Abstract
BACKGROUND:
Prostate transmembrane protein androgen-induced 1 (PMEPA1), a critical checkpoint of multiple signaling pathways, has been demonstrated to play a crucial role in various types of cancers. However, little is known about its function in non-small cell lung cancer (NSCLC).
OBJECTIVE:
Our objective is to explore the function of PMEPA1 and its potential mechanisms in NSCLC progression.
METHODS:
PMEPA1 expression and prognostic significance in adenocarcinoma of lung cancer (LUAD) and squamous cell carcinoma of lung cancer (LUSC) were determined using Gene Expression Profiling Interactive Analysis (GEPIA). Next, a series of cell assays were performed to examine whether overexpression or depletion of PMEPA1 affected the malignant behaviors of NSCLC H1299 cells, such as proliferation and migration. Luciferase reporter gene assays and SP600125 (a JNK inhibitor) were employed to ascertain the regulatory relationship between PMEPA1 and JNK.
RESULTS:
PMEPA1 is overexpressed in LUAD and LUSC tissues and portends a worse prognosis for cancer patients. Gain and loss of function experiments demonstrated that PMEPA1 executes oncogenetic function in H1299 cells. Mechanism studies elucidated that PMEPA1 stimulated the transcriptional activity of the JNK pathway.
CONCLUSION:
PMEPA1 increased the H1299 cell viability, proliferation, and migration which works, at least partially, by triggering the JNK activity. Hence, our findings support that the PMEPA1/JNK axis might be a promising therapeutic target for this challenging disease.
Introduction
Owing to its high incidence, mortality, and recurrence, lung cancer represents one of the most prevalent malignant tumors [1]. Recent epidemiologic studies demonstrated that cigarette smoking, air pollution, and genetic factors aggravated the burden of this malignancy [2]. Notably, non-small cell lung cancer (NSCLC) is a predominant subgroup of lung cancers, with an extremely dismal 5-year survival rate [3]. Although great strides have been made in the diagnosis and treatment of NSCLC [4, 5, 6], the early diagnostic rate and prognostic value remain unsatisfactory. Therefore, it is urgently required to verify the pathological and etiological mechanisms underlying NSCLC initiation and progression for future therapy.
PMEPA1 protein, located on chromosome 20q13, is known as a type Ib transmembrane protein and consists of a Smad interaction motif (SIM) and two PY motifs. It is important in cell proliferation and differentiation of epithelial tissues and is implicated in oncogenic transformation and tumorigenesis [7]. PMEPA1 expression is elevated in numerous human cancers and drives the malignancy of glioblastoma [8], ovarian [9], lung [10], prostate [11], and colorectal cancers [12]. For instance, overexpression of PMEPA1 was observed in esophageal adenocarcinoma (EAC) with a significantly poor clinical outcome [13], whereas knockdown of PMEPA1 suppresses breast cancer cell tumor growth and tumor cell extravasation and impairs lung metastasis [14]. In line with these results, the pro-tumor roles of PMEPA1 were observed in lung cancer both in vivo and in vitro [10] despite the overall tumorigenesis activities still remaining inadequately understood.
Mechanically, a large number of studies have indicated that PMEPA1 acts as a critical propagator of multiple tumor-related signaling pathways, including androgen, TGF-
Herein, PMEPA1 mRNA expression status and its prognostic values in LUAD and LUSC were unveiled using the GEPIA database. Furthermore, we examined the impact PMEPA1 exerted on the H1299 cell behaviors by employing loss and gain functional assays. Furthermore, the underlying mechanisms of PMEPA1 on tumorigenesis in NSCLC cells were investigated. Our studies might contribute to improved targeted therapeutic interventions for NSCLS.
Methods
Bioinformatics analysis
GEPIA database (
Cell culture, reagents, and plasmids
The NSCLC cell line H1299 and the human embryonic kidney cell 293 line (HEK293T) were purchased from the Cell Storage Center, Wuhan University (Wuhan, China). All cell lines were preserved in DMEM with 10% fetal bovine serum and 100 U/mL penicillin-streptomycin mixture under standard conditions. Anti-PMEPA1 antibodies were purchased from Abcam Trading Company Ltd. (Shanghai, China). Anti-Tag antibody, anti-
Construct and transfection
For overexpression of PMEPA1, the cDNA-encoding PMEPA1 derived from H1299 cells was obtained via RT-qPCR using primer F: ATGCACCGCTTGATGGGGGT primer R: AGAAAGGACACCCTCTCTAG. The amplified fragment was cloned into the pHAGE-6tag-puro vector to generate the pHAGE puro-flag/PMEPA1 vector for PMEPA1 overexpression. HEK293T cells at 50% confluence were transfected with 4ug pHAGE puro-flag/PMEPA1 or empty vector and packaging vectors using Lipofectamine 2000 reagent (Thermo Fisher, Shanghai, China). After 48 h, the produced lentivirus was infected subconfluent H1299 cells for 36 h. The cells were continuously cultured for seven days in the presence of 2
PMEPA1-deficient H1299 cell lines were generated using CRISPR-Cas9 knockout genomic editing. Briefly, a single guide RNA sequence against PMEPA1 exon 1 (sgRNA-1: GCCACTACAAGCTGTCTGCA, sgRNA-2: GAGGAGCCCCCACCCTACCA) was designed, annealed, and inserted into pGL3-U6-sgRNA-PGK-puromycin. The H1299 cells at 50% confluence were co-transfected with pGL3-U6-sgRNA-PGK-puromycin and pST1374-NLS-Flag-linker-Cas9 for 48 h before puromycin selection. The surviving cells were amplified, and the PMEPA1-deficient H1299 cells were further confirmed by western blotting. The PMEPA1-deficient H1299 cells (KO-1 and KO-2) were selected for the next assays. The normal H1299 cells were used as normal control.
CCK-8 assay
A total of 2
Colony formation assay
In this assay, 500 H1299 cells/well were plated in 6-well plates and cultured for 14 days. The culture plates were taken out and washed with PBS before treatment with 0.5% of crystal violet for 30 min. Finally, surviving colonies were calculated and imaged.
Anchorage-independent assay
A single cell suspension (3
Transwell experiments
About 1
Western blotting
The indicated cell lysates were collected, and protein concentration was quantified by the Bradford Protein Assay kit, according to the product or manufacturer’s protocol. An equal number of proteins was analyzed using 8 to 12% SDS-PAGE, and protein strips were transferred to the PVDF membranes. After incubating in the blocking buffer, the membranes were exposed to anti-Flag, anti-PMEPA1 (Cat. ab128006, 1:1000), and anti-GAPDH antibodies in TBST at 4
Transfection and luciferase reporter assays
Considering the alternation in the JNK signaling pathway in lung cancer carcinogenesis [19], we examined the activity of the MAPK/JNK signaling pathway following PMEPA1 treatment in the presence or absence of SP600125 (a JNK inhibitor). First, HEK293T (5
To further confirm the regulatory relationship between PMEPA1 and JNK, we cotreated/coinfected 5
Statistical analysis
GraphPad Prism version 8.0 (GraphPad Software, USA) for Windows was used for statistical analyses. Results are expressed as mean
Overexpression of PMEPA1 is associated with dismal prognosis in lung adenocarcinoma (LUAD) and lung squamous cell carcinoma (LUSC). Gene PMEPA1 was significantly upregulated in LUAD and LUSC tissues in comparison with adjacent normal lung tissues in the GEPIA cohort. B and C. The Kaplan-Meier survival plot displays the relationship between PMEPA1 mRNA expression and overall survival (OS) in the LUAD and LUSC cohorts.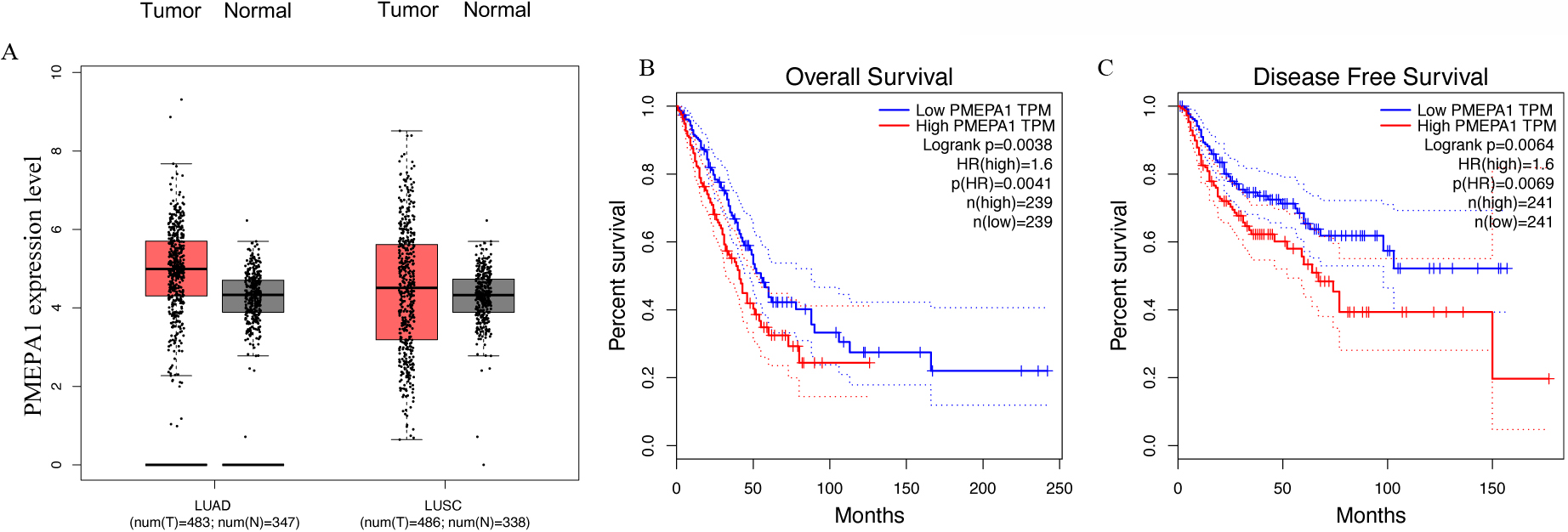
Upregulation of PMEPA1 is associated with poor prognosis in patients with NSCLC
To confirm the association of PMEPA1 expression with NSCLC, we first studied its expression in the GEPIA database, including 483 LUAD and 488 LUSC samples compared to their corresponding normal tissues. The gene expression of PMEPA1 in tumor tissues was enhanced when compared with that in matched adjacent noncancerous tissues (Fig. 1A). Survival analysis revealed that patients with high PMEPA1 expression had shorter disease-free survival (
Overexpression of PMEPA1 promotes H1299 viability, proliferation, and migration. A. Western blotting displays stable overexpression of PMEPA1 in the H1299 cell line using anti-Flag-antibody. B. The proliferative rate of H1299 cells was assessed by CCK8 assays. B and C. The colony-forming ability of H1299 cells was determined by colony-formation assay. D and E. The anchorage-independent growth of PMEPA1 overexpression in H1299 cell lines compared with its parent cells. F and G. The effect of PMEPA1 overexpression on the migration ability of H1299 cell lines.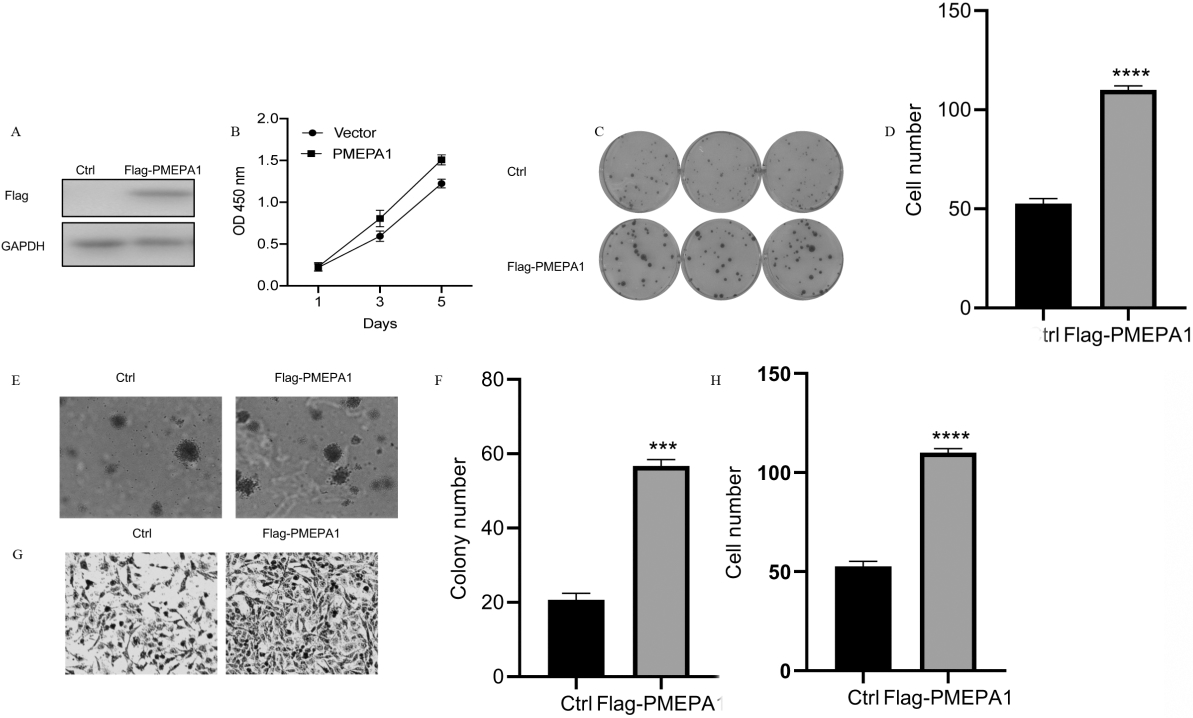
Considering the poor outcomes of PMEPA1 overexpression, we attempted to experimentally validate the oncogenic properties of PMEPA1 in NSCLC cell lines. The overexpression of PMEPA1 via viral vector-mediated gene transfer was first determined by western blotting (Fig. 2A). As seen in Fig. 2B and 2C, the exogenous expression of PMEPA1 can profoundly enlarge the proliferative and colony-forming capacities of H1299 cells. Furthermore, the migratory behavior was assessed using the Transwell assay, which revealed that the PMEPA1 overexpression markedly increased the migratory and metastatic abilities of H1299 cells (Fig. 2D and E). Our results demonstrated that PMEPA1 played a critical role in the initiation and promotion of NSCLC metastasis.
PMEPA1 deficiency impedes tumorigenesis in vitro
To further determine the oncogenic role of PMEPA1, we employed CRISPR-Cas9 to delete the transcribed unit of PMEPA1 in H1299 cell lines. Genetic inactivation of PMEPA1 in H1299 cells was confirmed by western blotting (Fig. 3A). Subsequently, functional assays were employed to investigate the cell phenotype changes in PMEPA1-deficient H1299 cells. As shown in Fig. 3B, the proliferative capacity was impaired in PMEPA1-deficient H1299 cells using the CCK8 assays. Colony formation efficiency was also curbed in PMEPA1-deficient H1299 cells (Fig. 3D). A similar outcome was observed in soft agar colony formation assays that demonstrated that the colony numbers in PMEPA1-deficient H1299 cells reduced in comparison to normal cells. Similarly, PMEPA1-deficient H1299 cells exhibited a sharply reduced migratory rate in Transwell assays compared with the untransfected cells. All these results show that the depletion of PMEPA1 diminishes tumorigenesis in H1299 cells.
Depletion of PMEPA1 suppressed cell lung cancer cell viability, proliferation, and migration. A. Western blotting was performed to assess the expression of PMEPA1 in wild type and PMEPA1-deficient H1299 cells. B. Depletion of PMEPA1 curbs the proliferative rate of H1299 cells tested by CCK8. C and D. Depletion of PMEPA1 impaired the colony-forming efficiency of H1299 cells. E and F. Soft agar colony-formation assay was performed to evaluate the anchorage-independent growth of untransfected and PMEPA1-deficient H1299 cells. G and H. Transwell experiment was used to compare the migration properties of wild type and PMEPA1-deficient H1299 cells.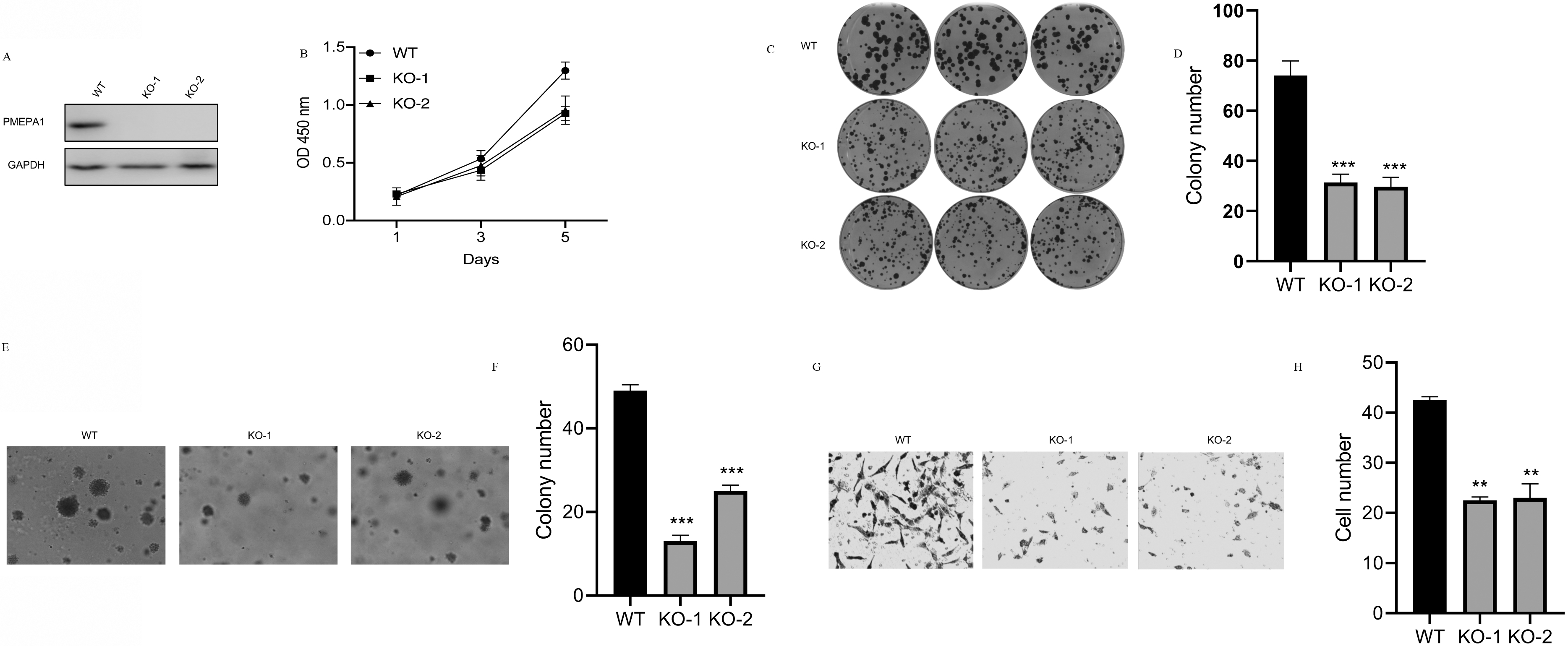
PMEPA1 positively regulates the MAPK/JNK signaling pathway. A. Luciferase experiments manifest the MAPK/JNK activity in HERK293T cells with increased PMEPA1 overexpression. B. Luciferase experiments display the MAPK/JNK activity in H1299 cells when these cells were transfected with an empty vector or increased pHAGE puro-flag/PMEPA1 and treated with different concentrations of SP600125 (a JNK inhibitor). C and D. RT-qPCR was performed to measure the CyclinD2 (C) and C-MYC (D) mRNA levels in H1299 cells upon PMEPA1 depletion. E. Western blot was performed measure the CyclinD2 and C-MYC expression levels in H1299 cells upon PMEPA1 depletion. F. Western blotting analysis of phosphorylation levels of JNK in the PMEPA1-overexpressing H1299 cells when the presence or absence of SP600125 (a JNK inhibitor).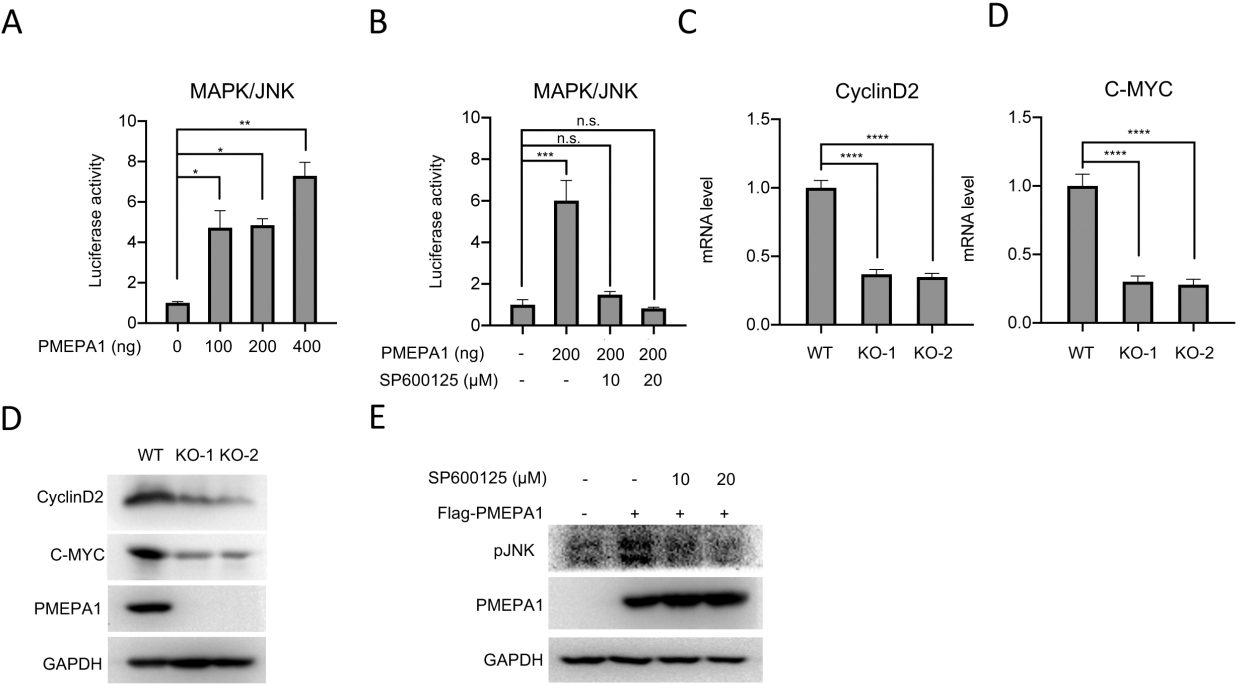
We next explored the transcriptional activity of the MAPK/JNK response to different doses of PMEPA1-overexpression plasmids. The dual-luciferase reporter gene assays indicated that PMEPA1 activated the transcriptional activity of the MAPK/JNK in HEK293T cells in a dose-dependent manner. To reconfirm the JNK involvement in the PMEPA1-mediated protumor role in NSCLC, SP600125, a JNK inhibitor, was employed to treat H1299 cells. As depicted in Fig. 4B, the same increased transcriptional activity of JNK was observed in H1299 cells treated only with 200 ng of PMEPA1-overexpressing vector; however, this induced transcriptional activity was inhibited when the JNK inhibitor was co-added to H1299 cells, especially at 20 ng. Moreover, reduction in the levels of Cyclin D2 and C-Myc, two critical downstream effectors of JNK signaling, was observed in KO-1 and KO-2 cells (Fig. 4C and D). More interesting, the overexpression of PMEPA1 could accumulate the phosphorylation level of JNK. However, this accumulation was abolished by the SP600125 treatment (Fig. 4E). These data suggested the implication of PMEPA1 in the tumor cell phenotype of H1299 cells by regulating the MAPK/JNK signaling cascade.
Discussion
In recent years, intensive studies suggested the prominent function of PMEPA1 in cancer [15]; however, its involvement in lung cancer remains incompletely determined and poorly understood. We noticed a high expression of PMEPA1 in LUAD and LUSC cancer tissues that contributed to an unfavorable prognosis in patients with LUAD and LUSC. Furthermore, we showed that PMEPA1 could influence the malignant behaviors of tumor cells and empower the tumorigenesis of NSCLC via activating the MAPK/JNK signaling pathway. These findings suggest that PMEPA1 might be a potential candidate/therapeutic target for lung cancer.
Previous studies have characterized PMEPA1 as an oncogenic factor in lung cancer [10, 18, 20, 21]. In our study, we analyzed that PMEPA1 was highly expressed in lung cancer tissues. Clinical relevance between the expression pattern of PMEPA1 and lung cancer by analyzing the data downloaded from GEPIA was also studied. The results suggested that PMEPA1 displayed aberrantly high expression in lung cancer tissues compared with matching adjacent normal lung tissues. This finding is consistent with that of previous studies on various solid cancers, except prostate cancer. In prostate cancer, five PMEPA1 isoforms (isoforms a, b, c, d, and e) were found to have different expression patterns and different functions. For example, PMEPA1-a (STAG1) is not significantly amplified in patients with prostatic cancer, correlating while PMEPA1-b expression was reduced in majority of cancer patients, both expression pattern are associated with cancer progression [11, 22]. Moreover, patients with high PMEPA1 showed a shorter overall survival rate compared with the low-PMEPA1 groups, reflecting the collective burden of lung cancer. Our finding was also consistent with other findings by Saadi et al., who reported that a high expression of PMEPA1 was related to the poor prognosis of most gastrointestinal tumors [13].
In the current work, in vitro gain and loss of function assays in lung cancer cell lines demonstrated that PMEPA1 significantly promoted viability, proliferation, and migration, further reinforcing the oncogenic potential of PMEPA1 in lung cancer. In this respect, our findings are consistent with several previous results, which demonstrated that the deficiency of PMEPA1 in various cancer cell lines reduced the tumorigenic activities both in vitro or in vivo [7, 23, 24]. These results substantiated the pro-tumor role of PMEPA1 in lung cancer.
To further explore the underlying mechanism of PMEPA1 mediation in tumorigenesis of NSCLC, we focused on the MAP/JNK signaling pathway, which reportedly drives the malignant phenotypes in lung cancer, including NSCLC [19]. Using the dual-luciferase reporter gene assay, we determined whether PMEPA1 affected the MAPK/JNK signaling pathway. The results showed that the overexpression of PMEPA1 significantly enhanced the transcriptional activity of the JNK pathway, indicating that PMEPA1 interfered with the proliferation and migration of H1299 cells via activating the JNK signaling pathway. Ying et al. found that ectopic PMEPA1 in 293T cells reduced the IRS-1 expression [21], whose degradation is critical in epithelial-mesenchymal transition (EMT). Furthermore, PMEPA1 is recognized as a TGF-induced EMT-promoting factor, contributing to post-EMT invasion and metastasis in lung cancer cells [25]. In contrast, the JNK signaling pathway is reportedly activated during EMT transformation, fostering the potency of invasive tumor proliferation in several cancer cell lines [26]. Significantly, the TGF and JNK signaling pathways reportedly crosstalk to modulate the EMT or tumorigenesis of lung cancer cells [27]. Therefore, we hypothesized that PMEPA1-mediated EMT might influence lung cancer cell invasiveness and metastatic potential. However, the involvement of PMEPA1 in the complex interplay still necessitates further investigation.
Conclusion
This study illuminates how the PMEPA1 expression was evaluated in LUAD and LUSC samples versus those in normal lung samples. The functional studies demonstrated that PMEPA1 enforces the tumorigenesis of NSCLC cells via activating the MAPK/JNK signaling pathway. However, more in-depth investigations in vitro are warranted to thoroughly explore the biological processes governed by PMEPA1 during the NSCLC malignancy.
Footnotes
Acknowledgments
This study was supported by the National Natural Science Foundation of China [81972851, 81802984] and Chongqing Postdoctoral Science Foundation [cstc2019jcyj-bshX0056].
Conflict of interest
All authors declared there was no conflict of interests involved.
Authors’ contributions
Conception and supervision: Zhenzhou Yang.
Interpretation and analysis of data: Benxu Tan and Yonghong Chen.
Preparation of manuscript: Lei Xia, Xian Yu, and Yuan Peng.
Revision for important intellectual content: Xiaoyue Zhang.
