Abstract
BACKGROUND:
Long noncoding RNA (lncRNA) are critical regulators of tumor progression.
OBJECTIVE:
To determine how the lncRNA membrane associated guanylate kinase, WW and PDZ domain-containing 2 (MAG12) antisense RNA 3 (MAGI2-AS3) and the phosphatase and tensin homolog (PTEN) gene function in regulating bladder cancer (Bca) progression.
METHODS:
Total RNA from 80 Bca tissues and 30 paired para-cancerous tissues from patients was sequentially extracted, quantified, purified, and reverse transcribed using RT-PCR. A library was constructed and sequenced. Four Bca cell lines and a normal urothelial cell line were transfected with lentiviral plasmids, and cell migration and invasion were assayed in vitro. An orthotopic mouse model of Bca was created for in vivo studies.
RESULTS:
MAGI2-AS3 expression was significantly downregulated in Bca, compared with normal tissues, and negatively associated with tumor stage and a poor prognosis. MAGI2-AS3 and its sense RNA MAGI2 showed significant and positive correlation. The expression of MAGI2 and its downstream gene, PTEN, increased in Bca cells overexpressing MAGI2-AS3, and interference by MAGI2 expression reversed the migration and invasion inhibited by MAGI2-AS3 overexpression.
CONCLUSION:
MAGI2-AS3 overexpression inhibited Bca cell progression by regulating the MAGI2/PTEN/epithelial-mesenchymal transition, offering novel insights into the mechanism of Bca progression.
Introduction
Bladder cancer (Bca) is one of the most prevalent genitourinary tumors in men [1], among which approximately 75% are non-muscle invasive (NMIBC) and approximately 25% are muscle-invasive (MIBC) Bca [2]. The standard treatment for MIBC and high risk NMINC is radical cystectomy (RC) [2]. However, tumors in approximately 50% of the patients with MIBC who undergo RC progress with local recurrence or distant metastases [3]. Moreover, the median survival rate for patients with metastatic Bca rarely exceeds 6 months [4]. Thus, the overall prognosis of patients with Bca remains unsatisfactory. Therefore, a better understanding of the molecular mechanisms and the identification of biomarkers involved in Bca progression are urgently needed.
Long non-coding (lnc) RNA are
Here, we analyzed the expression profiles of MAGI2-AS3 and its sense RNA MAGI2 in Bca tissues, and analyzed the correlations between the MAGI2-AS3 expression and clinical-pathological variables as well as prognosis. Moreover, we explored the functional importance and the detailed mechanisms of MAGI2-AS3 that influence Bca progression, and determined the significant correlation between MAGI2-AS3 and MAGI2 expression.
Materials and methods
Clinical samples
The Ethics Committee at the Affiliated Hospital of Jining Medical University approved the study, which conformed to The Code of Ethics of the World Medical Association (Declaration of Helsinki, 2013 amendment). All patients provided written, informed consent to collect resected tissue samples for research use. In all, 80 Bca tissues and 30 paired para-cancerous tissues, respectively, were obtained from patients diagnosed with Bca between March 2010 and May 2015, at the Department of Urology, Affiliated Hospital of Jining Medical University. All samples were immediately stored at
High-throughput transcriptome sequencing
Total RNA was sequentially extracted, quantified, and purified, and the integrity was confirmed. Total RNA was enriched using magnetic beads with oligo (dT) (New England BioLabs, Inc., Ipswich, MA, USA) after digesting DNA with DNase I (Bio-Rad Laboratories, Inc., Hercules, CA, USA). Short fragments that dissociated from the total RNA were sublimated and resolved in Elution buffer (Thermo Fisher Scientific, Inc., Waltham, MA, USA). Appropriate templates were amplified using OneTaq
Cell culture and transfection of lentiviral plasmids
The Bca cell lines, T24, J82, 5637, RT4, and the normal urothelial cell line, SV-HUC, (Cell Bank of the Chinese Academy of Sciences, Shanghai, China) were respectively cultured in RPMI-1640 and L-15 media (both: Invitrogen, Carlsbad, CA, USA containing 1% ampicillin (100 units/mL), streptomycin (100 units/mL), and 10% (v/v) fetal bovine serum (FBS) at 37
Quantitative real-time PCR
Total RNA was extracted from bladder tissues and cell samples using TRIzol™ Reagent (Thermo Fisher Scientific Inc., MA, USA) as described by the manufacturer. Vimentin, MAGI2-AS3, Snail1, E-cadherin, and N-cadherin expression was quantified using RT-qPCR and SYBR select master mix kits (Thermo Fisher Scientific Inc.) Expression levels of these genes were normalized to that of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Table S1 lists the primers for all genes. Data are presented as means of fold change compared with an internal reference.
Migration and invasion assays
Cell migration and invasion were determined using Transwell chambers. Chambers for invasion assays were inserted into Matrigel (Millipore Sigma Co., Ltd., Burlington, MA, USA), and activated under pure media at 37
Mouse model of orthotopic Bca
Six 6-week-old female NOD/SCID mice (Shanghai Laboratory Animal Center, SLAC, Shanghai, China) were anesthetized via an intraperitoneal injection of 10% Nembutal (45 mg/kg), then an indwelling catheter was placed in the mice with paraffin oil. Residual urine in bladders was removed, then the bladders were washed twice with PBS, and flushed with 30
Western blotting
Protein expression was quantified by western blotting. All samples were adjusted to a protein concentration of 1 g/L using the bicinchoninic acid (BCA) method, then 5
Immunohistochemistry (IHC)
Paraffin-embedded tissue samples from orthotopic Bca models were assessed by immunohistochemistry (IHC) using polyclonal antibodies against MAGI2 (diluted 1:500; Thermo Fisher Scientific Inc.) and PTEN (1:50; Abcam), followed by a standard IHC protocol. Three pathologists independently evaluated staining intensity and the proportions of positively stained tumors.
Statistical analyses
All data were statistically analyzed using GraphPad Prism version 5.0 (GraphPad Software In., San Diego, CA, USA) and SPSS version 17.0 (SPSS, Inc., Chicago, IL, USA). Data are presented as means
Long non-coding RNA MAGI2-AS3, MAGI2, and PTEN expression in bladder cancer tissues determined by RNA-seq. (a) Heatmap shows differentially expressed lncRNA in RNA-seq data. Red box, expression profile of n410887 (MAGIS-AS3). Histograms show significant differences in MAGI2-AS3 (b) and MAGI2 (c) and PTEN (d) among three tissues. **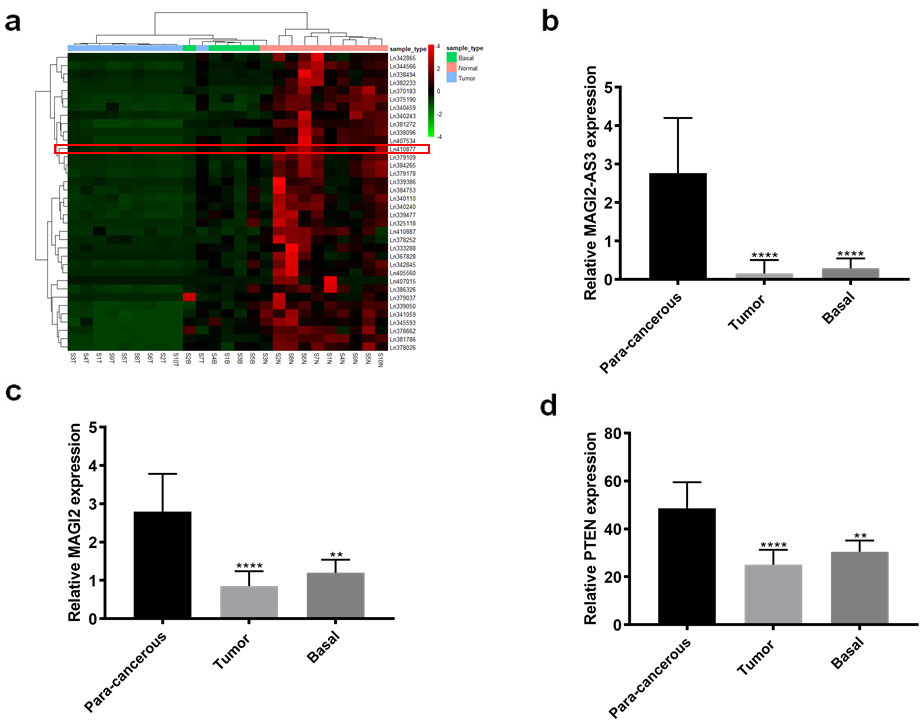
Expression of lncRNA MAGI2-AS3 is downregulated in Bca tissues and cell lines
The results of RNA-seq data from 10 paired of Bca and para-cancerous tissues, and 5 base tissues, showed that 36 lncRNA were significantly downregulated by at least 3-fold in Bca, compared with para-cancerous and corresponding base tissues. These downregulated lncRNA included LINC01082 (ln375190), ADAMTS9-AS2 (ln384265), and ADAMTS9-AS1(ln339386).MAGI2-AS3, defined as n410887 in the NONCODE database was also differentially expressed (Fig. 1a and b). Like that of MAGI2-AS3, expression of the sense RNA, MAGI2 (Fig. S1), and its downstream gene, PTEN, was also downregulated in Bca tissues (Fig. 1c and d). Thus, we speculated that MAGI2-AS3 correlated with its sense RNA MAGI2 in Bca. Consequently, further investigated MAGI2-AS3.
We confirmed the expression profiles of lncRNA MAGI2-AS3 in 80 Bca and 30 paired para-cancerous normal tissues by qPCR. The expression levels of MAGI2-AS3 were significantly lower in Bca, than in normal tissues (Fig. 2a). Similarly, the expression of MAGI2 and PTEN in tissues was consistent with the RNA-seq data; expression was downregulated in cancer, compared with normal tissues (Fig. 2b and c). Furthermore, expression of the three genes was obviously higher in NMIBC, than in MIBC tissues (Fig. 2d–f).
Expression profiles of lncRNA MAGI2-AS3, MAGI2, and PTEN in enlarged bladder cancer tissues validated by qRT-qPCR. Expression of (a) MAGI2-AS3, (b) MAGI2 and (c) PTEN is significantly decreased in Bca, compared with adjacent non-tumorous bladder tissues. Expression of (d) MAGI2-AS39, (e) MAGI2 and (f) PTEN is much lower in NMIBC, than in MIBC tissues. *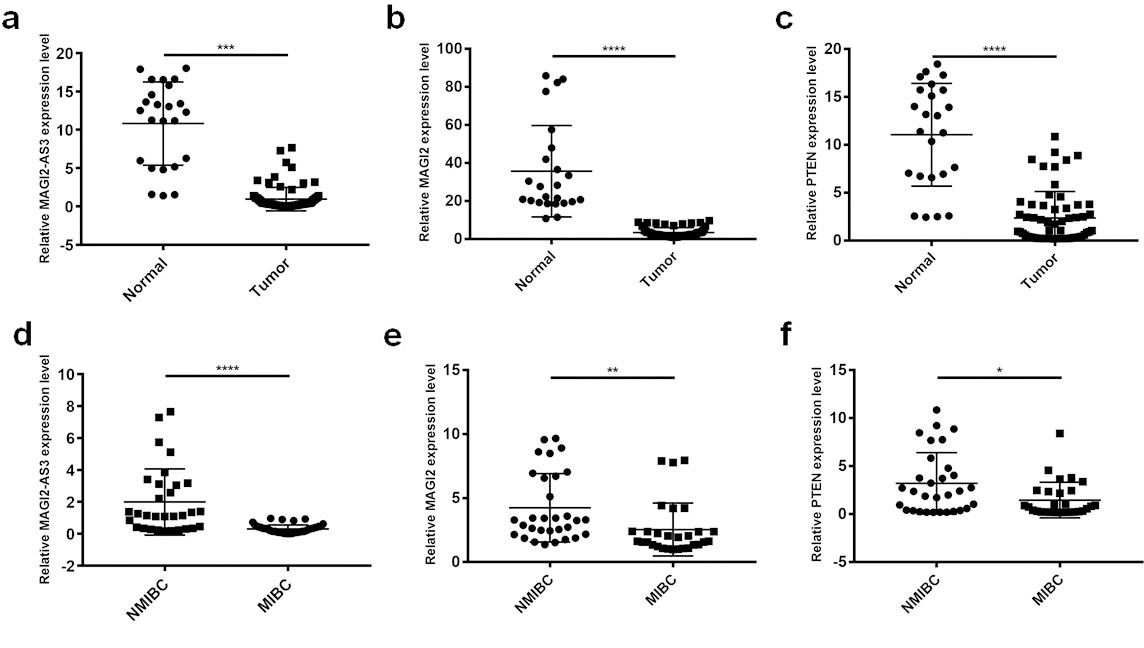
We classified the 80 patients with Bca into groups with high (
Associations between lncRNA MAGI2-AS3 expression and prognosis of patients with bladder cancer. Kaplan-Meier curves show differences in overall survival between patients with high and low lncRNA MAGI2-AS3 expression (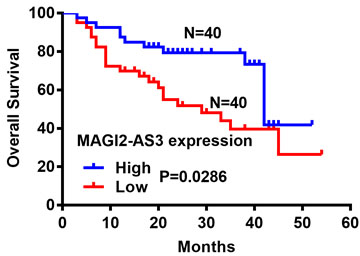
The expression of MAGI2-AS3 and MAGI2 mRNA significantly correlated (Fig. 4a). Moreover, expression of the latter positively correlated with that of PTEN mRNA (Fig. 4b). Subsequent qPCR results verified these correlations among the three genes (Fig. 4c and d). Analysis using the GEPIA webtool in TCGA datasets revealed that MAGI2-AS3 expression positively correlates with MAGI2 expression (Fig. 4e). The correlation between MAGI2 and PTEN was not statistically significant, as trends in expression were the same for both genes (Fig. 4f). Thus, we investigated whether MAGI2-AS3 influences Bca progression by regulating the sense strand RNA MAGI2 and its downstream gene PTEN expression.
Correlations between clinical-pathological characteristics and relative expression of MAGI2-AS3 in Bca tissues from 80 patients
Correlations between clinical-pathological characteristics and relative expression of MAGI2-AS3 in Bca tissues from 80 patients
Correlations between expression of MAGI2-AS3 and MAGI2, and MAGI2-AS3 and PTEN. (a) Significantly positive correlation between MAGI2-AS3 and MAGI2 expression (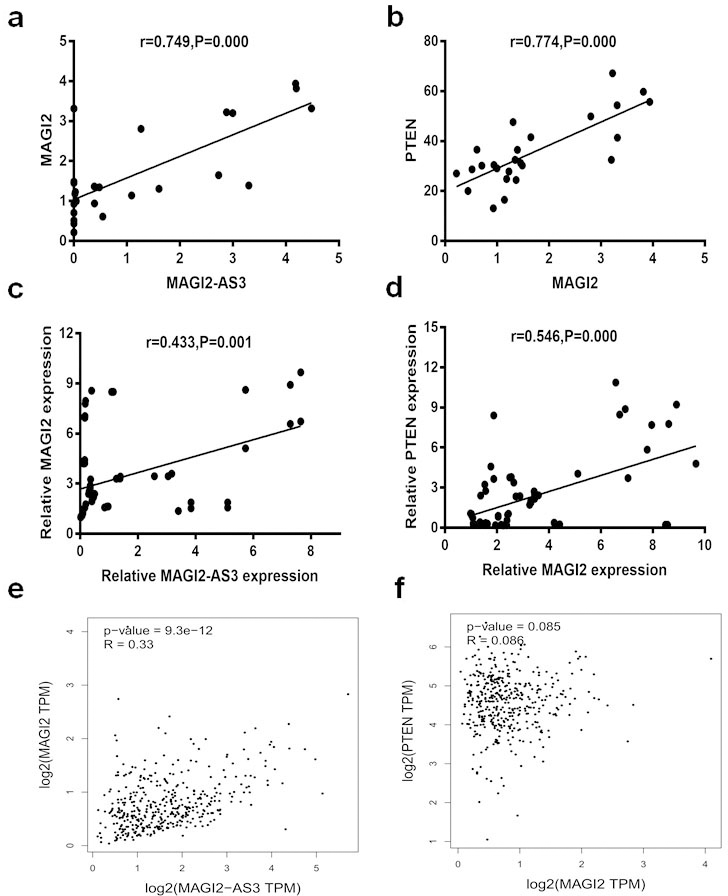
We applied western blotting and qPCR assays to determine changes in MAGI2 and PTEN expression while overexpressing MAGI2-AS3 in T24 and J82 Bca cells in vitro. WE found that MAGI2 and PTEN expression was upregulated when MAGI2-AS3 expression was increased (Fig. 5a and b). Metastatic sites occurred in the abdomen of orthotopic Bca models after 10 weeks in the T24-LV-NC, but not in the T24-LV-MAGI2-AS3 group (Fig. 5c). The IHC findings of the resected tumors revealed more MAGI2 and PTEN protein in the T24-LV-MAGI2-AS3, than the T24-LV-NC group (Fig. 5d). Furthermore, transwell assays showed significantly higher migration and invasion capacity in T24 Bca cells overexpressing MAGI2-AS3 together with interfering MAGI2, compared with T24 Bca cells overexpressing only MAGI2-AS3 (Fig. 6a). These findings indicated that the expression of interfering MAGI2 reversibly inhibited the migration and invasion capacity induced by overexpressing MAGI2-AS3. Finally, the results of western blotting and qPCR assays showed significantly downregulated MAGI2 and PTEN expression in the co-transfected group compared with the group overexpressing only MAGI2-AS3 (Fig. 6b and c).
MAGI2-AS3 regulates MAGI2 and PTEN expression. (a) Western blots show more MAGI2 and PTEN protein expression in LV-MAGI2-AS3 than in LV-NC groups. (b) Obviously upregulated expression of MAGI2 and PTEN in LV-MAGI2-AS3, compared with LV-NC groups validated by RT-qPCR. (c) Distant metastatic sites are more likely to arise in T24-LV-NC, than T24-LV-MAGI2-AS3 mouse models of orthotopic bladder cancer. (d) MAGI2 and PTEN proteins are more intensely stained according to IHC assays in LV-MAGI2-AS3, than LV-NC models of orthotopic bladder cancer (magnification: 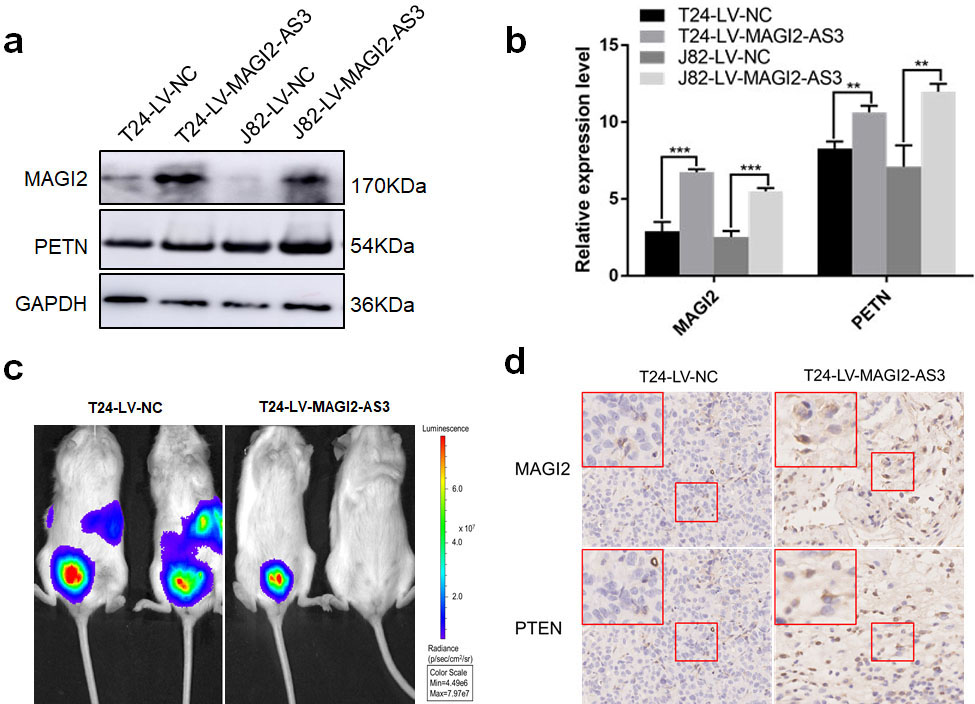
Functional correlation between MAGI2-AS3 and MAGI2 in terms of migration and invasion capacity of T24 Bca cells. (a) Findings of transwell migration and invasion assays show that fewer cells overexpressing (OE) MAGI2-AS3 combined with interfering MAGI2-AS3+si MAGI2 passed through polycarbonate membrane those OE MAGI2-AS3. (b) Western blots show significantly downregulated MAGI2 and PTEN expression in OE MAGI2-AS3+si MAGI2 than OE MAGI2-AS3. (c) Findings of RT-qPCR show significantly downregulated MAGI2 and PTEN expression in OE MAGI2-AS3+si MAGI2, than in OE MAGI2-AS3 group. *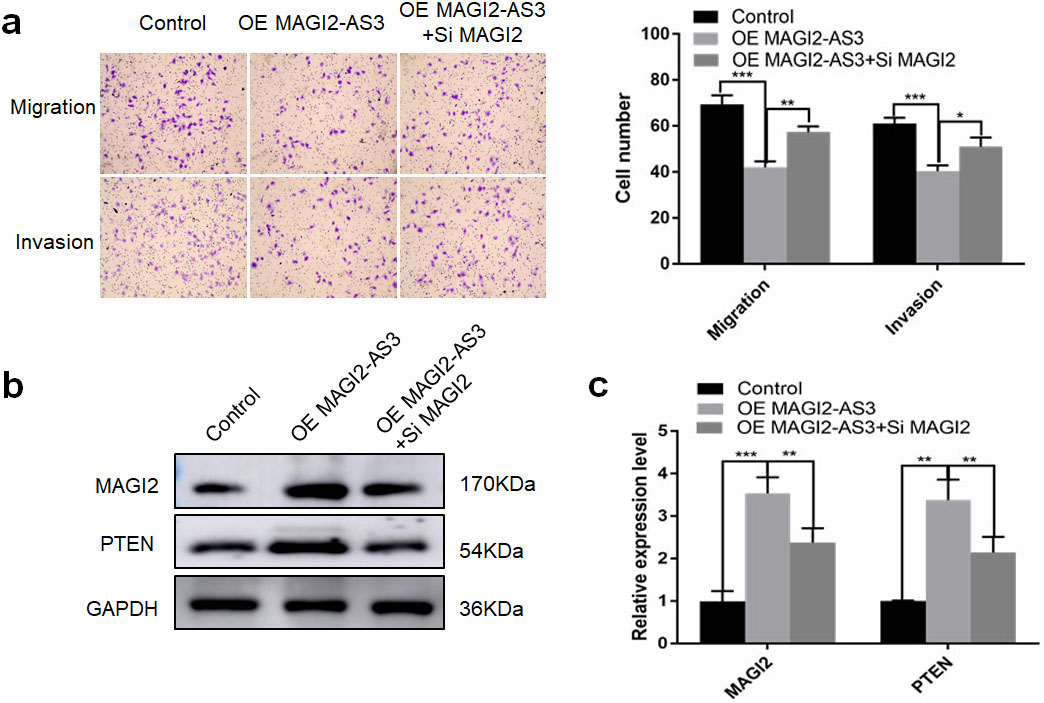
Inactivating PTEN induces the EMT process and tumor metastasis [17]. Furthermore, gene ontology (GO) enrichment analysis of RNA-seq data revealed that differentially expressed genes associated with MAGI2-AS3 were significantly enriched on the extracellular matrix (Fig. S2), and that protease recruitment was mostly caused by EMT activation [18]. Hence, lncRNA MAGI2-AS3 probably inhibits Bca progression by inducing EMT inactivation. We therefore selected the vital genes Vimentin, Snail1, E-cadherin, and N-cadherin that are involved in the EMT pathway, and measured their protein levels expressed in LV-MAGI2-AS3 and LV-NC groups in both cell lines by western blotting. The expression of vimentin, Snail1, and N-cadherin was much lower in the LV-MAGI2-AS3, than the LV-NC groups, whereas that of E-cadherin was obviously enhanced (Fig. 7a). The mRNA expression of the corresponding genes detected using qRT-PCR showed much more Vimentin, Snail1, and N-cadherin mRNA expression in the LV-MAGI2-AS3, than the LV-NC groups, while E-cadherin mRNA expression was significantly downregulated in the LV-MAGI2-AS3 groups (Fig. 7b and c). Changes in gene expression on the background of MAGI2-AS3 overexpression suggested inhibition of the EMT pathway.
Protein and mRNA expression of EMT-associated genes in LV-MAGI2-AS3 and LV-NC groups detected by western blotting and RT-qPCR. (a) Band intensities of vimentin, Snail1, and N-cadherin proteins are significantly lower in LV-MAGI2-AS3, than LV-NC groups in both cell lines, whereas E-cadherin protein expression is obviously enhanced. (b) Relative Vimentin, Snail1, and N-cadherin mRNA expression is significantly decreased in the T24-LV-MAGI2-AS3 compared with T24-LV-NC group. More E-cadherin mRNA is expressed in the T24-LV-MAGI2-AS3 group. (c) Relative expression of vimentin, Snail1, and N-cadherin mRNA expression is significantly decreased in J82-LV-MAGI2-AS3 than J82-LV-NC group. More E-cadherin mRNA is also expressed in J82-LV-MAGI2-AS3 group.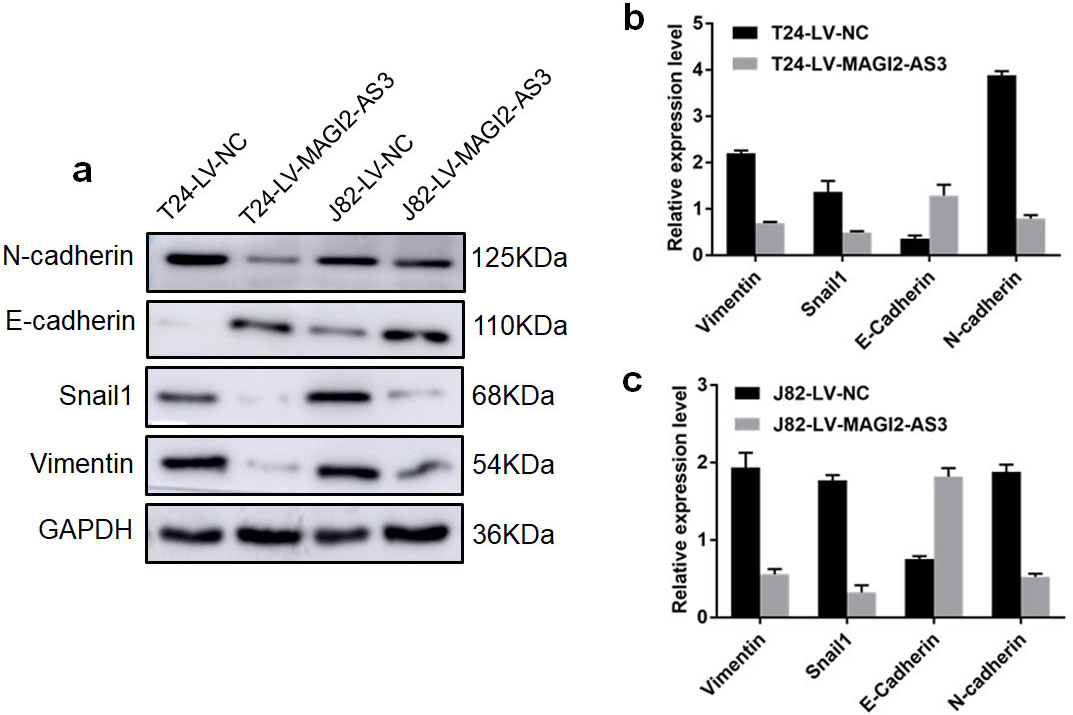
Long non-coding RNA can regulate changes in gene expression in cancer development by recruiting chromatin-modifying complexes and directly targeting transcriptional or posttranscriptional changes [19]. Indeed, many upregulated and downregulated lncRNA play crucial roles in carcinogenesis and Bca metastasis. For example, high levels of UCA1 are expressed in Bca tissues, but not in non-neoplastic cancers [21, 20]. In contrast, the imprinted maternally expressed gene 3 (MEG3) encodes an lncRNA that not found in Bca tissues; it might serve as a tumor suppressor to inhibit Bca proliferation and invasion through MEG3-induced apoptosis and increased PHLPP2 protein translation [23, 22]. MAGI2-AS3 was initially identified in Bca as a tumor suppressor [10], as its overexpression inhibited the proliferation, migration and invasion of BCa cells [14]. The present study investigated how MAGI2-AS3 regulates Bca progression. We found significantly downregulated MAGI2-AS3 expression in Bca tissues compared with adjacent non-tumorous tissues, and that its expression negatively correlates with number of tumors, grade, stage, and prognosis. We discovered that MAGI2-AS3 overexpression might improve the expression of the sense strand RNA MAGI2 and its downstream gene, PTEN, and subsequently enhance the active EMT process, leading to the inhibition of Bca progression.
Indeed, the function of MAGI2-AS3 in tumors is inconsistent. Some investigators have indicated that MAGI2-AS3 functions in promoting cervical squamous cell carcinoma (CSCC) and gastric cancer (GC); a higher expression level of which reflects an increased capacity for more aggressive malignant progression and, to some extent, a shorter OS [26, 24, 25]. However, antagonistic roles have been validated in most solid tumors, including Bca [28, 27, 10], HCC [29, 12], and NSCLC [11, 30]. The low expression of MAGI2-AS3 was closely associated with tumor progression and metastasis, and overexpression obviously inhibited cancer cell proliferation, migration, and invasion. Recent studies have generally focused on the MAGI2-AS3 pathway in which suppressing the ceRNA sponging of some miRNA could interfere with the expression of tumor-associated genes. For example, the MAGI2-AS3/miR-374b-5p/SMG1 axis is suppressed during HCC progression [12], and MAGI2-AS3 regulates CCDC19 expression by sponging miR-15b-5p, thus, suppressing Bca progression [15]. The present study also found that MAGI2-AS3 plays an anti-cancer role in Bca. More importantly, based on the correlation between MAGI2-AS3 and MAGI2, we discovered that MAGI2-AS3 overexpression positively regulates the expression of MAGI2 and its downstream gene, PTEN, which is an important regulator for EMT activation. Such positive regulation of PTEN might lead to the inhibition of migration and invasion of Bca cells.
The EMT has been regarded as the critical process for the development and metastasis of various tumors including Bca [31]. Several lncRNA stimulate the EMT process [32]. MAGI2-AS3 was originally identified as an EMT-related lncRNA in gastric cancer. Ectopic MAGI2-AS3 promotes tumor progression by sponging miR-141/200a and maintaining ZEB1 overexpression [26]. However, whether MAGI2-AS3 regulates the EMT as a tumor suppressor remains poorly understood. Studies initially confirmed that suppressing MAGI2 in tumors destabilizes PTEN in cancer cells [34, 35, 33]. The inactivation of PTEN induces EMT and tumor metastasis [17, 36, 38, 37]. Overexpressed MAGI2-AS3 inhibits breast cancer progression by decreasing miR-374a and enhancing PTEN expression, and the MAGI2-AS3/miR-374a/PTEN axis is important for regulating tumor progression [28]. Furthermore, the target of miRNA-23a-3p is PTEN [30], and miRNA-23a-3p is the target gene of MAGI2-AS3, indicating that MAGI2-AS3 upregulation suppresses the proliferative and invasive capacity of NSCLC via miRNA-23a-3p/PTEN axis. Thus, although the exact regulation axis seemed non-conforming, PTEN was a downstream target gene of MAGI2-AS3. Therefore, we believe that the MAGI2-AS3/MAGI2/PTEN axis regulates Bca progression by influencing EMT activation. Combined with the changes in mRNA and protein expression of EMT-associated genes, including vimentin, Snail1, E-cadherin, and N-cadherin, we affirmed that the EMT pathway is crucial for the MAGI2-AS3-mediated regulation of Bca cell migration and invasion.
Although this study revealed some important insights into the detailed mechanism of MAGI2-AS3 in Bca, it has several limitations. Our findings were based on relatively few clinical samples from a single center. More comprehensive and objective findings should be independently validated in a larger cohort derived from several centers. Models of metastatic Bca do not exist; thus we could not provide sufficient objective evidence that MAGI2-AS3 inhibits Bca metastasis in vivo. Thus, optimized orthotopic models are urgently required. The specific mechanisms involved in the MAGI2-AS3 regulation of the MAGI2 and EMT pathways are unknown. Further studies should investigate the regulatory mechanism between MAGI2-AS3 and MAGI2 using chromatin immunoprecipitation (ChIP), RNA immunoprecipitation (RIP), and Luciferase reporter assays.
In summary, the present study revealed that MAGI2-AS3 is a tumor suppressor in Bca, and that low MAGI2-AS3 expression positively correlates with numbers of tumors, grade, stage, and a poor prognosis. MAGI2-AS3 overexpression induced the expression of the sense strand RNA MAGI2 and its downstream gene, PTEN, which interfered with EMT activation, and led to the inhibition of Bca progression. These findings offer novel insights into the mechanism of Bca progression.
Footnotes
Supplementary data
The supplementary files are available to download from
