Abstract
BACKGROUND:
Lung adenocarcinoma (LUAD) is a primary cause of cancer-patient mortality throughout the world. Thyroid hormone receptor interactor 13 (TRIP13) is a gene that expresses a protein involved in cell division, including tumorigenesis. Its expression is high in various human tumors; however, its role in LUAD cells remains undetermined.
OBJECTIVE:
To investigate the TRIP13’s role in the development of LUAD.
METHODS:
Bioinformation analysis was used to analyze the expression of TRIP13 in LUAD tissues and the impact on the prognosis of LUAD; CRISPR/Cas9 was used to construct the cell lines; CCK-8 was used to explore the cell proliferation; Transwell assays was applied to exam the cell migration and cell invasion abilities; Western blot and immunoprecipitation was used to explore the relation between TRIP13 and AKT/mTORC1/c-Myc signaling pathway.
RESULTS:
By analyzing LUAD data from The Cancer Genome Atlas and the Gene Expression Omnibus databases, we determined that TRIP13 is highly expressed in LUAD tissues and that this expression level has a negative impact on the patient mortality. TRIP13 has also proved to promote LUAD cell proliferation, migration, and invasion. In this study, we demonstrated that TRIP13 activates AKT/mTORC1/c-Myc signaling in these cells.
CONCLUSION:
Our results have identified the role and potential mechanism by which TRIP13 affects LUAD cells, which may provide a useful marker for helping to diagnose this disease and create new therapies against it.
Introduction
Lung adenocarcinoma is a leading cause of cancer related death throughout the world. In 2019, there were 228,150 new cases of LUAD and 142,670 persons were projected to die from the disease in the United States [1]. Non-small-cell lung cancer (NSCLC) accounts for
Studies have indicated that the rapamycin complex (mTORC) signaling pathway in mammals regulates tumor cell growth, survival, and metabolism [6, 7]. MTOR, a serine/threonine protein kinase, creates mTOR complexes 1 (mTORC1) and mTORC2. mTORC1 is regulated by AMP-activated protein kinase (AMPK) and its activation results in the phosphorylation of protein-translation and cell-growth mediators, such as ribosomal S6 kinase 1 (S6K1) and 4EBP1 [8, 9]. Several studies have demonstrated that mTOR is frequently activated in tumors; therefore, mTOR is a promising target for tumor therapy [6, 10]. Studies have also shown that c-Myc expression and activity is mediated by mTOR [11, 12]. C-Myc is a proto-oncogene that encodes a nuclear phosphoprotein and helps regulate the cell cycle, apoptosis, and malignant transformation of tumors [13, 14, 15].
Thyroid hormone receptor interactor 13 (TRIP13) is a protein-coding gene in the AAA ATPase family. Recent studies have suggested that the AAA ATPase family is involved in the spindle-assembly checkpoint, DNA break/repair, and chromosome synapsis [16, 17]. TRIP13 in humans, which helps regulate mitosis, is located on chromosome 5 and encodes a protein comprising 432 amino acid residues. TRIP13 is overexpressed in various cancers, which indicates that it might be associated with tumor progression and poor survival [18, 19, 20]. For example, Tao et al. have reported that TRIP13 is overexpressed in multiple myeloma (MM), and that it impairs the spindle assembly checkpoint, a mechanism of mitosis, through the Akt pathway and is thus associated with a poor prognosis. Moreover, a small TRIP13 inhibitor has been identified based on the crystal structure of wild-type TRIP13 in humans. Wang et al. have determined that this inhibitor suppresses MM progression, which suggests that targeting TRIP13 can be effective therapy against MM. Li et al. have shown that TRIP13 overexpression is also associated with LUAD; however, the specific mechanism by which TRIP13 increases tumorigenesis in this disease must be further studied.
In the current study, we investigated TRIP13’s role in the proliferation, migration, and invasion of lung cancer cells. We evaluated at mechanistic levels the changes in mTORC1 and c-Myc after TRIP13 knockdown or overexpression to provide more information for creating a targeted therapy against lung cancers.
Materials and methods
Cell lines and cultures
Human LUAD cell lines SK-LU-1 and A549 were obtained from the cell bank at the Chinese Academy of Medical Sciences (Beijing, China) and maintained in 5% CO
Patients and LUAD samples
LUAD gene expression datasets with relevant clinicopathological information were collected from The Cancer Genome Atlas (TCGA) database with datasets to June 2019. Patients with clinicopathological and survival information were included for followup. We also searched “lung cancer” and “Homo sapiens” in the Gene Expression Omnibus (GEO) database (
Meta-analyses
To further confirm the accuracy of our results, we conducted a meta-analysis using Review Manager, which estimated the error from the heterogeneity of platform. Continuous outcomes were estimated as a standard mean difference with a 95% confidence interval.
CEPIA2 and GSEA
The correlation between TRIP13 expression and that of other genes was investigated using the CEPIA2 web server, which uses data on normal tissues from TCGA and GTEx (
CRISPR/Cas 9-mediated TRIP13 deletion
TRIP13 was knocked down using the clustered regularly interspaced short palindromic repeats (CRISPR)/ Cas9 system. Yahata et al. have previously desscribed the process of CRISPR gene editing. Briefly, the oligonucleotides were designed according to http:// crispr.mit.edu and cloned into the lentiCRISPR/Cas9 vector (Addgene, cat. no. 49535) using the Zhang laboratory methods. The single-guide (sg)RNAs for TRIP13 were as follows:
SgRNA-1-F 5’-CACCGTGAGTAGCTTTCTAAC ACTC-3’ SgRNA-1-R 5’-AAACGAGTGTTAGAAAGCTA CTCAC-3’ SgRNA-2-F 5’-AAACGAGTGTTAGAAAGCTA CTCAC-3’ SgRNA-2-R 5’-AAACTGGGCTTTGGGACAGC TTGGC-3’
SK-LU-1 and A549 cells were then infected for 72 h with lentiCRISPRv2 plasmids and selected with 10
PcDNA3.1-TRIP13(
Cell proliferation
Cell proliferation was measured using the Cell Counting Kit-8 (CCK-8) and colony-formation assays. For the CCK8 assay, 800 cells/well were planted into 96-well plates and cultured for 0, 24, 48, or 72 h, after which 10
Cell migration and invasion
The Transwell assay was used to assess cell migration and invasion. First, the Transwell filter chambers were precoated with or without Matrigel (BD Bioscience). Then, 5
Western blotting
The lung cell lines were collected and lysed using radioimmunoprecipitation assay buffer. The protein content of each lysate was assayed using bicinchoninic acid, after which 40
Statistical analyses
SPSS 23.0 (IBM Corp., Armonk, NY, USA) was used for the statistical analyses. The data on the continuous variables are presented as the mean
Thyroid hormone receptor interactor 13 (TRIP13) was overexpressed in human lung cancer tissues. (A) TRIP13 was significantly upregulated in lung adenocarcinoma (LUAD) tissues compared with that in normal tissues as indicated in The Cancer Genome Atlas database. (B) The analysis of the area under the curve of the expression of TRIP13 in LUAD. (C) Meta-analysis of TRIP13 mRNA expression in LUAD from the Gene Expression Omnibus database. (D) Funnel plot showing the heterogeneity of all studies included in the meta-analysis.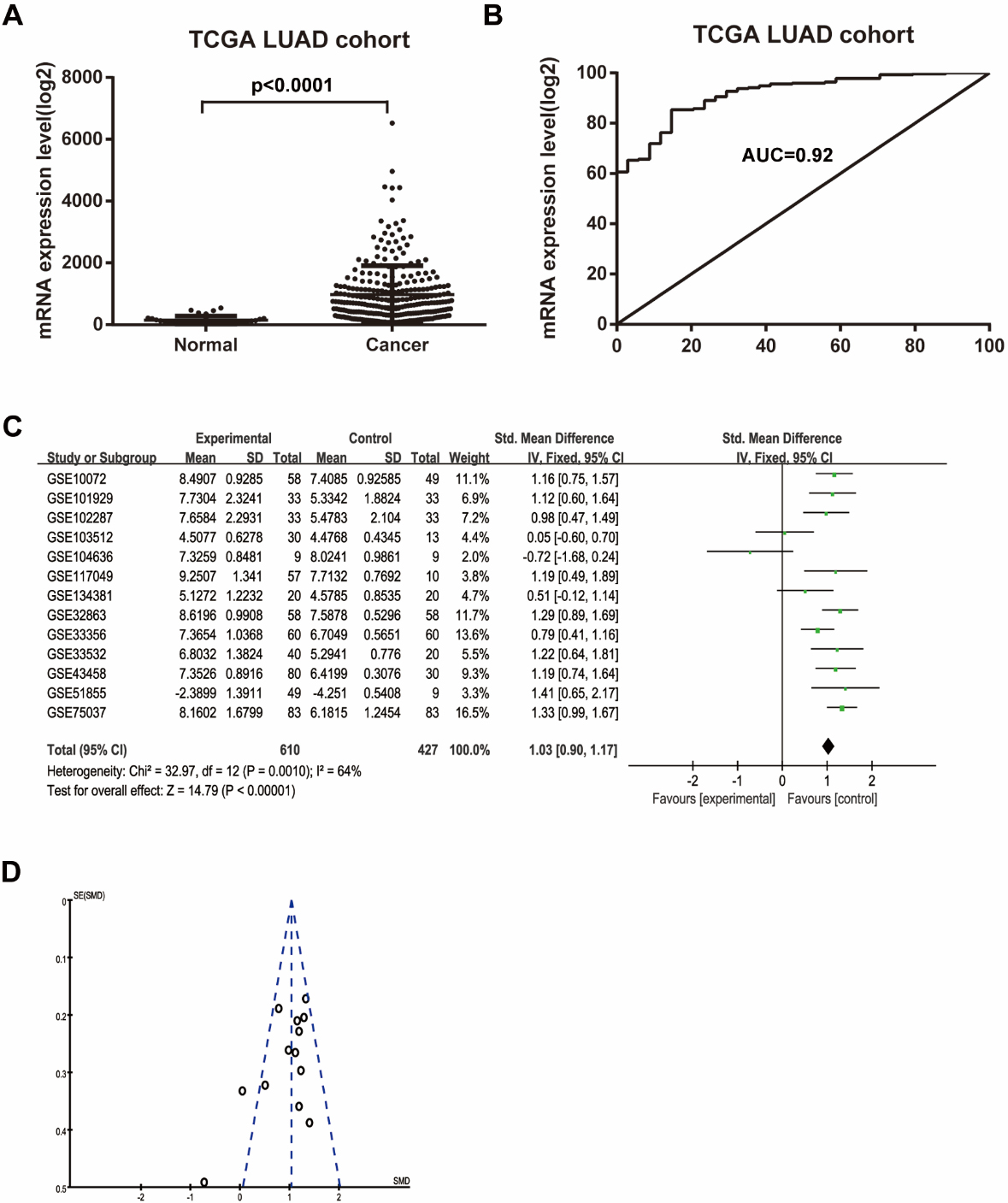
Thyroid hormone receptor interactor 13 (TRIP13) was associated with a poor prognosis and an independent prognostic factor in lung adenocarcinoma (LUAD) patients. (A) TRIP13 expression in cancers. (B) Expression of TRIP13 was significantly associated with LUAD stage. (C) Overall survival (OS) analysis of TRIP13 expression in 462 LUAD patients. (D) Analysis of progression-free survival (PFS) of TRIP13 in 462 LUAD patients.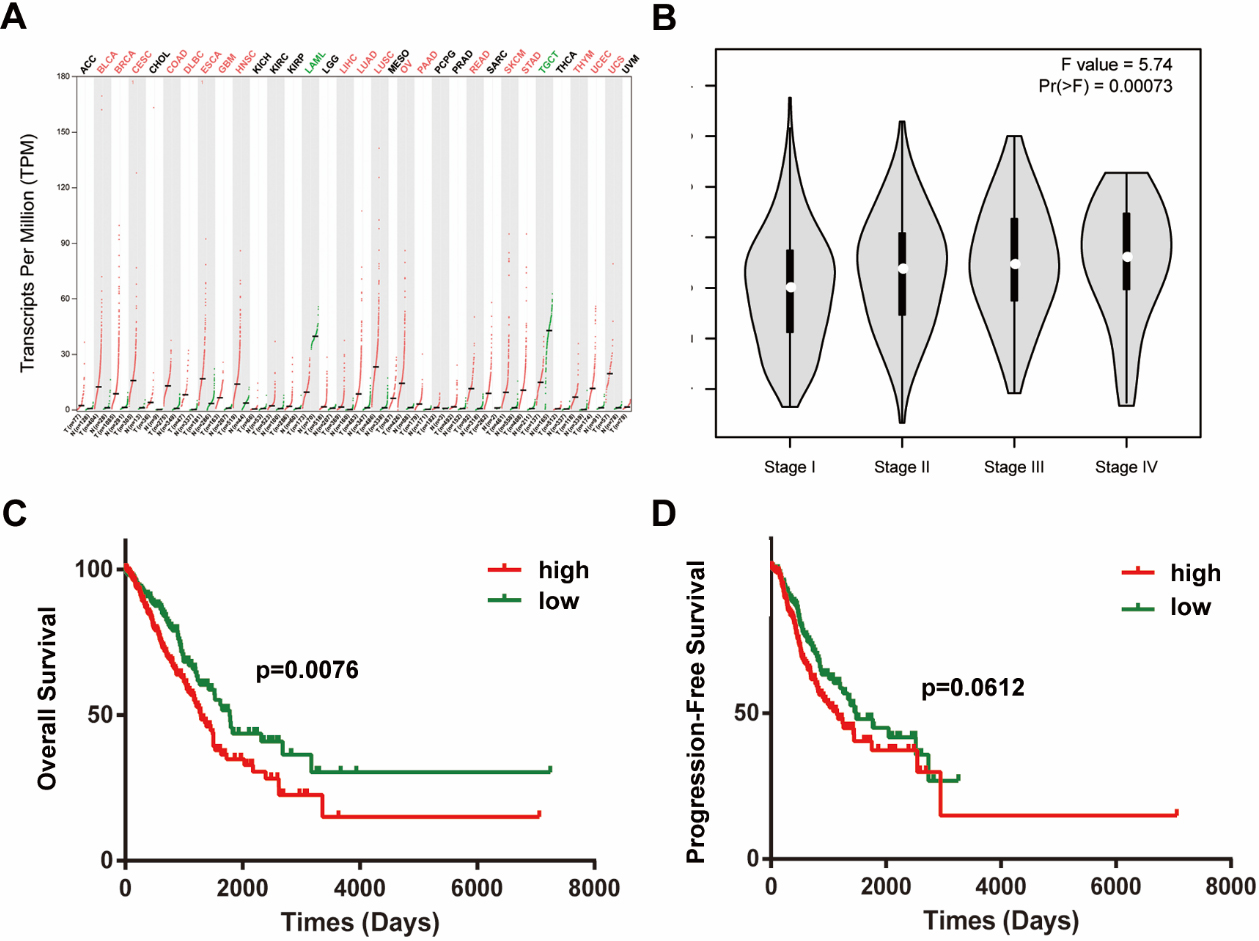
TRIP13 is overexpressed in human lung cancer tissues
We compared TRIP13 mRNA expression in LUAD (
The detailed information of LUAD chips
The detailed information of LUAD chips
Downregulated Thyroid hormone receptor interactor 13 (TRIP13) inhibited the lung adenocarcinoma (LUAD) cell proliferation, migration, and invasion in vitro. (A) Protein expression of TRIP13 in SK-LU-1 and A549 cell lines confirmed by Western blotting. (B) and (C) Knockdown of TRIP13 inhibited cell proliferation as determined by the Cell Counting Kit 8 (B) and colony-formation assays (C). (D) and (E) Knockdown of TRIP13 in SK-LU-1 and A549 cell lines decreased their migration (D) and invasion (E) abilities cells as determined by Transwell assays, respectively.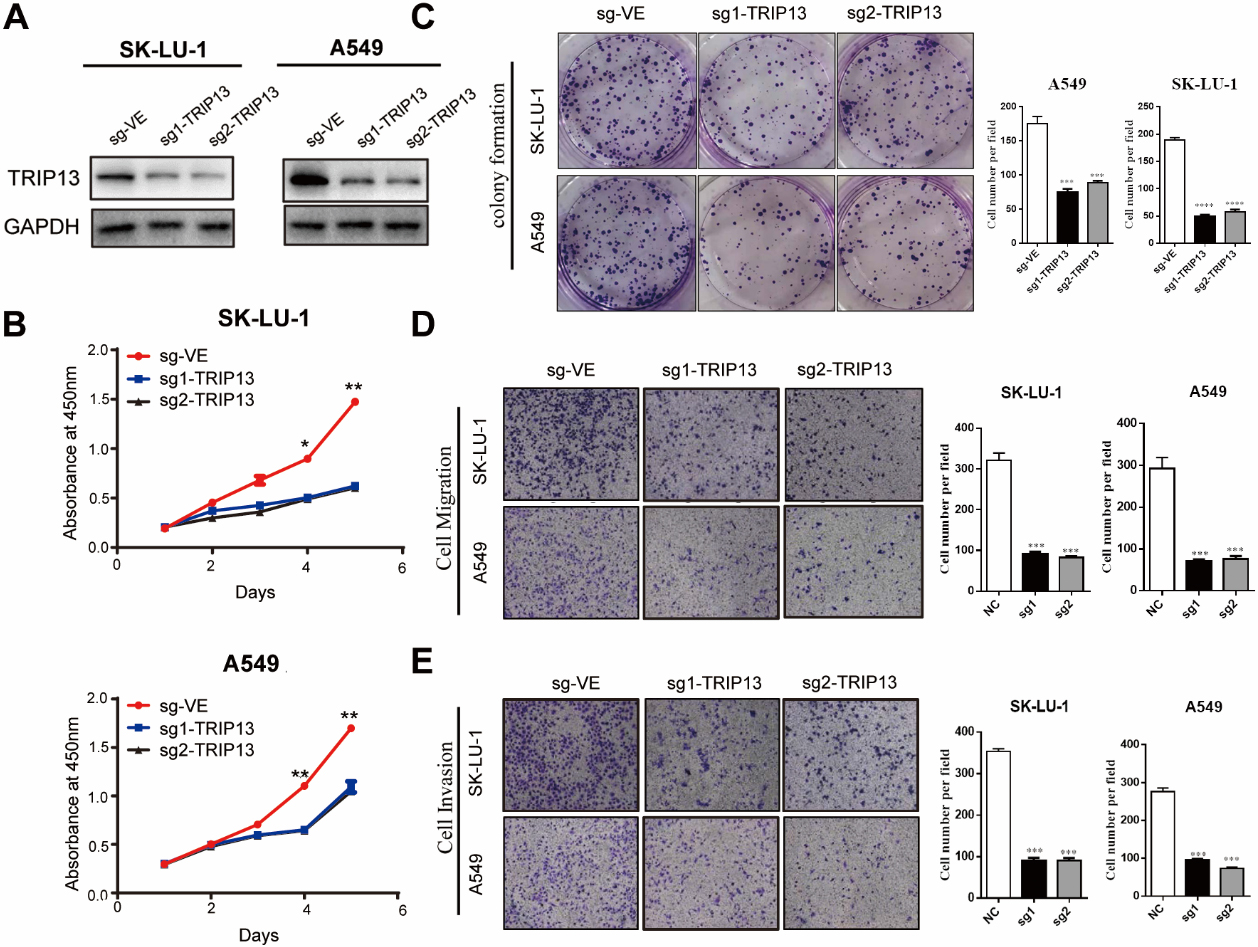
Upregulated Thyroid hormone receptor interactor 13 (TRIP13) promoted lung adenocarcinoma (LUAD) cell proliferation, migration, and invasion in vitro. (A) Validation of the efficiency of TRIP13 in SK-LU-1 and A549 cell lines using Western blotting. (B) and (C) TRIP13 overexpression increased cell proliferation as determined using Cell Counting Kit 8 (B) and colony-formation assays (C). (D) and (E) TRIP13 overexpression in SK-LU-1 and A549 cell lines enhanced their migration (D) and invasion (E) abilities as determined by Transwell assays, respectively.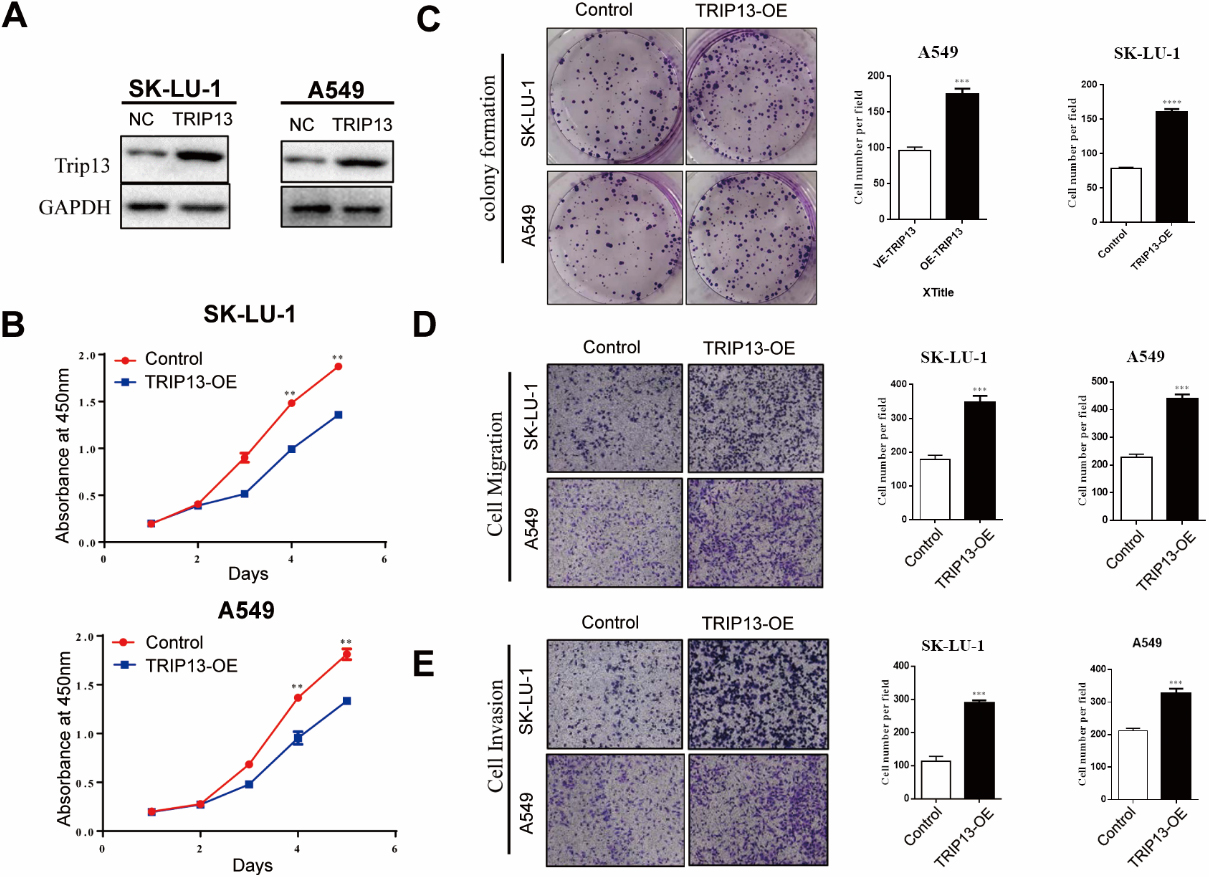
The CEPIA2 web server helped identify that TRIP13 expression increases most tumors, including lung cancer cells (Fig. 2A). Moreover, TRIP13 expression was significantly associated with LUAD stage (Fig. 2B). To determine the clinical significance of this increased expression, we examined the relationship between TRIP13 and widely recognized clinicopathologic factors. Patients were divided into high- and low-TRIP13 groups based on TRIP13 mRNA expression from the TCGA database, median expression of TRIP13 was used as the cutoff value. Patients in the high-TRIP13 group had a shorter overall survival time than those in the low group (
In addition, we found that TIRP13 expression was significantly correlated with lymph metastasis (
Comparison of clinicopathological profiles between low and high TRIP13 expression in LUAD patients
Comparison of clinicopathological profiles between low and high TRIP13 expression in LUAD patients
Biological analysis of Thyroid hormone receptor interactor 13 (TRIP13) – related pathway in lung adenocarcinoma (LUAD). (A) and (B) Gene set enrichment analysis results showed that TRIP13 was significantly related to cell cycle and mTOR signaling. (C) and (D) Expression of TRIP13 is positively correlated with mTOR and c-Myc expression based on the analysis of the CEPIA2 web server.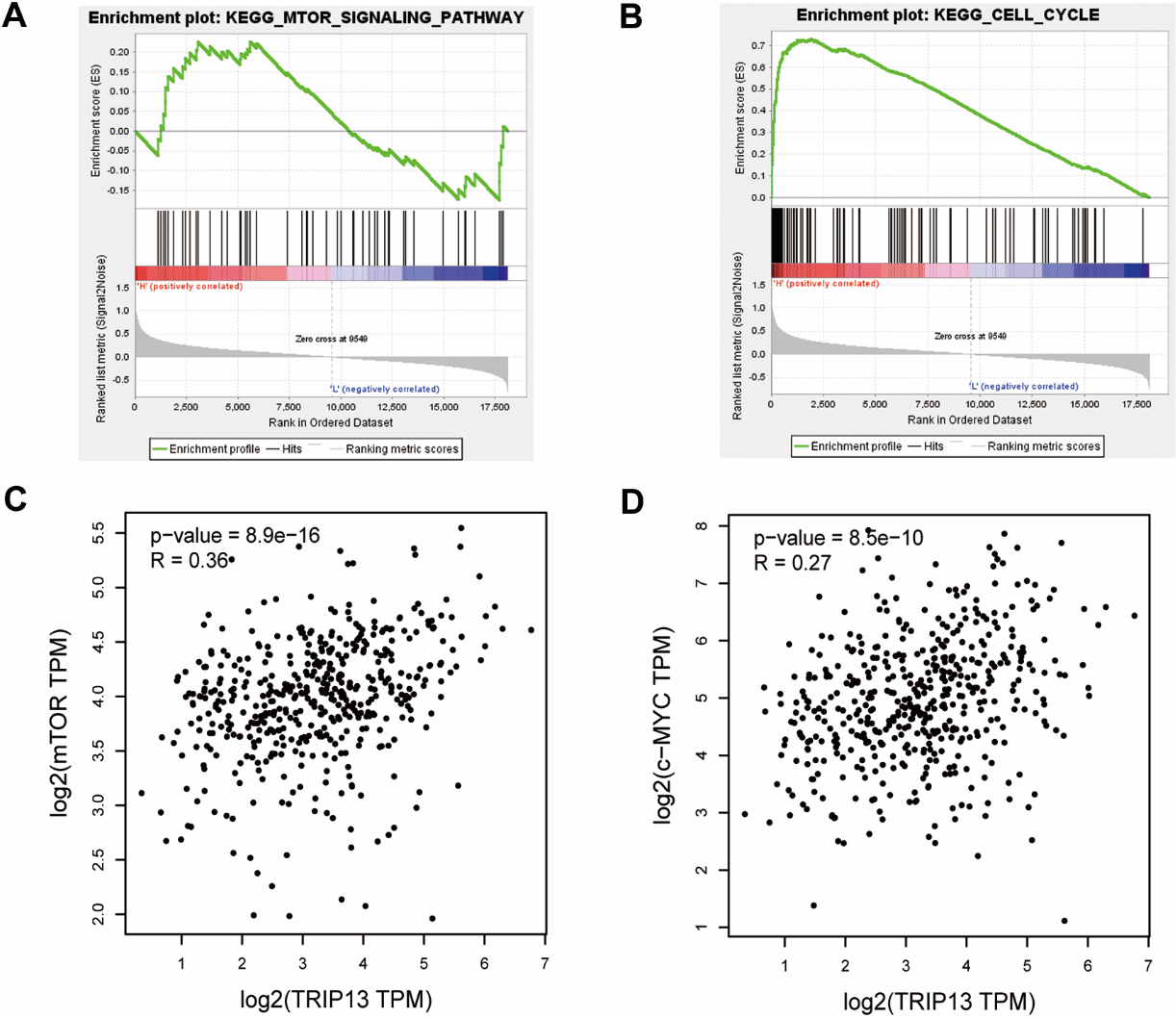
Thyroid hormone receptor interactor 13 (TRIP13) stimulated AKT/mTORC1/c-Myc signaling in lung adenocarcinoma (LUAD) cells. (A) TRIP13 downregulation reduced the level of phosphorylated AKT, mTOR, P70S6K, and c-Myc without changes in AKT, mTOR, and P70S6K. (B) TRIP13 overexpression enhanced the expression of phosphorylated AKT, mTOR, P70S6K, and c-Myc without changes in AKT, mTOR, and P70S6K.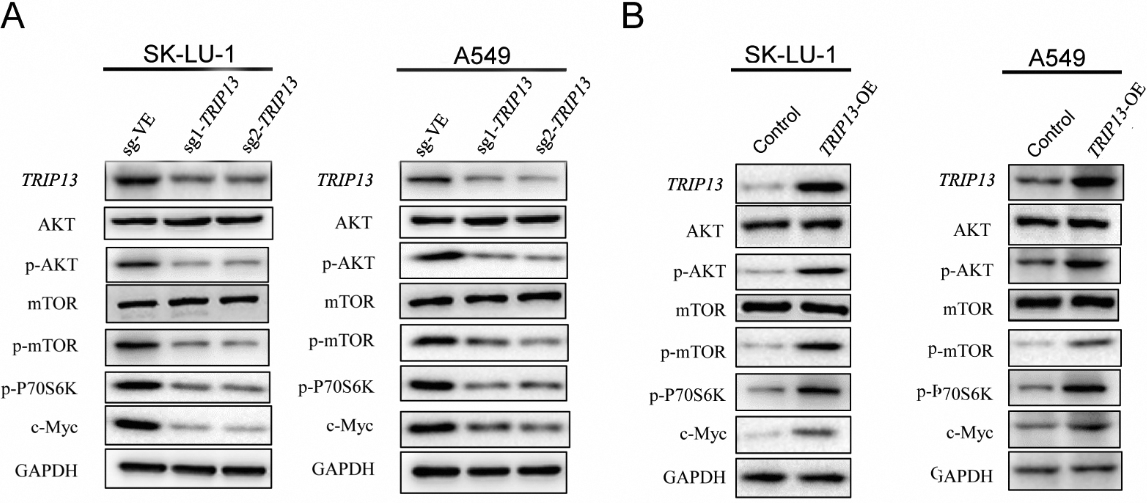
Thyroid hormone receptor interactor 13 (TRIP13) regulated the c-Myc-induced cyclin-dependent kinases (CDKs) and cyclins in lung adenocarcinoma (LUAD) cells. (A) The expression of CDK2, CDK4, cyclin D1, and cyclin E1 decreased with TRIP13 downregulation in LUAD. (B) The expression of CDK2, CDK4, cyclin D1, and cyclin E1 increased with TRIP13 overexpression in LUAD cells.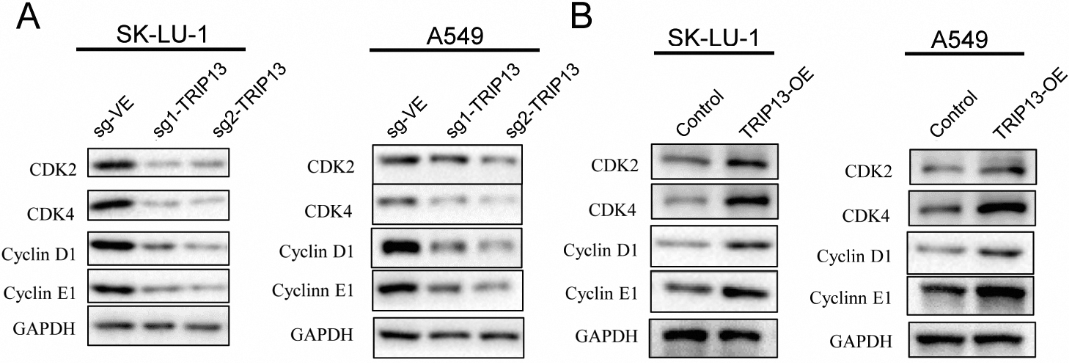
Thus, we suggest that TRIP13 can be used as a potential biomarker for predicting LUAD patient survival, providing additional prognostic information for clinical staging.
The CCK8 and colony-formation assays helped determine whether TRIP13 knockdown had any inhibitory effect on LUAD cell proliferation. The results of the CCK8 assay indicated that cell proliferative ability decreased when TRIP13 was downregulated in LUAD cells when compared with those in the control group (Fig. 3B). The colony-formation assay indicated that downregulation of TRIP13 expression significantly inhibited formation of SK-LU-1 or A549 cells (Fig. 3C). The results of the Transwell assays indicated that the number of invasive cells after TRIP13 silencing was significantly lower than that in the control group (Fig. 4D and E). These results suggested that TRIP13 knockdown could partly suppress the proliferative, migratory, and invasive abilities of LUAD cells.
Upregulated TRIP13 promoted LUAD cell proliferation, migration, and invasion in vitro
After overexpressing TRIP13 in SK-LU-1 and A549 cells, the results of the CCK-8 assays confirmed that cell proliferation was significantly facilitated compared with that in the control (Fig. 4B). In addition, TRIP13 significantly influenced the formation of cell colonies (Fig. 4C). The Transwell assays showed that TRIP13 upregulation significantly increased the number of SK-LU-1 and A549 cells migrating and invading the lower chamber, respectively, (both
Biological analysis of TRIP13-related pathway in LUAD
Previously studies have demonstrated that the AKT/ mTORC1 pathway and c-Myc, the wellcharacterized downstream target, are overexpressed in LUAD, promoted tumor proliferation and metastasis, and regulated LUAD-cell survival [22, 23, 24]. The mTOR signaling pathway plays an important role in cell proliferation, metastasis, survival, metabolism, and immunity [10, 25]; therefore, we hypothesized that TRIP13 regulated LUAD proliferation and metastasis through AKT/mTORC1/c-Myc signaling.
Firstly, GSEA [21] was conducted to explore the molecular mechanism of TRIP13-induced LUAD invasion and metastasis. Using this method, we identified that TRIP13 was significantly related to cell cycle (
TRIP13 promotes oncogenic progression in LUAD by stimulating AKT/mTORC1/c-Myc signaling
TRIP13 expression was downregulated in LUAD cells, and mTOR signaling-related proteins were explored using Western blotting. This downregulation reduced the level of phosphorylated AKT, mTOR, P70S6K, and c-Myc. Meanwhile, no obvious changes were observed in total AKT, mTOR, and P70S6K (Fig. 6A). Conversely, compared with the untreated cells, the levels of phosphorylated AKT, mTOR, and P70S6K increased when TRIP13 was overexpressed in the LUAD cells (Fig. 6B).
TRIP13 regulated the c-Myc-induced cyclin-dependent kinases and cyclins in LUAD cells
Wang et al. have reported that c-Myc upregulated the expression levels of cyclin-dependent kinases (CDKs) and cyclins in LUAD cells; therefore, we analyzed whether TRIP13 plays a role in the c-Myc-induced cell cycle. Our results showed that when TRIP13 was silenced, the expression of CDK2, CDK4, cyclin D1, and cyclin E1 decreased (Fig. 7A); however, when TRIP13 was overexpressed in the LUAD cells, the expression of CDK2, CDK4, cyclin D1, and cyclin E1 dramatically increased (Fig. 7B). Thus, TRIP13 significantly regulated the c-Myc–induced cell cycle in LUAD cells.
Discussion
Lu et al. [20] have indicated that TRIP13 is overexpressed in various human tumors. A study by Vader [17] has indicated that TRIP13 is a key player in cell progression and specific chromosomal events, such as controlling cell division. The classical transcription factor c-Myc is upregulated in many malignancies. This aberrant expression in the cancer cells promotes tumorigenesis by accelerating the cell cycle process [26, 27]. It is well known that activation of c-Myc can be regulated by the AKT/mTORC1 signaling pathway [11, 28]; therefore, we speculated that TRIP13 might regulate tumor progression through the AKT/mTORC1/c-Myc signaling pathway. To verify this, we transfected SK-LU-1 and A549 cells with a TRIP13-entiCRISPRv2 plasmid and pcDNA3.1-TRIP13 overexpression vector, respectively. The results showed that downregulation of TRIP13 in LUAD cells suppressed cell growth and metastasis in vitro, and Western blot analyses demonstrated that this downregulation decreased the expression of the AKT/mTORC1/c-Myc pathway. In addition, TRIP13 overexpression in LUAD cells had a directly opposite effect. These results indicated that TRIP13 plays an important oncogenic role in LUAD cells.
We determined that TRIP13 expression increased in most tumors, including lung cancer cells, a finding consistent with those of previous reports that TRIP13 is overexpressed in NSCLC [29], hepatocellular carcinoma [30], and colorectal cancer [31]. The results of our study indicated based on the TCGA database that TRIP13 was more upregulated in LUAD tissues than in normal tissues, which was further confirmed in the GEO datasets. From these results, we suggest that TRIP13 plays a critical role in LUAD progression. We then investigated the correlation between TRIP13 expression and the clinicopathological parameters in LUAD patients. Our results showed a positive correlation between TRIP13 and lymph metastasis and TNM stage. In addition, patients with a low TRIP13 expression had longer overall and disease-free survival rates than those with a high TRIP13 expression. These results suggest that TRIP13 is significantly associated with a patient’s prognosis and acts as an oncogene in LUAD.
Tumor metastasis and unlimited cell growth are the main causes of failed cancer treatment. Various interrelated biological events, such as proliferation, migration, invasion, angiogenesis, and adhesion, promote these processes [31, 32]. Previous studies have reported that TRIP13 promotes hepatocellular carcinoma growth and metastasis [30], and that high levels of TRIP13 promote cell proliferation and drug resistance in bladder cancer [20]. The data from our current study demonstrated that TRIP13 downregulation inhibited LUAD cell proliferation, migration, and invasion. In addition, TRIP13 overexpression distinctly enhanced LAUD cell proliferation, migration, and invasion. These findings suggest that TRIP13 plays a vital role in LUAD tumorigenesis; however, the molecular mechanism by which TRIP13 regulates this progression was unclear. We thus applied GSEA to identify the biological signaling associated with TRIP13 expression. Interestingly, the results showed that TRIP13 was significantly related to mTOR signaling and regulated the cell cycle. Substantial evidence has demonstrated that mTOR signaling plays an important role in cancer proliferation, invasion, and metastasis [33], and that aberrated mTOR signaling was observed in various tumors, such as pancreatic cancer [34], bladder cancer [35], and especially in LUAD [36]. In the represent study, our Western blot analyses, conducted to investigate the expression levels of mTOR-related protein, indicated that TRIP13 downregulation reduced p-AKT, p-mTOR, p-P70S6K, and c-Myc expression, although there were no obvious changes in the total amounts of AKT, mTOR, and P70S6K. In addition, TRIP13 overexpression in LUAD had the opposite effect. These results indicated that TRIP13 may promote the progression of LUAD through the AKT/mTORC1/c-Myc signaling pathway [37].
C-Myc is a transcription factor upregulated in several cancers [38]. It promotes tumor-cell progression by regulating the cell cycle [39, 40]; therefore, we speculated that the expression of cell cycle-related proteins would be adjusted to the expression of c-Myc. Previous studies have shown that the expression of CDK2, CDK4, cyclin D1, and cyclin E1 was consistent with the expression of TRIP13 in LUAD. When TRIP13 was downregulated, CDK2, CDK4, cyclin D1, and cyclinE1 were also downregulated and when TRIP13 was overexpressed in LUAD, these proteins were also upregulated. These results demonstrated that TRIP13 may regulate the LUAD cell cycle through the c-Myc-dependent pathway; however, the mechanisms for this must be further explored.
Conclusions
Our results demonstrate that TRIP13 is significantly related to LUAD patient prognosis and mediates the proliferation, invasion, and metastasis of LUAD cells through AKT/mTORC1/C-Myc signaling. Our study demonstrated that TRIP13 has a significant effect on the survival and progression of LUAD cells and may promote LUAD progression through AKT/mTORC1/c-Myc signaling pathway, regulate the LUAD cell cycle through the c-Myc-dependent pathway, and serve as a potential biomarker for LUAD progression to create a therapeutic target against LUAD; however, more detailed mechanisms by which these activities are implemented must be explored in additional studies.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
Funding
This work was supported by the Scientific Research Foundation of the First Affiliated Hospital of Wenzhou Medical University (Grant No. FHY2019002) and the Wenzhou Science and Technology Bureau (Y2020168).
