Abstract
Background:
The pretreatment prognostic nutritional index has been considered a potential prognostic biomarker in patients with non-small cell lung cancer (NSCLC), but this remains controversial. Therefore, we performed a meta-analysis to systematically assess the prognostic value of the prognostic nutritional index in patients with NSCLC.
Methods:
We systematically searched PubMed, EMBASE, Web of Science, and CNKI. The hazard ratios (HRs) with their corresponding 95% confidence intervals (CIs) were used to evaluate the link between the prognostic nutritional index and the oncological outcomes of patients with NSCLC, including overall survival, disease-free survival/recurrence-free survival, and progression-free survival.
Results:
Fifteen studies were included in this meta-analysis. Twelve of these studies explored the association between the prognostic nutritional index and the overall survival of patients with NSCLC. Our pooled analysis indicated that a low prognostic nutritional index was significantly related to adverse overall survival (HR 1.61; 95% CI 1.44, 1.81; P < 0.001). Our results also showed that the prognostic nutritional index was a negative predictor for disease-free survival/recurrence-free survival, and progression-free survival in patients with NSCLC.
Conclusion:
Our meta-analysis demonstrated that there was a close association between the prognostic nutritional index value and prognosis in NSCLC patients and that the prognostic nutritional index may act as a useful prognostic biomarker in NSCLC patients.
Introduction
Lung cancer is still an important cause of death among malignant neoplasms, which can be divided into non-small cell lung cancer (NSCLC) and small cell lung cancer (SCLC), with the proportion of the former being approximately 85%.1,2 Although much progress in early detection and treatment has been made in past decades, the 5-year survival rates of NSCLC patients remain unsatisfactory. 3
Numerous researchers have shown that nutrition and immune status are closely associated with tumor progression and prognosis.4,5 It was reported that the prognostic nutritional index (PNI), calculated by combining serum albumin levels and the total circulating lymphocyte count, 6 could reflect the nutritional and immunological status of patients with cancer. Recently, a body of studies showed that pretreatment PNI was correlated with the prognosis of patients with a variety of human malignancies, such as esophageal squamous cancer, colorectal cancer, gastric cancer, and lung cancer.7-12 However, most of the relevant studies about NSCLC were conducted with a small sample size, which limits the statistical power of their conclusions. Therefore, we performed this meta-analysis to provide more persuasive evidence that supports the prognostic significance of PNI in NSCLC.
Materials and methods
The present meta-analysis was undertaken according to the PRISMA statement issued in 2009. 13
Search strategy
Eligible studies were identified through searching PubMed, EMBASE, Web of Science, and CNKI from 1980 to April 29, 2018. The following search terms were used: “prognostic nutritional index,” “cancer or tumor or carcinoma or neoplasm or malignant or malignancy or adenocarcinoma,” and “lung or pulmonary.”
Eligibility criteria
Studies meeting the following criteria were included: (a) the relationships between PNI and survival of NSCLC patients were analyzed; (b) the literature was published in Chinese or English; and (c) the full text was available for extracting necessary data.
The criteria for the exclusion of studies were: (a) reviews, letters, comments, case reports, animal studies, and meeting abstracts; and (b) studies that did not provide hazard ratios (HRs) and confidence intervals (CIs) that assessed the relationships between PNI and the survival of NSCLC patients, or did not present sufficient data for directly calculating HRs.
Data extraction
Data were independently extracted by two investigators. Any incompatibility regarding data extraction between the investigators was resolved by discussion among the two reviewers and the other authors. The extracted data included the first author’s name, country, recruitment time, median age, number of patients, clinical stage, treatment type, cut-off value, follow-up duration, treatment type, and HRs with their 95% CIs for overall survival (OS), disease-free survival/recurrence-free survival (DFS/RFS), or progression-free survival (PFS).
Quality assessment
The methodological quality of each included study was evaluated using the Newcastle-Ottawa Scale (NOS) system with total scores that varied from 0 to 9. 14 In this meta-analysis we used a score of no less than 6 as the threshold for high-quality studies, and those with 5 points were considered moderate quality.
Statistical analysis
All statistical processes were conducted using STATA version 12.0 (Stata Corporation, College Station, Texas, USA). The association between low PNI and prognosis was assessed using HRs and their corresponding 95% CIs. Heterogeneity was evaluated using Cochran’s Q and Higgins I2 statistics, and we regarded P < 0.05 and I2 > 50% to indicate significant heterogeneity in this meta-analysis. When there was significant heterogeneity, HRs were subjected to pooling analysis using a random-effects model, and a fixed-effects model was applied to pool the data. When the HR (low/high PNI) was more than 1, and its corresponding 95% CI did not overlap 1, we considered that a low PNI was associated with a poorer prognosis. According to the country, the number of patients, the tumor, node, metastasis (TNM) stage, the cut-off value, the method for determining cut-off value, and the treatment type, we undertook subgroup analyses to test the robustness of the pooled HR for OS. Additionally, a sensitivity analysis was carried out to further verify the robustness of our pooled result for OS by omitting one study in each step. The Begg’s and Egger’s tests were used to assess the extent of publication bias.15,16 When the Begg’s funnel plot was asymmetrical or the P value of the Egger’s test was less than 0.05, we considered that there was significant publication bias. When there was significant publication bias, we applied a trim-and-fill method to assess the effects of publication bias on the reliability of our pooled result for OS. 17 A P value less than 0.05 indicated statistical significance.
Results
Study characteristics
The detailed process of literature selection is presented in Figure 1. During the initial search, we identified 126 potentially relevant studies. After excluding 23 duplicated studies, the titles and abstracts of the remaining 103 studies were further screened, and 77 studies were excluded: 4 SCLC reviews, 26 conference abstracts and comments, and 47 irrelevant studies. Subsequently, 26 publications remained for full-text screening, and 11 articles were further excluded for lack of sufficient data (n = 6), enrolling duplicated cases (n = 3), and not providing HRs from multivariate analysis (n = 2). Finally, based on the inclusion and exclusion criteria mentioned above, 15 studies were included in this meta-analysis.2,18-31
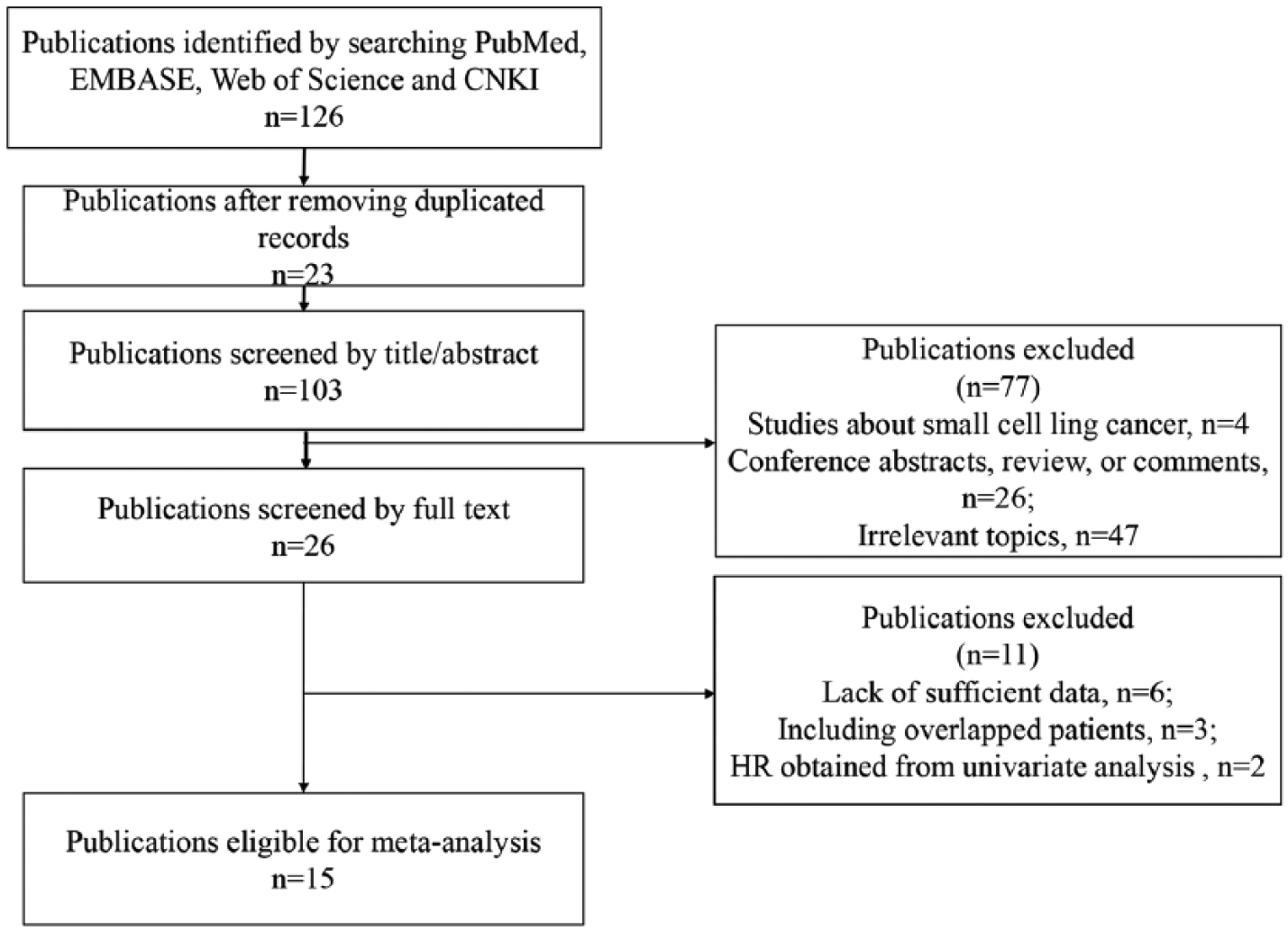
Flow diagram of literature selection process.
The main characteristics of the 15 eligible studies are presented in Table 1. Of all the included studies, seven were performed in China, six were conducted in Japan, and one study each was performed in Turkey and Korea. In addition, according to the NOS, all the included studies had a score of 6 or more, indicating that they were high quality.
The main characteristics of the included studies.
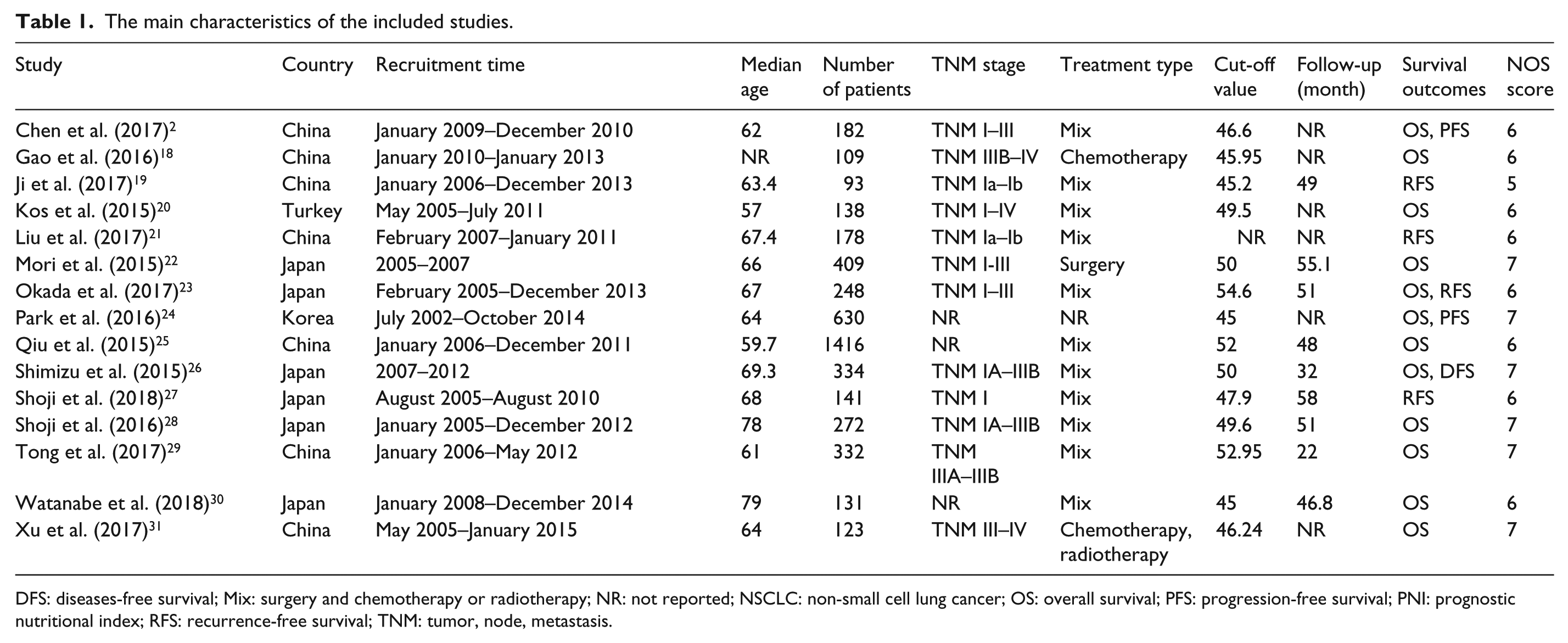
DFS: diseases-free survival; Mix: surgery and chemotherapy or radiotherapy; NR: not reported; NSCLC: non-small cell lung cancer; OS: overall survival; PFS: progression-free survival; PNI: prognostic nutritional index; RFS: recurrence-free survival; TNM: tumor, node, metastasis.
Meta-analysis
Correlation between PNI and OS in NSCLC
Twelve studies provided HRs for OS of patients with NSCLC.2,18,20,22-27,29-31 The fixed-effects model was used to combine the HRs for OS. The result revealed that a lower preoperative PNI was closely associated with worse OS of NSCLC patients (HR 1.61; 95% CI 1.44, 1.81; P < 0.001, Figure 2).
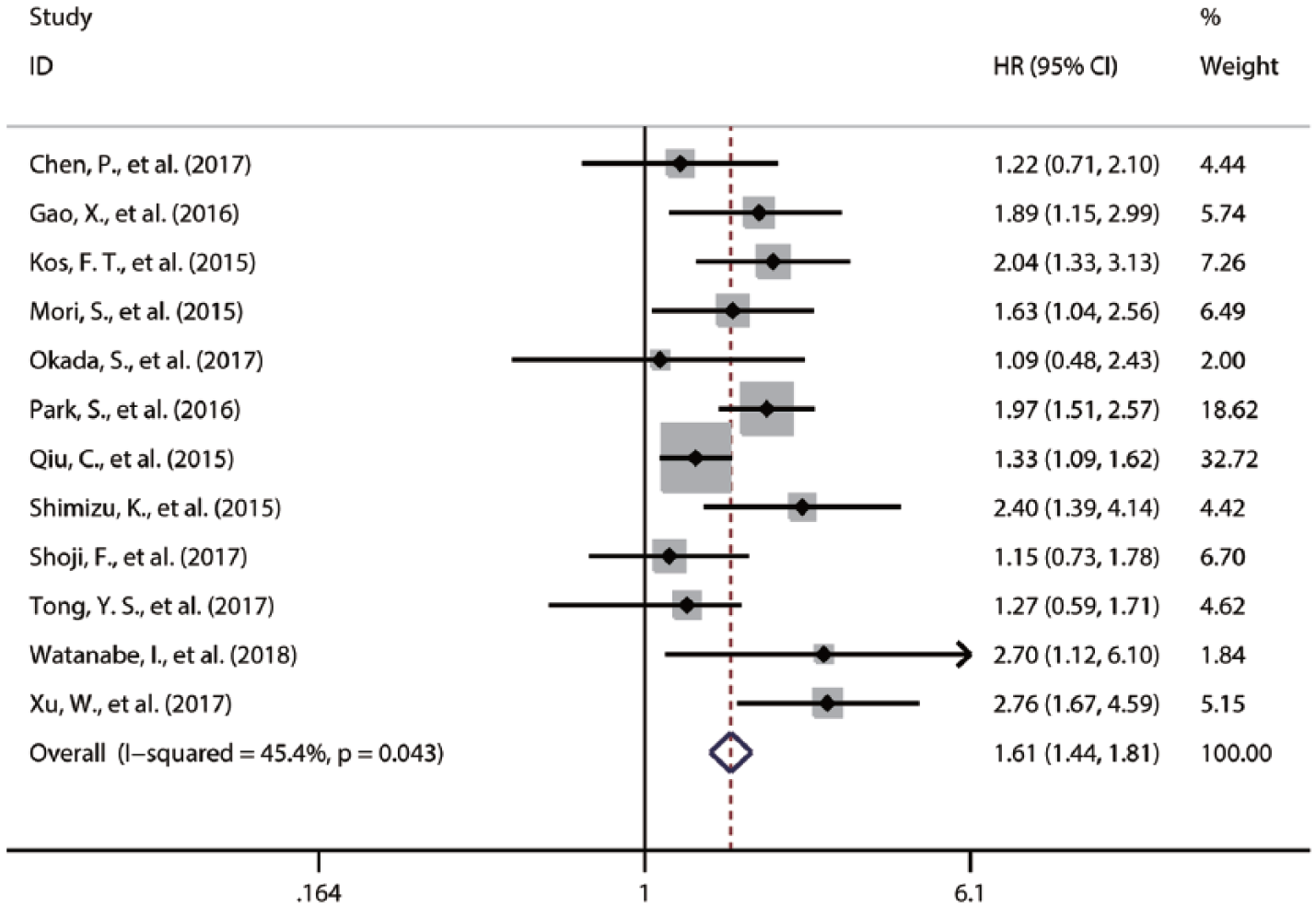
Forest plot of the HR assessing the correlation between the PNI and OS in NSCLC patients. HR: hazard ratio; NSCLC: non-small cell lung cancer; OS: overall survival; PNI: prognostic nutritional index.
Considering the potential effects of heterogeneity on our pooled result for OS, we performed subgroup and sensitivity analyses to confirm the reliability of this result. From the results of subgroup analysis, we still observed a significant association between a lower PNI and worse OS in each subgroup (Figure 3). In addition, our sensitivity analysis showed that there were no significant fluctuations for the pooled HR for OS (Figure 4). Taken together, these results demonstrated that our pooled result for OS was reliable.
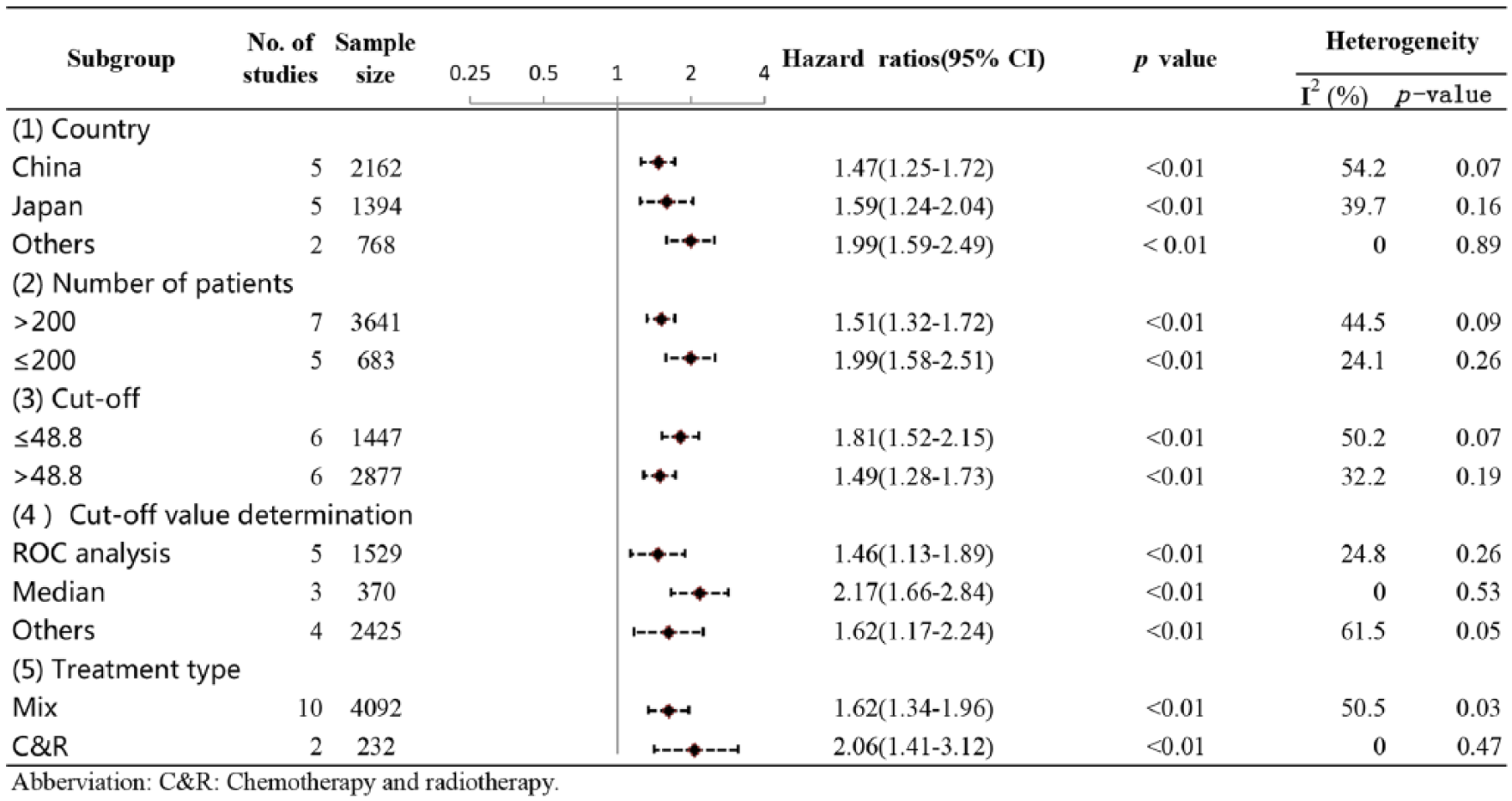
Subgroup analyses for exploring the sources of heterogeneity of the pooled HR for OS in NSCLC patients. HR: hazard ratio; NSCLC: non-small cell lung cancer; OS: overall survival.
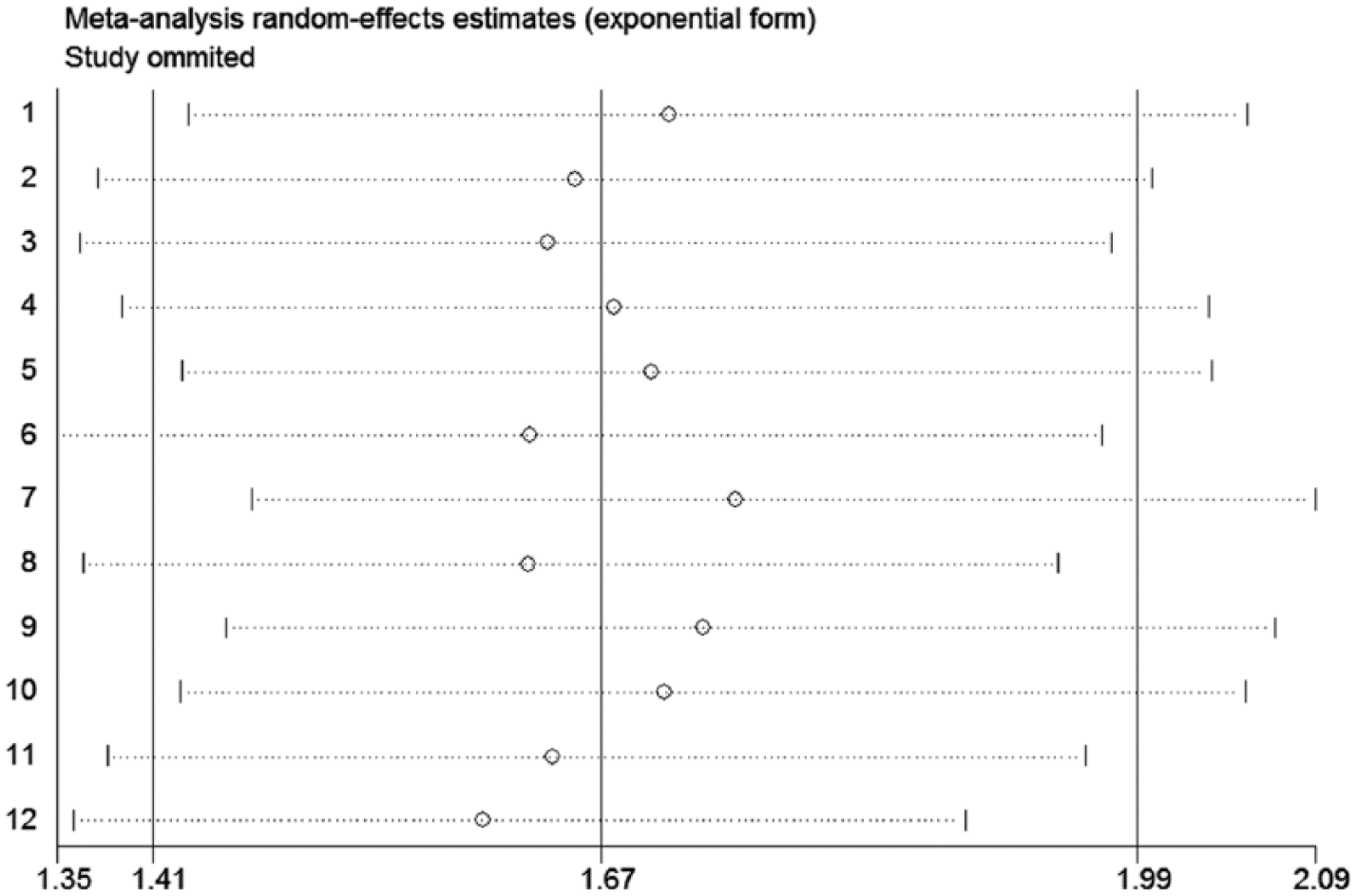
Sensitivity analyses for testing the robustness of the pooled HR for OS in NSCLC patients. HR: hazard ratio; NSCLC: non-small cell lung cancer; OS: overall survival
Correlation between PNI and DFS/RFS in NSCLC
Five studies investigated the relationship between PNI and DFS/RFS.19,21,23,26,28 As significant heterogeneity (I2 = 72.7%; P = 0.005) was detected, the random-effects model was used to pool the HRs for DFS/RFS. The results revealed an obvious correlation between a lower PNI and worse DFS/RFS (HR 2.27; 95% CI 1.40, 3.69; P < 0.01; Supplement 1).
Correlation between PNI and PFS in NSCLC
We successfully extracted available data from only three included studies that referred to the correlation between PNI and PFS.2,18,24 As no significant heterogeneity (I2 = 0.00%; P = 0.781) was observed, we applied the random-effects model to pool the HRs for PFS. The results indicated that a lower PNI was significantly linked with a more unfavorable DFS/RFS (HR = 1.52; 95% CI 1.26, 1.83; P = 0.002; Supplement 2).
Publication bias
The Begg’s and Egger’s tests were used to assess publication bias. The symmetry of the funnel plot of the Begg’s tests indicated that there was no significant bias for the pooled HR for OS (Supplement 3), which was confirmed by the P values of the Begg’s and Egger’s tests (Begg’s test, P = 1.000; Egger’s test, P = 0.365). However, the assessment of publication bias was not available for the pooled HRs for DFS/RFS and PFS owing to the limited number of eligible studies exploring the relationship of PNI to DFS/RFS and PFS.
Discussion
A body of studies has suggested that low PNI correlated with the adverse prognosis of patients with various cancers, including NSCLC. Nevertheless, no meta-analyses have been performed to systematically assess the association between PNI and survival of NSCLC patients. Therefore, we performed a systematic review and a meta-analysis to validate the notion that a lower PNI predicts a worse prognosis in NSCLC patients.
In our meta-analysis, 15 eligible studies investigating the correlation of PNI with the prognosis of patients with NSCLC were included. The data on OS, DFS/RFS, and PFS of the individual studies were pooled for statistical analysis. Overall, we confirmed the concept that a lower PNI is a predictor of worse OS, DFS/RFS, and PFS in patients with NSCLC. The prognostic role was not undermined by subgroup analysis based on country, sample size, cut-off value, cut-off value determination, and treatment type. Furthermore, our sensitivity analyses demonstrated the robustness of the pooled result for OS. Given these data, PNI is a promising prognostic factor in patients with NSCLC.
There are some potential mechanisms for the inverse association of PNI value and survival in NSCLC patients. First, PNI reflects immunological and nutritional status, which has been associated with the survival of patients with cancer. 32 Second, it is well known that many clinicopathological features, such as clinical stage, lymph node metastasis, and histological type, are closely associated with prognosis of patients with cancer. Our results indicated that a low PNI was associated with more advanced TNM stages, implying that low PNI correlated with tumor progression, and thus worsened the survival of NSCLC patients. Third, emerging evidence demonstrated that poor nutritional and immune conditions were significantly associated with postoperative complications.33,34 Moreover, poor immune and nutritional conditions may result in the delay of postoperative adjuvant therapy, which therefore may also partly explain the association between a low PNI and poor survival of patients with NSCLC.
This meta-analysis provided evidence for the prognostic significance of PNI in patients with NSCLC. However, some limitations in our meta-analysis need to be emphasized. First, although this study demonstrated an association between PNI and survival in NSCLC patients by pooling the data of previously published literature, only 12 of the included studies provided available data for synthetically analyzing the link between PNI and OS, and many less eligible studies reported data regarding DFS/RFS and PFS. Thus, in future, more well-designed studies enrolling more patients should be conducted to validate our findings. Second, this meta-analysis included a cluster of heterogeneous patients owing to the limitations of the included studies, which may affect the reliability of our pooled results. The heterogeneity among the patients may partly stem from disease stage or treatment type. Additionally, all the included studies referred only to Asian populations. Therefore, it is imperative to conduct ad hoc studies to explore the prognostic value of pretreatment PNI in NSCLC patients with different backgrounds. Third, only studies published in English and Chinese were included, which might introduce a degree of bias. Fourth, all the included studies were retrospective observational studies, which might also result in uncontrolled bias and heterogeneity. Fifth, the cut-off values for PNI differed across the included studies, and an optimal cut-off value is needed for guiding clinical decisions.
In conclusion, our meta-analysis demonstrated that there was a close association between PNI value and survival of NSCLC patients, and that PNI may act as a useful prognostic biomarker in NSCLC patients.
Supplemental Material
Supplemental_Figures – Supplemental material for Prognostic value of pretreatment prognostic nutritional index in non-small cell lung cancer: A systematic review and meta-analysis
Supplemental material, Supplemental_Figures for Prognostic value of pretreatment prognostic nutritional index in non-small cell lung cancer: A systematic review and meta-analysis by Yuanyuan Hu, Jie Shen, RuiKe Liu, ZhiMei Feng, ChangNing Zhang, Li Ling and LiBo Chen in The International Journal of Biological Markers
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
