Abstract
Lung cancer is one of the deadliest types of cancer worldwide due to its high mortality rate. Adenocarcinoma constitutes 20%–30% of all lung cancers. In recent years, studies on the mechanisms of lung tumorigenesis and development have in part focused on the microRNAs for their crucial role in the progress of different cancers. As for our study, we demonstrated that miR-519d was differently downregulated and eIF4H was significantly overexpressed in lung adenocarcinoma via the detection of quantitative real-time polymerase chain reaction compared with the adjacent normal tissues. Furthermore, Cell Counting Kit-8 assay, colony formation assay, xenograft tumor experiment, Ki67 immunohistochemistry assay and transwell assay were performed to explain that the upregulated miR-519d could inhibit the proliferation and invasion of A549 and H1299 cells. To further advance our understanding of the mechanisms of miR-519d, we performed the bioinformatics analysis and the luciferase report assay. The results from these procedures revealed eIF4H to be one of the targets of miR-519d. Downregulated eIF4H was analogous to the overexpressed miR-519d obtained from miR-519d agomir and si-eIF4H transfection. In summary, it can be concluded that miR-519d targets eIF4H in lung adenocarcinoma to inhibit cell proliferation and invasion. This mechanism may offer new insights into the tumorigenesis and development of lung adenocarcinoma.
Introduction
Cancer is a leading public health problem for entire population worldwide. 1 In particular, lung cancer is responsible for one of the highest cancer-caused mortality rates globally.2,3 Adenocarcinoma is categorized as a form of non-small-cell lung cancer and constitutes 20%–30% of all lung cancers.4,5 Cigarette smoking is considered to be a major risk factor for lung cancer. 6 A previous study reported that cigarette smoking contributed to the increase of morbidity of lung adenocarcinoma. 7 Studies have been conducted on the pathogenesis of lung adenocarcinoma, more specifically focusing on the genetic and molecular mechanism. In recent years, improved diagnosis and surgery along with new treatments made considerable progress in enhancing patient survival.8,9 However, up to now, the molecular mechanisms of genesis and development of lung adenocarcinoma are still unclear. 10 When it comes to the definitive diagnoses and treatments for lung adenocarcinoma, there are much more waiting to be explored and studied.
MicroRNAs (miRNAs) are non-coding endogenous RNAs.11–13 They regulate the expression of messenger RNAs (mRNAs) and gene expression by targeting complementary sequences located on the 3′-UTR. 13 MiRNAs play an especially significant role in complicated cell progress such as cell differentiation, development, morphogenesis, invasion, metabolism, and migration.14,15 In lung adenocarcinoma, the expression of miRNAs differs markedly between tumor tissues and the adjacent normal tissues. 16 Additional studies discovered that miRNAs can regulate gene expression in lung adenocarcinoma. For example, the expression of let-7 miRNA is reduced in human lung adenocarcinoma. At the same time, the restoration of let-7 expression inhibited lung cancer cell growth in vitro. 17 A second example is observed in miR-873, which can directly target SRCIN1 to induce proliferation and migration of lung adenocarcinoma cells. 18 A third example can be seen in miR-29b, which was significantly downregulated in primary lung adenocarcinoma involving in the Src-ID1 signaling pathway. 19 Undoubtedly, miRNAs provide a valuable entry point into further researches on the genetic changes underlying lung adenocarcinoma.
Among all the miRNAs, emerging evidence reveals that the expression of miR-519 plays a notable role in the regulation of cells in various cancers. For example, miR-519d-3p is detected to be overexpressed in patients with Preeclampsia, where its overexpression was found to suppress the invasion and migration of trophoblast cells. 20 In cervical cancer, miR-519d facilitates the metastasis and progression including the apoptosis and proliferation of cancer cells by targeting Smad7. 21 Based on the whole studies on the effects of miR-519d in varied tumor disease, we aimed to determine the expression level of miR-519d in human lung adenocarcinoma cell lines and tumor tissues. We analyzed the possible mechanisms by identifying the potential target genes of miR-519d.
Materials and methods
Clinical patients
To investigate the expression of miR-519d and eukaryotic translation initiation factor 4 (eIF4H) mRNA in tumor tissues, 69 cases of lung adenocarcinoma tissues and adjacent normal tissues were collected at surgery from the First Affiliated Hospital of Zhengzhou University from February 2015 to January 2016, where each sample was immediately snap frozen in liquid nitrogen. Prior to the tissue collection, each patient has signed the informed consent form and the study has been approved by the ethics committee.
Cell culture and transfection
A549 and H1299 cell lines purchased from the cell bank of the Chinese Academy of Sciences (Shanghai, China) were some of the most widely used cell lines in the research of lung adenocarcinoma.22,23 Normal human bronchial epithelial (NHBE) cells were also obtained from the same cell bank and served as the normal control. Cells were incubated at 37°C in a humidified 5% CO2 atmosphere and grown in Dulbecco’s modified Eagle’s medium (DMEM; Gibco, USA) containing 10% fetal bovine serum (FBS; Gibco) and 2 mM mixture of penicillin (100 U/mL) and streptomycin (100 µL/mL; Invitrogen, USA).
When it comes to cell transfection, cells were incubated in the six-well plates at a density of 5 × 104 cells/well. Once the cell confluence reached 60%–80%, we conducted the transient transfection using Lipofectamine™ 2000 (Invitrogen) following the manufacturer’s instructions. In our study, the miR-519d agomir (GenePharma, China), miR-519d scramble (GenePharma), and si-eIF4H (GenePharma) were transfected into the A549 and H1299 cells, giving rise to four experimental groups: the miR-519d agomir group, the miR-519d scramble group, the si-eIF4Hgroup, and the Blank group, which was not transfected. The sequences of eIF4H siRNA used for transfection were as follows: 5′-GGAUCUCAGCAUAAGGAGU-3′ (sense) and 5′-ACUCCUUAUGCUGAGAUCC-3′ (antisense).
RNA isolation and quantitative real-time polymerase chain reaction
In tissues and cells, the expression of miR-519d and eIF4H mRNA was detected by quantitative real-time polymerase chain reaction (qRT-PCR) assays. Total RNA in lung adenocarcinoma tissues and adjacent normal tissues was extracted using trizol. Total RNA in A549 and H1299 cells was isolated through the application of the E.Z.N.A.® Total RNA Kit I (Omega Bio-tek, USA). The RNA was reversely transcripted to complementary DNA (cDNA) using a First Strand cDNA Synthesis Kit (Qiagen, Germany). All of these operations were performed strictly according to the manufacturer’s instructions. The cDNA was stored in a −80°C refrigerator, and the qRT-PCR was performed in a 30-µL ABI Power SYBR® Green PCR reaction mixture (Applied Biosystems, USA). During the qRT-PCR process, the relative expression was calculated by the 2−ΔΔCt method, the expression level of eIF4H was normalized using glyceraldehyde 3-phosphate dehydrogenase (GAPDH), and the expression level of miR-519d was normalized using U6. PCR primer used was as follows—eIF4H: forward 5′-GACTTCGACACCTACGACGAT-3′ and reverse 5′-TAGCCGTACACTCCTTATGCTG-3′; GAPDH: forward 5′-ACAACAGCCTCAAGATCATCAGC-3′ and reverse 5′-CACGCCACAGTTTCCCGGAG-3′; miR-519d: RT-primer 5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGTTTCACG-3′, forward 5′-TCCGAGTGAGATTTCCCTC-3′, and reverse 5′-GTGCAGGGTCCGAGGT-3′; U6: RT-primer 5′-GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAATA-3′, forward 5′-TCCGATCGTGAAGCGTTC-3′, and reverse 5′-GTGCAGGGTCCGAGGT-3′.
Western blotting
Protein samples weighing 50 µg were extracted from A549 and H1299 cells which had been transfected for 24 h and separated on a 10% sodium dodecyl sulfate (SDS) acrylamide gel (Bio-Rad Laboratories, USA) for 1.5 h at 90 V and transferred to a nitrocellulose membrane (Whatman, UK) for 2 h at 220 mA. Next, the membrane was blocked in 5% fat-free milk for 2 h and treated with the diluted primary antibody (1:2000, polyclonal rabbit anti-human eIF4H antibody; Santa Cruz Biotechnology, USA) overnight, followed by another treatment with the diluted secondary antibody (1:10,000, horseradish-peroxidase-conjugated goat anti-rabbit IgG; Santa Cruz Biotechnology) for 1 h at room temperature. After being washed with tris-buffered saline with Tween 20 (TBST) for five times, all blots were visualized by a chemiluminescence detection kit (Amersham Pharmacia Biotech, USA) using the ECL Plus (GE Healthcare, USA). β-actin was served as control. The experiment was repeated at least three times.
Luciferase reporter assay
According to the seed region researched through miRanda and TargetScan, we were ready to conduct the luciferase reporter vectors. The mutant type of eIF4H 3′-UTR fragments was amplified by Overlap PCR, and the wild-type eIF4H 3′-UTR fragments were amplified by regular PCR. When it comes to the Overlap PCR, the primer sequences of eIF4H were synthesized by Sangon Biotech (China). The primer sequences were as follows: forward 5′-AAAGAGCTCAGCGTTTCCCCCCATCCCTGTTGC-3′ and reverse 5′-CTCCTCGAGCTCAAAAGCCTAACCCCTAGCACT-3′ and mutant forward 5′-AATCTCCGTGAAAACTCATTTCCTTTGATAAATTGTACTATG-3′ and mutant reverse 5′-ATGAGTTTTCACGGAGATTCTAGACCCATTTTGGCTCTGAAC-3′. Then, we combined the fragments above with the pmirGLO vector in order to create the mutant and wild-type dual-luciferase reporter vectors. Next, we co-transfected either the mutant or the wild-type eIF4H 3′-UTR luciferase reporter vectors with either miR-519d agomir or miR-519d scrambles into the A549 and H1299 cells, respectively. After co-transfection for 24 h, relative luciferase activity was detected using the Dual-Luciferase Reporter Assay Kit (Promega, USA) according to the manufacturer’s instructions.
Cell Counting Kit-8 assay
The Cell Counting Kit-8 (CCK-8) assay was one of the methods we employed to examine the proliferation condition of A549 and H1299 cells. In this way, cells were cultured in a 96-well plate and divided into three groups, the miR-519d agomir group, the miR-519d scramble group, and the Blank group through cell transfection described in the “Materials and methods” section. After being treated for 0, 24, 48, and 72 h, the cell viability was measured at 450 nm by a microplate reader (Model 680; Bio-Rad Laboratories).
Colony formation assay
We examined the proliferation status of the A549 and H1299 cells using the colony formation assay. Cells were cultured in a six-well plate and divided into three groups: the miR-519d agomir group, the miR-519d scramble group, and the Blank group through cell transfection described in the “Materials and methods” section. After being incubated for 2 weeks, any colonies formed were stained using crystal violet, where cell clusters containing >50 cells were defined as surviving colonies. The cloning efficiency was calculated by dividing the average number of colonies per dish by the number of cells plated.
Xenograft tumor experiment
The xenograft tumor experiment allowed us to study the effect of miR-519d in vivo. In all, 30 of 6-week-old female BALB/c nude mice were purchased and assigned into three groups (10 per group): the mice injected with cells belonged to the miR-519d agomir group, the mice injected with cells belonged to the miR-519d scramble group, and the mice injected with normal saline belonged to the control group. The A549 cells were stably transfected with luciferase, and the cell viability and cell number were calculated by trypan blue; finally, 1 × 106 live cells were injected subcutaneously into the mice in different groups. With the passage of time, the luciferase signal intensity was visualized by IVIS Spectrum Imaging System (PerkinElmer, USA) according to the manufacturer’s instructions; the Guide for the Care and Use of Laboratory Animals has been followed during all of our animal study (National Institutes of Health).
Ki67 immunohistochemistry assay
After the xenograft tumor experiment, the tumors from the nude mice in three groups, including miR-519d agomir group, miR-519d scramble group, and the normal control group, were embedded in paraffin cut into 3-µm-thick sections and mounted onto adhesion microscope slides (Thermo LabServ, USA). For Ki67 immunohistochemistry, slides were incubated in 0.01 M sodium citrate (PH 6.0) which was heated to 100°C for 20 min to expose antigen. After washing with phosphate-buffered saline (PBS) for three times, 0.3% H2O2 was used to block endogenous peroxidase activity through 10 min incubation. Next, 1% normal goat serum in 0.01 M PBS was used to block non-specific binding. Then, slides were treated with 100 µL diluted rabbit anti-Ki67 antibody (1:200; Zhongshan Golden Bridge Biotechnology, China) which is a marker for cell proliferation at 4°C overnight. After washing with PBS for three times, slides were visualized using a SP staining kit (Zhongshan Golden Bridge Biotechnology, China) using diaminobenzidine (DAB) as a chromogen and counterstained with hematoxylin according to the manufacturer’s instructions. We counted the Ki67-positive cells by selecting at random 10 view fields per filter with a light microscope.
Transwell assay
Transwell insert chambers (Corning, USA) were applied in the detection of cell invasion. After cell starvation for 24 h, 5 × 104 cells were cultured in the upper chambers, which had been precoated with matrigel (Corning, USA), in serum-free DMEM medium in triplicate. In the lower chamber, DMEM medium containing 5% FBS was added. During the cell incubation process, the invasive cells would move from the upper chambers to the lower chambers. After being cultured for 48 h, we removed the filters containing the cells that migrated to the lower surface and suspended them in 70% ethanol. After 30 min, the cells were stained with 0.2% crystal violet. After another 20 min, we counted the migrated cells by selecting at random 10 view fields per filter with a light microscope.
Statistical analysis
SPSS 21.0 was the software used in the statistical analysis process. A least significant difference (LSD)
Results
The expression of eIF4H mRNA was upregulated and the expression of miR-519d was downregulated in lung adenocarcinoma tissues
In our study, 69 lung adenocarcinoma tissues were collected, representing the expression levels of miR-519d and eIF4H mRNA in variant conditions. The data obtained from these 69 samples indicate that the expression level of eIF4H mRNA was higher in the lymph node metastasis–positive group relative to that in the lymph node metastasis–negative group (0.645 ± 0.147 vs 0.580 ± 0.150;
The expression level of miR-519d and eIF4H found in lung adenocarcinoma tissues with various clinicopathological characteristics, taken from 69 lung adenocarcinoma patients.
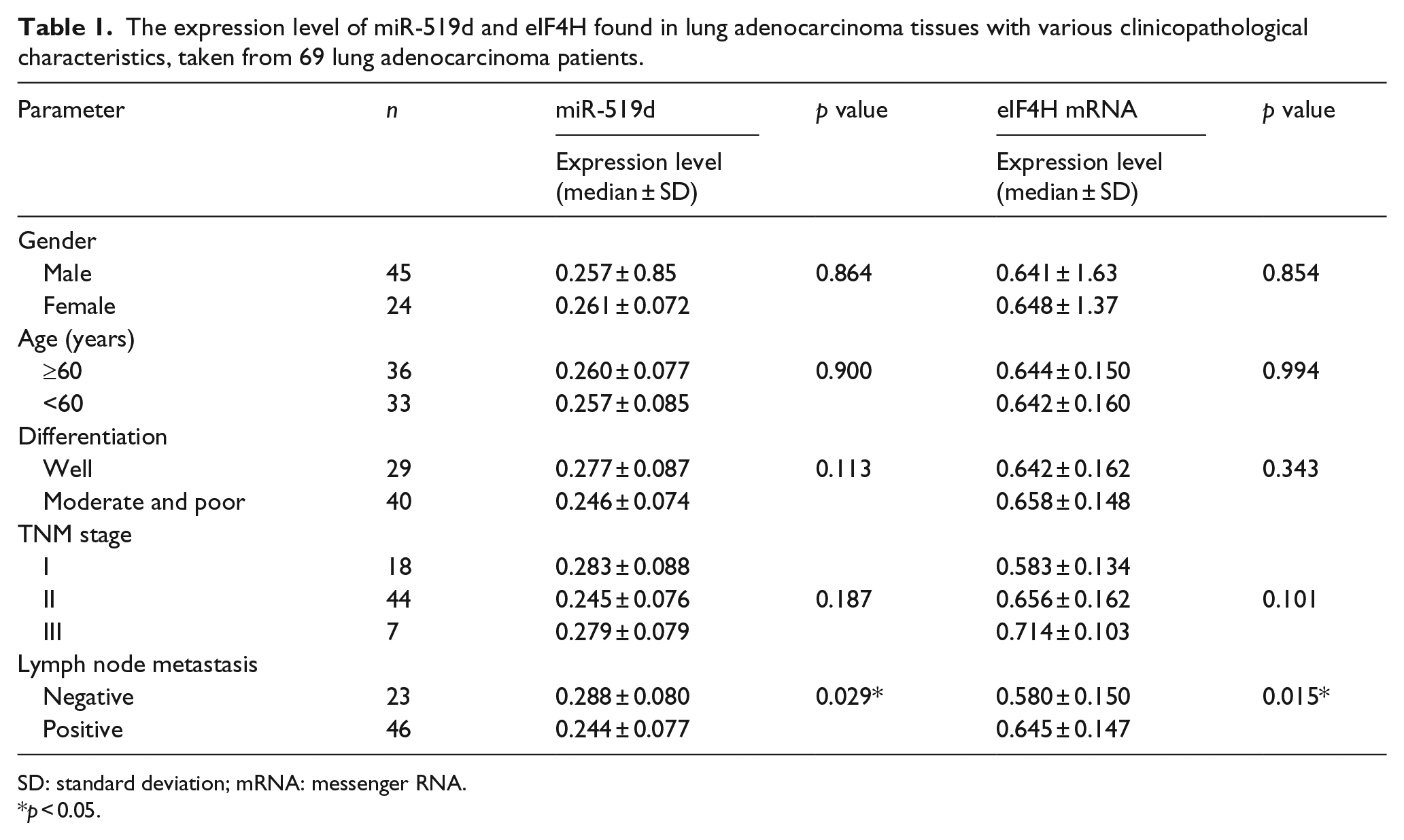
SD: standard deviation; mRNA: messenger RNA.
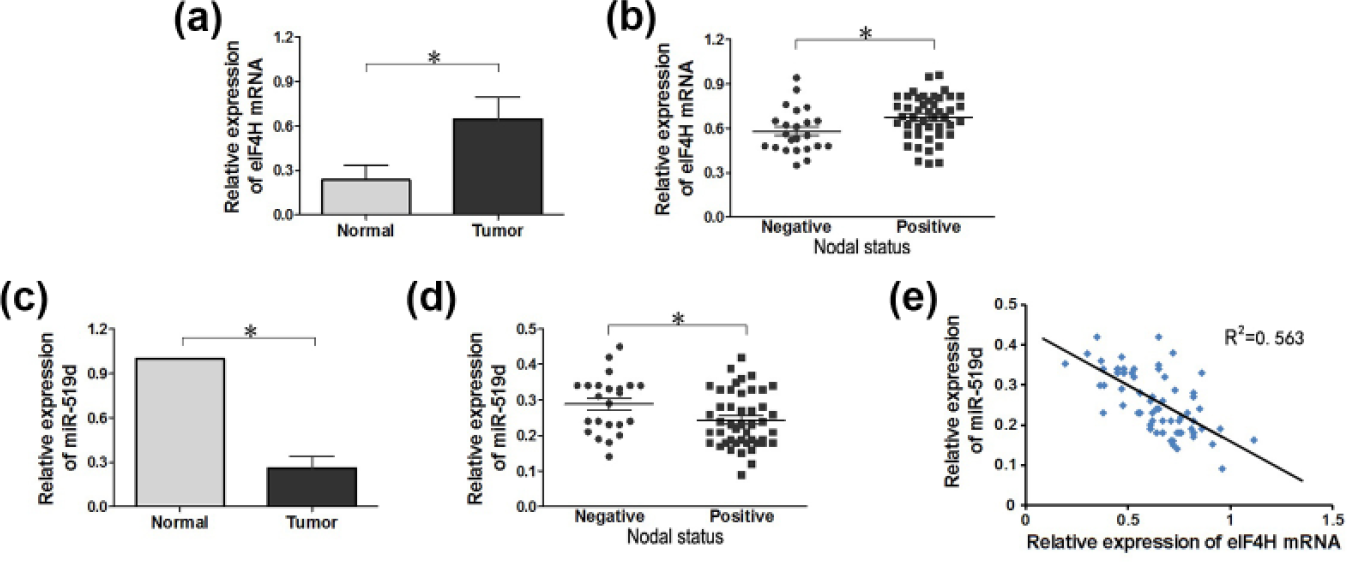
The expression of miR-519d and eIF4H mRNA in lung adenocarcinoma tumor tissues and cells as determined by qRT-PCR. (a) Relative expression of eIF4H mRNA in lung adenocarcinoma tumor tissues and adjacent normal tissues showing a statistically significant difference (
eIF4H was one of the targets of miR-519d and was negatively regulated by miR-519d
To further evaluate the relationship between miR-519d and eIF4H mRNA, bioinformatics analysis was applied and found the presence of a seed region between miR-519d and the eIF4H mRNA 3′-UTR (Figure 2(a)). Next, two kinds of lung cancer cell lines, A549 and H1299, were used to carry on the ensuing procedure research. The expression of eIF4H protein was visibly reduced in the miR-519d agomir group when compared to the negative control (NC) group and the miR-519d scramble group (

eIF4H is a target of miR-519d in A549 and H1299 cells. (a) The putative miR-519d binding sequence in the eIF4H 3′-UTR. (b) Western blotting analysis of eIF4H protein expression in translated A549 and H1299 cells. (c) The relative luciferase activities were measured after transfection. In cells co-transfected with mutation (Mt) eIF4H, luciferase activities of cells co-transfected with miR-519d had no obvious change compared with those of cells in the scramble group. However, in cells co-transfected with wild-type (Wt) eIF4H, the luciferase activities of cells co-transfected with miR-519d were significantly lower than those of cells in the scramble group (*
Overexpressed miR-519d restricted cell proliferation in A549 and H1299 cells
The level of miR-519d was measured by qRT-PCR among Blank group, the miR-519d scramble group, and the miR-519d agomir group to identify the transfection rate. The miR-519d agomir group showed a significantly higher level of miR-519d than the other two groups in both the A549 cell lines and the H1299 cell lines. The level of miR-519d in NHBE cells was served as normal control (

Overexpressed miR-519d restricted cell proliferation in A549 and H1299 cells. (a) The expression level of miR-519d after cell transfection. Cells transfected miR-519d agomir showed a significantly higher expression level than the Blank group and scramble group. (b and c) A CCK-8 assay was performed to detect cell proliferation with the passage of time (0, 24, 48, and 72 h). (d) A colony formation assay was conducted to detect cell proliferation (*
Overexpressed miR-519d restricted tumor growth in vivo
Tumor growth assays were conducted to further examine whether the overexpressed miR-519d could inhibit the growth of a xenografted tumor. As shown in Figure 4(a) and (b), the xenograft tumor experiment showed that the bioluminescence signal of A549 cells in the miR-519d agomir group was significantly weaker than that of cells in both the Blank group and the miR-519d scramble group (

Overexpression of miR-519d restricted tumor growth in vivo. (a and b) The luciferase signal of miR-519d agomir group cells was relatively weaker than the signal shown by the Blank group and the scramble group in A549 cells. (c and d) The luciferase signal of the cells in the miR-519d agomir group was significantly weaker than the signal shown by the Blank group and the scramble group in H1299 cells. (e) Paraffin-embedded tissue sections of the nude mice transplanted tumor tissues were stained with antibodies against ki-67 (20× magnification). The sections were counterstained with hematoxylin (*
Overexpressed miR-519d restricted cell invasion in A549 and H1299 cells
A transwell assay was conducted to evaluate the regulation in invasion activity of miR-519d in A549 and H1299 cells. As displayed by the number of cells penetrating the transwell membrane, there was no significant difference between the miR-519d scramble and the Blank groups (
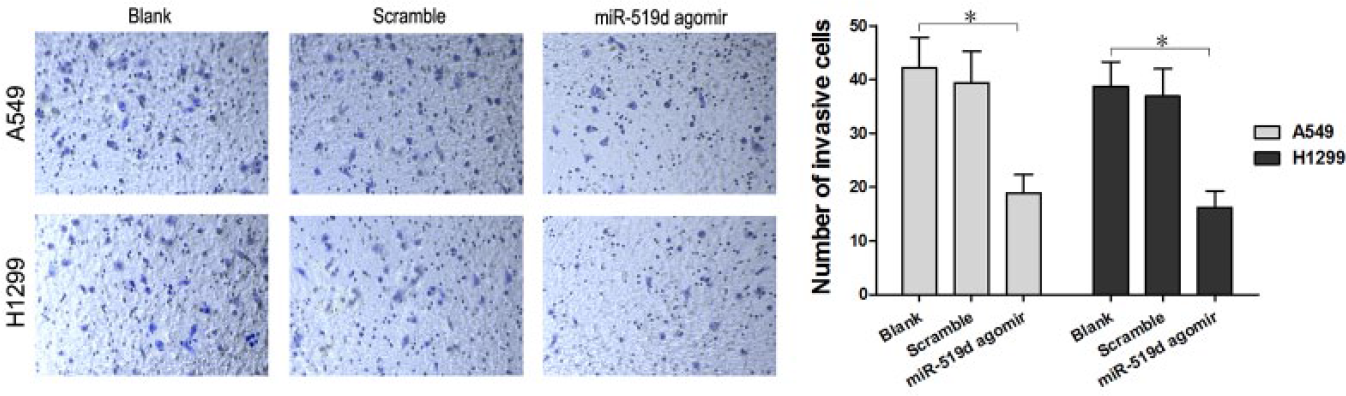
The overexpression of miR-519d restricted cell invasion in A549 and H1299 cells. The number of cells penetrating the transwell membrane in the miR-519d agomir group was significantly reduced compared to the Blank group and scramble group, as measured by the transwell assay (*
Downregulation of eIF4H and upregulation of miR-519d share the similar biological effects in A549 and H1299 cells
So far in our study, we have observed that the upregulation of miR-519d may play an inhibitory role in cell proliferation and invasion. Additionally, we also found that eIF4H was one of the targets of miR-519d and was negatively regulated by miR-519d. Along the same line, we then studied whether the downregulation of eIF4H would have similar biological effects as the upregulation of miR-519d, in order to further confirm our finding that miR-519d participates in the direct regulation of eIF4H. We transfected si-eIF4H to A549 and H1299 cells and found that compared to the Blank group and the miR-519d scramble group, eIF4H protein expression in si-eIF4H group was at a low level similar to that observed in the miR-519d agomir group through western blotting assay. CCK-8 assay and colony formation assay were performed and showed that the miR-519d agomir group and the si-eIF4H group both possessed visibly lower cell numbers than the Blank group and the miR-519d scramble group in the A549 cell lines, as well as in the H1299 cell lines (
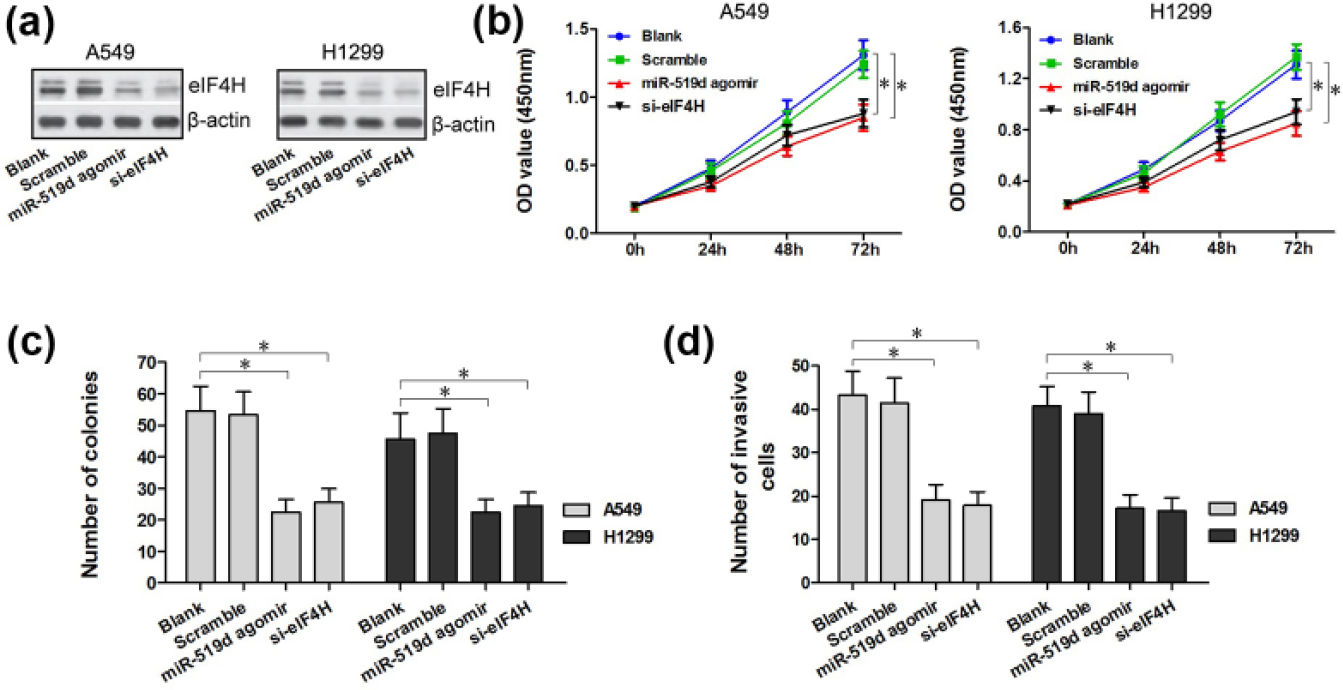
The downregulation of eIF4H was analogous to the overexpression of miR-519d. (a) Western blotting analysis of eIF4H protein expression in translated A549 and H1299 cells. (b–d) si-eIF4H in A549 and H1299 cells led to inhibitory cell proliferation and invasion, as measured by CCK-8, colony formation, and transwell assays. The differences are significant when compared with the cells in the Blank group and the scramble group (*
Discussion
In general, the reason that lung adenocarcinoma occurs in such high incidence and causes such high mortality rate lies in its wide invasion and metastasis. Recently, the mechanisms involved in the invasion and metastasis processes of the specific gene expression and regulation pathways have been widely explored in different cancers, such as colon cancer, esophageal cancer, breast cancer, and chronic myeloid leukemia.24–28 Due to findings from recent studies, many genetic factors, including lncRNA, 29 miRNA 30 as well as matrix metalloproteinase (MMP) family members, 31 have become associated with the metastasis of lung cancer.
MiR-519d is reported to be downregulated in lung cancer. 32 When miR-519d is upregulated, it provides beneficial effect of suppressing cancer progression, 33 and when considering its mechanisms of the regulatory ways, some studies have suggested that it is related with the expression of MMP-2 32 or STAT3. 33 In our experiments, we found that the expression of miR-519d was low enough to achieve statistical significance in lung adenocarcinoma tissues from A549 as well as H1299 cells undergoing lymph node metastasis. MiR-519d agomir was used to upregulate the expression level of miR-519d, and its subsequent inhibitory effects on cell proliferation and invasion were observed through a series of experiments, including the CCK-8 assay, the colony formation assay, the xenograft tumor experiment, the Ki67 immunohistochemistry assay, and the transwell assay. Later, more experiments were performed to explore a specific gene that may involve in the regulatory mechanisms of miR-519d.
The overexpression of eIF4H contributes to tumor promotion in stimulating protein translation in 93% of cancers. 34 Therefore, the detection of eIF4H is a potential therapeutic target for human cancer. 35 Fiume et al. 36 studied that eIF4H is under the transcriptional control of p65/NF-κB. Along the same vein, we explored whether eIF4H could be under the transcriptional control of a miRNA. The results we obtained demonstrated that eIF4H was statistically significantly upregulated in lung adenocarcinoma tissues and A549 as well as H1299 cells, and the statistics difference was associated with the presence of lymph node metastasis. We then discovered the inverse relationship between miR-519d and eIF4H, with the help of linear regression tests. To further investigate this relationship, bioinformatics analysis and luciferase report assay were applied and revealed that eIF4H was one of the targets of miR-519d and was negatively regulated by miR-519d. Moreover, we knockdown the expression of eIF4H and found that the downregulation of eIF4H and the upregulation of miR-519d share similar inhibitory effects in cell proliferation and invasion in A549 and H1299 cell lines. This finding provided further evidence for the assumptions that miR-519d affects the progression of lung adenocarcinoma at least partly by targeting the expression of eIF4H.
Conclusion
We concluded that miR-519d is downregulated in lung adenocarcinoma and that eIF4H serves as one of the targets of miR-519d. In A549 and H1299 cells, upregulated miR-519d inhibits cell proliferation and invasion via its association with eIF4H. The regulation of eIF4H as a target for miRNAs can serve as a novel direction in the targeted cancer diagnosis or therapy. Consequently, this brand new angle of research invites more detailed and deepened research efforts in the future.
Footnotes
Acknowledgements
The authors thank all staff at the research center who contributed to this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from Ministry of Major Science & Technology of Henan (201401005).
