Abstract
BACKGROUND:
Potassium channels, encoded by more than seventy genes, are cell excitability transmembrane proteins and become evident to play essential roles in tumor biology.
OBJECTIVE:
The deregulation of potassium channel genes has been related to cancer development and patient prognosis. The objective of this study is to understand the role of potassium channels in lung cancer.
METHODS:
We examined all potassium channel genes and identified that KCNN4 is the most significantly overexpressed one in lung adenocarcinoma. The role and mechanism of KCNN4 in lung adenocarcinoma were further investigated by in vitro cell and molecular assay and in vivo mouse xenograft models.
RESULTS:
We revealed that the silencing of KCNN4 significantly inhibits cell proliferation, migration, invasion, and tumorigenicity of lung adenocarcinoma. Further studies showed that knockdown of KCNN4 promotes cell apoptosis, induces cell cycle arrested in the S phase, and is associated with the epithelial to mesenchymal transition (EMT) process. Most importantly, we demonstrated that KCNN4 regulates the progression of lung adenocarcinoma through P13K/AKT and MEK/ERK signaling pathways. The use of inhibitors that targeted AKT and ERK also significantly inhibit the proliferation and metastasis of lung adenocarcinoma cells.
CONCLUSIONS:
This study investigated the function and mechanism of KCNN4 in lung adenocarcinoma. On this basis, this means that KCNN4 can be used as a tumor marker for lung adenocarcinoma and is expected to become an important target for a potential drug.
Abbreviations
Introduction
Lung cancer is one of the malignant tumors in the world which has high mortality and morbidity. As the leading cause of cancer-related death, lung cancer has been categorized into small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), of which the latter represents 85% [1, 2, 3]. The NSCLC mainly includes squamous cell carcinoma (LUSC), large cell carcinoma (LCLC), and adenocarcinoma (LUAD), which accounts for approximately 40% of lung cancers. Many risk factors are associated with lung cancer development, including smoking, air pollution, chronic infections in the lungs, and some genetic factors [4, 5]. The characteristics of lung cancer are difficult to be identified during the early stage, so more than 70% of patients are diagnosed in advanced stages [6]. Currently, surgical resection, chemotherapy, immunotherapy and targeted drugs such as Gefitinib, Crizotinib, Alectinib are the mainstay of treatment for lung cancer [7, 8]. However, like most targeted drugs, these can also produce resistance and a range of toxic effects. Although the advancement of LUAD treatment has made significant progress, patients’ five-year survival rate is still meager [9, 10, 11]. Therefore, it is particularly important to study the mechanism of LUAD development and to create an effective therapeutic drug.
More than seventy genes encode potassium channels, including four classes of K+ channels, calcium-activated potassium channels, inward rectifier potassium channels, two-pore-domain potassium channels, and voltage-gated potassium channels. Voltage-gated potassium channels are the largest subset of potassium channels encoded by 40 genes in humans [12, 13]. Potassium channels are cell excitability transmembrane proteins and become evident to play essential roles in tumor biology. The deregulation of potassium channel genes has been related to cancer development and patients’ prognosis. To investigate the potential role of potassium channels in lung cancer, we examined all potassium channel genes and identified that KCNN4 is the most significantly overexpressed one in lung adenocarcinoma. KCa 3.1 channel, encoded by KCNN4, is the potassium calcium-activated channel. After activation of KCNN4, the cell membrane appears hyperpolarized, promoting the flow of calcium ions into the membrane. Previous studies reported that KCNN4 is usually expressed in the hematopoietic system, lung, pancreas, and colon tissues, which is crucial for the modulation of the immune system [14, 15, 16, 17, 18]. Recently, KCNN4 has been shown to induce poor prognosis in lung and other cancers [19, 20]. However, the function and mechanism of KCNN4 in the progression of LUAD remain unclear.
In this study, we aimed to investigate the role of KCNN4 in LUAD. Our analysis revealed that KCNN4 is significantly overexpressed in LUAD, and its upregulation induces proliferation and metastasis of LUAD cells. Furthermore, we found that KCNN4 induces its oncogenic effect in LUAD through the activation of PI3K/AKT and MEK/ERK signaling pathways and affects EMT. To sum up, KCNN4 is involved in the development and progression of LUAD via P13K/AKT and MEK/ERK signaling pathways. Therefore, KCNN4 is a promising biomarker and therapeutic target for lung adenocarcinoma, and blocking AKT and ERK signaling pathways may provide a novel therapeutic strategy for KCNN4 overexpressing tumors.
Materials and methods
Reagents
RNA extraction kit was purchased from Tiangen. SYBR, CCK8, and TUNEL kits were obtained from YEASEN. AKT inhibitor (MK2206) and ERK inhibitor (U1206) were obtained from MCE. The SABC-HRP from Beyotime Biotechnology was used for immunohistochemistry. Anti-Vimentin, anti-E-cadherin, anti-GAPDH, and anti-KCNN4 antibodies were all purchased from Proteintech. AKT, Phospho-AKT, ERK1/2 and Phospho-p44/42 MAPK (ERK1/2) antibodies were obtained from Cell Signaling Technology.
siRNA transfection
siRNA was used to diminish the expression of KCNN4 mRNA in A549 cells. Briefly, A549 cells were seeded into six-well plates (2
Plasmids and stable cell line
pLKO.1-shScramble and pLKO.1-shKCNN4 plasmids were purchased from Sigma-Aldrich (St. Louis, MO, USA). pLenti-CMV-GFP/puro and pLenti-CMV-KCNN4 plasmids were bought from Vigene Biosciences. All recombinant vector plasmids have been sequenced before being used. To generate a stable cell line, the LUAD cells were seeded into a six-well plate (2
RNA extraction and quantitative RT-PCR
Total RNA was extracted from lung cancer cells and tissues using TRIzol reagent. 5
Cell migration and invasion assays
The ability of cells to invade and migrate was mainly measured by whether cells could pass through the cell chambers. For migration assay, the cells were starved for 12 hours in a serum-free RPMI-1640 medium before the inoculation. 3
Animal models
The animal experiments were approved by the Animal Core Facility of Nanjing Medical University. Four-week-old male nude mice and NCG mice (Model Animal Research Center of Nanjing University) were divided into two groups. One group was injected with sh-KCNN4-A549 cells (5 million cell/mice in 200
The database and analysis
The databases used in this study are derived from TCGA and GEO databases (GSE101929, GSE116959, GSE75037). The Wilcox rank-sum test was used to analyze the expression differences of KCNN4 in tumor and normal tissue samples. The analysis of the relationship between the overall survival rate and the expression of KCNN4 was based on GEPIA2 analysis in the TCGA dataset. Use cluster profiler to conduct pathway enrichment analysis on KCNN4.
Statistics analysis
In this study, all experiments were repeated more than three times, and all experimental data were expressed in the form of Mean
KCNN4 is overexpressed in lung adenocarcinoma. (A) The heatmap showed the expression pattern of more than seventy potassium channel genes in LUAD and matched normal tissues. The potassium channel genes were examined from the clinical samples sequencing datasets. (B) KCNN4 was identified to be the most significantly overexpressed potassium channel gene in lung adenocarcinoma. (C) H&E and IHC staining of paraffin sections of LUAD patients showed an elevation of KCNN4 expression. (D) Compared with normal lung cells, KCNN4 expression was up-regulated in LUAD cells. (E) KCNN4 expression in lung adenocarcinoma patients compared to control in three different datasets from GEO. The expression of KCNN4 in LUAD patients was significantly higher than that in control ones. (F) KCNN4 expression in TCGA-LUAD patients was highly expressed compared to normal samples. (G) The survival curve of LUAD-TCGA data predicted the association of KCNN4 with poor patients’ prognosis.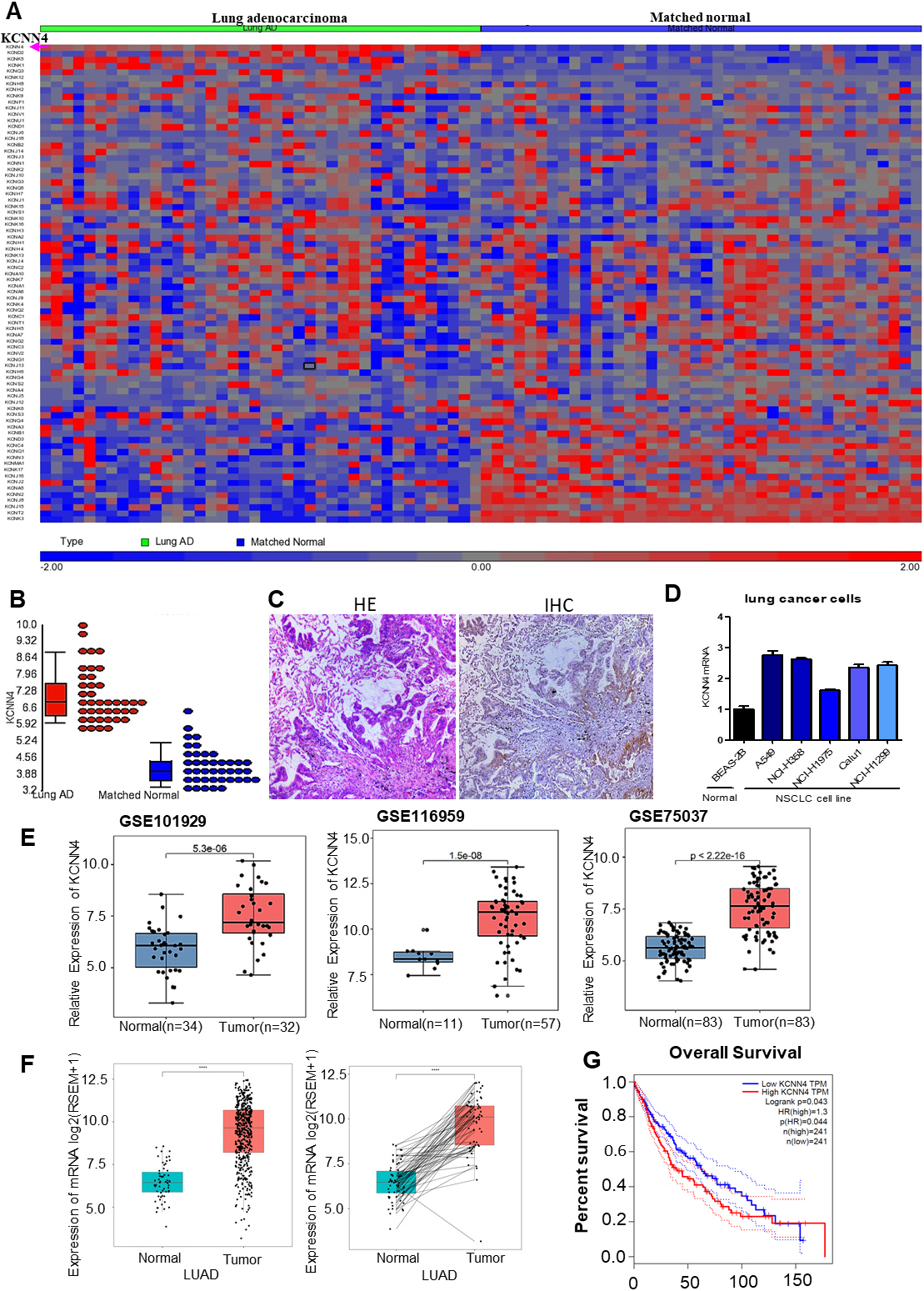
Silencing KCNN4 inhibits different LUAD cell growth. (A) and (B) Knockdown of KCNN4 by siRNA and shRNA in A549 cells, cell proliferation was measured using cell counting and CCK8 assays. (C) The proliferation of NCI-H1299 cells slowed down after the knockdown of KCNN4. (D) The growth of knockdown-KCNN4 in NCI-H1975 was inhibited.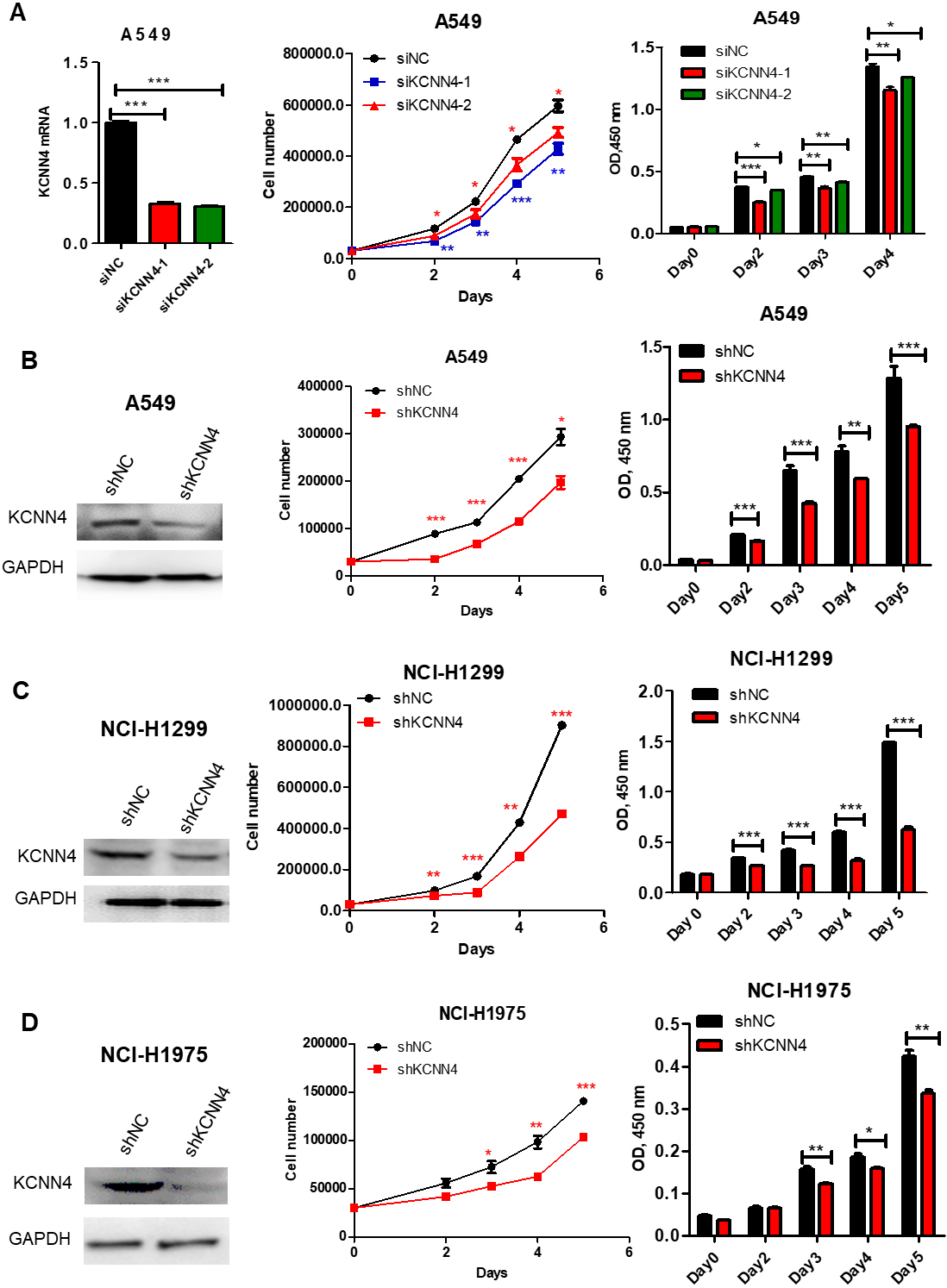
KCNN4 is identified to be significantly overexpressed in lung adenocarcinoma
To understand the role of potassium channels in lung cancer, we examined all potassium channel gene expression levels from the clinical samples sequencing datasets. The heatmap showed the expression pattern of more than seventy potassium channel genes in LUAD and matched normal tissues. Among them, KCNN4 was the most significantly overexpressed potassium channel gene in LUAD (Fig. 1A and B). The overexpression of KCNN4 was further validated in another group of tumor tissues by immunohistochemical staining (Fig. 1C). The results showed that the positive rate of KCNN4 expression in LUAD tissues was 58.62%, significantly higher than that in normal and adjacent non-cancerous tissues, with statistical significance. We also found that the transcriptional level of KCNN4 in LUAD cell lines was higher than that in normal lung cells (Fig. 1D). To validate the overexpression of KCNN4 in lung adenocarcinoma, we further analyzed its expression in LUAD using The Cancer Genome Atlas (TCGA) datasets and Gene Expression Omnibus (GEO) datasets. GEO dataset analysis showed that KCNN4 was significantly up-regulated in LUAD (Fig. 1E). TCGA database analysis also revealed that the expression level of KCNN4 in LUAD patients was higher compared to the normal tissues (Fig. 1F). Moreover, the high expression of KCNN4 is significantly related to the poor prognosis of LUAD (Fig. 1G). KCNN4 is one kind of six calcium-activated potassium channels. TCGA database analysis also showed that the expression of six different calcium-activated potassium channels in LUAD, which indicated that only KCNN4 was significantly overexpressed in lung adenocarcinoma. These data indicated the high expression of KCNN4 in LUAD and the information of different biological processes (Figs S1 and S2). Therefore, overexpression of KCNN4 in LUAD may play an important role in tumor progression.
Silencing KCNN4 inhibits LUAD cell growth
To reveal the biological functions of KCNN4 during the development of lung adenocarcinoma, we used CCK8 and cell counting assays to test the effect of KCNN4 on the proliferation of LUAD cells. As shown in Fig. 2A and B, the proliferation ability of A549 slowed down significantly after silencing KCNN4. To further prove this result, sh-KCNN4 and shNC plasmids were respectively transfected into NCI-H1299 and NCI-H1975. The growth rate of KCNN4-knockdown NCI-H1299 was significantly slower than the control group (Fig. 2C), similar results were shown in NCI-H1975 cells (Fig. 2D). Consistent with these, the high expression of KCNN4 promoted cell proliferation (Fig. 7A and B). Taken together, these data consistently suggest that KCNN4 promotes LUAD cell growth and may function as a lung cancer activator.
Knockdown of KCNN4 blocks cell cycle in S phase and promotes apoptosis
Rapid cell proliferation is commonly associated with cell cycle dysregulation and inhibited apoptosis. Therefore, we explored the role of KCNN4 in the cell cycle and cell apoptosis. As shown in Fig. 3A, when knockdown KCNN4 in A549 cells, the number of cells increased in the S phase and decreased in the G1 phase. Besides, compared with normal tissues, the percentage of apoptosis in A549 and NCI-H1299 was increased after knockdown of KCNN4 (Fig. 3B). TUNEL staining assay also confirmed that the ratio of apoptosis cells was increased after silencing KCNN4 (Fig. 3C and D). In summary, our results demonstrate that KCNN4 can inhibit the process of cell apoptosis and block the cell cycle in the S phase, which may be the reason for regulating cell proliferation.
Knockdown of KCNN4 blocks the cell cycle in the S phase and promotes apoptosis. (A) Knockdown of KCNN4 in A549 cells, the cell cycle was measured by Flow cytometry. (B) Silencing KCNN4 in A549 and NCI-H1299 cells, the percentage of apoptotic cells was assessed by Annexin V-FITC/PI staining assay. (C) TUNEL assay analysis showed that percentage of apoptotic cell of KCNN4-knockdown cells was increased. (D) Silencing KCNN4 induced apoptosis in NCI-H1299 cells by TUNEL assay.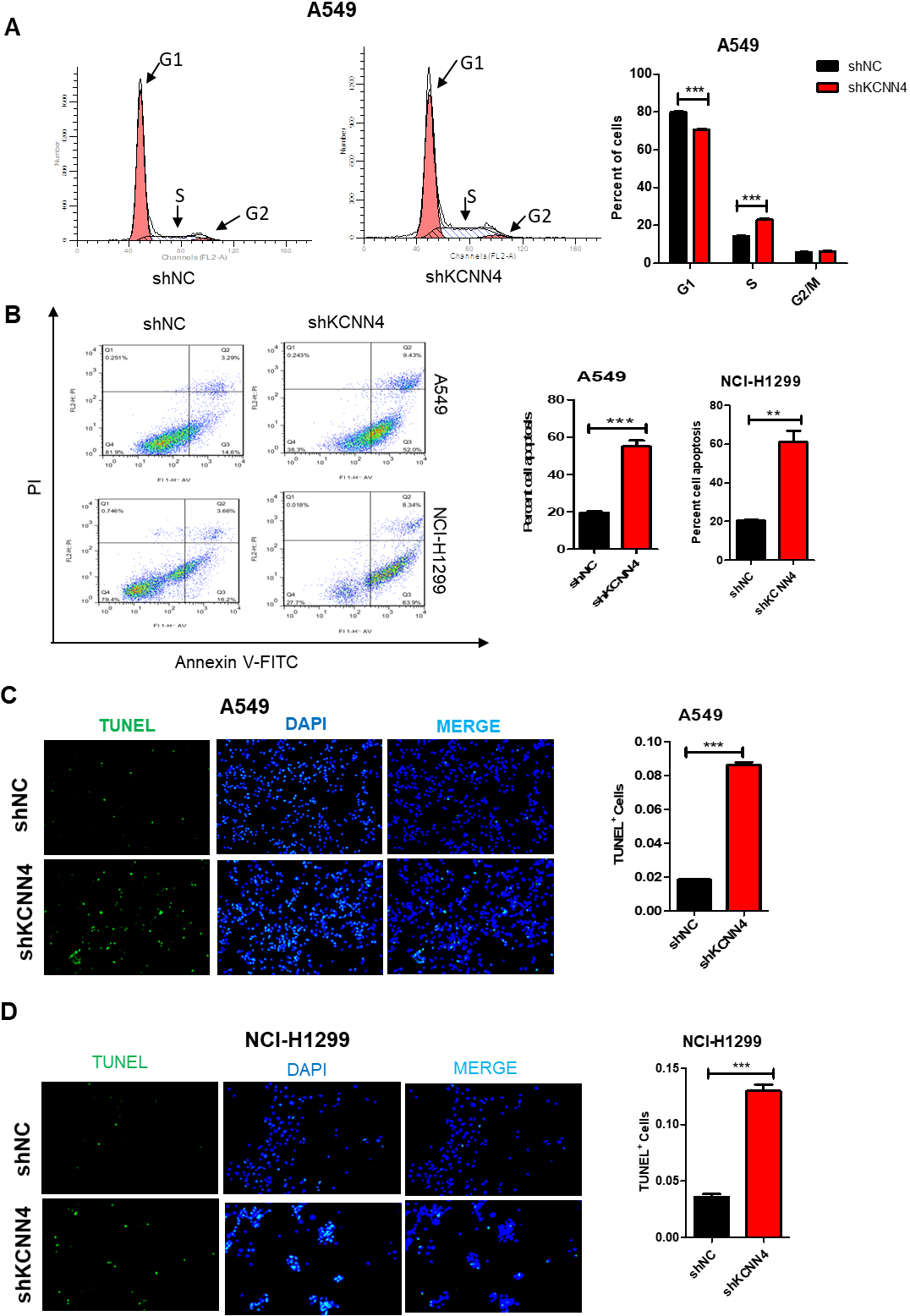
Knockdown of KCNN4 inhibits the migration and invasion of LUAD. (A) Knockdown of KCNN4 inhibits the migration and invasion of A549 cells. (B) Knockdown of KCNN4 blocked the migration and invasion of NCI-H1299 cells. (C) Wound healing assay showed that the migration ability of KCNN4-knockdown A549 cell was reduced. (D) The migration ability of NCI-H1299 was weakened after KCNN4 silencing.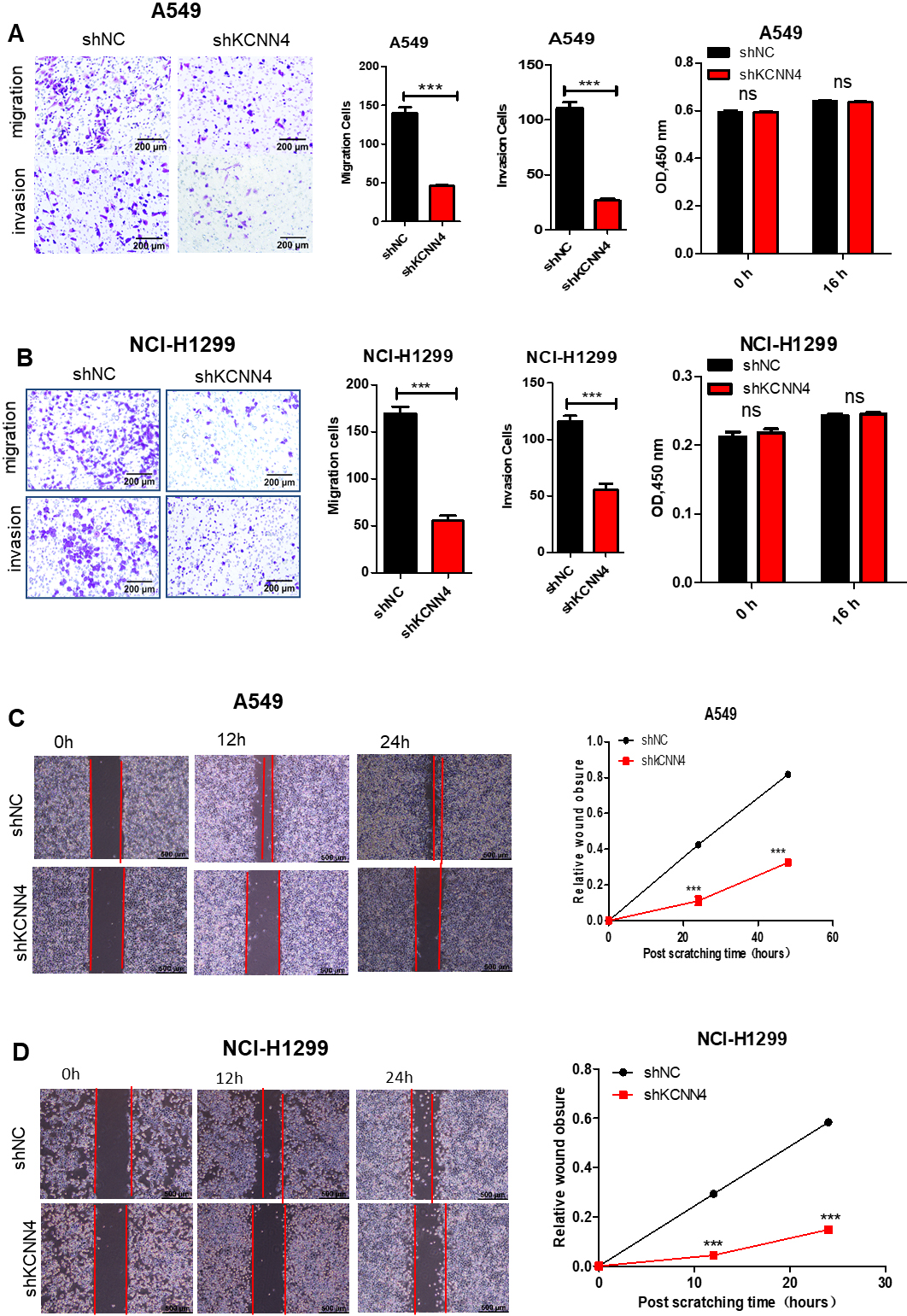
Knockdown of KCNN4 inhibits LUAD cell colony formation and tumor growth in the xenografted in vivo. (A) and (B) Knockdown of KCNN4 in A549 (A) and NCI-H1299 (B) cells restricted their clone forming ability. (C) KCNN4-knockdown and control A549 cells were injected subcutaneously into the BALB/C nude mice, the tumor size was measured for five consecutive weeks. (D) Knockdown of KCNN4 in A549 cells can also significantly slow down the growth of transplanted tumors in the NCG mice. (E) and (F) Overexpression of KCNN4 in A549 (E) and NCI-H1299 (F) cells promotes their clone forming ability. Data are shown as mean 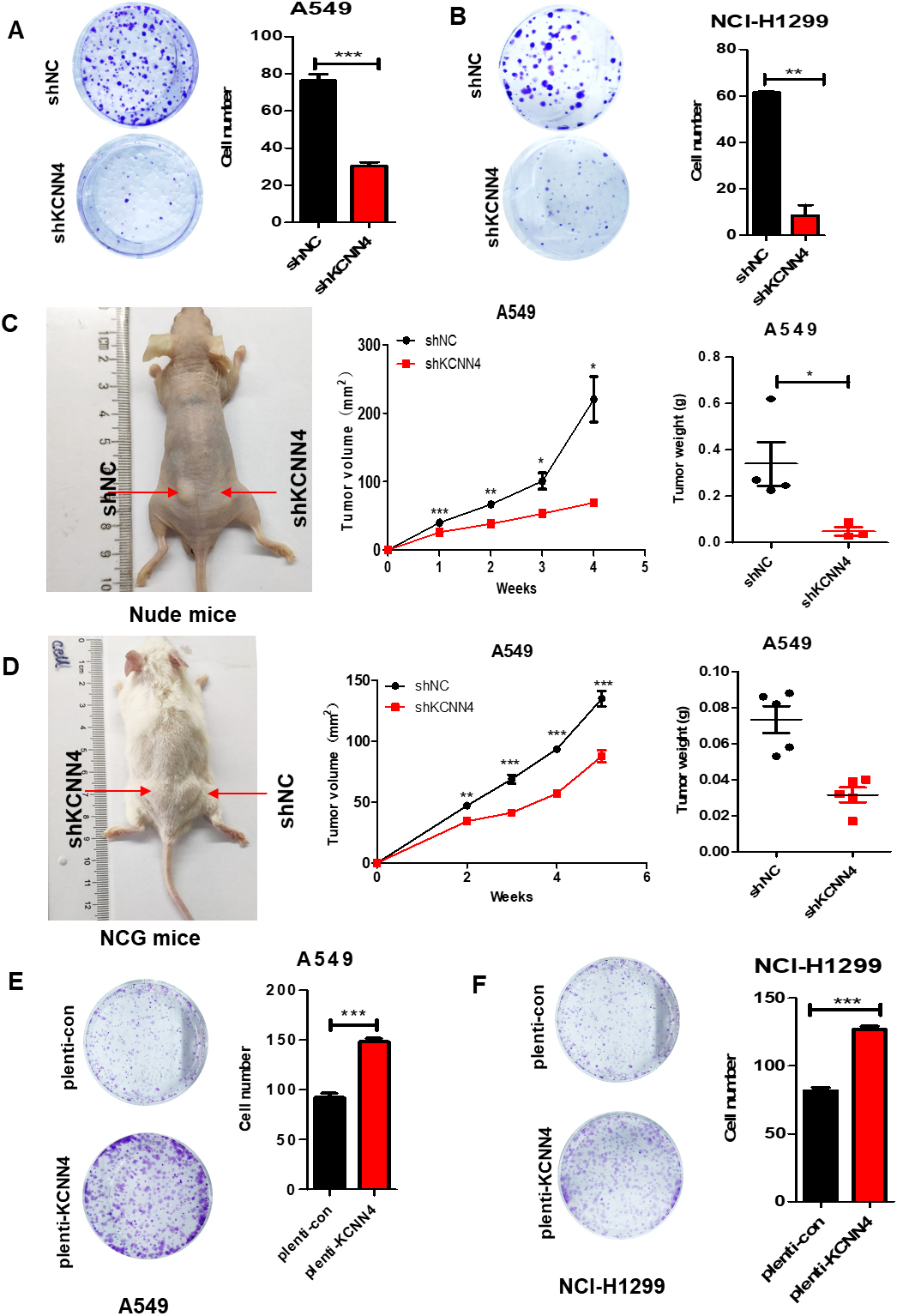
KCNN4 is associated with the activation of the AKT/ERK signaling pathway. (A) Bioinformatics analysis shows that there is a significant correlation between KCNN4 and PI3K/AKT signaling pathways. (B) Western blotting analysis of KCNN4-knockdown and KCNN4-overexpressed in LUAD cells. KCNN4 was positively correlated with p-AKT and p-ERK level, while other tested pathways were little affected. (C) Western blotting assay showed that knockdown of KCNN4 in different LUAD cells reduced Vimentin expression and increased E-cadherin expression while KCNN4 overexpression caused the adverse result.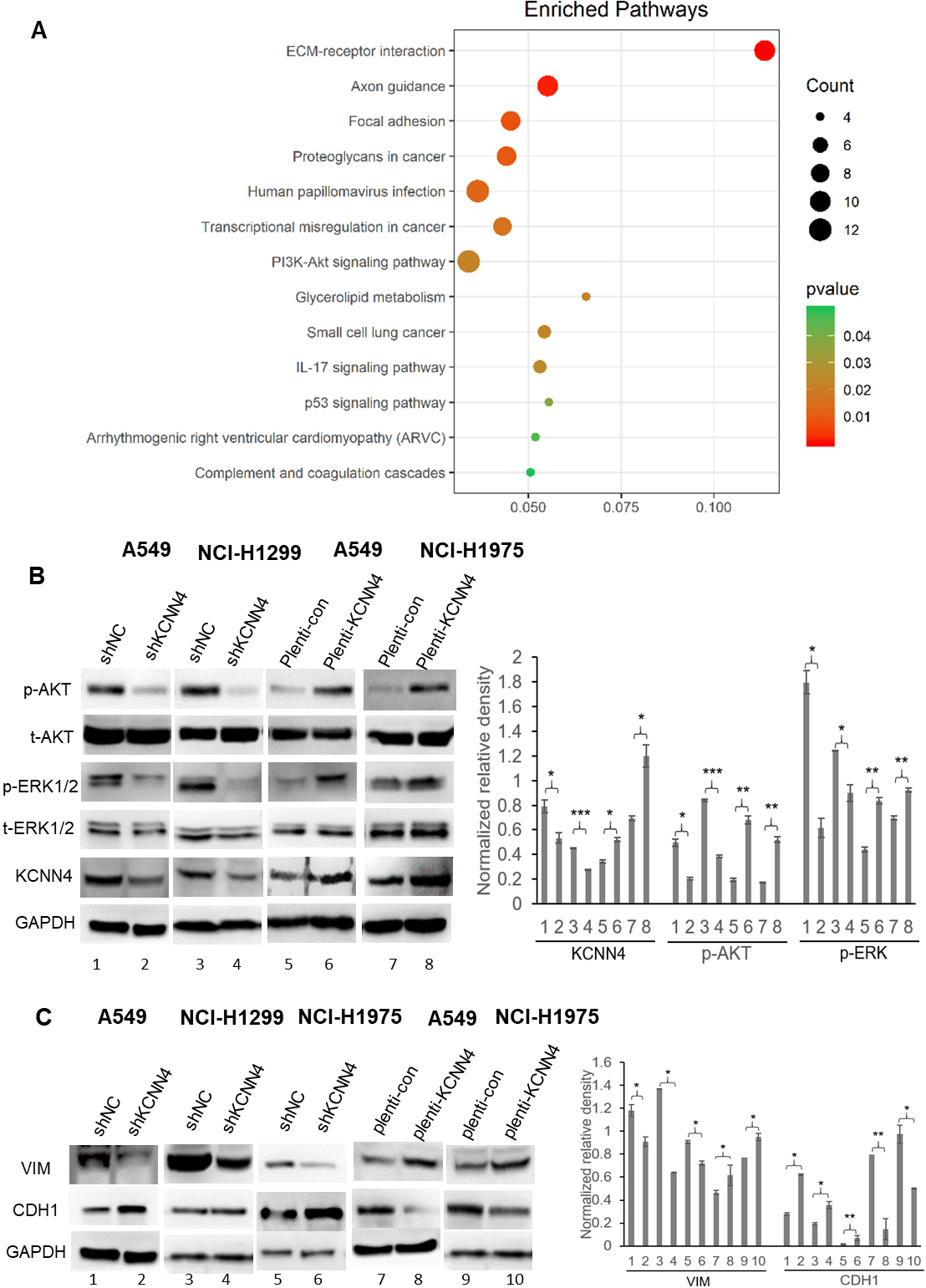
Blocking AKT and ERK signaling pathways with small molecular inhibitors can significantly overcome KCNN4 induced cell proliferation and migration. (A) and (B) KCNN4 was overexpressed in A549 and NCI-H1299 cell lines. The upregulation of KCNN4 resulted in the acceleration of malignant cell proliferation. (C) and (D) Overexpression of KCNN4 in A549 and NCI-H1299 cell also significantly promote cell migration. (E) and (F) The ERK inhibitor (U0126) and AKT inhibitor (MK2206) inhibit the invasion and migration of A549 cells after overexpression KCNN4. (G) and (H) ERK inhibitor (U0126) and AKT inhibitor (MK2206) inhibit the proliferation in KNCC4-overexpressing A549.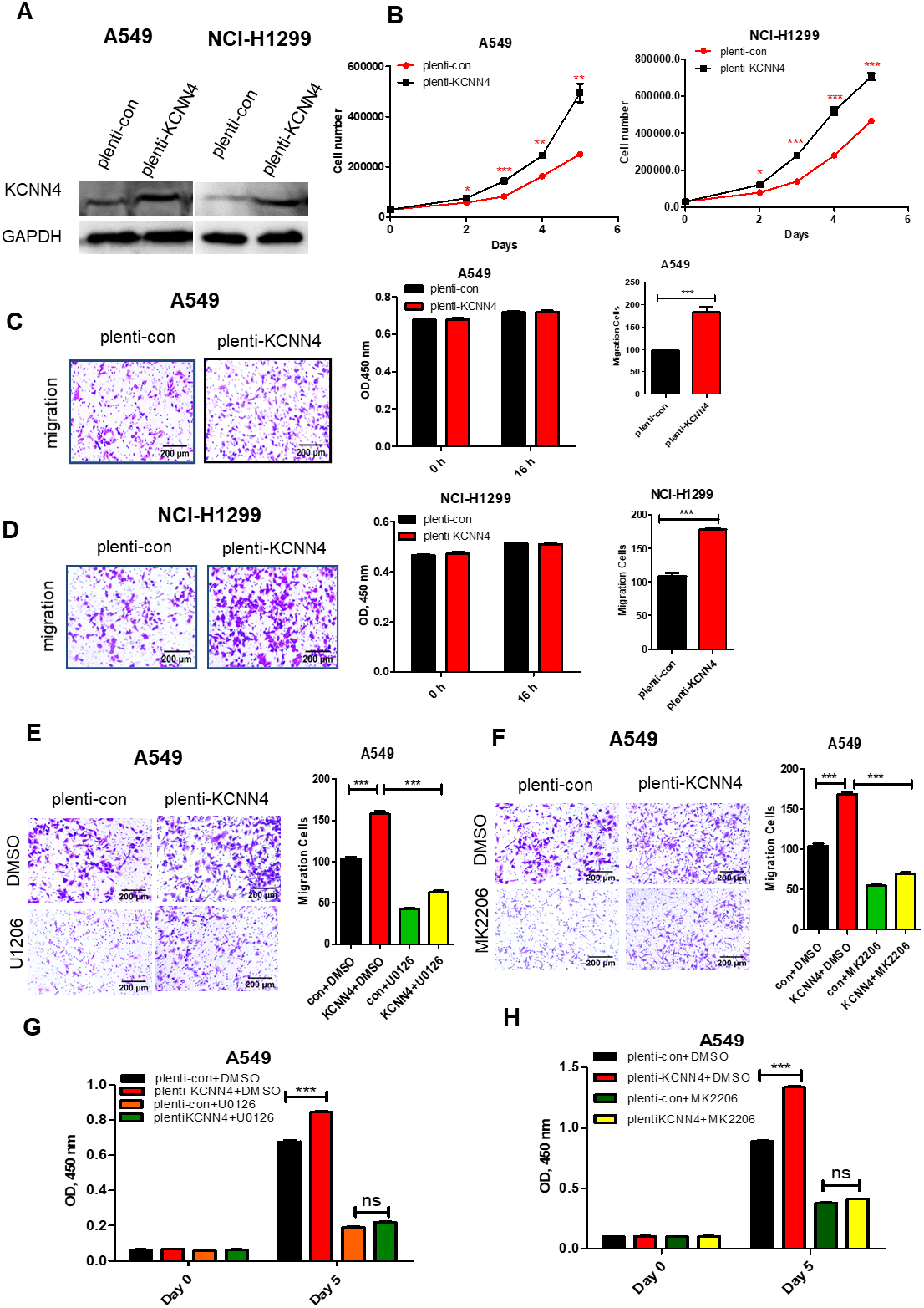
Fast metastasis is an essential reason for the high malignancy of lung cancer. The function of KCNN4 in the metastasis of LUAD was measured by migration, invasion, and wound healing assays. The results showed that the migration and invasion ability of shKCNN4-A549 and shKCNN4-NCI-H1299 was significantly weakened (Fig. 4A and B). By contrast, the migration ability of A549 and NCI-H1299 was enhanced after overexpression of KCNN4 (Fig. 7C and D). As shown in Fig. 4C and D, the wound healing results were consistent with the previous results. The process of tumorigenesis is usually associated with the occurrence of EMT. EMT can cause the loss of polarity of epithelial cells, increasing the invasiveness, metastatic, and anti-apoptotic ability [21, 22]. KCNN4 knockdown can enhance the expression of E-cadherin while reducing the expression of Vimentin in epithelial cells. Contrary experimental results were obtained after mandatory expression of KCNN4 (Fig. 6D). Taken all together, these data suggested that knockdown of KCNN4 inhibits migration, invasion ability and EMT of lung adenocarcinoma.
Knockdown of KCNN4 inhibits LUAD cell colony formation and tumorigenicity in vivo
To further understand the role of KCNN4 in the tumorigenicity of LUAD cells, we conducted colony formation and in vivo tumor formation assay. The result showed that knockdown of KCNN4 in A549 and NCI-H1299 cells restricted their clone forming ability (Fig. 5A and B), and the colony formation ability was significantly improved after overexpressed KCNN4 (Fig. 5E and F). To confirm this conclusion, we inspected the effect of KCNN4 on tumor growth in vivo by injecting sh-KCNN4 A549 cells and negative controls into mice (4 weeks). Interestingly, the tumor volume and tumor weight produced by knockdown-KCNN4 cells were significantly smaller than the control group in vivo (Fig. 5C and D). All in all, KCNN4 promotes the proliferation and tumorigenesis of LUAD cells, which further confirms the regulatory relationship between KCNN4 and the development of lung adenocarcinoma.
KCNN4 activates the AKT and ERK signaling pathways in LUAD cells
Based on the previous results, we paid close attention to the mechanism of KCNN4 in the LUAD. Pathway enrichment analysis showed that genes related to KCNN4 expression were significantly enriched in the PI3K/AKT signaling pathway in LUAD (Fig. 6A). Beyond that, we use KCNN4 knockdown and KCNN4 overexpressed stable cells to detect classic tumor signaling pathways. The results revealed that when knockdown the expression of KCNN4, both p-AKT and p-ERK1/2 was significantly inhibited compared to the control samples. In contrast, the levels of p-AKT and p-ERK 1/2 were increased after elevating the KCNN4 level (Fig. 6B). Such results specified that the activation of both AKT and ERK kinases might be a critical event affecting the KCNN4-induced LUAD development. Moreover, MEK/ERK and PI3K/AKT have been reported to affect cell proliferation, apoptosis, and EMT [23]. Therefore, KCNN4 activates the AKT and ERK signaling pathways and mediated EMT in LUAD cells, contributing to cell growth, migration, and metastasis of tumor cells.
Blocking AKT and ERK signaling pathways with small molecular inhibitors significantly overcome KCNN4-induced cell proliferation and migration
To further clarify the role of KCNN4 in LUAD, KCNN4 was also overexpressed in A549 and NCI-H1299 cell lines. The upregulation of KCNN4 consistently resulted in the acceleration of malignant cell proliferation (Fig. 7A and B) and promoted cell migration (Fig. 7C and D). We next investigated whether the use of AKT inhibitor (MK2206) and ERK inhibitor (U1206) would reduce the LUAD proliferative and migrative abilities or not. Figure 7E–H showed that both cell proliferation and migration were inhibited in KCNN4-overexpressing cells after using the AKT and ERK inhibitors, suggesting that blocking AKT and ERK signaling pathways with small molecular inhibitors can significantly overcome KCNN4 induced cell proliferation and migration, which revealed that KCNN4 promotes LUAD progression depending on AKT and ERK signaling pathways.
Discussion
According to previous studies, lung cancer has gradually become the leading cause of cancer-related deaths, whether in both men or women worldwide [24, 25]. According to the data provided by WHO, 2.09 million patients were diagnosed with lung cancer worldwide, and 1.76 million patients died in 2018. Due to the limitation of detection means, most lung cancers are diagnosed in the middle and late stages, among which NSCLC is the most common histological type of lung cancer. Although targeted therapy and immunotherapy have made some progress, the results are still discouraging [26, 27]. Some studies have shown that the differential expression of ion channels between normal and cancer cells can regulate tumor proliferation and apoptosis and is closely related to tumor progression and metastasis [28]. KCNN4 has been reported to be abnormally expressed in a variety of tumors and associated with poor prognosis, but the current study on LUAD has not been in-depth [29, 30]. Therefore, we aimed to focus on the potential functions and mechanisms underlying KCNN4 in lung adenocarcinoma.
In this study, we observed abnormal overexpression of KCNN4 in our clinical samples of lung cancer. We also got similar findings from analyzing TCGA data and previously published GEO datasets. As a result, we speculated whether the abnormal expression of KCNN4 is related to the occurrence and development of LUAD. Our results showed that KCNN4 knockdown could inhibit LUAD cell proliferation and metastasis in vitro and in vivo. Furthermore, it could significantly induce cell cycle arrest and promote cell apoptosis. These results revealed that KCNN4 may act as a proto-oncogene in the development of lung adenocarcinoma. To further support this conclusion, we investigated the mechanism of KCNN4-induced LUAD. So using western blotting, we analyzed the classic cancer signaling pathway after knockdown or and overexpression of KCNN4 in LUAD cell lines. PI3K/AKT and MEK/ERK pathways are important signaling pathways in cells, promoting cell proliferation and inhibiting apoptosis by influencing the activation state of a variety of downstream effector molecules. Therefore, they are most closely related to tumor occurrence and development [31, 32, 33]. In recent years, KCNN4 has been reported to regulate MEK/ERK signaling pathway in tumors and can be used as a marker of poor tumor prognosis. We demonstrated that knockdown of KCNN4 in LUAD cells significantly reduces the expression levels of p-AKT and p-ERK1/2 proteins. In contrast, the p-AKT and p-ERK1/2 protein levels were elevated after KCNN4 induced overexpression. Also, the use of AKT and ERK inhibitors attenuated the growth, migration, and invasion of KCNN4-overexpressing cells. In summary, the results further confirmed that KCNN4 regulates the proliferation and apoptosis of LUAD cells by activating the P13K/AKT and MEK/ERK signaling pathways.
The invasion and metastasis of malignant tumors are complex processes and responsible for 90% of cancer-related mortality, in which the formation of new blood vessels and induction of epithelial-mesenchymal transformation (EMT) are the key events [34, 35, 36]. Besides, the induction of vascular aggregation is dependent on vascular endothelial growth factor (VEFG). We found that KCNN4 silencing could raise E-cadherin level and reduce Vimentin expression while overexpressing KCNN4 showed opposite results. Several studies have shown that VEGF and EMT are regulated by the feedback of the P13K/AKT signaling pathway [37, 38]. These results can reasonably explain the relationship between KCNN4, EMT, and P13K/AKT. KCNN4 may participate in the EMT process through the P13K/AKT signaling pathway, thereby promoting the development and metastasis of lung adenocarcinoma. However, in the current research, how KCNN4 regulates the P13K/AKT and MEK/ERK signaling pathways are still an unresolved problem. The regulation of the transcription factors involved in these processes requires further investigation by nextgeneration sequencing [39].
In conclusion, our results revealed that KCNN4 can promote the proliferation and metastasis of LUAD cells, dependent on the activation of P13K/AKT and MEK/ERK signaling pathways. This provides evidence that KCNN4 may be a potential target for the treatment of LUAD in the future.
Author contributions
Conception: Ping Xu, Xiao Mo, Hongping Xia.
Interpretation or analysis of data: All authors.
Preparation of the manuscript: Ping Xu, Hongping Xia.
Revision for important intellectual content: All authors.
Supervision: Guoren Zhou, Yijie Zhang, Yongsheng Wang, Hongping Xia.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-201045.
sj-docx-1-cbm-10.3233_CBM-201045.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-201045.docx
Footnotes
Acknowledgments
This study was supported by grants from The Recruitment Program of Overseas High-Level Young Talents, “Innovative and Entrepreneurial Team” (No. (2018)2015), Science and Technology Grant (BE2019758) and the Six Talent Peaks Project (TD-SWYY-007) of Jiangsu Province and High-Level Talents Program of Nanjing Medical University; Funding of Postdoctoral Funding of Nanjing Drum Tower Hospital Affiliated to Medical School of Nanjing University, The Medical Interdisciplinary Research Funding of Henan University (NO. CJ1205A0240011).
Conflict of interest
All authors declare that there are no conflicts of interest.
