Abstract
BACKGROUND:
Cancer will become the leading cause of death worldwide in the 21st century, meanwhile, immunotherapy is the most popular cancer treatment method in recent years. COPI Coat Complex Subunit Beta 1 (COPB1) relates to human innate immunity. However, the role of COPB1 in pan-cancer remains unclear.
OBJECTIVE:
The purpose of this study was to explore the relationship between COPB1 mRNA expression and tumor infiltrating lymphocytes and immune examination sites in pan-cancer.
METHODS:
Data from multiple online databases were collected. The BioGPS, UALCAN Database, COSMIC, cBioPortal, Cancer Regulome tools, Kaplan-Meier Plotter and TIMER website were utilized to perform the analysis.
RESULTS:
Upregulation of COPB1 has been widely observed in tumor tissues compared with normal tissues. Although COPB1 has poor prognosis in pan-cancer, COPB1 high expression was beneficial to the survival of ESCA patients. Unlike ESCA, COPB1 expression in STAD was positively correlated with tumor infiltrating lymphocytes, including B cells, CD8
CONCLUSIONS:
COPB1 may be a prognostic biomarker for pan-carcinoma, and also provide an immune anti-tumor strategy for STAD based on the expression of COPB1.
Abbreviations
Introduction
Cancer incidence is rapidly growing and cancer mortality become the leading cause of death worldwide. A recent study assessed the tendence of various cancers incidence and mortality in 185 countries, and found the number of new cancer cases each year increased from 12.7 million in 2008 to 22 million in 2030 all over the world [1]. In order to improve the quality of life and prolong the survival time of cancer patients, immunotherapy targeting the interaction of tumor infiltrating lymphocytes (TILs) or immune checkpoints (PD-L1/CTLA4) with tumor cells has been developed, which can reactivate the adaptive and innate immune systems to produce a strong anti-tumor immune response. In recent years, immunotherapy is the most popular cancer treatment method, considered as the third revolution in cancer treatment. However, there are still many cancer patients who failed to respond to immunotherapy or have acquired drug resistance [2, 3, 4]. Thus, it is urgent to predict the efficacy of cancer immunotherapy and the prognosis of patients receiving immunotherapy timely and effectively, so as to guide the individualized immunotherapy of cancer patients more comprehensively and accurately. To solve this problem, growing studies focused on discovering the immune-related genes with prognostic value.
COPB1 is a subunit of coatomer protein complex consisting of the alpha, beta, beta’, gamma, delta, epsilon and zeta subunits. Glycolysis inhibition in combination with other anticancer therapies is a potential cancer treatment. 2-deoxyglucose (2DG), as a possible anti-tumor drug, has been shown to inhibit various tumor types and has been studied for nearly half a century [5, 6, 7, 8]. It has been found that silencing COPB1 expression can increase the sensitivity of tumor cells to 2DG toxicity [9]. It is well known that CD4 and MHC-I molecules play an important and irreplaceable role in the immune response. Previous studies have shown that COPB1 can promote the degradation of Nef cell target CD4 and MHC I antigens [10]. So far, COPB1 has only been reported to play a role in breast cancer metastasis [11]. There’s no study focused on the role of COPB1 in other tumors, and its expression in pan-cancer is unclear.
In this study, we utilized multiple online tools whose data are mainly from GEO, EGA, TCGA databases to visualize the expression, mutations, and prognosis of COPB1 in pan-cancer. Then, we used the TIMER to explore the potential relationship between COPB1 expression or mutation and immune cell infiltration levels or immune checkpoints (PD-L1 and CTLA4). The results of this study suggested that COPB1 may predict the efficacy and prognosis of immunotherapy in cancer patients through the interaction with infiltrating immune cells and immune examination sites.
Materials and methods
The Bio gene annotation portal (BioGPS)
BioGPS (
UALCAN database
UALCAN (
Catalog of somatic mutations in cancer (COSMIC)
COSMIC (
The cBio cancer genomics portal (cBioPortal)
cBioPortal (
Integrative data visualization
On the basis of the TCGA dataset, the Cancer Regulome tools (
Kaplan-Meier Plotter
The Kaplan-Meier Plotter (
Tumor Immune Estimation Resource (TIMER)
TIMER (
Results
The expression of COPB1 mRNA in cancer
The tissue specific expression pattern of COPB1 mRNA can indicate important clues to gene function. As shown in the BioGPS results in Fig. 1, higher expression of COPB1 was observed in B cells, dentritics cell (DCs), CD8
Expression profile for COPB1 in human tissues displayed by the Bio gene annotation portal (BioGPS).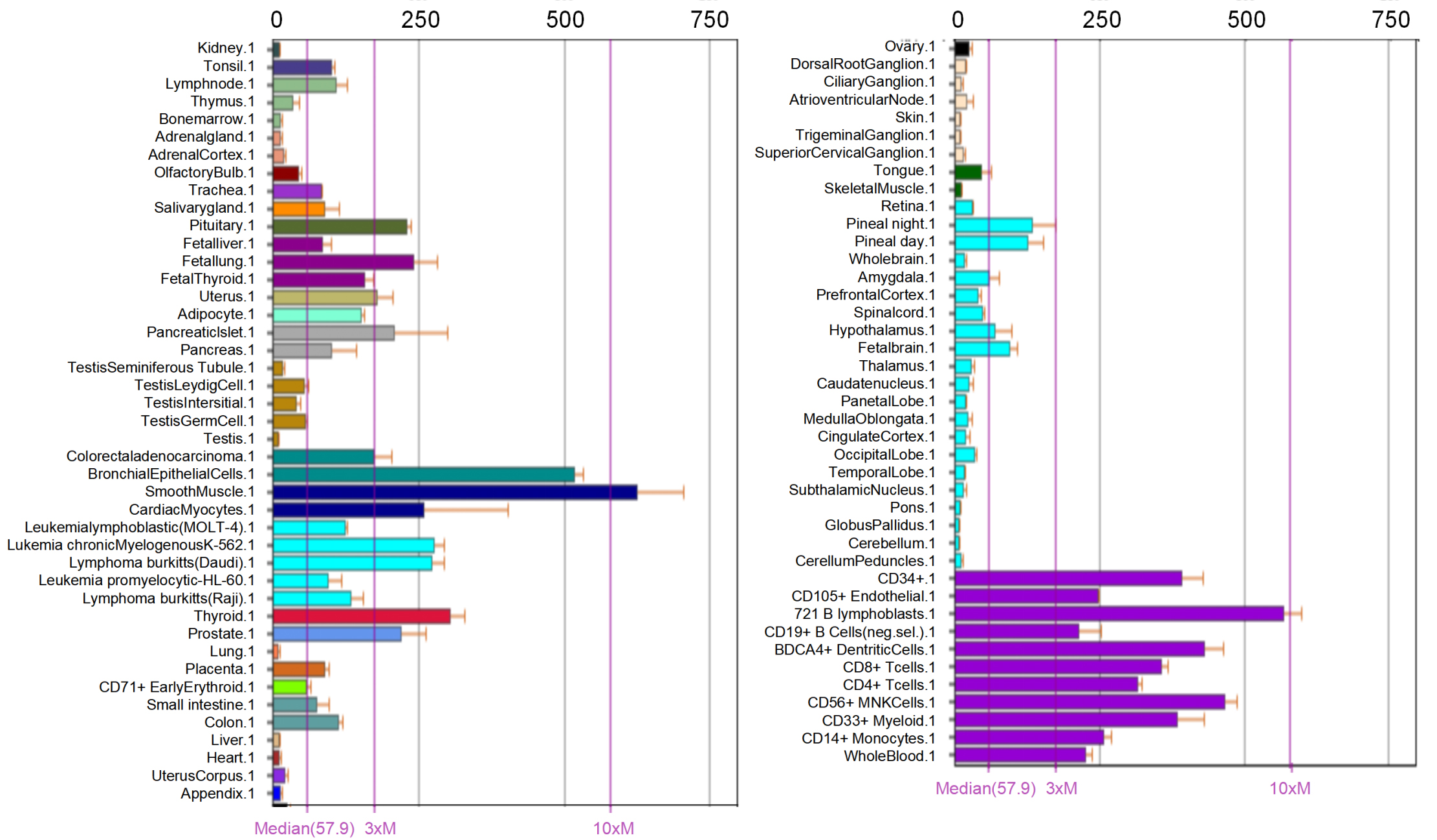
The mRNA expression level of COPB1 in normal tissues vs. tumor tissues in human cancers (UALCAN). 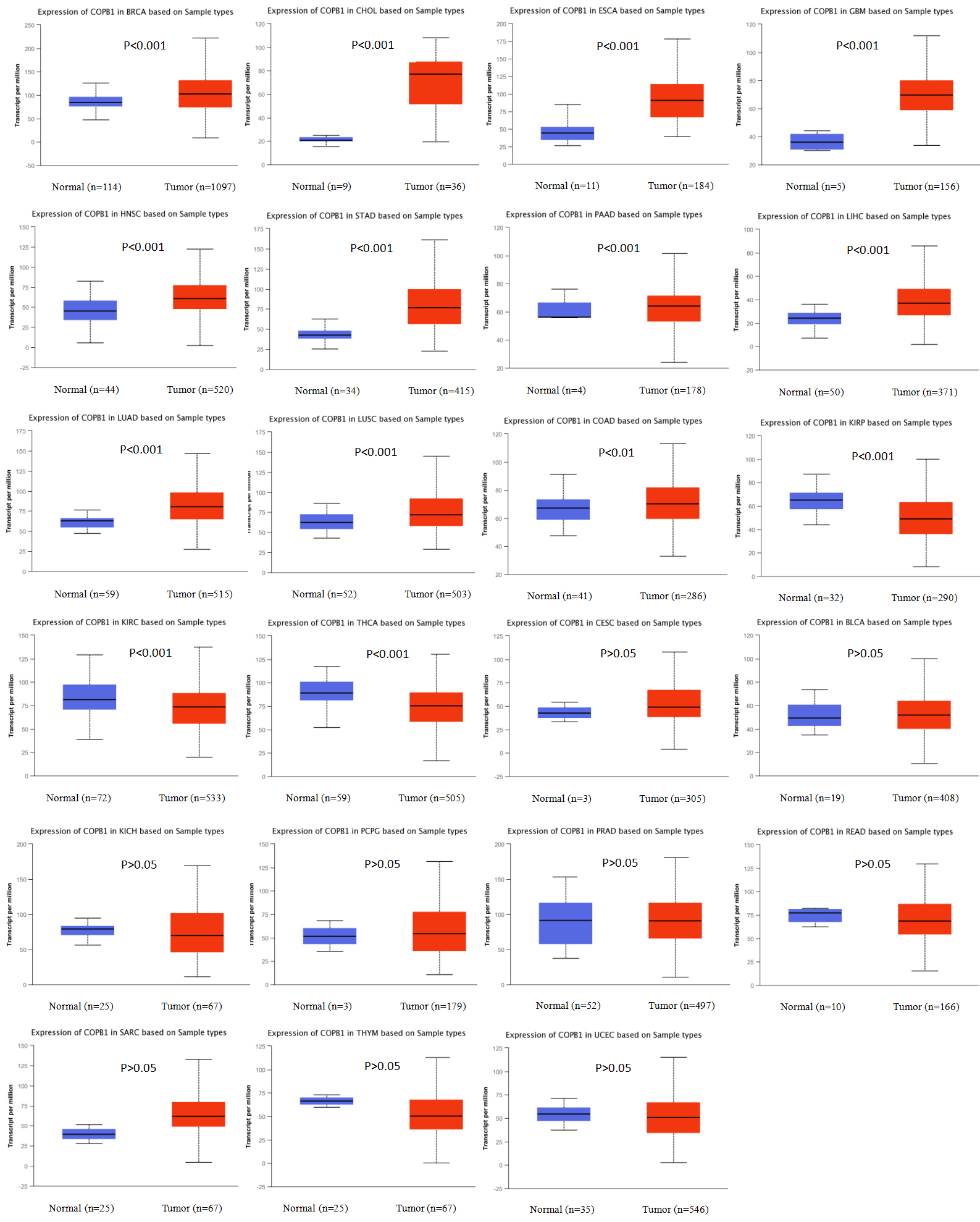
Data from 466 whole genome and large-scale systematic screens publications, as well as open-access data from The Cancer Genome Atlas (TCGA) and the International Cancer Genome Consortium (ICGC) was included in COSMIC. It provides information on COPB1 mutations in different cancers, including missense mutations, nonsense mutations, and synonymous mutations, and the results are presented in pie charts. Missense mutation caused the change of a certain amino acid in the peptide chain, affecting, altering or losing the function of the protein or making the protein gain new functions. Nonsense mutation made the translation of polypeptide chain stop in mutation, which often leads to the inactivation of protein. Synonymous mutation caused no change in the polypeptide and therefore have no effect. Nonsense substitutions were found in endometrial cancer (10%), biliary tract cancer (11.11%), kidney cancer (7.14%), large intestine cancer (4.6%), lung cancer (3.28%), pleura cancer (100%), stomach cancer (2.94%), upper aerodigestive tract cancer (8.33%) and urinary tract cancer (14.29%). Moreover, missense mutations were observed in endometrial cancer (60%), breast cancer (22.22%), central nervous system cancer (63.16%), cervix cancer (100%), hematopoietic and lymphoid cancer (15%), kidney cancer (57.14%), large intestine cancer (55.17%), liver cancer (11.68%), lung cancer (54.1%), esophageal cancer (29.03%), ovary cancer (25%), pancreas cancer (3.57%), prostate cancer (16.67%), salivary gland cancer (100%), skin cancer (69.23%), soft tissue (100%), stomach cancer (35.29%), thyroid cancer (80%), upper aerodigestive tract cancer (66.67%) and urinary tract cancer (42.86%). Additionally, synonymous substitution mutations appeared in endometrial cancer (20%), breast cancer (8.33%), central nervous system cancer (21.05%), kidney cancer (7.14%), large intestine cancer (11.49%), liver cancer (19.12%), lung cancer (22.95%), ovary cancer (25%), pancreas cancer (7.14%), skin cancer (19.23%), stomach cancer (14.71%), thyroid cancer (20%), upper aerodigestive tract cancer (16.67%) and urinary tract cancer (42.86%) (Fig. 3). G
Pie chart showing the percentage of the different mutation types of COPB1 in human cancers (COSMIC). Mutation types mainly include missense mutations, nonsense mutations, and synonymous mutations.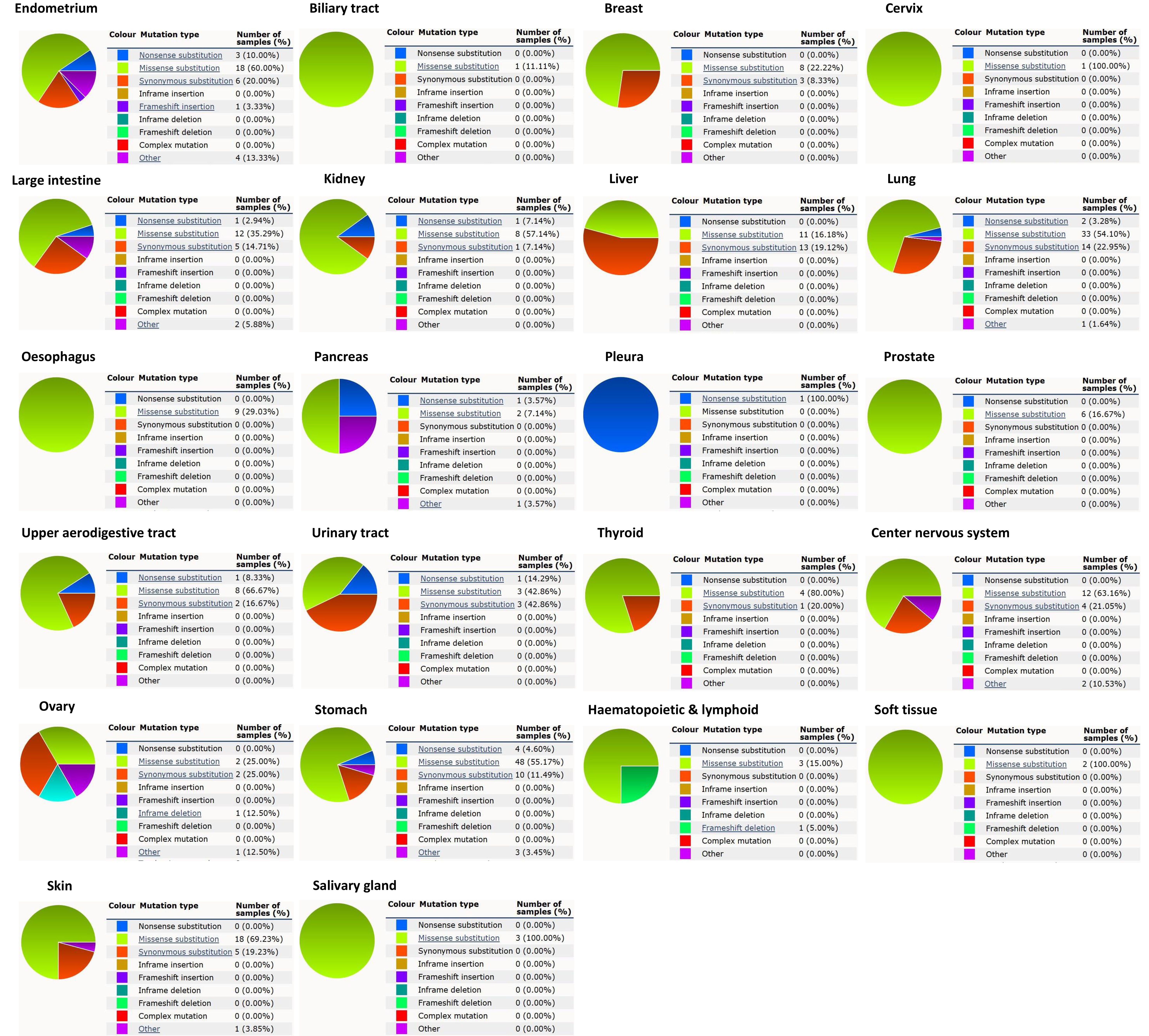
The relationship between COPB1 and mutation in human cancers. (A) COPB1 mutation level in pan-cancer; (B) Mutation diagram of COPB1 in different cancer types across protein domains (cBioPortal).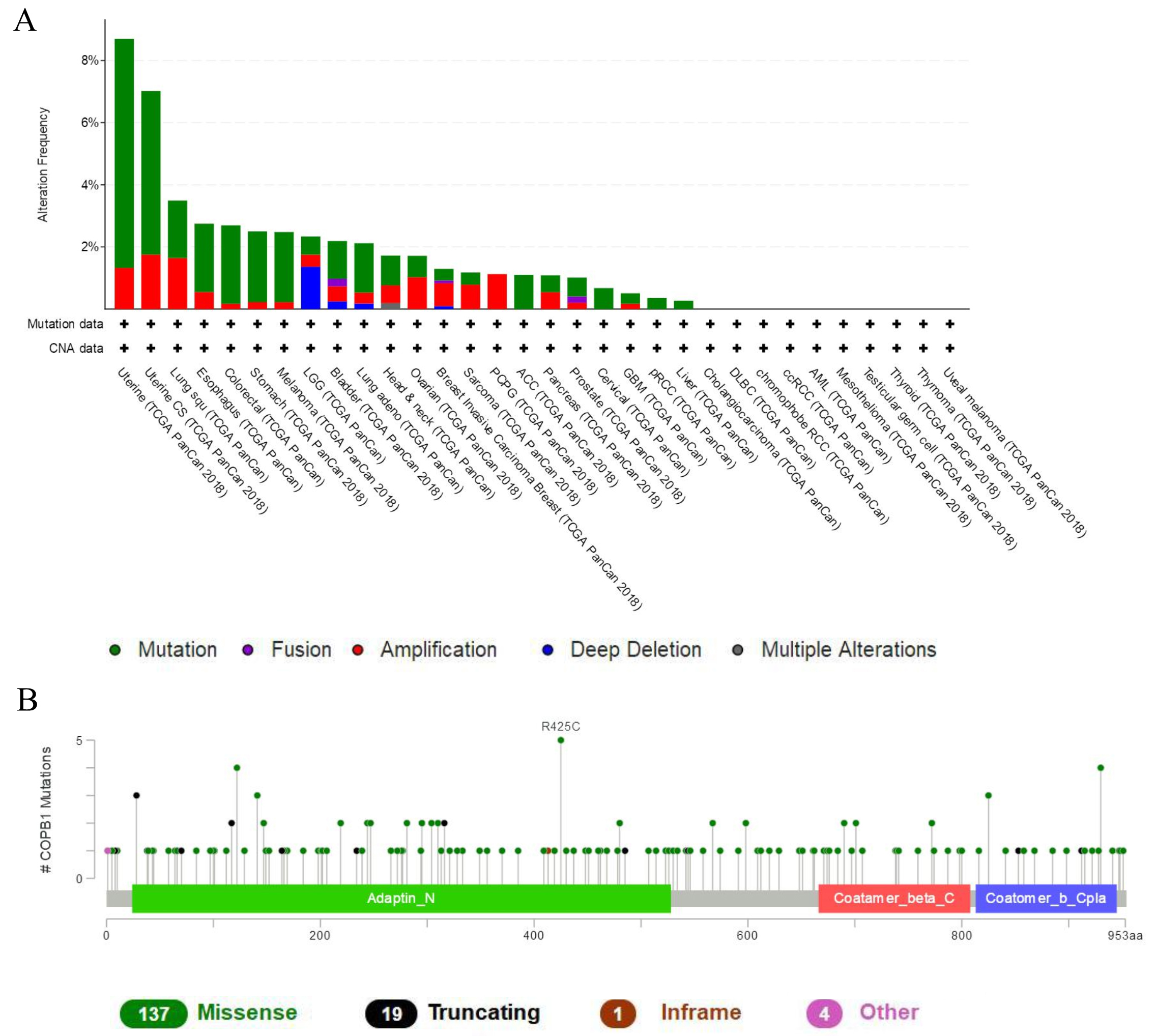
A total of 161 mutations sites were detected between amino acids 0 and 953 by online analysis of cBioPortal, percentage of samples with a somatic mutation in COPB1 was 1.2% (Fig. 4B). Data from TCGA pan-carcinoma studies had shown that the mutation rates of COPB1 were all at high levels in endometrial carcinoma, esophageal squamous cell carcinoma, non-small cell lung cancer, colorectal adenocarcinoma, melanoma, diffuse glioma, esophagogastric adenocarcinoma, bladder urothelial carcinoma, head and neck squamous cell carcinoma, ovarian epithelial tumor, breast invasive carcinoma, sarcoma, adrenocortical carcinoma, pancreatic adenocarcinoma, prostate adenocarcinoma, cervical squamous cell carcinoma, glioblastoma, renal non-clear cell carcinoma, hepatocellular carcinoma (Fig. 4).
We further analyzed the localization of the human genome and the association of certain genes with COPB1 in human cancers via using the Regulome Explorer. Circus diagrams were drawn to show the interrelationships between COPB1 and other genes based on the links between genes, somatic mutations, somatic copy numbers, DNA methylations and protein levels. The result had shown that the correlation between COPB1 and other genes could be detected in adrenocortical carcinoma, breast invasive carcinoma, colorectal cancer, bladder urothelial carcinoma, endometrial carcinoma, esophageal carcinoma with gastric esophageal carcinoma, gastric carcinoma, gliobastoma, kidney clear cell carcinoma, lung adenoma and squamous carcinoma, lower grade glioma and thyroid carcinoma (Fig. 5). Details had shown in Supplementary Tables 1 to 14.
The correlation between COPB1 and other genes from the TCGA database (Regulome program). Note: The circular layout displays the associations as edges in the Center connecting the features (with genomic coordinates) displayed around the perimeter. The outer ring displays cytogenetic bands. The inner ring displays associations that contain features lacking genomic coordinates.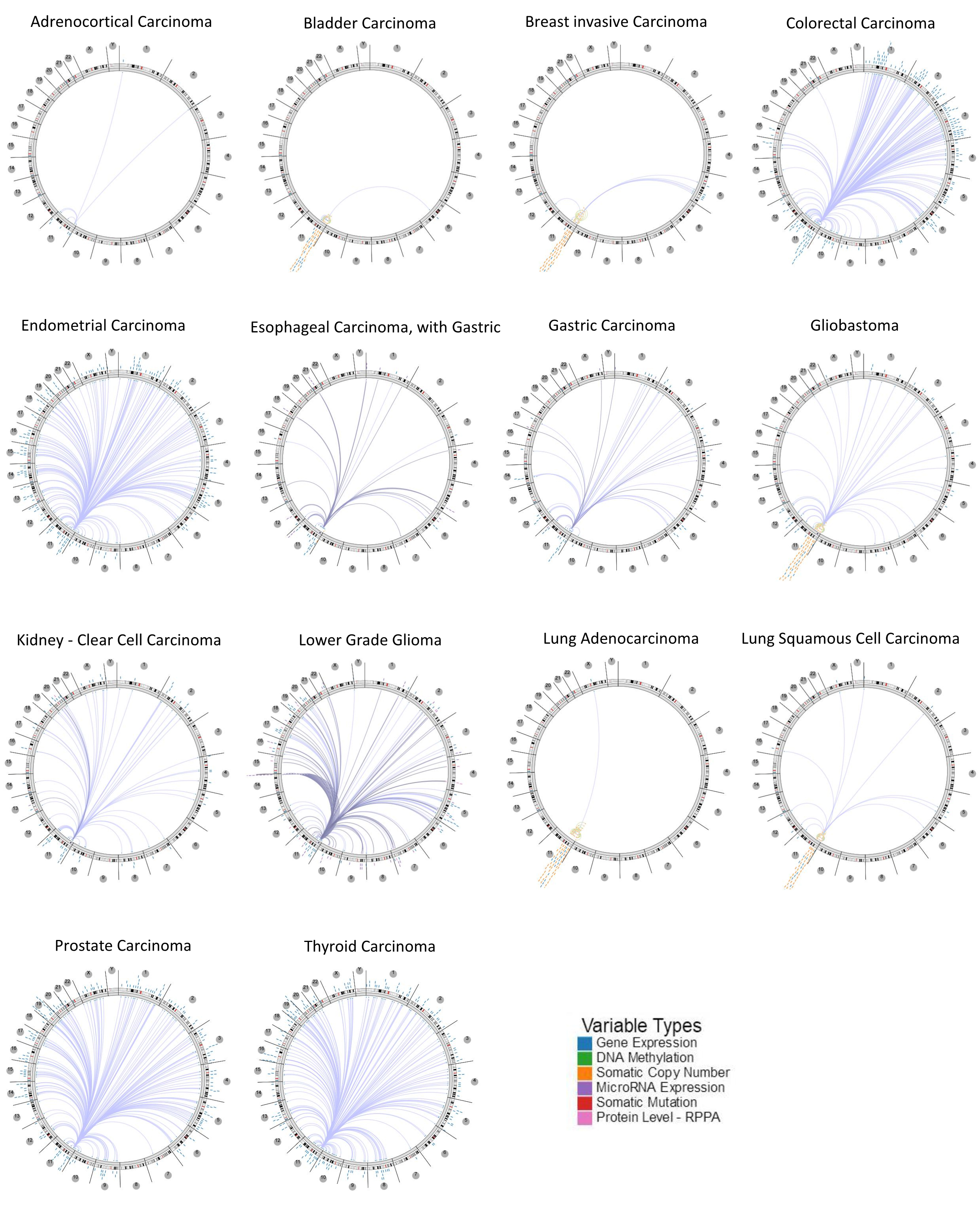
Different expression levels of COPB1 mRNA will result in different overall survival rates in cancers (Kaplan-Meier plotter). The mRNA expression of COPB1 in cancers was divided into high and low groups according to median. 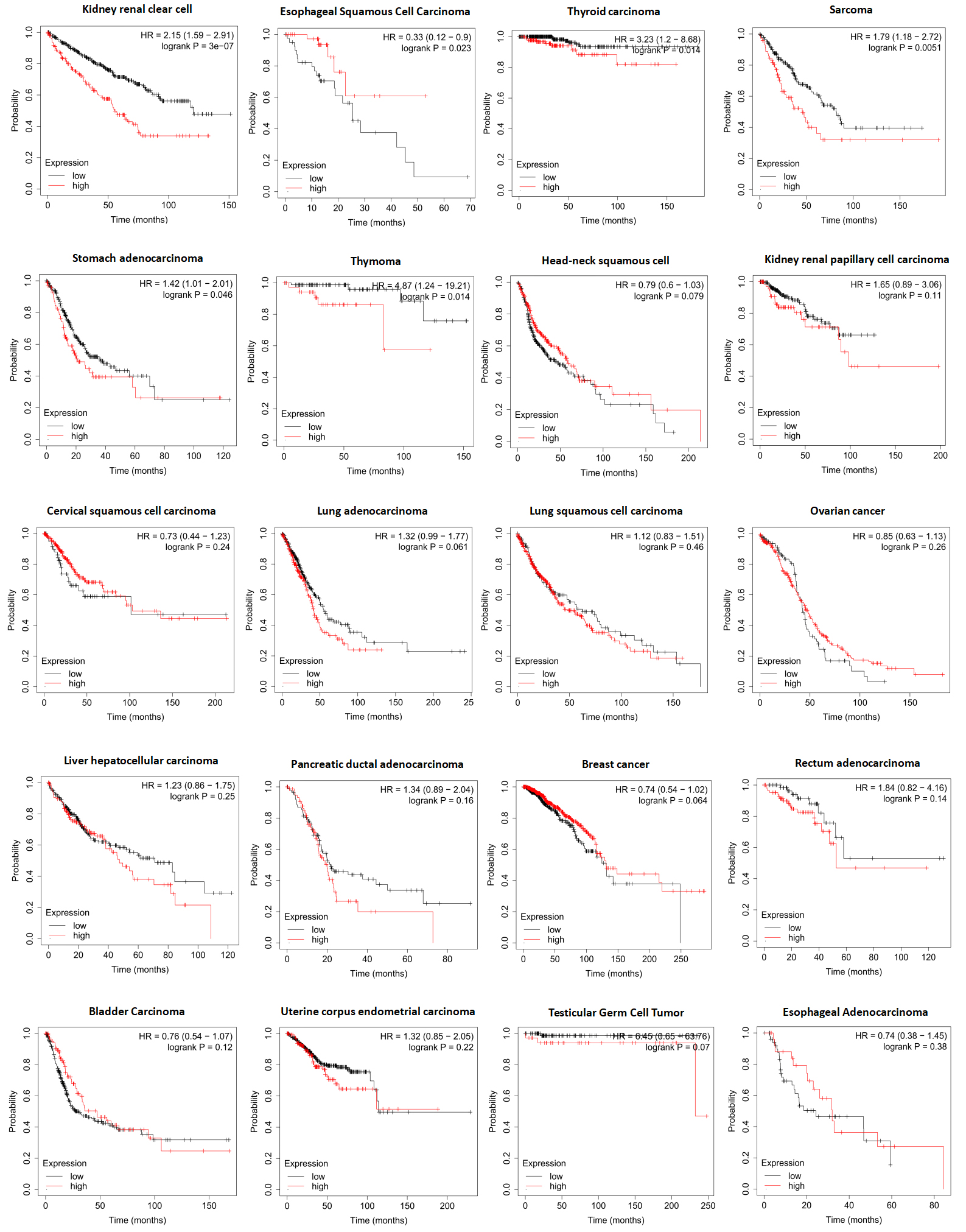
According to the Kaplan-Meier analysis results, higher expression COPB1 indicated worse OS in kidney renal clear cell carcinoma (
Correlations between COPB1 expression/ mutation and immune cells
Immune cells in tumor microenvironment can influence the survival of patients, and the above findings support the prognostic role of COPB1 in pan-carcinoma. Therefore, it is of great significance to explore the relationship between immune infiltration and COPB1 expression or mutation. Based on the above findings of the Kaplan-Meier plotter, we selected several cancer types whose prognosis was associated with COPB1 expression level, including kidney renal clear cell carcinoma (KIRC), sarcoma (SARC), stomach adenocarcinoma (STAD), thymoma (THYM) and thyroid carcinoma (THCA), and esophageal squamous cell carcinoma (ESCA), and then performed the correlation of COPB1 expression/mutation with immune infiltration level in these cancers via TIMER.
The results indicated that COPB1 expression was positive significantly correlated with the infiltration levels of CD8
Correlations between COPB1 expression and immune cells
Correlations between COPB1 expression and immune cells
Next, we used TIMER to analyze and visualize the effect of gene mutations on immune infiltration distribution in the mutant vs wildtype tumors, and the results were depicted in violin plots. Ultimately, we found that only mutations of COPB1 in ESCA and STAD had an effect on immune cell infiltration. In ESCA, the infiltration levels of CD8
The relationship between COPB1 and immunity. (A) Correlations between COPB1 mutation and immune cells; (B) Correlations between COPB1 expression and PD-L1; (C) Correlations between COPB1 expression and CTLA4 (TIMER). 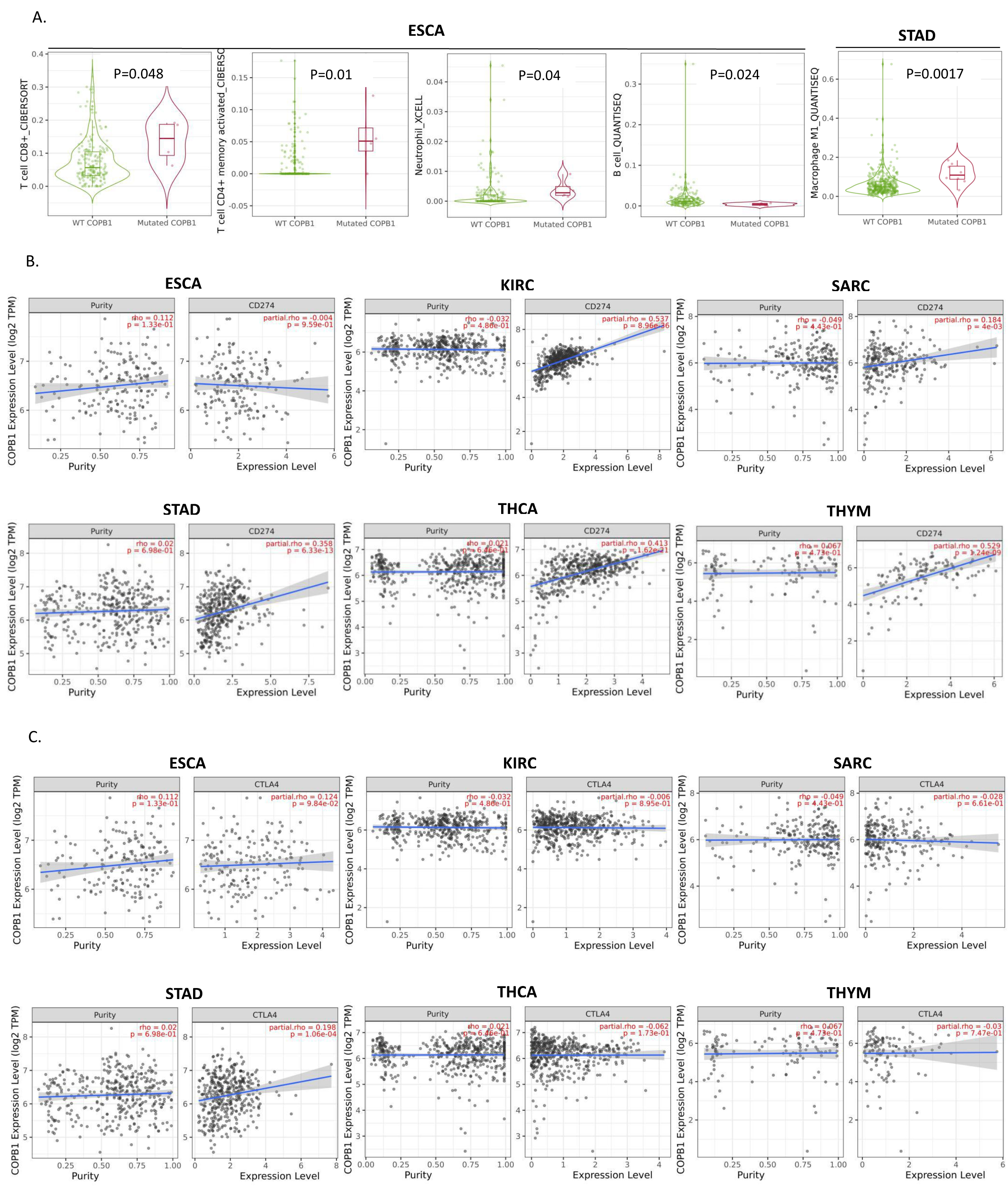
The TIMER was used to visualize the correlation of COPB1 expression with the expression level of PD-L1, CTLA4 in diverse cancer types. Then the correlation between the expression of COPB1 and PD-L1, CTLA4 in these cancers was further analyzed. The result indicated that the COPB1 expression level had significant positive correlations with the PD-L1 expression level in KIRC (
Discussions
The sheer volume of TCGA cancer genomics data, with its availability in different data formats, makes in-depth analyses difficult for clinicians and cancer researchers who lack bioinformatics or programming skills. In order to facilitate basic queries of the data, various analytical tools have been developed (Supplementary Table 15). In this study, different online tools were used to analyze the expression, mutation and prognosis of COPB1 in different tumors. We found that the expression of COPB1 was up-regulated in eleven cancer tissues, but down-regulated in three cancer tissues. In addition, patients with high expression of COPB1 had poor overall survival in KIRC, SARC, STAD, THYM and THCA, whereas in ESCA it’s showed the opposite result. According to the expression analysis results from BioGDP, COPB1 expressed higher in deverse immune-related cells than that in most other human tissues. Currently, there are no relevant reports about COPB1 and immune infiltration or immunotherapy. Therefore, we used TIMER to explore the relationship between the expression and mutation of COPB1 and tumor immune cells. Except SARC, the expression of COPB1 was positively correlated with tumor immune cells in other five cancers. Compared to the wild-type tumor tissues, COPB1 mutation tumor tissues were found to have higher infiltration levels of CD8
It is well known that genetic mutations play a very important role in the development of cancer. More than 90% of missense variants in public databases identified by clinical genetic testing are listed as variants of uncertain significance (VUS) [21, 22]. Missense variants with definitive pathogenic or neutral status can inform clinical management, prevention, and treatment. For example, patients with missense mutation of TP53 in triple negative breast cancer are often associated with poor prognosis [23]. Nonsense and missense mutation of mitochondrial ND6 gene promotes cell migration and invasion in human lung adenocarcinoma [23]. In many cases, cancer development is driven by somatic mutations and the number of somatic mutations per gene indicates the gene’s involvement in cancer development [24]. A recent study by Martincorena et al. [25] used normalized ratio of non-synonymous to synonymous mutations to identify genes under positive or negative selection in cancer evolution. The authors noted that about half of the identified driver mutations “occur in yet-to-be-discovered cancer genes”. So, it’s of great importance to recognize the type and number of mutations in different genes. In this study, it was found that COPB1 had nonsense mutations in nine cancer types and missense mutations in twenty cancer types.
To the best of our knowledge, the relationship between COPB1 expression and tumor cell metastasis has been proved in breast cancers [11, 26]. Moreover, it is acknowledged that autophagy is closely related to human immunity. Information from GeneCards indicates that COPB1 may participate in autophagy and play a role in early endosomal function. Meanwhile, it may also be involved in regulating the human’s innate immunity.
In the past 10 years, the research of tumor immunology has made great progress. New theories and understandings have emerged, and new tumor treatment strategies have been applied in clinical trials. The aim of tumor immunotherapy is to control and kill tumor cells by stimulating or activating human immune function, so as to re-educate tumor microenvironment and enhance anticancer immunity. To date, there’re three directions focused on cancer immunotherapy including checkpoint antibody therapy, adoptive cell immunotherapy (ACT), and tumor neogenic antigen vaccine. Among them, PD-L1 antibody is one of the most famous immunocheckpoint inhibitors. As a new drug in tumor immunotherapy, it has become the most important breakthrough in the field of tumor therapy. PD-L1 inhibitor has been shown to be effective against various cancers, particularly those caused by carcinogens or viral infections [27]. Currently, FDA has approved five antibodies of PD-L1 for clinical use. The third PD-L1 antibody to be proved effective is Duravulumab, which was suitable for the treatment of various solid tumors such as lung cancer, urothelial carcinoma, head and neck cancer, pancreatic cancer, breast cancer, gastrointestinal tract carcinoma, hepatobiliary system malignancy and urinary system malignancy. And it is currently being used in clinical trials in both solid carcinoma and hematologic tumors [28, 29, 30]. However, there are still many cancer patients not respond to PD-L1 inhibitors treatment, and the specific mechanisms need further study. In addition, data from numerous clinical trials have also shown that tumor cells could develop acquired resistance to PD-L1 inhibitors [2, 3, 4]. Therefore, it is urgent to improve the efficiency of PD-L1 inhibitor and reduce resistance.
Additionally, the expression level of PD-L1 was limited in predicting the efficacy of immunocheckpoint inhibitors [31]. Previous studies had shown that the expression of PD-L1 combined with TILs could elucidate the characteristics of tumor immune microenvironment better than PD-L1 alone, so as to predict the efficacy and prognosis of immunotherapy more effectively [32]. Multi-drug combination has also become a hot spot and treatment trend of immunotherapy. For example, the combination of PD-L1 and CTLA4 (such as Nivolumab and Ipilimumab) can prolong the progression-free survival of patients with advanced renal cell cancer and lung cancer [33, 34], which are highly complementary. In this study, we found that COPB1 was significantly overexpressed only in STAD tumor tissues and was associated with worse OS. Moreover, the expression of COPB1 was positively correlated with PD-L1, CTLA4 and TILs. Previous studies have shown that PD-L1 and CTLA4 were associated with the occurrence of STAD, and both of them were up-regulated in patients with this tumor. In vitro amplification of TILs and specific T cells in peripheral blood of patients with STAD demonstrated antitumor effects [35]. Schaefer et al. found that COPB1 was involved in the internalization of CD4 molecules and MHC-I molecules on the surface of HIV-infected T cells and accelerated their degradation in lysosomes, thus helping HIV-infected T cells evade immune surveillance and ultimately leads to HIV infection and transmission [10]. Additionally, it has been reported that suppression of COPB1 inhibits the invasion and metastasis of breast cancer cells [11]. So we speculate that COPB1 overexpression in cancer cells may help cancer cells to evade the immune system, resulting in tumor growth and metastasis. To date, this study was the first to explore the role of COPB1 in STAD and discovered a new prognostic marker and immune prognostic target for STAD. In addition, Kono et al. isolated TILs from primary metastatic lymph nodes and autologous gastric cancer ascites, and found that TILs can specifically bind to gastric cancer antigens in vitro experiments [36]. Combined with the above findings, it suggested that COPB1 was likely to become a new marker that can effectively predict the efficacy and prognosis of immunocheckpoint inhibitor therapy in STAD.
However, although data from multiple databases were consolidated and analyzed, this study has limitations. First, a large amount of chip and sequencing data were collected via analyzing tumor tissue information. Thus, cell-level analysis of immune cell markers may lead to systematic bias. To overcome it, higher resolution studies, such as single-cell RNA sequencing, should be undertaken in the future [37, 38]. Second, this study only conducted bioinformatics analysis on COPB1 expression and patient survival in different databases, and did not conduct in vivo or in vitro experiments. Future studies on the mechanisms of COPB1 at the cellular and molecular levels will help clarify the role of COPB1. Third, although COPB1 expression was found to be associated with TILs and patient survival in some cancers, we were unable to prove that COPB1 affected patient survival through immune infiltration. Prospective studies of COPB1 expression and immunotherepy or TILs in cancer populations may help provide definitive answers in the future.
Conclusions
In summary, COPB1 was generally overexpressed in pan-cancer and affected patients’ prognosis. In particular, for STAD, the expression of COPB1 was positively correlated with TILs, PD-L1 and CTLA4. Thus, COPB1 may not only be a prognostic biomarker for pan-carcinoma, but also provide a new insights on immune anti-tumor strategy based on the expression of COPB1.
Supplementary data
The supplementary files are available to download from http://dx.doi.org/10.3233/CBM-200398.
sj-xlsx-1-cbm-10.3233_CBM-200398.xlsx - Supplemental material
Supplemental material, sj-xlsx-1-cbm-10.3233_CBM-200398.xlsx
sj-docx-1-cbm-10.3233_CBM-200398.docx - Supplemental material
Supplemental material, sj-docx-1-cbm-10.3233_CBM-200398.docx
sj-docx-2-cbm-10.3233_CBM-200398.docx - Supplemental material
Supplemental material, sj-docx-2-cbm-10.3233_CBM-200398.docx
Footnotes
Acknowledgments
We thank The Cancer Genome Atlas, Gene Expression Omnibus and European Genome-phenome Archive for sharing the large amount of data. The authors also thank National Natural Science Foundation of China, NSFC (grant No. 81702632) for funding to do this work.
Conflict of interest
The authors declare no conflicts of interest.
