Abstract
BACKGROUND:
Non-small cell lung cancer (NSCLC) is the main type of lung cancer. While miR-186 is significantly reduced in lung cancer tissues and cells, its role in NSCLC has not been completely elucidated.
MATERIAL AND METHODS:
We used qRT-PCR and western blot methods to investigate the levels of miR-186 and YY1 in 21 pairs of NSCLC tissues. Dual luciferase reporter gene assays were performed to detect whether miR-186 directly targets YY1. Next, the roles of miR-186 and its target gene (YY1) in determining the proliferation, apoptosis and migration capabilities of selected cell lines (A549 and HCC827) were investigated by using miR-186 mimics or YY1 siRNA.
RESULTS:
Our results showed that miR-186 was downregulated and YYI was upregulated in NSCLC tissue, and miR-186 expression was negatively associated with YY1. Similarly, miR-186 was also downregulated and YY1 expression also was upregulated in both A549 and HCC827 cells; furthermore, miR-186 was found to directly target YY1. Cell proliferation, invasion, and migration, as well as apoptosis induction were more strongly inhibited by YY1 siRNA than by miR-186.
CONCLUSION:
Our results suggest that miR-186 and its target gene (YY1) could possibly serve as new prognostic biomarkers and therapeutic targets for treating NSCLC in humans.
Background
Lung cancer is one of the most common malignant tumors in China. Data released by the National Cancer Center of China in 2015 showed that lung cancer ranked first in both incidence and mortality among all malignant tumors in China during 2011 [1]. Additionally, according to the American Cancer Society, the latest statistics for a population of Asian Americans, Native Hawaiians, and Pacific Islanders (AANHPIs) estimate 3460 and 3030 new cases of lung cancer among males and females, respectively. This means that in 2016, lung cancer will account for 14% of all new cancers in males, 9% of all new cancers in females, and rank second and third in cancer incidence among males and females, respectively. The numbers of deaths among male and female lung cancer patients are expected to be 2290 and 1780, respectively, and will account for 27% of all cancer-related deaths among males and 21% of all cancer-related deaths among females, making lung cancer the leading cause of cancer-related death [2]. Non-small cell lung cancer (NSCLC) accounts for 80% to 85% of all lung cancers, and includes subgroups of squamous cell carcinoma, lung adenocarcinoma, and large cell carcinoma [3, 4]. Most cases of NSCLC have already metastasized when they are diagnosed [5, 6, 7], making treatment of advanced stage patients the fastest growing group of lung cancer patients in recent years. Surgery, radiotherapy, and chemotherapy are the traditional methods used to treat lung cancer. While surgery is the most common treatment for NSCLC patients, combined radiotherapy plus chemotherapy is the primary treatment for patients with advanced NSCLC. However, the clinical outcomes of these treatment methods are often not ideal, and the overall prognosis for NSCLC patients remains poor, as the 5-year survival rate is only 5%–15.8% [8, 9, 10]. In recent years, significant progress has been made in studying the molecular genetics of lung cancer. As a result molecular targeted therapy is playing an increasingly important role in treating individual patients with advanced lung cancer.
MicroRNAs (miRNAs) are naturally occurring, short, non-coding RNAs that negatively regulate gene expression [11]. In mammals, mature miRNAs consist of 21–24 nucleotides, integrate into RNA-inducing silencing complexes, and pair with the 3
Yin Yang 1 (YY1) is a relatively conserved transcription factor that plays important roles in embryonic development, differentiation, and cell proliferation. In addition, it plays a direct regulatory role in the metastasis and invasion of solid tumors [18]. However, little is known about how the YY1 gene is regulated.
MiR-186 expression is significantly downregulated in colorectal cancer cell lines SW620 and LoVo, and also in colon cancer tissue when compared with its expression in adjacent tissues. Transfection with the recombinant lentiviral vector PLVTHM-miR186 significantly inhibits cell proliferation, migration, and invasion, and suppresses YY1 protein expression in SW620 cells [20]. The goal of the current study was to determine whether miR-186 inhibits cell proliferation, migration, and invasion by directly targeting YY1 in NSCLC cells.
Material and methods
Specimens
Twenty-one pairs of NSCLC tissue specimens and adjacent normal tissue specimens were obtained from 21 different patients who had visited the Shenzhen People’s Hospital. All patients who participated in this study provided their written informed consent. The investigation and experimental protocol were approved by the Ethics Committee of Shenzhen People’s Hospital.
Cell culture
Human lung cancer cell lines A549, 95-D, HCC827, and NCI-H1650, and lung epithelial cells (HPAEpiC) were purchased from ATCC (Manassas, VA, USA). All cells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM; Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco), 100
Transfection
MiR-186 mimics, small-interfering RNAs targeting YY1 (si-YY1), and their respective negative controls were obtained from Gene Pharma (Shanghai GenePharma Co. Ltd, Shanghai, China). Cells were seeded into six-well plates (2
Primer sequences used for miRNA and mRNA expression analysis
Primer sequences used for miRNA and mRNA expression analysis
The binding sites for miR-186 within YY1 mRNA were predicted, and the wild-type YY1 3’-UTR (wt 3’-UTR) fragment containing the predicted miR-186 binding sites was amplified by PCR. Mutant YY1 3’-UTR (MUT 3’-UTR) was obtained by mutating the conserved binding sites for miR-186. Fragments which including the wt 3’-UTR or MUT 3’-UTR regions of YY1 were cloned into a psiCHECK-2 vector (Promega, Madison, WI, USA) that contained both Renilla and firefly luciferase reporter genes. The psiCHECK-2 vectors with the wt 3’-UTR or MUT YYI 3’-UTR regions were transfected into HEK23 cells that had been previously transfected with miR-186 mimics or negative control mimics, respectively, by using Lipofectamine 2000 (Invitrogen, USA). After 24 hours, the firefly and Renilla luciferase activities in the cells were determined with a dual-luciferase reporter assay system (Promega) in accordance with the manufacturer’s instructions.
qRT-PCR
After treatment, total RNA was extracted using TRIZOL (Invitrogen). The stem-loop primers used for PCR amplification were synthesized by RiboBio (Beijing, China). The relative level of miR-186 expression was normalized against the level of U6 expression. Real-Time PCR reactions were conducted using TaKaRa SYBR Premix Ex Taq II (TaKaRa, Shiga, Japan) on an Applied Biosystems 7500 Real-Time PCR System (ABI, Foster City, CA, USA). The level of YY1 gene expression was normalized to the level of GAPDH expression within each sample using the
Western blot studies
Tissue specimens or cells were homogenized, and their total proteins were extracted with ice-cold SDS lysis buffer (Beyotime, Haimen, China). The protein concentration of each sample was measuring using a Bio-Rad protein assay. Next, 30
CCK-8 assay
Cell proliferation was determined by using a Cell Counting Kit-8 (CCK-8) (Beyotime, China). In brief, cells were cultured to a density of 2
Differential expression of miR-186 and YY1 in specimens of NSCLC tissue and adjacent normal tissue. (A) Relative expression of miR-186 in NSCLC tissue and adjacent normal tissue, 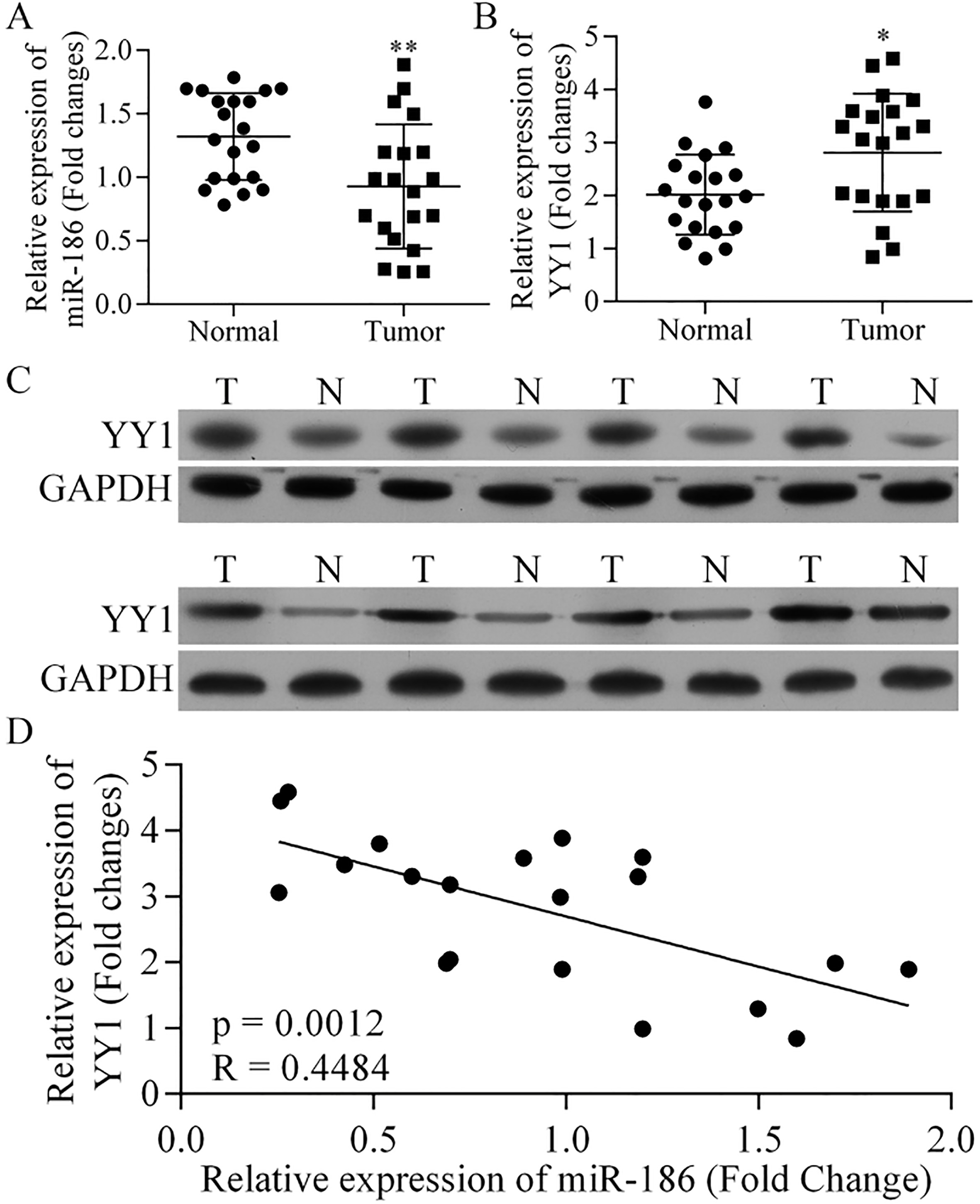
Cell apoptosis was assessed by using an Annexin V-FITC Apoptosis Detection Kit (BD Pharmingen, San Diego, CA, USA). Annexin V-FITC (5
Migration and invasion assay
Cell migration and invasion assays were performed by using a Transwell chamber (Corning Inc., Corning, NY, USA). The Transwell chamber was precoated with 30
Statistical analysis
All data were analyzed using SPSS Statistics for Windows, and results are expressed as the mean value
Results
Expression of miR-186 and YY1 in NSCLC tissue
The levels of miR-186 and YY1 expression were examined in 21 pairs of NSCLC tissue and adjacent normal tissues by qRT-PCR (Fig. 1A and B). Our results showed that the levels miR-186 expression in the tumor tissues were significantly lower than those in the normal tissues (
MiR-186 directly targeted YY1
To investigate whether YY1 was a target of miR-186, the levels of miR-186 and YY1 expression in several human lung cancer cell lines (A549, 95-D, HCC827, and NCI-H1650) and lung epithelial cells (HPAEpiC) were examined to identify the appropriate cells to be studied. The levels of miR-186 in A549 and HCC827 cells were lower than those in HPAEpiC, (Fig. 2A), while YY1 was more highly expressed in A549, 95D, and HCC827 cells than in HPAEpiC (Fig. 2B). Thus, A549 and HCC827 cells were selected for use in the subsequent studies.
YY1 is the target of miR-186. (A) Expression of miR-186 and (B) YY1 in various human lung cancer cell lines, including A549, 95-D, HCC827, and NCI-H1650, and in lung epithelial cells (HPAEpiC). 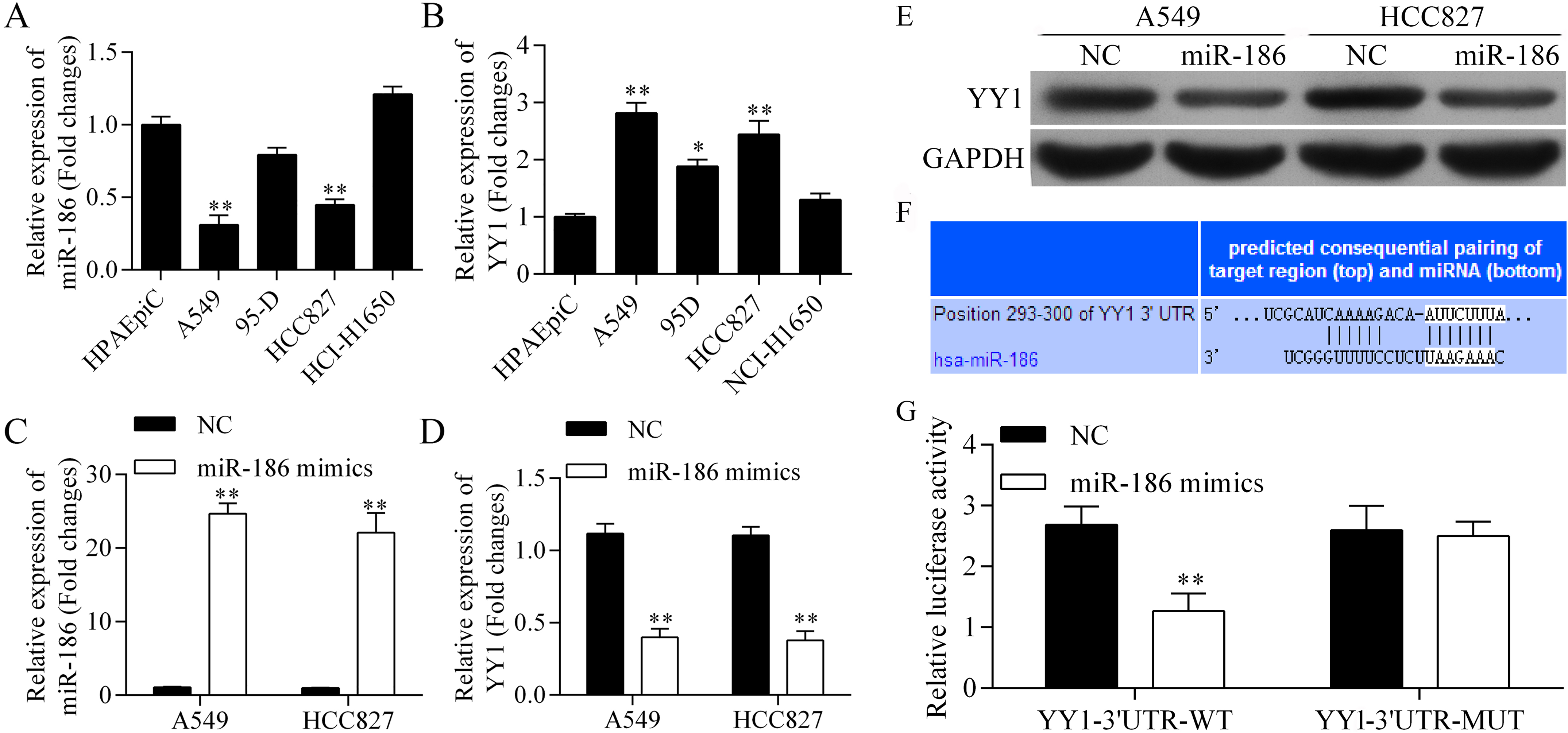
Our data showed that transfection with miR-186 mimics upregulated miR-186 expression in the A549 and HCC827 cells (Fig. 2C), and downregulated miR-186 YY1 mRNA expression in both of those cell lines (Fig. 2D). Moreover, western blot studies showed that YY1 protein expression was downregulated by miR-186 (Fig. 2E). Those findings further confirmed that YY1 could be downregulated by miR-186.
TargetScan was used to predict the miR-186 seed region in the YY1 3’UTR (Fig. 2F). The luciferase reporter assay system showed that the relative level of luciferase activity in YY1-3’UTR-WT (wild type) was decreased by the miR-186 mimics, but remained unchanged in the YY1-3’UTR-MUT (mutant type) (Fig. 2F). These results indicated that miR-186 directly targets YY1.
To examine the roles played by miR-186 and its target gene YY1, YY1 expression was knocked down by siRNA, and the resultant effects on cell proliferation and apoptosis were examined (Fig. 3). We found that YY1 siRNA significantly downregulated the levels of YY1 mRNA (Fig. 3A) and protein expression (Fig. 3B), verifying that YY1 had been knocked down by YY1 siRNA. Next, cell proliferation was detected with the CCK-8 assay (Fig. 3C). Our results showed that the miR-186 mimics significantly inhibited A549 and HCC827 cell proliferation. Moreover, YY1 siRNA significantly inhibited the proliferation of both A549 and HCC827 cells. However, cell proliferation was more strongly inhibited by YY1 siRNA than by the miR-186 mimics.
Silencing of YY1 by siRNA and its roles in cell proliferation and apoptosis. After transfection with YY1 siRNA, knockdown of YY1 was confirmed by qRT-PCR (A) and western blot (B). Next, the levels of cell proliferation in cultures of NC-, YY1 siRNA (si-YY1)-, and miR-186 mimics-transfected (C) A549 cells and (D) HCC827 cells were detected by the CCK-8 assay. Also, the rates of apoptosis among NC-, YY1 siRNA-, and miR-186 mimics-transfected A549 cells and HCC827 cells were detected by flow cytometry (D), and then calculated (E). 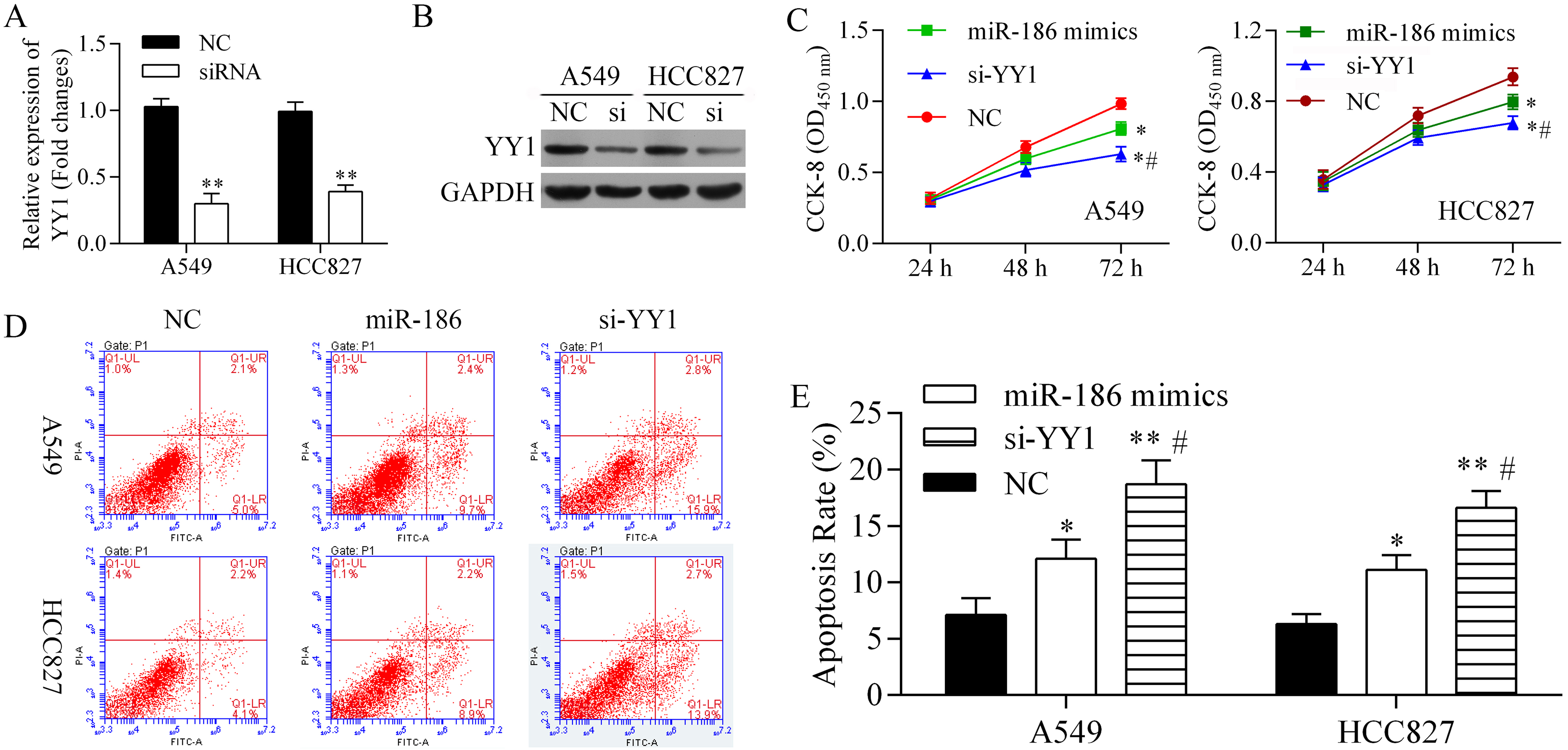
Effects of miR-186 and YY1 on the migration and invasion capabilities of A549 cells and HCC827 cells. Transwell assays were performed to quantitate the migration and invasion capabilities of NC-, YY1 siRNA-, and miR-186 mimics-transfected (A) A549 cells and (B) HCC827 cells. 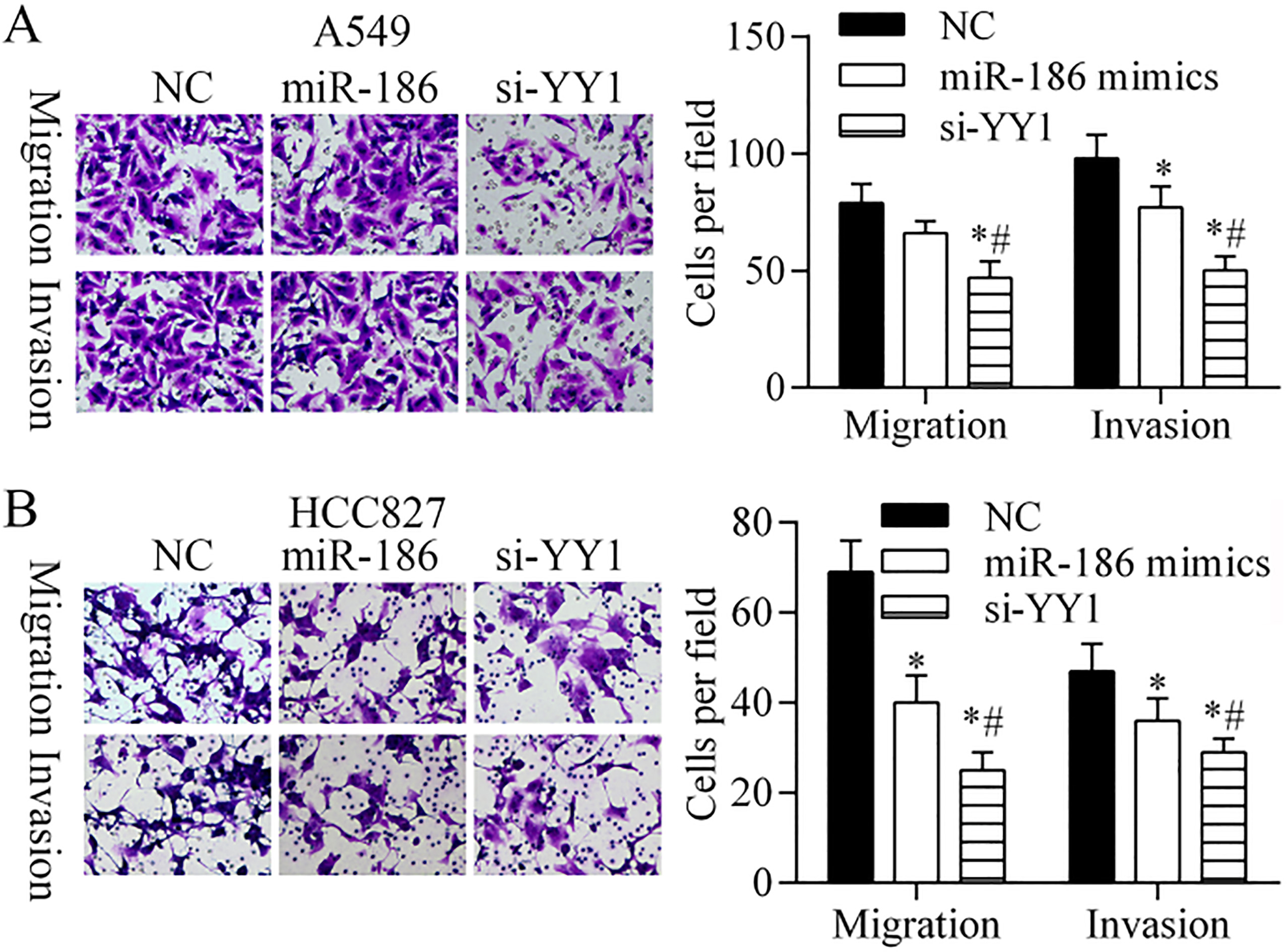
The effects of miR-186 and YY1 siRNA on cell apoptosis were also evaluated (Fig. 3D and E). Both miR-186 and YY1 siRNA significantly increased the rates of apoptosis among A549 and HCC827 cells. However, the amount of apoptosis induction produced by YY1 siRNA was greater than that produced by miR-186 mimics.
When taken together, our results showed that miR-186 and YY1 siRNA inhibited cell proliferation, but promoted cell apoptosis. The degrees cell proliferation inhibition and apoptosis induction produced by YY1 siRNA were greater than those produced by miR-186. Our findings suggest that miR-186 inhibits cell proliferation, but promotes cell apoptosis by downregulating YY1.
Transwell assays were performed to examine whether miR-186 inhibited cell migration and invasion by downregulating YY1 (Fig. 4). In A549 cells (Fig. 4A), cell migration was inhibited by YY1 siRNA, and cell invasion was significantly inhibited by both miR-186 and YY1 siRNA. In HCC827 cells (Fig. 4B), cell migration and cell invasion were significantly inhibited by both miR-186 and YY1 siRNA. Furthermore, the degrees cell migration and invasion inhibition produced by YY1 siRNA were greater than those produced by miR-186 mimics. These findings suggest that miR-186 inhibits cell invasion and migration by downregulating YY1.
Discussion
In the present study, we found that miR-186 expression was suppressed in samples of NSCLC tissue, and negatively associated with YYI expression those tissues. Furthermore, our data showed that miR-186 inhibited cell proliferation, migration, and invasion, but induced apoptosis in NSCLC cells by directly targeting YY1, and may function as a tumor suppressor during NSCLC development. Thus, miR-186 and its target gene (YY1) could possibly serve as novel prognostic biomarkers and therapeutic targets for treatment of human NSCLC.
Recently, numerous oncogenes and tumor suppressors have demonstrated their involvement in regulating NSCLC development. In addition to putative protein-coding genes, non-coding miRNAs contain extremely well conserved RNA sequences of 21–24 nucleotides that play important roles in the development of different tumors by binding to the 3’-UTR of their target mRNAs. MiR-186 has been identified in various cancer tissues, and found to be either downregulated or upregulated in those tissues. Among the cancers in which miR-186 was downregulated were prostate cancer, colorectal neuroendocrine tumors, ovarian cancer, and NSCLC [15, 21, 22]. A single type of miRNA can target several different genes. Ectopic overexpression of miR-186 was shown to inhibit gastric cancer cell proliferation, invasion, and migration. In contrast, inhibition of miR-186 expression was shown to enhance cell proliferation, invasion, and migration by causing it to target twist1 [14]. Golgi phosphoprotein 3 (GOLPH3) is a target for miR-186. Overexpression of miR-186 was shown to arrest cells in their G0/G1 phase, and decrease their p21 and p27 levels by targeting GOLPH3 [15]. Another study found that miR-186 expression was significantly reduced in lung cancer tissues and A549 and HCC827 cells [17]. Knockdown of the miR-186 target gene MAP3K2 by RNAi inhibited lung cancer cell proliferation, migration, and invasion, and promoted cell apoptosis [17]. Consistent with those studies, we found that miR-186 levels were significantly reduced in lung cancer tissues, and also in A549 and HCC827 cells. We also identified YYI as new target gene for miR-186 and demonstrated that YY1 is involved in regulating cell proliferation, apoptosis, migration, and invasion. YY1 is also directly involved in regulating Snail1 gene transcription, and suppresses E-cadherin synthesis induced by the epithelial-mesenchymal transition process [23]. Snail is an important transcription factor induced by epithelial-mesenchymal transition [24], which is believed to ultimately promote tumor metastasis [25]. Knockdown of YY1 might inhibit cell migration and invasion through Snail in a downstream signaling pathway.
YY1 is a relatively conserved transcription factor that plays important roles in embryonic development, differentiation, cell proliferation, and metastasis [18], and helps regulate the invasion and metastasis of solid tumors [19]. Moreover, recent studies have shown that YY1 is involved in carcinogenesis, and can play either an oncogenic or suppressor role in tumor development and progression [26, 27].
Besides miR-186, YY1 is also the direct target of several other miRNAs, including miR-190 [28], miR-206 [29], miR-29 [30], miR-635 [31], and miR-34 [32]. MiR-635 transfection can significantly decrease endogenous YY1 expression, and thereby mimic the effects produced by inhibition of cell invasion caused by siRNA-mediated YY1 knockdown. This suggests that YY1 may be a direct target of miR-635 [31]. YY1 is also a target of miR-34, and contributes to gastric carcinogenesis [32]. A previous study using NUGC-3 cancer cells showed that overexpression of miR-34 family members suppressed carcinogenesis by downregulating YY1 expression. Alternatively, a study with SC-MI and AZ521 cancer cells showed that knockdown of miR-34 family members promoted tumorigenesis by up-regulating YY1 expression. In addition to miR-186, it is possible that other miRNAs can also regulate YY1, as the degrees of cell proliferation, migration, and invasion inhibition produced by YY1 siRNA, and also levels of cell apoptosis induced by YY1 siRNA, were greater than those produced by miR-186 mimics. Taken together, our results indicate that miR-186 inhibits the proliferation, migration, and invasion of non-small cell lung cancer cells by targeting Yin Yang 1.
Footnotes
Conflict of interest
The authors have no conflicts of interest to declare.
