidelines of the Declaration of Helsinki. All enrolled participants signed written informed consent for the use of serum samples and clinical data.
RNA isolation and RT-qPCR
Total RNA was isolated from serum samples using a miRNeasy Serum/Plasma Kit (QIAGEN Inc., Valencia, CA, USA) according to the manufacturer’s protocol. Complementary DNA (cDNA) was synthesized using a First Strand cDNA synthesis kit (Fermentas; Thermo Fisher Scientific, Inc., Pittsburgh, PA, USA). RT-qPCR was performed in an Applied Biosystems
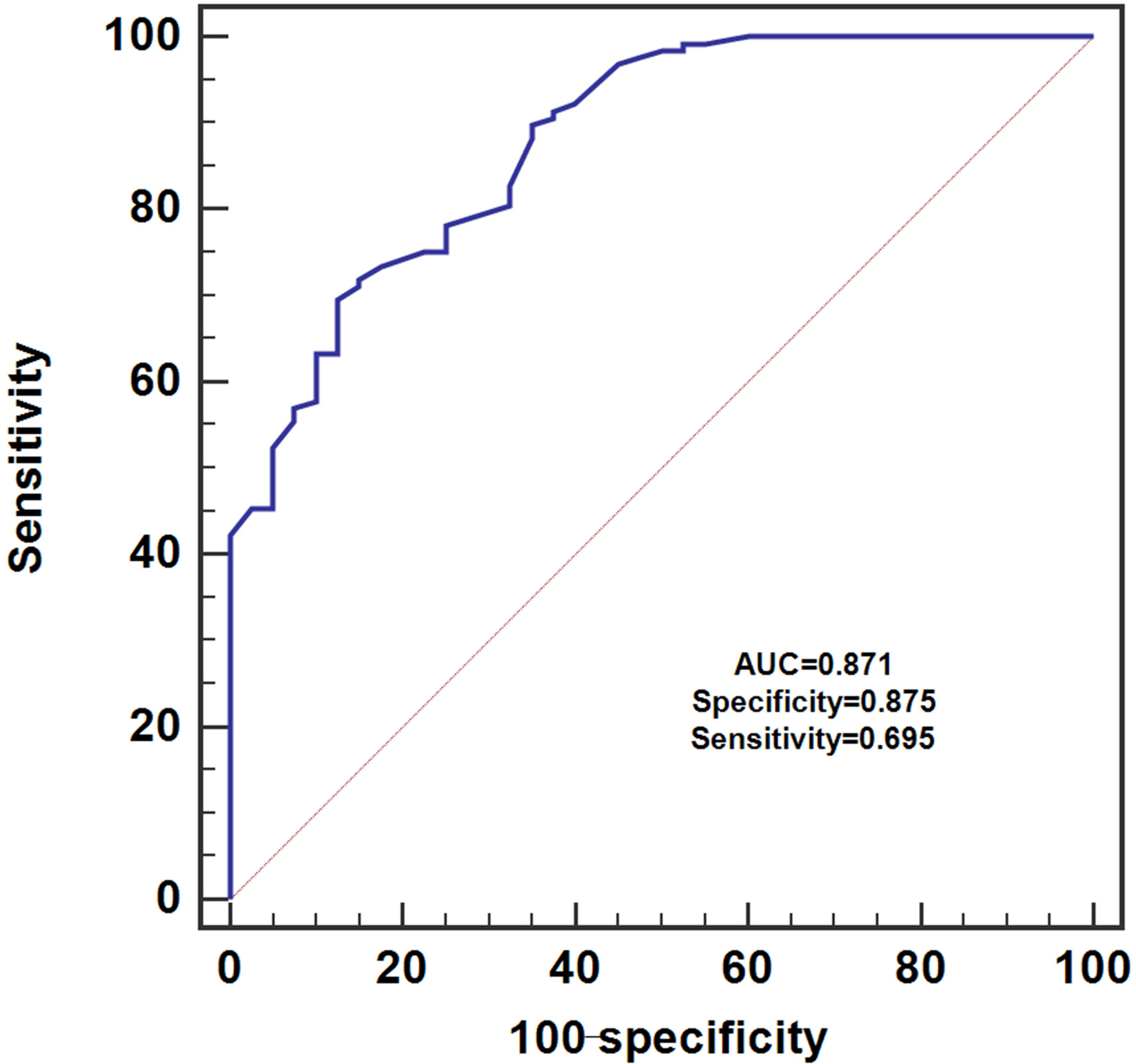
Serum miR-138 effectively discriminated ESCC patients and healthy controls.
The expression levels of miR-138 in both serum and tissue sample do not subject to normal distribution. The differences in the relative expression of tissue /serum miR-138 between ESCC patients and the controls were analyzed using the Mann-Whitney U test. Pearson correlation analysis was performed to evaluate the association between tissue and serum miR-138 levels. The receiver operating characteristic (ROC) curve was used to evaluate the discriminative performance of serum miR-138 in ESCC. The median value of tissue/serum miR-138 was used as a cut-off point to divide the ESCC patients into two groups, namely high tissue/serum expression group and low tissue/serum expression group. The relationship between tissue/serum miR-138 expression and clinicopathological factors of ESCC patients was analyzed by chi-square test. The five year overall survival curves were generated by the Kaplan-Meier method and compared using the log-rank test. The Cox proportional hazards model was used to detect independent prognostic factors. GraphPad Prism 6 (GraphPad Software Inc., La Jolla, CA, USA) was used for the data analysis and two tailed
Results
The expression level of tissue and serum miR-138 in ESCC
The expression levels of miR-138 were compared between tumor and their paired paracancerous tissue samples in 128 ESCC patients by using qRT-PCR. Our results showed that miR-138 levels were significantly reduced in tumor samples compared with non-adjacent normal tissues (
Correlation with tissue/serum miR-138 and clinicopathological parameters of ESCC
Chi-square test was used to find out the association between tissue/serum miR-138 and clinicopathological features of ESCC. Our results showed that reduced tissue miR-138 was associated with T stage (
Association of tissue miR-138 expression with clinicopathologic features of patients with ESCC
Association of tissue miR-138 expression with clinicopathologic features of patients with ESCC
As regards to serum miR-138, it was associated with lymph node metastasis (
Association of serum miR-138 expression with clinicopathologic features of patients with ESCC
Kaplan-Meier analysis was performed to evaluate the correlation between tissue/serum miR-138 levels and five year overall survival in ESCC. Overall survival was significantly poorer in patients with low tissue miR-138 expression than those with high tissue miR-138 expression(
Multivariate analysis demonstrated that low tissue miR-138 expression (HR
Multivariate analyses of prognostic variables associated with overall survival
Multivariate analyses of prognostic variables associated with overall survival

The association between tissue/serum miR-138 levels and overall survival in patients with ESCC.
ESCC remains to be a significant public health issue around the world especially in China [18, 19]. In this study, our results showed that both tissue and serum miR-138 was significantly reduced in patients with ESCC. A strong positive correlation was found between tissue and serum miR-138 levels. Serum miR-138 was able to differentiate ESCC patients from healthy volunteers. Reduced tissue/serum miR-138 was strongly associated with worse clinicopathological variables and poorer clinical outcome. Taken together, miR-138 might play a tumor suppressive role in the progression of ESCC and downregulation of miR-138 promote the carcinogenesis process. Therefore, miR-138 might serve as a promising prognostic biomarker for ESCC.
The findings are consistent with the results in the previous study, miR-138 was remarkably reduced in ESCC tissue samples. In addition, reduced miR-138 levels were correlated with worse prognosis in ESCC. Moreover, overexpression of miR-138 suppressed the oncogenic activities of ESCC cells both in vitro and in vivo [20]. One of our novel findingsis that serum miR-138 has similar clinical significance compared with tissue miR-138. Although tissue biopsy remains the gold standard for the diagnosis of many human diseases including cancer, it has many shortcomings. For instance, tissue biopsy is a invasive approach which might lead to infection or cause other complications. In addition, it is impossible to monitor the disease status as multiple tissue biopsies are not practical in most cases. Moreover, the results of tissue biopsy might not reflect the correct information because of the inherent heterogeneity of tumor tissue [21]. Detecting the biomarkers in the circulation system is an effective strategy to solve this problem. miRNAs are highly stable in the serum samples [22]. To the best of our knowledge, this is the first study to demonstrate the clinical value of serum miR-138 in ESCC. Examining the serum miR-138 levels in patients with ESCC might guide the clinical treatment. One limitation of our current study was the relative sample size, which might have potential effects on our conclusion. Further studies need to be done with a larger sample size and in different areas to validate our findings.
Similarly, miR-138 has also been shown to function as a tumor suppressor in other types of cancer. miR-138 was significantly reduced in NSCLC tissues and cell lines. In addition, downregulation of miR-138 was correlated with poorer clinicopathological parameters. Upregulation of miR-138 inhibited the proliferation, migration and invasion capacity of lung cancer cells and vice versa [23]. The expression level of miR-138 was downregulated in osteosarcoma tissues especially in the cancer tissues with metastasis. Overexpression of miR-138 suppressed proliferation and invasion, and promoted apoptosis of human osteosarcoma cells. Opposite results were observed when miR-138 was underexpressed [24]. As far as we know, no study has demonstrated miR-138 play an oncogenic role in the development of cancer. Thus downregulation of miR-138 might be a common phenomenon in cancer, regardless of the specific tissue microenviroment and downstream targets.
In conclusion, both tissue and serum miR-138 were reduced in ESCC and decreased tissue/serum miR-138 were correlated with unfavorable clinical outcome. Taken together, tissue and serum miR-138 might be novel prognostic biomarkers for ESCC.
Footnotes
Conflict of interest
None.
