Abstract
BACKGROUND:
To investigate the prognostic role of lung immune prognostic index (LIPI) in extensive-stage small-cell lung cancer (ES-SCLC) patients treated with platinum plus etoposide chemotherapy.
METHODS:
Data were obtained from two randomized controlled trials (NCT00119613 and NCT00363415). Overall survival (OS) and progression-free survival (PFS) was assessed according to LIPI score through Kaplan-Meier analysis. Univariate and multivariate Cox-regression analysis were performed to investigate predictors for OS and PFS.
RESULTS:
A total of 911 patients with ES-SCLC treated with platinum plus etoposide chemotherapy (CT) were included for analysis. The median age at diagnosis was 62 years, and 760 (83.4%) had performance status of 1 or less. 1-year OS for ES-SCLC with poor, intermediate, and good LIPI was 20%, 30% and 31%, respectively, and 1-year PFS was 7%, 15% and 21%, respectively. Cox-regression analysis showed that the PFS and OS of ES-SCLC with a poor LIPI score was significantly worse than those with good LIPI scores (HR 1.81, 95% CI: 1.38–2.36;
CONCLUSION:
Prognosis of patients with pretreatment LIPI score of 2 is poorer than those with LIPI score of 0–1 among ES-SCLC who received first-line platinum plus etoposide chemotherapy; Further studies are still recommended to confirm our findings in prospective studies.
Introduction
According to the GLOBOCAN estimates of cancer incidence and mortality, lung cancer remains the most commonly diagnosed cancer (11.6% of the total cases) and the leading cause of cancer death (18.4% of the total cancer deaths) in 2018 [1]. Small-cell lung cancer (SCLC) accounts for approximately 15% of all new lung cancers, with an annual incidence of overall 300,000 new cases worldwide. Generally, SCLC is a highly aggressive disease, which is characterized by rapid tumor growth and early distant metastasis [2, 3]. In the past decades, although major improvements have been achieved in the treatment of solid tumors including non-small-cell lung cancer, the current standard first-line treatment for extensive-stage(ES)-SCLC remains platinum-based doublet chemotherapy [4, 5]. Despite the initial response rate to first-line chemotherapy is approximately 70%, nearly all of ES-SCLC patients would inevitably develop chemotherapy resistance and disease progression, and the prognosis of ES-SCLC remains poor, with 5-year survival less than 10% [2]. Therefore, a better understanding of prognostic factors for survival would be of particular importance to assist physicians to identify SCLC patients who are considered at risk for unfavorable outcomes and perform an individualized therapy for such patients in advanced.
Cancer-associated inflammation leads to poor survival [6, 7]. Recently, the associations between inflammatory markers and the prognosis of lung cancer have been extensively investigated [8, 9, 10]. In 2018, Prof. Mezquita [11] developed a lung immune prognostic index (LIPI) based on dNLR greater than 3 and LDH greater than ULN, and characterized into 3 groups (good, 0 factors; intermediate, 1 factor; poor, 2 factors). The authors found that pretreatment LIPI was correlated with worse outcomes for non-small-cell lung cancer (NSCLC) treated with immune checkpoint inhibitor (ICI), but not for chemotherapy. However, the sample size of NSCLC treated with chemotherapy was relative small (
Materials and methods
About PDS and study cohorts
Project data sphere was an independent, not-for-profit data-sharing platform, which provided individual patient-level data from prospective clinical trial in order to advance future cancer research (
Data collection
The available data of the phase III trial contains data about age at diagnosis, baseline ECOG performance status, sex, race, baseline neutrophil and white blood cell, baseline lactate dehydrogenase (LDH) and alkaline phosphatase (ALP), baseline weight and height. Moreover, data about disease response rate, progression-free survival status and overall survival status were recorded. Based on the inclusion criteria for clinical trials, all included patients in the present study should have adequate organ function and acceptable performance status.
Statistical consideration
The baseline characteristics of included patients was simply described by using frequencies and percentages. The LIPI composite scores were calculated based on the dNLR (absolute neutrophil count/[white blood cell count – absolute neutrophil count]) and the baseline LDH level according to Mezquita et al. report [11]. In addition, we also investigated the prognostic role of dNLR and baseline LDH level for ES-SCLC in this patient population. Disease control rate (DCR) and objective response rate (ORR), Overall survival (OS) and progression-free survival (PFS) was assessed according to LIPI scores through Kaplan-Meier analysis. Multiple comparisons of survival times between LIPI score are compared by Log-rank tests. Univariate and multivariate Cox-regression analysis were performed to investigate predictors for overall survival and progression-free survival. Factors significantly associated with risk of OS and PFS in the univariate analysis (
Results
Patients characteristics
A total of 934 patients from two phase III trials were identified (NCT00363415481: 455 patients and NCT00119613: 479 patients, Supplemental Table 1), 23 patients were excluded for the present analysis due to lack baseline data of albumin or LDH. Finally, 911 patients were included for analysis and the baseline characteristics were shown in Table 1. All of the chemotherapy-naïve ES-SCLC patients received platinum plus etoposide therapy. The median age at diagnosis was 62 years. 568 (62.3%) patients aged less than 65, while 343 (37.7%) patients older than 65; Among these patients, and 760 (83.4%) had performance status of 1 or less. 632 were male and 279 were female patients; In addition, a dNLR greater than 3 were observed 263 patients, while the other 648 patients less than 3. As for baseline LDH value, 674 patients were greater than upper limits of normal (ULN), and 237 patients were less than UNL (Table 1).
Baseline characteristics of 911 included patients
Baseline characteristics of 911 included patients
Abbreviation: ECOG PS, Eastern Cooperative Oncology Group Performance Score; dNLR, derived neutrophil to lymphocyte ratio (leukocytes minus neutrophils); ULN, upper limits of normal; BMI, Body Mass Index; ALP, Alkaline phosphatase; LDH, lactate dehydrogenase.
The LIPI was evaluable for 911 patients, giving 174 patients (19.1%) in the poor group, 536 (58.8%) in the intermediate group, and 201 (22%) in the good LIPI group. Median PFS was 5.16 months (95% CI, 4.90–5.30 months). According to LIPI scores, median PFS was 5.50, 6.13, and 6.57 months, respectively. And 1-year PFS was 7%, 15% and 21%, respectively, and there was significant difference of PFS among LIPI groups (
Cox-regression analysis showed that the PFS of ES-SCLC with a poor LIPI score was significantly worse than those with good LIPI scores (HR 1.81, 95% CI: 1.38–2.36;
Univariate and multivariate cox regression analysis for factors predicting PFS
Univariate and multivariate cox regression analysis for factors predicting PFS
Abbreviation: ECOG PS, Eastern Cooperative Oncology Group Performance Score; dNLR, derived neutrophil to lymphocyte ratio (leukocytes minus neutrophils); ULN, upper limits of normal; BMI, Body Mass Index; ALP, Alkaline phosphatase; LDH, lactate dehydrogenase; HR, hazard ratio.
Progression-free survival according to LIPI scores.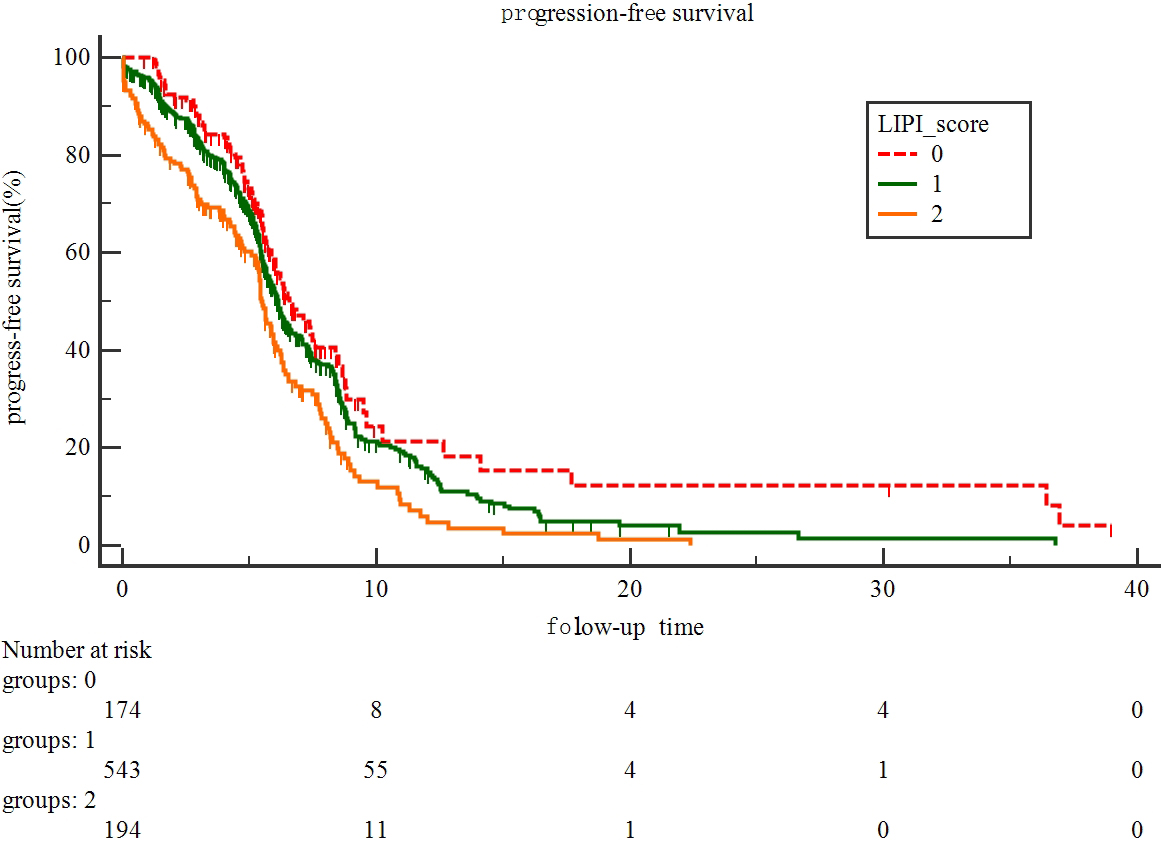
The LIPI was evaluable for 911 patients, giving 174 patients (19.1%) in the poor group, 536 (58.8%) in the intermediate group, and 201 (22%) in the good LIPI group. Median OS was 7.52 months (95% CI, 7.16–7.83 months). According to LIPI scores, median OS for ES-SCLC with poor, intermediate, and good LIPI was 7.6 months, 9.07 months, and 8.81 months, respectively. And 1-year OS for ES-SCLC with poor, intermediate, and good LIPI was 31%, 30% and 20%, respectively, and there was significant difference of OS among LIPI groups (
Overall survival according to LIPI scores.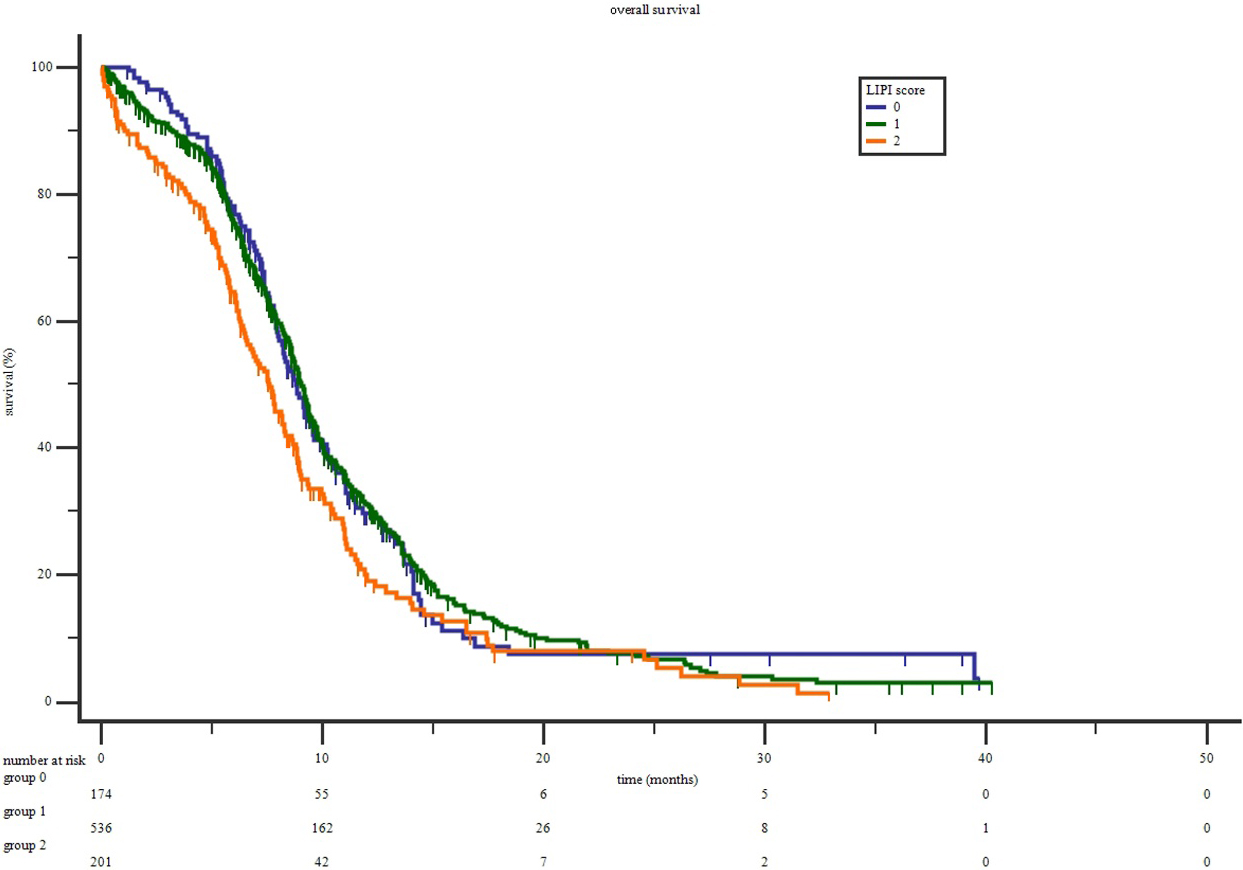
Cox-regression analysis showed that the OS of ES-SCLC with a poor LIPI score was significantly worse than those with good LIPI scores (HR 1.35, 95% CI: 1.07–1.72,
Univariate and multivariate cox regression analysis for factors predicting OS
Abbreviation: ECOG PS, Eastern Cooperative Oncology Group Performance Score; dNLR, derived neutrophil to lymphocyte ratio (leukocytes minus neutrophils); ULN, upper limits of normal; BMI, Body Mass Index; ALP, Alkaline phosphatase; LDH, lactate dehydrogenase; HR, hazard ratio.
A total of 693 ES-SCLC patients were available for ORR and DCR analysis, the ORR was observed in 58 patients (426%) in the poor group, 193 (46.7%) in the intermediate group, and 81 (563%) in the good LIPI group. Similarly, the DCR in the poor group (551%) was significantly lower than that in the intermediate group (62.0%), and (771%) in the good LIPI group. And significant correlation was observed between LIPI scores and ORR or DCR (both
Response rate and disease control rate according to LIPI groups
Response rate and disease control rate according to LIPI groups
Abbreviations: CR, complete response; PR, partial response; SD, stable disease; PD, progression disease; NA, not available.
Emerging evidence indicates that the mediators and cellular effectors of cancer-related inflammation play an important role in cancer development and disease progression [15]. However, the specific mechanism of inflammation involves this procedure remains unclear. Possible mechanism is that the inflammatory cells and cytokines found in tumors are more likely to contribute to tumor growth, progression, and immunosuppression than they are to initiate an effective host anti-tumor response [16, 17]. As a result, the prognostic value of multiple peripheral hematological markers has been extensively investigated in various tumors. Indeed, more and more evidence have showed that biomarkers of inflammation, which derived from peripheral blood, such as neutrophil-to-lymphocyte ratio (NLR), derived neutrophil-to-lymphocyte ratio (dNLR) and lymphocyte-to-monocyte ratio (LMR) is significantly associated with survival for various tumors including lung cancer [18, 19, 20, 21, 22, 23, 24].
Interesting, baseline LDH level has been associated with poor prognosis in solid tumors including SCLC [25]. It has been well known that hypoxic microenvironments frequently exist in many solid tumors with oxygen levels fluctuating temporally, and the hypoxic microenvironment of a tumor can increase the conversion of pyruvic acid to lactic acid [26]. While LDH is an important enzyme that catalyzes the hydrogen transfer reaction between lactic acid and pyruvic acid [27]. Therefore, unregulated LDH levels ensure proficient anaerobic glycolytic metabolism in the tumor environment, leading to reduced oxygen dependence. Numerical studies indicated that LDH expression increased with tumor progression, and the prognostic role of LDH in cancers has been reported in respectable studies, including SCLC [25, 28, 29, 30, 31, 32], although most of these studies are retrospective analysis with relative small sample size. However, the LDH level in the present study seemed to be prognostic only for PFS, but not for OS. One possible explanation for this findings is that all patients were included in clinical trials with adequate hematological and liver function, and there might be potential biases.
The LIPI composite scores are calculated based on the dNLR and the baseline LDH level, both of which can be conveniently obtained in clinical practice. This LIPI scoring system was initially established to reflect immune status and identify patients who would benefit from treatment with ICI, but the sample size is relative small. Mezquita et al. found that Pretreatment LIPI was correlated with worse outcomes for ICI, but not for chemotherapy [11]. Subsequently, Kazandjian et al. [12] perform a larger sample size retrospective studies of 11 clinical trials and find that LIPI scores are important prognostic biomarkers not only for metastatic NSCLC patients treated with ICI, but also for targeted therapy or cytotoxic chemotherapy, although the prognostic value of LIPI seems greater for patients treated with immunotherapy. However, the prognostic role of LIPI scores in SCLC remains undetermined. As a result, we conduct the present study to investigate role of LIPI in ES-SCLC patients who treated with platinum plus etoposide chemotherapy.
To our best knowledge, the present study is the first to demonstrate prognostic role of pre-treatment LIPI for survival of ES-SCLC patients with a large sample size (
We acknowledge that there are several potential limitations of our study. First of all, despite of the randomized, prospective nature of the included studies, our study is a retrospective analysis of the pooled dataset of these included studies. Thus there might be potential biases due to missing trials and laboratory values. Second, the present study is lack of external validation for SCLC, although the score system has been validated in mNSCLC. However, our study has several strong points: 1) this is a secondary analysis of two prospective phase III trial, detailed information about clinical characteristics, performance status and disease progression of the present analysis is reliable; 2) this is the first and largest individual analysis to validate the prognostic role of LIPI in ES-SCLC treated with first-line platinum plus etoposide chemotherapy. Thirdly, none of the included patients received immunotherapy, thus the prognostic role of LIPI score in SCLC who receiving immunotherapy remains undetermined, which needed to be investigated in further studies. Finally, the present study uses the bivariable selection (BVS) for selecting variables to be used in multivariable analysis, this type of variable selection is inappropriate according to previous publication if the relationship between an outcome and a risk factor is confounded by any confounder and when this confounder is not properly controlled. However, no obviously confounder between risk factors were observed in the present study.
Conclusion
In conclusion, the preset study confirm that prognosis of patients with pretreatment LIPI score of 2 is poorer than those with LIPI score of 0–1 among ES-SCLC who received first-line platinum plus etoposide chemotherapy. However, further studies are still recommended to confirm our findings in prospective studies.
Ethical approval
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
As our study was a second-analysis of previously published trials, informed consent was waved in the present study.
Funding
None.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-201502.
sj-docx-1-cbm-10.3233_CBM-201502.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-201502.docx
Footnotes
Acknowledgments
This publication was based on research using information obtained from www.projectdatasphere.org, which was maintained by Project Data Sphere, LLC. Neither Project Data Sphere, LLC nor the owner(s) of any information from the website had contributed to, approved or were in any way responsible for the contents of this publication.
Conflict of interest
None.
