Abstract
Background:
Emotional distress (ED) is associated with worse survival outcomes in patients with melanoma and non–small-cell lung cancer (NSCLC) treated with immune checkpoint inhibitors (ICIs). However, several preclinical studies suggest the association between stress and cancer treatment extends beyond ICIs alone.
Objectives:
To systematically evaluate the relationship between ED and NSCLC patient prognosis.
Design:
This study is an individual patient data (IPD) meta-analysis.
Data sources and methods:
Relevant studies were identified from the PubMed, Web of Science, The Cochrane Library, Embase, and Vivli databases to obtain IPD up to February 2025. The hazard ratios (HRs) and 95% confidence intervals (CIs) were calculated to estimate the association between ED and patient prognosis. Subgroup analysis was conducted based on patient treatment methods.
Results:
Kaplan–Meier analysis shows that compared with patients without ED, patients with ED had worse survival outcomes, regardless of ICIs or chemotherapy (CT) (HR of overall survival (OS), 1.21 (1.11–1.33), p = 0.01; hazard ratio of progression-free survival (PFS), 1.19 (1.07–1.31), p = 0.01). IPD meta-analysis also supported results above (HR of OS, 1.18, 95% CI, 1.07–1.30; HR of PFS, 1.15, 95% CI, 1.03–1.28).
Conclusion:
Our research suggests that ED is only a prognostic biomarker in NSCLC, rather than a predictive biomarker for ICIs.
Introduction
Emotional distress (ED) is a negative psychological state prevalent in cancer patients, commonly manifested as anxiety and depression. 1 Recent studies have shown considerable interest in the association between ED and survival outcomes of cancer patients and demonstrated that ED associate with worse survival outcomes in patients with melanoma and non–small-cell lung cancer (NSCLC) treated with immune checkpoint inhibitors (ICIs).2,3
While preclinical research has demonstrated that ED can promote inflammation and immune evasion,4,5 mechanisms that likely explain its impact on ICI treatment outcomes, additional studies have shown that ED also stimulates the release of stress-related hormones, including adrenaline, noradrenaline, and glucocorticoids.6,7 These hormones have been implicated in tumor proliferation, metastasis, and immune suppression, indicating that the association between ED and survival outcomes may extend beyond immunotherapy and may potentially influence cancer progression more broadly. To verify broader connection between ED and survival outcomes in NSCLC patients, we conducted an individual patient data (IPD) meta-analysis.
Materials and methods
The study was conducted in accordance with Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines and the respective checklist has been provided as a Supplemental File 1. This study is registered with PROSPERO (CRD42025649467).
Search strategy
Studies of potential interest were identified without language restriction by querying the electronic databases PubMed, Web of Science, The Cochrane Library, Embase, and Vivli, up to February 2025. For the database queries, three search term groups were used, with at least one term from each group required to match: (1) “emotional distress”[All Fields] OR “psychological distress”[All Fields] OR “anxiety”[All Fields] OR “depression”[All Fields] OR “stress”[All Fields]; (2) “NSCLC”[All Fields] OR “non-small-cell lung cancer”[All Fields] OR “lung cancer”[All Fields]; (3) “immune checkpoint inhibitors”[All Fields] OR “ICI”[All Fields] OR “immunotherapy”[All Fields]. No geographical restrictions were applied. We excluded studies that were written in non-English, abstracts, letters, reviews, commentary articles, opinion articles, case reports, studies involving early-stage lung cancer, studies involving pediatric populations, animal and in vitro studies, studies that cannot obtain individual patients data, as well as studies that did not utilize the EORTC QLQ-C30 scale to evaluate patients. Finally Seven clinical trials were included: BIRCH (NCT02031458), 8 FIR (NCT01846416), 9 POPLAR (NCT01903993), 10 OAK (NCT02008227), 11 IMpower130 (NCT02367781), 12 IMpower131 (NCT02367794), 13 and IMpower150 (NCT02366143), 14 all of them provided IPD.
Cohort definition
For each chosen randomized controlled trials, patients who received atezolizumab were assigned into the ICI cohort regardless of whether they had received platinum-based chemotherapy (CT) prior to that period, patients who did not receive atezolizumab were assigned to the CT cohort. For each single-arm trials, divided all individual patients into ICI cohorts. A total of 12 cohorts were obtained, of which 7 cohorts were ICI cohort and 5 cohorts were CT cohort.
Method of assessing ED status
EORTC (European Organisation for Research and Treatment) QLQ-C30 version 3 was applied to evaluate ED status of patients. The QLQ-C30 includes 30 items, which can be summarized into five functional scales: physical functioning (PF), role functioning (RF), social functioning (SF), emotional functioning (EF), and cognitive functioning (CF); three symptom scales: fatigue (FA), pain (PA) and nausea/vomiting (NV); six single items: appetite loss (AP), dyspnea (DY), insomnia (SL), diarrhea (DI), constipation (CO), and financial difficulties (FI) and a global health status/Qol (QL). After being evaluated by the questionnaire, the patient will receive a raw score, which can be transferred to a standard score (0–100) through the range reduction method with linear transformation. Previous studies have matched the EF scale of the QLQ-C30 with patients’ actual ED status, ultimately drawing conclusions that patients with standard score >71 were designated as the non-ED group and ⩽71 as the ED group.2,15,16
Kaplan–Meier analysis
We integrated all IPD and first-line treatment patient data, grouped them into four groups according to ED status and treatment methods of patients separately: ED-ICI group; ED-CT group; non-emotional distress (NED)-ICI group and NED-CT group. The median overall survival (mOS) and progression-free survival (mPFS) is obtained by using Kaplan–Meier analysis to estimate the survival function, plotting the survival curves and identifying the time at which the survival probability reaches 50% with overall survival (OS) and progression-free survival (PFS) as the primary endpoints respectively. Calculating and comparing mOS/mPFS, hazard ratios (HRs), 95% CI, and p value between four groups (details see “Statistical analysis”).
Methodology for meta-analysis
The IPD were aggregated through a two-stage meta-analysis framework. 17 First, we utilized the Cox proportional hazards model to estimate HRs and 95% CIs for OS and PFS of each cohort. Second, the fixed-effects model meta-analysis was conducted to generate summary estimates of results of aggregated data. We selected the I 2 statistic as the statistical measure for heterogeneity testing. Meta-analysis chose the fixed-effect model as final result when I 2 statistic ⩽40% and p value of heterogeneity testing >0.1; otherwise, the random-effects model was chosen. 18 Publication bias of cohorts were assessed by Egger’s test, which was considered as absence of evidence for publication bias if p value > 0.05. The stepwise elimination method was employed to assess stability of results. 18
Analyses of independent risk factor
We sorted out other variables that could be obtained from the collected data and included them in the directed acyclic graph (DAG) used to identify confounding factors (Supplemental Figures 1 and 2).19 –26 These variables include age, sex, BMI, Tumor Node Metastasis (TNM) staging, and metastasis status. Ultimately, it was determined that all of them can serve as confounding factors for ED affecting tumor prognosis.
To identify independent risk factors, we first performed a univariate Cox regression analysis to assess the individual association of each variable with the outcome of interest. Then, the variables above were then included in a multivariable Cox regression model. In the multivariable analysis, we considered all relevant variables simultaneously to determine their independent contributions to the outcome, adjusting for potential confounders. Variables that remained statistically significant in both the univariate and multivariable analyses were considered as independent risk factors for the outcome.
The impact of ED status on prognosis
Given that the QLQ-C30 scores are relatively fixed, every specific value were considered as thresholds for Cox regression analysis. For each score, values above the threshold were categorized as non-ED, while those below the threshold were categorized as ED. Cox regression analyses were then conducted separately for both groups. The results of all analyses were summarized and compared to observe the impact of ED status on the outcome.
Statistical analysis
The Cox proportional hazards model was applied to estimate HRs and 95% CIs between ED group and non-ED group for each cohort, and the two-sided log-rank test was used to compare OS and PFS among the two groups, with the significance level alpha = 0.05. The validity of the proportional hazards assumption was assessed by Schoenfeld residuals. Analysis was conducted in R version 4.4.1. The package we utilize are as follows: the “Matefor” package (version 4.6-0); and the “survminer” package (version 0.4.9).
Results
Characteristics of included studies and patients
Initially, 61 items were identified and screened, of which 7 were duplicates. Following a screening of titles, abstracts, and full texts (where applicable), 19 items had remained for searching of individual patients data (Figure 1). Finally, we identified seven clinical trials which involve patients treated with atezolizumab, including two single-arm trials (BIRCH, FIR) and five randomized controlled trials (POPLAR, OAK, IMpower130, IMpower131, IMpower150).8 –14 Among a total of 4632 participants, 2753 (59.43%) received first-line treatment; 3162 (68.26%) received ICIs; 1802 (38.90%) classified as ED and 2830 (61.10%) classified as non-ED (Table 1).
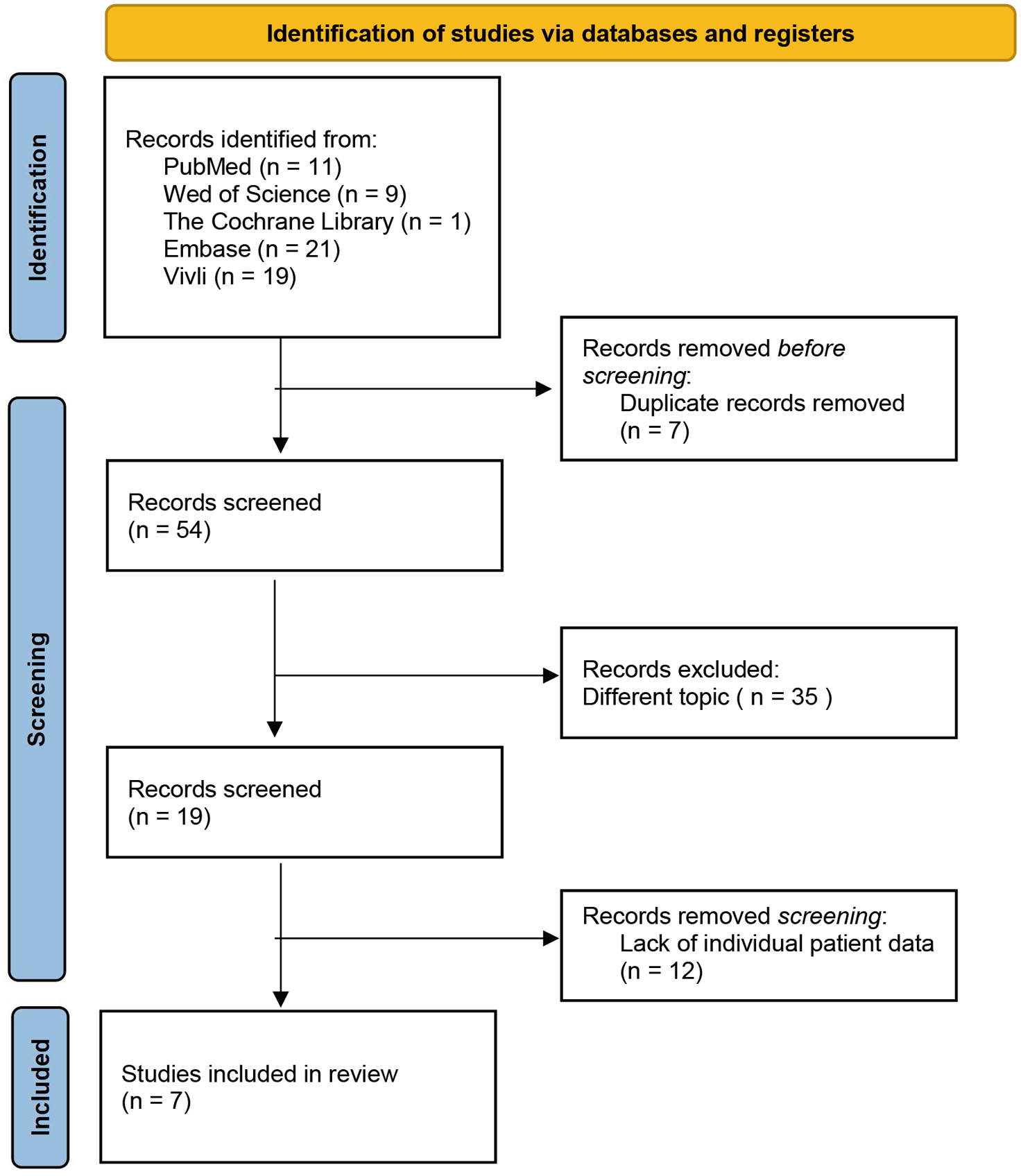
Flowchart of trial identification and selection.
Baseline characteristics of NSCLC patients of ICI and CT.

CT, chemotherapy; ED, emotional distress; ICI, immune checkpoint inhibitor.
Analyses results of OS
Kaplan–Meier analysis indicates that ED patients associate with worse survival outcomes, regardless of ICIs or CT (HROS = 1.21 (1.11–1.33), p = 0.01; HRPFS = 1.19 (1.07–1.31), p = 0.01). Different from previous findings, which suggested ED as a predictive biomarker for ICIs, our results show that ED is only a prognostic biomarker in NSCLC. Compared with non-ED, ED patients had worse OS in both ICIs (ED vs non-ED, mOS, 13.34 month (m) vs 16.07 m; p = 0.01; HR, 1.21 (1.09–1.34)) and CT (ED vs non-ED, mOS, 12.02 m vs 13.93 m; p = 0.01; HR, 1.19 (1.04–1.37); Figure 2(a)). IPD meta-analysis indicates that the ED group exhibited worse OS outcome (HR = 1.18 (1.07–1.30), p = 0.01). Subgroup analysis confirmed this association in both ICIs (HR = 1.18 (1.04–1.34), p = 0.01) and CT groups (HR = 1.18 (1.00–1.39), p = 0.05; Figure 2(b)).

Kaplan-Meier analyses and IPD meta-analyses for OS by ED status and treatment. (a) OS for two different treatments in the subgroups of ED status. Survival curves were compared using two-sided log-rank test. Results show that patients treated with ICIs exhibit a significantly improved OS in both ED and non-ED group, and compared with patients with non-ED, those with ED have worse outcomes in both treated with ICIs and chemotherapy. (b) Forest plots illustrate the findings of IPD meta-analysis and subgroup analysis by treatment. Data are presented as the HR with error bars showing 95% CI. A total of 12 cohorts consistently showed a trend indicating that ED patients experience worse outcomes, aligning with the results from the IPD meta-analysis and subgroup analysis. These analyses of OS remind the association between ED and worse OS outcome.
To further ensure the reliability of the meta-analysis results, we conducted another analysis of the random-effects model for OS in accordance with Cochrane guidelines (Supplemental Figure 3), and the results were consistent with the fixed-effects model. At the same time, the values of τ2 and its standard error were small, indicating low heterogeneity between studies, which is consistent with the heterogeneity analysis results based on I2.
Analyses results of PFS
We excluded clinical trials in which PFS not primary outcome, and investigated the association with ED and PFS in first-line clinical trials. Similarly, Kaplan–Meier analysis shows ED patients had worse PFS regardless of ICIs (mPFS, 5.52 m vs 5.58 m; p = 0.01; HR, 1.19 (1.06–1.33)) or CT (mPFS, 5.55 m vs 5.59 m; p = 0.06; HR, 1.17 (0.99–1.37); Figure 3(a)). IPD meta-analysis of PFS also supported the aforementioned results (HR = 1.15 (1.03–1.28), p = 0.02; Figure 3(b)). The seven trials included in the IPD meta-analysis had been validated as free from publication bias, and the results of the IPD meta-analysis had been passed the heterogeneity and sensitivity analyses.

Kaplan-Meier analyses and IPD meta-analyses for PFS by ED status and treatment. (a) Survival curves of PFS for two different treatment methods in the subgroups of ED status. Similar to results of analysis of OS, ED patients had worse PFS regardless of ICIs or chemotherapy, and patients treated with ICIs exhibited a significantly improved PFS in both ED and non-ED group. (b) Results of IPD meta-analysis and subgroup analysis for PFS were shown as forest plot. Data are presented as the HR with error bars showing 95% CI. A total of eight cohorts show that ED patients have a worse PFS, and results of IPD meta-analysis and subgroup analysis support this finding. Analysis results of comprehensive OS and PFS illustrate that patients with ED have a worse survival outcome compared with patients with non-ED.
Similarly, we conducted another analysis of the random-effects model for PFS (Supplemental Figure 4), and obtained the same result. The values of τ2 and its standard error were also small, indicating low heterogeneity between studies.
Analyses results of independent risk factor
We performed independent risk factor analysis with OS and PFS as endpoints. The analysis included potential confounding factors such as age, sex, BMI, tumor stage, and the number of metastatic lesions. The results of the OS analysis showed that in the univariate analysis, ED status was significantly associated with OS (HR = 1.209, 95% CI: 1.158–1.346, p < 0.001). In the multivariable analysis, after adjusting for other confounders, ED status remained independently associated with OS (HR = 1.200, 95% CI: 1.107–1.301, p < 0.001). The PFS analysis showed similar findings, with ED status being a significant independent risk factor for survival both in the univariate analysis (HR = 1.186, 95% CI: 1.087–1.292, p < 0.001) and the multivariable analysis (HR = 1.137, 95% CI: 1.038–1.246, p = 0.006; Table 2).
Univariate and multivatiable analysis for OS and PFS.

ED, emotional distress; OS, overall survival; PFS, progression-free survival. *, p < 0.05; **, p < 0.01; ***, p < 0.001.
The impact of ED status on prognosis in patients
We divided patients into ED and non-ED group based on different scores on the scale as thresholds. The results showed that regardless of which threshold was chosen, the HR value was greater than 1, indicating that patients with ED faced a higher risk of death (Figure 4). The HR values range from 1.26 to 1.53, and the 95% CIs of all thresholds do not include 1, further demonstrating the independent prognostic value of ED status in survival outcomes.

HR and 95% CI across different score thresholds. This figure shows the hazard ratio (HR) and 95% confidence intervals (CI) for different score thresholds. The HR remains consistently greater than 1, indicating higher risk in the ED group.
Discussion
In this IPD meta-analysis encompassing 12 cohorts of 7 trials, we elucidate a significant association between ED and adverse survival outcomes among patients with NSCLC regardless of CT or ICIs. Compared with previous related studies, this study innovatively proposes that ED is a prognostic biomarker for NSCLC by incorporating a large amount of IPD.
Preclinical studies provide supplementary evidence for the association between ED and the prognosis of NSCLC in our research. Neuroendocrine regulation is currently the main mechanism used to explain the link between ED and tumor progression. 27 ED typically activates the hypothalamic–pituitary–adrenal (HPA) axis, promoting the synthesis and secretion of cortisol. Hara et al. 28 have confirmed that cortisol inhibits p53 function by increasing the activity of MDM2, thereby suppressing DNA damage repair and promoting tumor cell progression. Additionally, ED also activates the sympathetic nervous system (SNS) axis, leading to the secretion of catecholamines. Previous studies have shown that catecholamines can activate Ras-related protein 1 (Rap1) via the ®ARs-cAMP-PKA pathway, and inhibit extracellular signal-regulated kinases (ERKs), thereby promoting tumor cell migration, invasion, and growth. 29 These findings demonstrated that ED is directly associated with tumorigenesis and progression, rather than merely affecting tumor immunotherapy; thus, providing support for our research results.
In comparison to the methodologies employed by the STRESS-LUNG study, which utilized the Patient Health Questionnaire-9 (PHQ-9) and Generalized Anxiety Disorder 7-item (GAD-7) scale to assess ED status, our approach with EORTC QLQ-C30 presents a more streamlined and clinically feasible alternative for evaluating ED, as informed by the PRADO study. 2 This simplification enhances the applicability of our findings in clinical practice, empowering clinicians to assess the psychological well-being of patients throughout their treatment journeys.
Most clinical guidelines recommend the use of GAD-7 and PHQ-9 to assess anxiety and depression in cancer patients.30 –32 For patients admitted for the first time, the Distress Thermometer (DT) is often employed as a simple initial screening tool, and only those who screen positive undergo further evaluation with GAD-7 and PHQ-9. Although the EF scale of the QLQ-C30 is not a conventional instrument for diagnosing anxiety or depression specifically, previous studies have shown that the ED status derived from QLQ-C30 can also serve as a predictor of patient prognosis. Moreover, it requires only five questions to assess a patient’s status, making it more practical for clinical implementation. However, since QLQ-C30 is not a specific diagnostic scale, it remains uncertain whether ED identified through QLQ-C30 can be equated directly with clinically diagnosed anxiety or depression. Therefore, clinicians must still determine on a case-by-case basis whether a more specific psychological evaluation is warranted. Regardless of these limitations, there is no doubt that patients experiencing ED generally face a poorer prognosis.
Our statistical analyses further emphasize the importance of ED as a prognostic factor. Both univariate and multivariable analyses demonstrated that ED is an independent predictor of poor survival in NSCLC patients, even after adjusting for other clinical variables. This reinforces the role of ED as a significant and independent prognostic biomarker for survival outcomes, irrespective of the treatment received (CT or ICIs).
Moreover, our use of the EORTC QLQ-C30 allowed us to assess ED in a nuanced manner by stratifying patients based on different threshold scores. Regardless of the specific cutoff point used to define ED severity, the results consistently showed that ED is associated with worse survival outcomes. This finding underscores the robustness of ED as a prognostic factor, suggesting that even lower levels of ED are clinically meaningful and should be considered in the management of NSCLC patients.
Our findings indicate that patients with ED face a poor prognosis regardless of the treatment method employed. This suggests that clinicians should consider implementing early interventions during the diagnostic and therapeutic processes to either prevent the onset of ED or assist patients in overcoming it. First, regular assessments of a patient’s ED status should be conducted throughout the treatment process. For patients identified as having ED, more specific evaluations of depression and anxiety should be carried out, with priority given to addressing depressive symptoms. Commonly used treatment approaches include cognitive-behavioral therapy, mindfulness-based stress reduction, and psychosocial interventions.30,33 If a patient’s depressive symptoms persist or worsen, pharmacological treatment may be warranted. Furthermore, maintaining open and effective communication with both patients and their families throughout the treatment process is equally essential. 34
This study has some limitations. In our research, once the patient has taken atezolizumab, they will be assigned to the ICIs group, regardless of whether they have previously received other treatments. While limitations of our study have been addressed through the integration of a multi-institutional clinical trials, further prospective studies and preclinical studies are imperative to validate the relationship between ED status and clinical outcomes not only in NSCLC but also in other cancer types. Additionally, the method chosen in this study to define the ED state is not a traditional ED-specific measures, but a relatively simpler and easier to operate evaluation method, which may have some differences in the details of classifying the patients with ED. One other limitation of our study is that we did not formally assess the proportional hazards (PH) assumption underlying the Cox regression models, due to constraints in accessing the necessary residual diagnostics. While this limits our ability to confirm the time-invariance of HRs, several factors mitigate this concern. First, the main objective of our study is to evaluate whether patients with ED consistently exhibit worse survival outcomes than those with NED. This was confirmed under multiple cut-off values, with hazard ratios remaining directionally consistent. Second, visual inspection of Kaplan–Meier survival curves did not reveal any substantial crossing between groups, suggesting that violations of the PH assumption, if present, are unlikely to substantially alter our conclusions. Nonetheless, the absence of formal testing represents a methodological limitation, and the results should be interpreted with appropriate caution.
Conclusion
In conclusion, to our knowledge, this study is the first to propose correlation between ED and prognosis of NSCLC regardless of CT or ICIs. Therefore, we recommend the implementation of ED status assessment in clinical practice for NSCLC patients to improve their survival outcomes.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359251371826 – Supplemental material for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials
Supplemental material, sj-pdf-1-tam-10.1177_17588359251371826 for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials by Xin Li, Qisha Li, Su-Han Jin, Xiaofei Chen, Juanyan Shen, Hu Ma and Jian-Guo Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251371826 – Supplemental material for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials
Supplemental material, sj-pdf-2-tam-10.1177_17588359251371826 for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials by Xin Li, Qisha Li, Su-Han Jin, Xiaofei Chen, Juanyan Shen, Hu Ma and Jian-Guo Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_17588359251371826 – Supplemental material for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials
Supplemental material, sj-pdf-3-tam-10.1177_17588359251371826 for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials by Xin Li, Qisha Li, Su-Han Jin, Xiaofei Chen, Juanyan Shen, Hu Ma and Jian-Guo Zhou in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-4-tam-10.1177_17588359251371826 – Supplemental material for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials
Supplemental material, sj-pdf-4-tam-10.1177_17588359251371826 for Association between pretreatment emotional distress and survival outcomes in patients with advanced non–small-cell lung cancer: an individual patient data meta-analysis of 4632 patients in seven trials by Xin Li, Qisha Li, Su-Han Jin, Xiaofei Chen, Juanyan Shen, Hu Ma and Jian-Guo Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
