Abstract
BACKGROUND:
MicroRNAs (miRNAs) emerge as important regulators involved in malignant progression in some tumors. MiR-181a has been found to function as a tumor suppressor in some tumors including non-small cell lung cancer (NSCLC). However, the functional role of miR-181a in NSCLC still needed to be investigated.
METHODS:
The expression of miR-181a were determined by qRT-PCR, the association between miR-181a and clinicopathological data were performed by chi-square test and survival analysis were evaluated by Kaplan-Meier curve and log rank test. Cell proliferation and invasion were assessed by CCK8, cell colony formation and transwell assays. Luciferase reporter assay demonstrated that CDK1 was a target of miR-181a. Western blot assay detected the relative protein expression.
RESULTS:
In the study, our results showed that miR-181a was significantly down-regulated in non-small cell lung cancer (NSCLC) tissues and cell lines. MiR-181 expression levels were significantly associated with histological grade, N status and TNM stage in the patients and lower miR-181a predicted a poor prognosis in NSCLC patients. Furthermore, upregulation of miR-181a significantly suppressed the NSCLC cell proliferation, colony formation, and cell invasion capacities. Moreover, upregulation of miR-181a inhibited CyclinB1 and CyclinD1 expression in NSCLC cells. Luciferase activity assay results demonstrated CDK1 was a direct target of miR-181a and miR-181a inhibited cell proliferation by regulating the mRNA and protein levels of CDK1 in NSCLC cells.
CONCLUSION:
These data suggested that miR-181a plays a tumor suppressor and may be a potential therapeutic target for NSCLC patients.
Introduction
Lung cancer is the leading cause of cancer-related deaths both in men and women around the world and non-small cell lung cancer (NSCLC) accounts for the most cases of lung cancer [1]. About 65% lung cancer patients who are diagnosed present locally advanced or metastatic disease [2, 3]. More than fifty percent lung cancer patients showed advanced stage disease and are unsuitable to surgery treatment, the others patients who are treated with undergoing surgery and definitive chemoradiation [4, 5, 6]. Although the survival time for lung cancer has increased because the advances of in diagnosis and treatment, the prognosis of NSCLC remains poor. The 5- and 10-year survival rates remain at
Over the past twenty years, a large of microRNAs (miRNAs), small noncoding RNAs, have been showed t be involving in the processes regulating tumorigenesis, acts as a biological marker of prognosis [8]. Accumulating evidence has identified that aberrant miR-181a expression is pronounced and correlated with tumor genesis and progression, enforced expression of miR-181a had been found to promote gastric cancer cell proliferation ability [9]. MiR-181a-5p is decreased in hepatocellular carcinoma and suppresses motility, invasion and branching-morphogenesis by directly targeting c-Met [10]. MiR-181a-5p-mediated reduction of MMP-14 levels is sufficient to decrease cancer cell migration, invasion, and activation of pro-MMP-2 [11]. MiR-181a-5p is also reported to inhibit non-small cell lung cancer A549 cells proliferation and migration by targeting KRAS [12]. However, it is still unclear the functional role of miR-181a in NSCCL.
In the study, miR-181a was significantly down-regulated in NSCLC tissues and lower miR-181a predicted a poor prognosis of NSCLC patients. Furthermore, we demonstrated miR-181a inhibited the cell proliferation by targeting CDK1. Thus, the results indicated that miR-181a plays a tumor suppressor and functions as a potential therapeutic target in NSCLC patients.
Methods
Clinical tissue samples and cell culture
Seventy-eight cases of NSCLC tissues and paired adjacent non-tumor tissues were collected from March 2012 to January 2014 at Department of Oncology, Fuzhou Pulmonary Hospital of Fujian. Tissues were immediately snap-frozen in liquid nitrogen and stored at
Cell transfecting
MiR-181a inhibitor, miR-181a mimic and miR negative control were purchased from Ribobio (Guang zhou, China). Cells were transfected with 50 nM using Lipofectamine™3000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. The sequences of siRNAs used in the study are as follows: si-CDK1: 5
RNA extraction and real-time PCR
Total RNA extracted from cultured cells and the tissues using Trizolv (Invitrogen) according to the manufacturer’s instructions. RNA was reverse transcribed to cDNAs using a Prime Script
Cell proliferation assay
Cell proliferation was tested by Cell Counting Kit-8 assay (Dojindo, Kumamoto, Japan) according to the manufacturer’s instructions. Briefly, 5
Colony formation and cell invasion assays
Cells were seeded in 12-well plates at a density of 100 cells/well. After 24 h, cells were transfected with with miR-181a mimic or miR-NC. After 7 day of incubation, the medium was removed and cell colonies were fixed with methanol and stained with crystal violet and counted with a cell counter. Images of stained tumor cell colonies were recorded. For transwell invasion assays, 5
Correlation between miR-181a expression and clinicopathologic parameters in NSCLC patients
Correlation between miR-181a expression and clinicopathologic parameters in NSCLC patients
Cells were lysed using a RIPA buffer (Sigma) with protease inhibitor PMSF (Sigma). Equal amounts of protein were separated by 10% SDS-PAGE and then transferred onto polyvinylidene difluoride membranes (Millipore).The membranes were blocked with 5% nonfat milk and then incubated with the indicated specific antibodies including CDK1 (dilution, 1:1000, Cell Signaling Technology, USA), CyclinB1 (dilution, 1:1000, Abcam, USA), CyclinD1 (dilution, 1:1000, Cell Signaling Technology, USA) and GAPDH (dilution, 1:3000, Cell Signaling Technology, USA). Membranes were incubated with horseradish peroxidase-conjugated goat anti-rabbit (Santa Cruz Biotechnology, Inc.) at room temperature for 1 h. The blots were analyzed with an ECL chemiluminescence kit (CWBIO, Beijing, China).and exposure in a Molecular Imager
Luciferase reporter assay
The 5
MiR-181a is down-regulated in NSCLC tissues and cells. (A) The expression levels of miR-181a in 78 cases of human NSCLC tissues and corresponding adjacent tissues relative were determined by qRT-PCR assays. (B) The Kaplan-Meier survival curves was used to evaluate the association between miR-181a expression and the over survival time in NSCLC patients.(C) The expression levels of miR-181a in 16HBE cell, A549 and H1299 cells were determined by qRT-PCR assays. Data are represented as mean 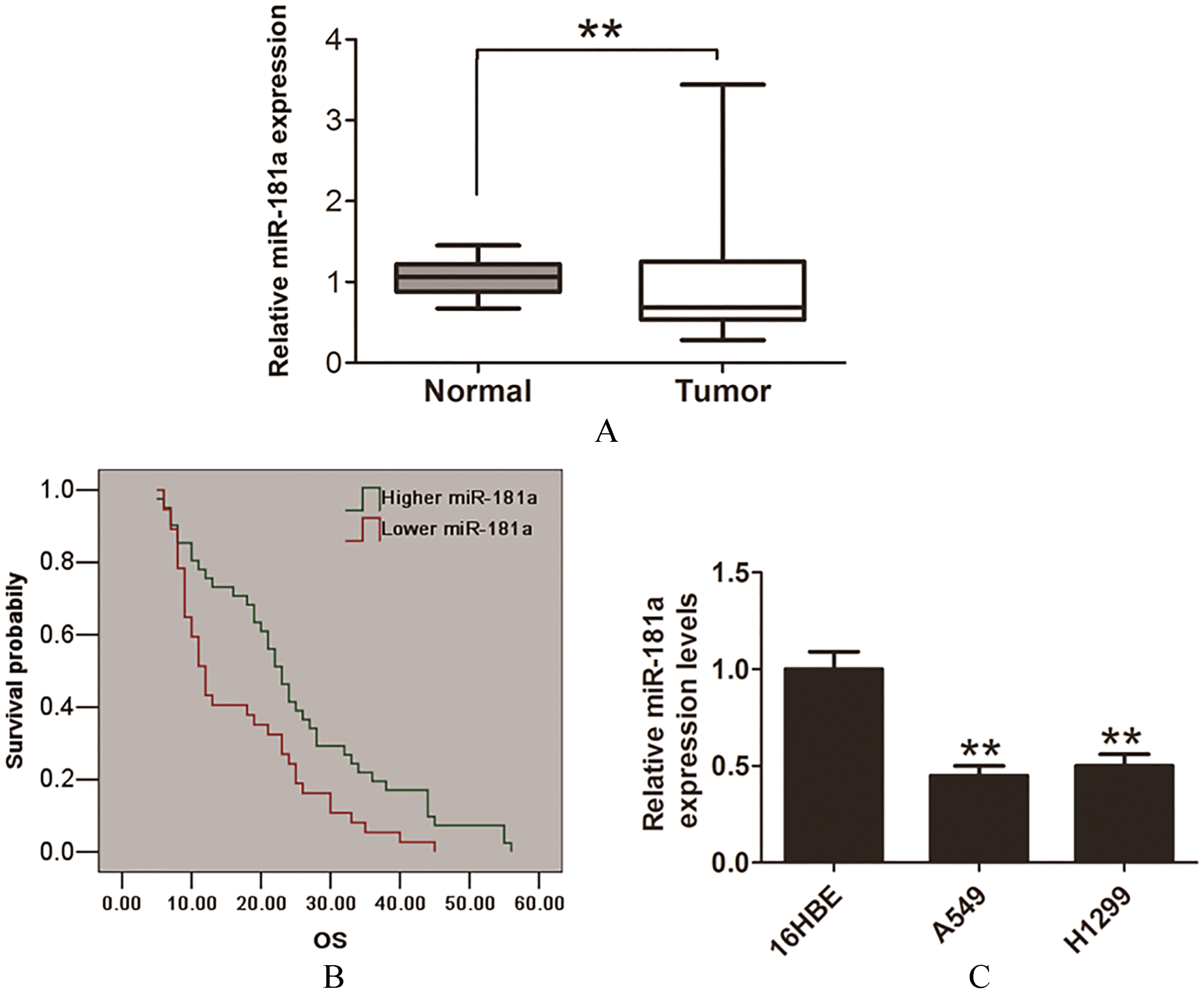
The quantitative data are showed as the means
MiR-181a inhibited cell proliferation ability NSCLC cells. (A) Cell proliferation was showed by CCK8 assay after transfected with miR-NC or miR-181a mimic into 549 cells. (B) Cell proliferation was showed by CCK8 assay after transfected with miR-NC or miR-181a inhibitor into H1299 cells. (C)–(D) Cell proliferation was showed by colony formation assay after transfected with miR-NC or miR-181a mimic into 549 cells. (E)–(F) Cell proliferation was showed by colony formation assay after transfected with miR-NC or miR-181a inhibitor into H1299 cells. Data are represented as mean 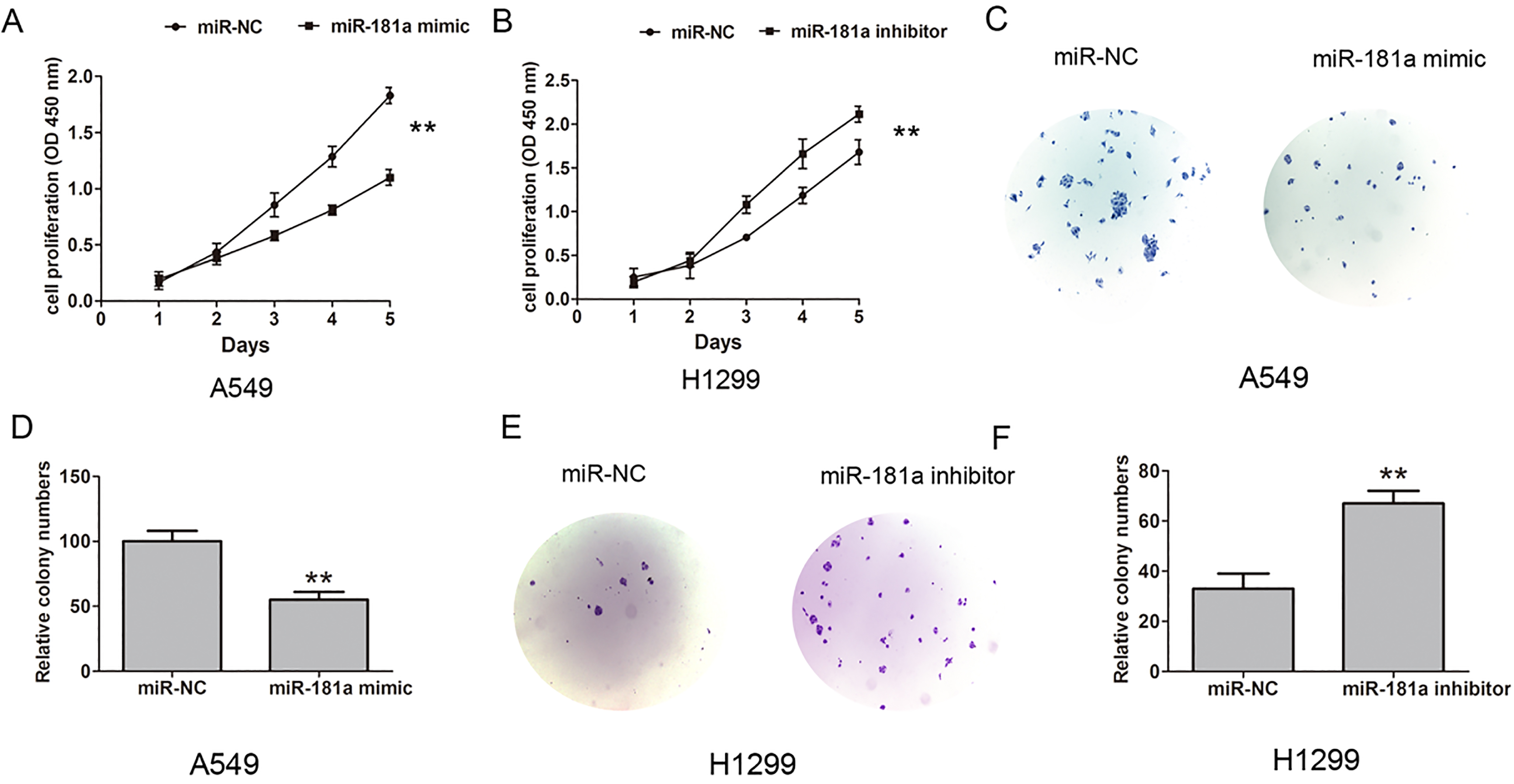
MiR-181a inhibited cell invasion ability and cell proliferation relative protein expression in NSCLC. (A)–(B) Cell invasion and cell invasive number were showed by transwell assays after transfected with miR-NC or miR-181a mimic into A549 cells. (C)–(D) Cell invasion and cell invasive number were showed by transwell assays after transfected with miR-NC or miR-181a inhibitor into H1299 cells. (E) The relative protein expression levels of CyclinB1 and CyclinD1 were showed by western blot assay after transfected with miR-NC or miR-181a mimic into A549 cells. (F) The relative protein expression levels of CyclinB1 and CyclinD1 were showed by western blot assay after transfected with miR-NC or miR-181a inhibitor into H1299 cells. Data are represented as mean 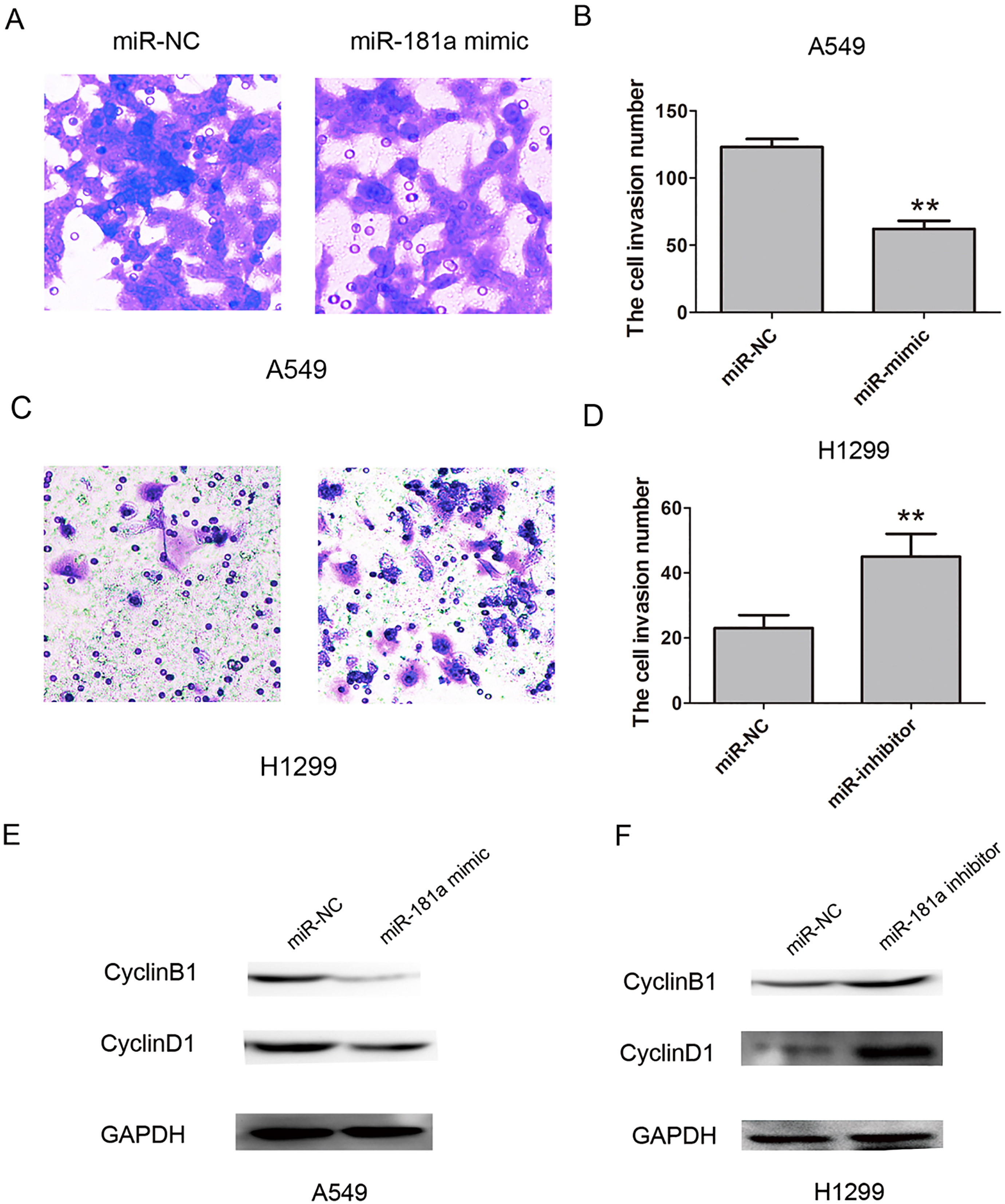
MiR-181a is down-regulated in NSCLC tissues and cells
We evaluated the expression of miR-181a in 78 pairs of NSCLC tissues and corresponding adjacent normal lung tissues by quantitative reverse transcriptase PCR (qRT-PCR) assays. The results showed that miR-181a was significantly down-regulated in NSCLC tissues compared with adjacent normal lung tissues (Fig. 1A). Furthermore, we analyzed the correlation between clinicopathological data miR-181a expression and the results revealed that miR-181 expression levels were significantly associated with histological grade, N status and TNM stage in the patients (Table 1), but no associated with the other clinicopathological factors (Table 1). In addition, compared with higher miR-181a expression in NSCLC patients, the lower miR-181a had a poor over survival time (Fig. 1B). Furthermore, we also evaluated miR-181a expression in two NSCLC cell lines (A549 and H1299 cells) and a normal human bronchial epithelial cell line (16HBE). The results showed that the relative expression levels of miR-181a were lower in tumor cells, compared with that in 16 HBE cells (Fig. 1C). Thus, these findings indicated that miR-181a could play a tumor suppressor in NSCLC.
MiR-181a inhibits NSCLC cell proliferation, colony formation and cell invasion in vitro
To further detect the effect of miR-181a expression on cell proliferation and invasion, we performed the function-gain and-lose assays, the miR-181a mimic or miR-181a inhibitor were transfected into the A549 and H1299. Cell Counting Kit-8(CCK8) assays were performed to assess the role of miR-181a in NSCLC cell proliferation. The results showed that the miR-181a mimic inhibited the cell proliferation ability compared with the miR-NC group in A 549 cells, but miR-181a inhibitor promoted cell proliferation ability compared with the miR-NC group in H1299 cells (Fig. 2A and B). Next, we tested the effects of miR-181a on cell colony formation, the results showed that colony formation was inhibited by miR-181a mimic in A549 cells, but was enhanced by miR-181a inhibitor in 1299 cells (Fig. 2C and F). Furthermore, transwell invasion assays showed the cell invasive ability number were inhibited when miR-181a mimic was introducing into A549 cells, but miR-181a inhibitor promoted the cell invasion in H1299 cells, compared with the miR-NC group (Fig. 3A and D). Moreover, the cell proliferation related protein CyclinB1 and CyclinD1 was down-regulated when miR-181a mimic was introducing into A549 cells, but miR-181a inhibitor promoted their expression in H1299 cells (Fig. 3E and F). Thus, our results showed that MiR-181a inhibits NSCLC cell proliferation and invasion in vitro.
MiR-181a inhibits the cell proliferation by targeting CDK1
To determine potential target genes of miR-181a in NSCLC, the predicted software targetscan and miRanda were used in the study. We found that 3
MiR-181a inhibited cell proliferation by target CDK1 in NSCLC cells. (A) MiRanda software predicted that CDK1 were putative target of miR-181a. The potential seed sequences of miR-181a within the wild-type and mutant 3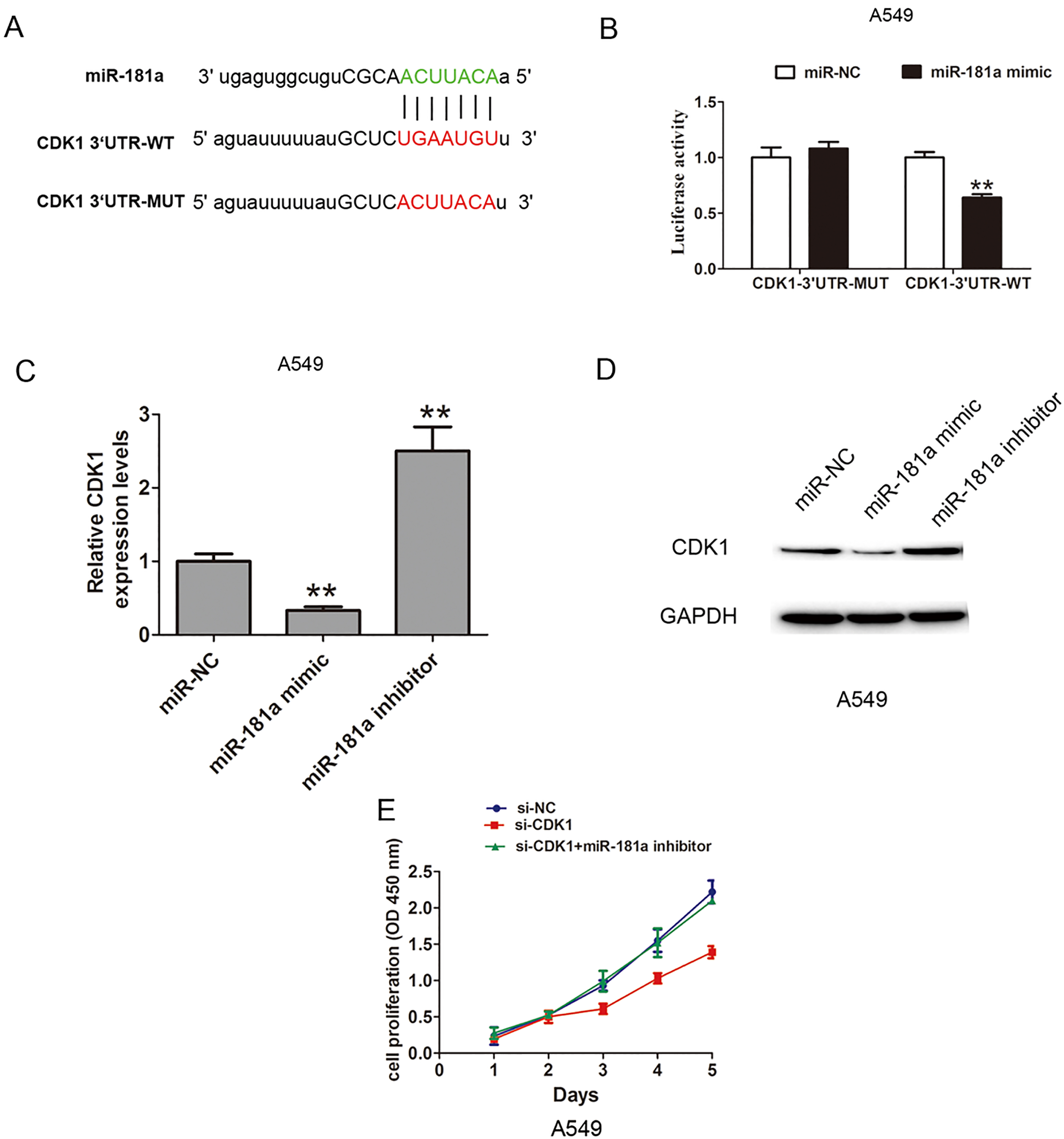
We further detected the mRNA and protein expression of CDK1 after upregulation or downregulation of miR-181a. The results showed that the mRNA expression of CDK1 was inhibited by miR-181a mimic, but was increased by miR-181a inhibitor (Fig. 4C). Besides, we analyzed the protein expression of CDK1 and the results verified that the protein expression of CDK1 was inhibited by miR-181a mimic, but was increased by miR-181a inhibitor (Fig. 4D). Thus, these results showed that miR-181a inhibited the cell proliferation by targeting the CDK1. To evaluate whether CDK1 mediated the effects of miR-181a on NSCLC proliferation, we performed the CCK8 cell proliferation assays. The results showed that si-CDK1 inhibited the cell proliferation, but cotransfection of si-CDK1 and miR-181a inhibitor dismissed the effects (Fig. 4E). Thus, these results showed that miR-181a inhibited the cell proliferation by regulating the CDK1 expression in NSCLC cells.
MicroRNAs (miRNAs) are a class of small non-coding RNAs that regulate the gene expressions at the posttranscriptional level and subsequently control key physiological processes [13, 14]. MiR-181a had been found to be involved in some tumors and served as tumor-suppressor genes or oncogenes. In previous studies, microRNA-181a-5p inhibited cancer cell migration and angiogenesis via down-regulation of matrix metalloproteinase-14 (MMP14). Down-regulation of microRNA-181a suppressed cell proliferation and cell invasion and promoted cell apoptosis in cervical cancer through the PTEN/Akt/FOXO1 signaling pathway [15]. MiR-181a enhanced sorafenib resistance in hepatocellular carcinoma cells through downregulation of RASSF1 expression [16]. Gao et al. demonstrated that low miR-181a expression levels group compared with higher miR-181a expression levels group in NSCLC patients had a poor survival in NSCLC patients [17]. Ma et al. reported that miR-181a-5p was significantly decreased in NSCLC tissues and cell lines and inhibits cell proliferation and migration by targeting Kras in non-small cell lung cancer A549 cells [12]. Our results showed that miR-181a was lower in NSCLC tissues and were significantly associated with histological grade, N status and TNM stage in the patients. Lower miR-181a predicted a poor prognosis in NSCLC patients. Upregulated miR-181a inhibited cell proliferation colony formation, and cell invasion in NSCLC cells.
Furthermore, we found that CyclinB1 and CyclinD1 was downregulated by miR-181a mimic in NSCLC cells and CDK1 was the potential targets of miR-181a by online predicted soft. Luciferase activity assay results demonstrated CDK1 were direct targets of miR-181a. The mRNA and protein expression of CDK1 were decreased by over-expressing miR-181 in NSCLC cells. CDK1 had been reported to promote the tumor progression, such as, Cucurbitacin-D-induced CDK1 mRNA up-regulation causes proliferation arrest of a non-small cell lung carcinoma cell line (NSCLC-N6) [18]. CCL21/CCR7 promotes G2/M phase progression via upregulating the expression of cyclin A, cyclin B1, or CDK1 and activating the ERK pathway in human non-small cell lung cancer cells [19]. We also demonstrated that knockdown of CDK1 inhibited the cell proliferation but co-transfected with si-CDK1 and miR-181a dismissed the effects. Thus, these results showed that miR-181a inhibited the cell proliferation by regulating CDK1 expression.
In conclusion, our results suggested the miR-181a was down-regulated in NSCLC and miR-181a inhibited the cell proliferation by regulating CDK1 expression. Thus, our findings indicated that miR-181a plays a tumor suppressor and functions as a potential therapeutic target in NSCLC patients.
Conflict of interest
The authors declare no conflict of interest.
Footnotes
Acknowledgments
We thank the members at Department of Oncology, Fuzhou Pulmonary Hospital of Fujian for their help.
