Abstract
Background
Exploring effect biomarkers that monitor tumor progression and predict the prognosis could benefit the clinical management of bladder cancer and improve the postoperative life of patients. This study aimed to estimate the function of long non-coding (lnc)RNA RHPN1-AS1 (RHPN1-AS1) in bladder cancer and the potential molecular mechanism.
Methods
The expression of RHPN1-AS1 was evaluated in bladder cancer tissues from 115 patients and cells by polymerase chain reaction. The clinical significance of RHPN1-AS1 was assessed and its effect was also estimated in cell proliferation, migration, and invasion. The underlying molecular mechanism was explored by the dual-luciferase reporter assay.
Results
The expression of RHPN1-AS1 was 2.91-fold elevated in bladder cancer, which showed a close correlation with advanced tumor node metastasis stage (P = 0.013) and the presence of lymph node metastasis (P = 0.018). RHPN1-AS1 also served as a poor prognostic indicator (hazard ratio = 2.563) for bladder cancer. The knockdown of RHPN1-AS1 significantly suppressed the proliferation and metastasis ability of bladder cancer cells. Moreover, miR-485-5p was found to mediate the function of RHPN1-AS1 in bladder cancer, which was considered the underlying regulatory mechanism.
Conclusions
RHPN1-AS1 serves as a prognostic biomarker and tumor promoter in bladder cancer via modulating miR-485-5p, which might be a reliable target of bladder cancer therapy.
Introduction
Bladder cancer is a frequently occurring malignant tumor and a leading cause of cancer-related death. Due to the similar symptoms of bladder cancer and urinary tract infection, the diagnosis of bladder cancer is always delayed. 1 Although great progress has been made in the therapeutic strategy of bladder cancer, which really improved the clinical management of bladder cancer patients at early stages, the treatment of patients with advanced stages is still poor, which results in an unsatisfying survival rate.2,3 With the development of molecular biology, correlated molecular therapeutic targets of bladder cancer attracted special attention, and the identification of effective biomarkers that indicate tumor progression and clinical outcome gradually became a research hotspot.
In the past decades, the function of non-coding RNAs has become a focus of tumor research. Among various types, long non-coding RNAs (lncRNAs) have been widely demonstrated to be involved in the development of various cancers.4,5 The lncRNA expression profile could leak out abnormally expressed lncRNAs, which have the potential to serve as biomarkers of cancers.6,7 A previous lncRNA expression profile reported a number of dysregulated lncRNAs in bladder cancer, among which lncRNA RNFI44A-AS1 was found to be upregulated and promoted bladder cancer progression. 8 The dysregulation of lncRNA RHPN1-AS1 (RHPN1-AS1) was also revealed in the expression profile in bladder cancer, while its function was also revealed in various cancers.8–10
The molecular mechanism underlying the biological effect of RHPN1-AS1 in various cancers has been uncovered mainly through sponging correlated miRNAs. For example, miR-485-5p was demonstrated to be a direct target of RHPN1-AS1 that mediates its promoted effect in hepatocellular carcinoma and ovarian cancer.11,12 Additionally, it was suggested that miR-485-5p could suppress the development of bladder cancer by targeting HMGA2. 13 Therefore, miR-485-5p was speculated to mediate the potential regulatory effect of RHPN1-AS1 in bladder cancer.
Methods
Study subject
This study had been approved by the Ethics Committee of Shandong Provincial Third Hospital. A total of 115 patients diagnosed with bladder cancer who received resection treatment at Shandong Provincial Third Hospital were included from 2012 to 2015. No patients had received antitumor therapy, and all had signed informed consent. Tissue samples were resected during surgery and confirmed by at least two pathologists. Collected tissues were stored in liquid nitrogen for the following analyses. A follow-up survey of 5 years was conducted after patients’ surgery.
Real-time quantitative polymerase chain reaction
The RHPN1-AS1 and miR-485-5p expression levels were evaluated by real-time quantitative polymerase chain reaction (RT-qPCR) after the isolation of total RNA using TRIpure (BioTeke, Beijing, China). The evaluation was conducted with cDNA generated from extracted RNA by Super M-MLV Reverse Transcriptase (BioTeke). The RHPN1-AS1 expression level was detected with the SYBR Green (Sigma, Omaha, NE, USA) and calculated by the 2−ΔΔCt method with GAPDH (for RHPN1-AS1) and U6 (for miR-485-5p) as the reference genes. The PCR reaction was conducted according to the following conditions: 95°C for 6 min, followed by 25 cycles at 95°C for 30 s, 55°C for 30 s, and 72°C for 30 s, then one final extension at 72°C for 10 min.
Online databases information
The expression of RHPN1-AS1 in bladder cancer was estimated with the data from the GEPIA database (http://gepia.cancer-pku.cn/detail.php?gene = RHPN1-AS1###) and the starBase database (https://rnasysu.com/encori/panGeneDiffExp.php).
The downstream targets of miR-485-5p were predicted from TargetScan (https://www.targetscan.org/vert_80/), miRDB (https://mirdb.org/), miRWalk (http://mirwalk.umm.uni-heidelberg.de/), and starBase databases (https://rnasysu.com/encori/agoClipRNA.php?source = mRNA), and the function of overlapped genes was enriched through the DAVID database (https://david.ncifcrf.gov/summary.jsp).
Cell transfection
Human bladder cancer cell lines, including RT4, T24, 5637, and HT1197 cells, and the normal epithelial cell SVHUC-1 cell (Shanghai Institute of Cell Biology, Shanghai, China) were used for in vitro cell experiments in the present study. All cells were maintained in the DMEM culture medium (containing 10% fetal bovine serum (FBS) and 1% penicillin or streptomycin) at 37°C arriving at the mature period. Then, the cultured cells were transfected with small interference RNA of RHPN1-AS1 (si-RHPN1-AS1), miR-485-5p mimic, or miR-485-5p inhibitor. Cell transfection was conducted with the help of Lipofectamine™ 2000 (Invitrogen, Waltham, MA, USA) and estimated by PCR.
Cell proliferation assessment
The proliferative potential of RT4 and T24 cells was evaluated by the CCK8 assay after 48 h of cell transfection. Cells (1 × 103 cells/well) were maintained in 96-well with completed culture medium for 0, 24, 48, and 72 h followed by adding CCK8 reagent (Beyotime, Haimen, Jiangsu, China). OD450 was detected with a microplate Reader (BIOTEK, Shoreline, WA, USA) for the proliferation evaluation.
Dual-luciferase reporter assay
The binding sites between RHPN1-AS1 and miR-485-5p were predicted (http://starbase.sysu.edu.cn/index.php) and used to establish the wild-type vector of RHPN1-AS1 (RHPN1-AS1 WT). While the mutant-type vector was established with the mutant 3ʹUTR by point mutation (RHPN1-AS1 MT). The fragments were cloned into the pmirGLO vectors and verified by DNA sequencing. Cells were co-transfected with miR-485-5p mimic, miR-485-5p inhibitor, or negative controls and established vectors with the Lipofectamine 2000 (Invitrogen). The relative luciferase activity was measured with the Dual Luminescence Assay Kit after 48 h of the transfection with Renilla as the reference.
Cell migration and invasion assessment
In the upper chamber of the transwell plates (Matrigel-coated for invasion assay, BD Biosciences, La Jolla, CA, USA), the RT4 and T24 cells (5 × 104 cells/well) were seeded. The seeded cells were supplied with an FBS-free culture medium, while the completed culture medium was placed in the lower chamber. After an incubation at 37°C for 24 h, cells on the upper surface were removed. The migrated and invasive cells were counted with a light microscope (OLYMPUS, Tokyo, Japan).
Statistical analysis
All data were expressed as mean value ± SD obtained from triplicate independent experiments. The differences were assessed by Student's t-test (for two groups, especially in the clinical samples) or one-way ANOVA (for multiple groups, especially in cell experiments) followed by the Turkey post-hoc test. The Chi-square test, Kaplan–Meier curve, and multivariate Cox regression analysis were performed to evaluate the clinical significance of RHPN1-AS1. The statistically significant difference was indicated by P < 0.05.
Results
The upregulation of RHPN1-AS1 was found in bladder cancer
The significant upregulation of RHPN1-AS1 was observed in collected tumor tissues compared with matched normal tissues (P < 0.001, Figure 1(A)). The upregulation of RHPN1-AS1 was validated by the online databases. Data from the GEPIA database included 404 tumor tissues and 28 normal tissues suggesting the upregulation of RHPN1-AS1 in bladder urothelial carcinoma (Figure 1(B)). Another online database, starBase database composed of 411 cancer and 19 normal samples also confirmed the increased RHPN1-AS1 in bladder cancer (Figure 1(C)). Consistently, the expression level of RHPN1-AS1 in bladder cancer cells was also found to be much higher than that in normal cells (P < 0.001, Figure 1(D)). The mean value of RHPN1-AS1 expression in tumor tissues was used as the cutoff to group enrolled patients.
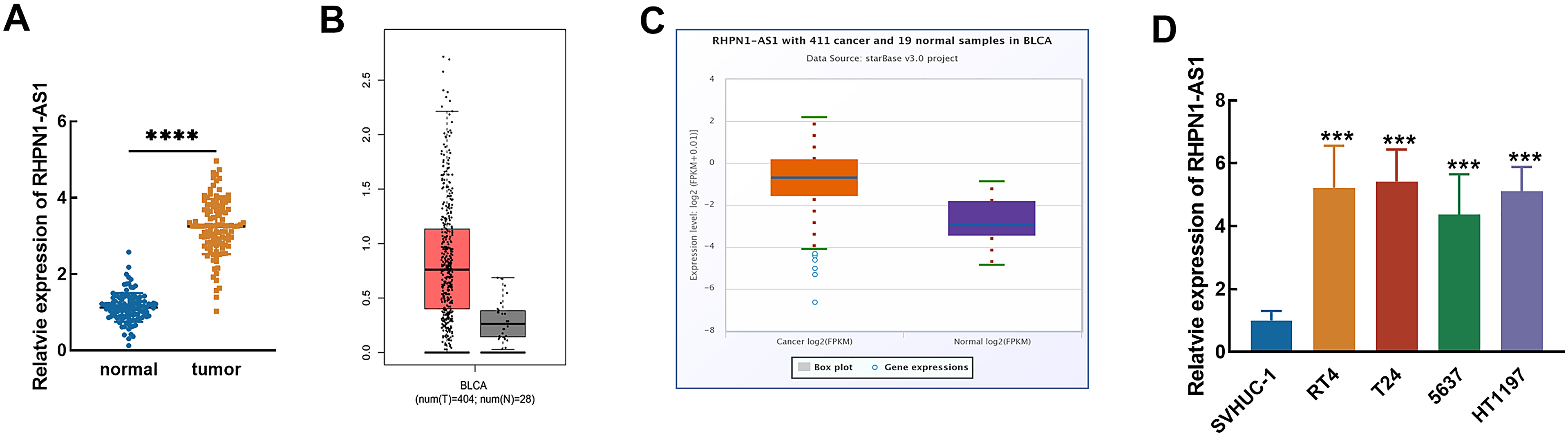
Expression of RHPN1-AS1 in bladder cancer. The significant upregulation of RHPN1-AS1 was observed in bladder cancer tissues (A), which was consistent with the dysregulation in the GEPIA database (B) and starBase database (C). RHPN1-AS1 was also upregulated in bladder cancer cell lines (D). ***P < 0.001, ****P < 0.0001.
The involvement in disease development and prognostic value of RHPN1-AS1 was demonstrated in bladder cancer
Patients were grouped into the low RHPN1-AS1 group (36 males and 16 females) and the high RHPN1-AS1 group (37 males and 26 females). The clinicopathological features were compared between the low-ASB16-AS1 group and the high-ASB16-AS1 group. It was found that most patients who showed positive lymph node metastasis (P = 0.013) and advanced tumor node metastasis (TNM) stage (P = 0.018) were found to show a relatively high expression of RHPN1-AS1, while the significant association of these clinicopathological features and RHPN1-AS1 expression was also demonstrated (Supplementary Table 1).
Additionally, the Kaplan–Meier curve showed that the relatively high expression of RHPN1-AS1 was found to possess a close correlation with the poor survival rate of patients (log-rank P = 0.024, Figure 2). Furthermore, the prognostic value of RHPN1-AS1 was also estimated by multivariable Cox regression analysis. It showed that RHPN1-AS1 could act as an independent prognostic indicator of bladder cancer patients with the hazard ratio (HR) value of 2.563 (95% confidence interval (CI) = 1.121, 5.861, P = 0.026), as well as TNM stage (HR = 2.768, 95% CI = 1.107, 6.922, P = 0.029, Table 1).
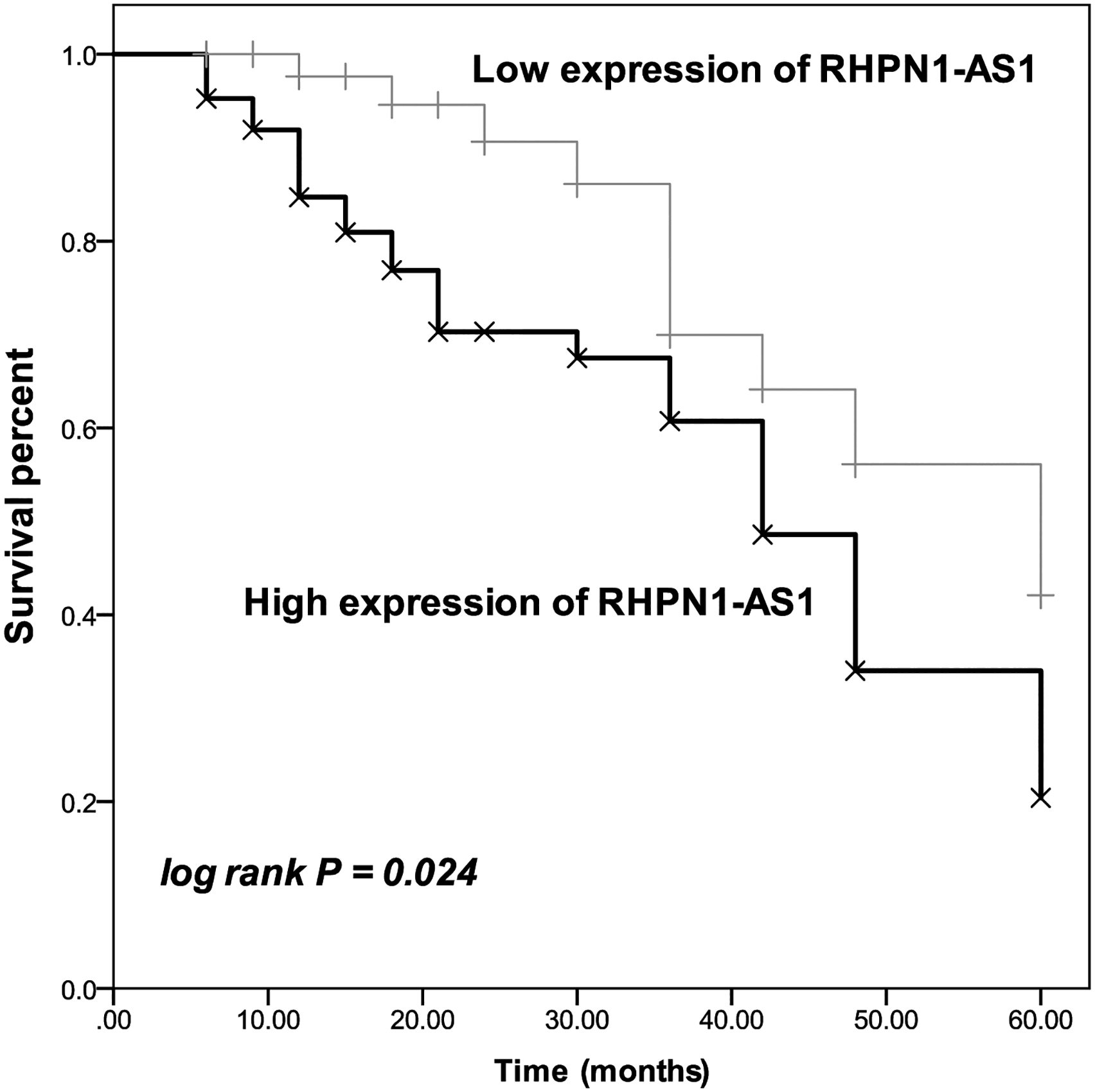
Kaplan–Meier analysis of included patients based on the average expression of RHPN1-AS1 in bladder cancer tissues. The relatively high RHPN1-AS1 expression was significantly associated with the worse overall survival of the patient. Log-rank P = 0.024.
Multivariate Cox regression evaluation on the prognostic value of clinicopathological features.

CI: confidence interval; HR: hazard ratio; TNF: tumor nod metastasis.
The tumor promoter role of RHPN1-AS1 in bladder cancer was mediated by mir-485-5p
The relative luciferase activity of the RHPN1-AS1 WT vector containing the binding sites between RHPN1-AS1 and miR-485-5p was significantly reduced by the overexpression of miR-485-5p and enhanced by miR-485-5p knockdown (P < 0.001, Figure 3(A)), while the luciferase activity of the RHPN1-AS1 MT vector was not affected (P > 0.05). In contrast, the knockdown of RHPN1-AS1 in RT4 and T24 cells increased the expression of miR-485-5p, which was reversed by miR-485-5p inhibitor transfection (P < 0.01, P < 0.001, Figure 3(B)).
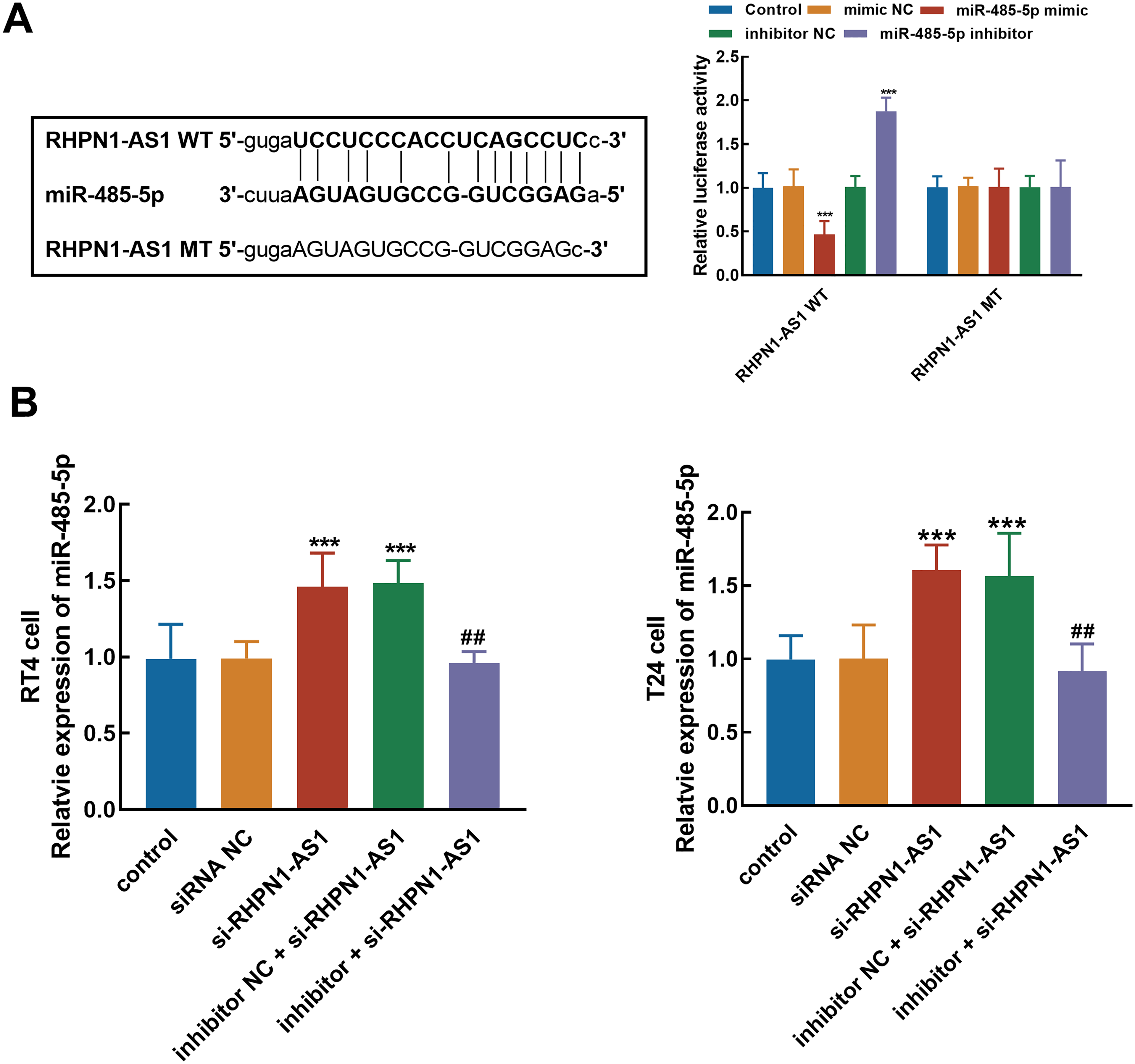
Evaluation of the interaction between RHPN1-AS1 and miR-485-5p. (A) Predicted binding sites and the effect of miR-485-5p on the relative luciferase activity of RHPN1-AS1. (B) The knockdown of RHPN1-AS1 significantly elevated the expression of miR-485-5p, which was reversed by miR-485-5p inhibitor. ***P < 0.001 relative to the control group; ##P < 0.01 relative to the si-RHPN1-AS1 group.
Silencing RHPN1-AS1 significantly suppressed the proliferation (Figure 4(A)), migration (Figure 4(B)), and invasion (Figure 4(C)) of RT4 and T24 cells. Furthermore, the regulatory effect of miR-485-5p was illustrated during the promoted effect of RHPN1-AS1 on bladder cancer cellular processes, as the knockdown of miR-485-5p could reverse the inhibition of cell proliferation (Figure 4(A)), migration (Figure 4(B)), and invasion (Figure 4(C)) by RHPN1-AS1 knockdown (P < 0.01, P < 0.001).
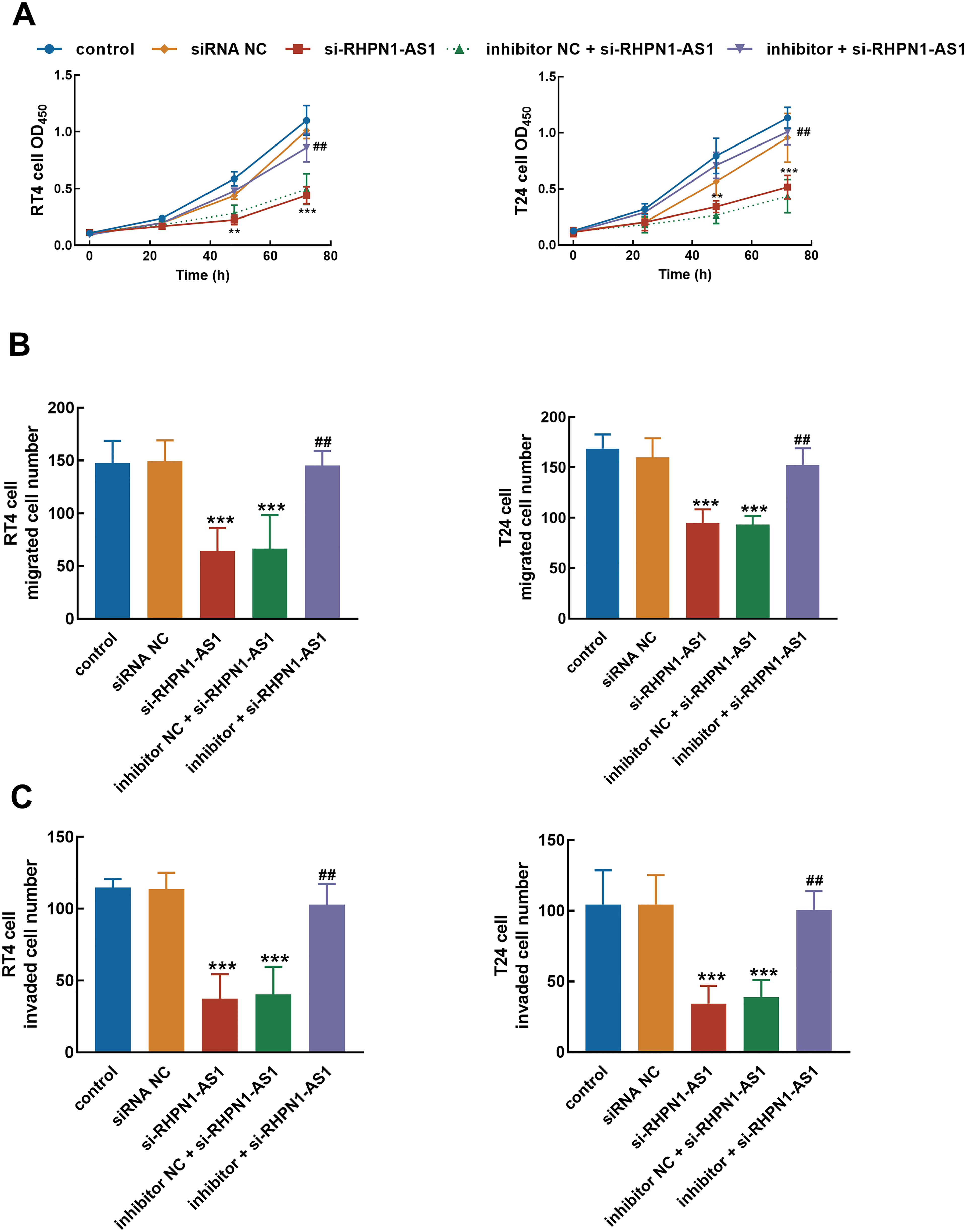
miR-485-5p mediated the tumor-promoted effect of RHPN1-AS1. The inhibitory effect of RHPN1-AS1 knockdown on the proliferation (A), migration (B), and invasion (C) of bladder cancer cells was reversed by the silencing of miR-485-5p.***P < 0.001 relative to the control group; ##P < 0.01 relative to the si-RHPN1-AS1 group.
The downstream targets of miR-485-5p were predicted from prevalent public databases. The number of targets predicted from miRWalk, starBase, TargetScan, and miRDN databases was 16,872, 1764, 448, and 923, respectively, and 83 targets overlapped in the four databases (Figure S1A). The overlapped targets were mainly involved in phosphorylation, protein serin kinase activity, protein phosphorylation, peptidyl-threonine phosphorylation, and protein autophosphorylation (Figure S1B).
Discussion
The lack of typical symptoms of bladder cancer always results in late diagnosis and invisible development. Screening of biomarkers that indicate tumor progression and patients’ prognosis could benefit the clinical management of bladder cancer. Previously, the role of lncRNAs in human cancers, such as colorectal cancer, breast cancer, and lung cancer has been widely demonstrated.14–16 In bladder cancer, several lncRNAs have been revealed to modulate disease development. For instance, lncRNA SLC16A1-AS1 was found to promote the invasiveness of bladder cancer through complexing with its transcription factor, which enhanced cancer metabolic reprogramming. 17 Downregulated lncRNA MORT could suppress critical cellular processes of bladder cancer by regulating miR-146a-5p. 18 Dysregulation of lncRNAs always implies their significant potential in monitoring cancer development and indicating clinical outcomes. The significant upregulation of RHPN1-AS1 has been observed in the present study, which is slightly different from its expression in a previous profile but was validated by the public data from a large sample size and by the expression levels in cells. Previously, the identified biomarkers for bladder cancer were demonstrated to indicate progressive tumor development as their close association with TNM stage and other severity-related indexes. For instance, a recent study revealed the potential of EZH2 in representing bladder cancer progression, due to its significant correlation with TNM stage lymph node metastasis, muscle invasion, and tumor size of patients. 19 BLACAT1 was suggested to increase in the invasive stage of bladder cancer and to indicate an unfavorable prognosis. 20 Notably, the elevated expression level of RHPN1-AS1 was found to be closely associated with positive lymph node metastasis and advanced TNM stage of patients. The lymph node metastasis and TNM stage always represent the malignant and severe disease development of patients. The significant relationship between these indicators and RHPN1-AS1 upregulation in bladder cancer suggested that RHPN1-AS1 participates in cancer progression and could serve as a biomarker.
The prediction of clinical prognosis could provide a reference for adjusting the therapeutic strategy of patients and therefore improve patients’ survival and ameliorate their quality of life. The prognostic value of lncRNAs has also been indicated in various cancers, including bladder cancer. For example, lncRNA PCAT6 was illustrated to distinguish bladder cancer and predict the overall survival of patients. 21 Wu et al. 22 dug out eight lncRNAs, which showed significant prognostic value and were considered as candidate prognosis predictors. Herein, the increased expression of RHPN1-AS1 was found to indicate a poorer survival rate of patients and was identified as an independent prognostic factor of bladder cancer.
Additionally, the functional role of RHPN1-AS1 in the cellular processes of bladder cancer was also investigated. The regulation of cell growth and motility by ncRNAs is a critical part of revealing their function in bladder cancer. lncRNA RMRP was illustrated to promote bladder cancer cell proliferation, migration, and invasion, which was considered to support its oncogene role. 23 The enhancement of bladder cancer tumorigenesis and ferroptosis resistance by lncRNA RP11-89 was hypothesized the major mechanism underlying its tumor promoter role. 24 RHPN1-AS1 was identified as a tumor promoter in ovarian cancer enhancing cancer onset and development through suppressing anti-oncogene or promoting oncogene.9,25 The enhancement was also observed in colorectal cancer, gastric cancer, breast cancer, and cervical cancer, where RHPN1-AS1 accelerated cancer deterioration and resulted in worse overall survival.26–29 In the present study, RHPN1-AS1 was found to boost major cellular processes of bladder cancer, which are closely associated with tumor progression. miR-485-5p was reported to be the sponge of RHPN1-AS1, and their interaction was revealed as the molecular mechanism underlying the promoted effect of RHPN1-AS1 in hepatocellular carcinoma and ovarian cancer.11,12 miR-485-5p was reported to serve as a tumor suppressor of bladder cancer and mediated the regulatory effect of lncRNAs in various human cancers.13,30 The sponging of miR-485-5p by RHPN1-AS1 was validated in the present study. The overexpression of miR-485-5p could reduce the activity of RHPN1-AS1 in dual luciferase assay, while the knockdown of RHPN1-AS1 could facilitate the expression of miR-485-5p. Moreover, miR-485-5p was found to modulate the regulatory effect of RHPN1-AS1 in bladder cancer cells, as the knockdown of miR-485-5p could alleviate the inhibition of cell proliferation, migration, and invasion by RHPN1-AS1 downregulation. Therefore, sponging miR-485-5p was hypothesized as the potential mechanism through which RHPN1-AS1 exerted its tumor-promoted effect in bladder cancer. With the help of several public databases, the downstream targets of miR-485-5p were predicted. The predicted targets were mainly involved in the phosphorylation process of proteins, which has been involved in the development of cancers and has also been considered a critical molecular mechanism underlying the regulatory effect of ncRNAs.31–34 Future studies would be devoted to exploring the specific involved gene in the regulatory effect of the RHPN1-AS1/miR-485-5p axis on bladder cancer.
With the development of targeted therapy, the identification of molecules involved in tumor progression provides potential therapeutic targets. Silencing RHPN1-AS1 can be considered a potential targeted therapeutic strategy for bladder cancer, to which future studies would pay special attention.
Conclusions
According to the above findings, the upregulation of RHPN1-AS1 was leaked out in bladder cancer in the present study, which was associated with cancer development and the clinical prognosis of patients. RHPN1-AS1 was identified as a tumor promoter facilitating cellular processes of bladder cancer by suppressing miR-485-5p.
Supplemental Material
sj-docx-1-jbm-10.1177_03936155241281076 - Supplemental material for Alteration of lncRNA RHPN1-AS1 predicts clinical prognosis and regulates the progression of bladder cancer via modulating miR-485-5p
Supplemental material, sj-docx-1-jbm-10.1177_03936155241281076 for Alteration of lncRNA RHPN1-AS1 predicts clinical prognosis and regulates the progression of bladder cancer via modulating miR-485-5p by Jingmin Zhou, Jinshan Xu, Lin Cheng, Shuhui Li, Deqi Jiang, Jianchao Zhang and Yulong Sheng in The International Journal of Biological Markers
Supplemental Material
sj-tif-2-jbm-10.1177_03936155241281076 - Supplemental material for Alteration of lncRNA RHPN1-AS1 predicts clinical prognosis and regulates the progression of bladder cancer via modulating miR-485-5p
Supplemental material, sj-tif-2-jbm-10.1177_03936155241281076 for Alteration of lncRNA RHPN1-AS1 predicts clinical prognosis and regulates the progression of bladder cancer via modulating miR-485-5p by Jingmin Zhou, Jinshan Xu, Lin Cheng, Shuhui Li, Deqi Jiang, Jianchao Zhang and Yulong Sheng in The International Journal of Biological Markers
Footnotes
Acknowledgments
None.
Author contributions
Jingmin Zhou and Jinshan Xu contributed equally to this work.
Availability of data and materials
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study had been approved by the Ethics Committee of Shandong Provincial Third Hospital. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
