Abstract
Background:
Proton pump inhibitors (PPIs) have been shown to regulate the gut microbiome and affect the response to immune checkpoint inhibitors (ICIs). Contradictory results on survival have been observed in patients concomitantly treated with ICIs and PPIs. We performed a systematic review and meta-analysis to determine the association between PPI use and survival outcomes in ICI-treated cancer patients.
Methods:
EMBASE, MEDLINE/PubMed, Cochrane Library databases, and major oncology conference proceedings were searched. Studies comparing overall survival (OS) and progression-free survival (PFS) between PPI-treated and PPI-free groups of ICI-treated cancer patients were included. Data regarding study and patient characteristics, ICI and PPI treatments, and survival outcomes were extracted. Hazard ratios (HRs) with 95% confidence interval (CI) were pooled using random effects models. Subgroup meta-analyses and meta-regressions were performed to explore possible factors of heterogeneity among the studies.
Results:
A total of 33 studies were included, comprising 7383 ICI- and PPI-treated patients and 8574 ICI-treated and PPI-free patients. The pooled HR was 1.31 (95% CI, 1.19–1.44; p < 0.001) for OS and 1.30 (95% CI, 1.17–1.46; p < 0.001) for PFS, indicating a significant negative association between PPI use and survival in ICI-treated patients. Subgroup meta-analyses by factors including cancer type, ICI type, and time window of PPI use revealed that ICI and PPI use impacted survival in patients with non-small cell lung or urothelial cancer, patients treated with anti-PD-1/PD-L1 antibodies, and patients receiving PPI as baseline treatment or 60 days before ICI treatment initiation.
Conclusions:
PPI use in patients treated with ICIs was associated with shorter OS and PFS, especially in several specific subgroups of cancer patients. PPIs should be strictly controlled and appear to not impact survival if given temporarily after ICI initiation. These observations could provide the basis for clinical guidelines for concomitant PPI and ICI use.
Introduction
Currently, immune checkpoint inhibitors (ICIs) that target programmed cell death protein 1 (PD-1) or its ligand (PD-L1), alone or in combination with ICIs that target cytotoxic T lymphocyte-associated antigen 4 (CTLA-4), have achieved remarkable results in the management of certain cancers, particularly melanoma, lung cancer, and microsatellite instable colon cancer.1–3 Although ICIs are effective, they are limited by a low response rate, which represents approximately 20–30% of patients.4,5 Thus, there is an unmet medical need to identify factors that affect the efficacy of ICIs and their underlying mechanisms.
A growing number of studies have demonstrated that an altered gut microbiome can impact homeostasis, systemic immune response, and ICI treatment efficacy in various cancer patient populations, though the exact underlying mechanisms remain unclear. The gut microbiome interacts with cytokines to regulate antigen presentation and by partial functional regulation of effector T cells or myeloid cells in the tumor microenvironment.6,7
Proton pump inhibitors (PPIs), among the 10 most widely prescribed drugs globally, are also frequently given to cancer patients. 8 Significant gut microbiome dysregulations and reduced bacterial richness are associated with PPIs.9,10 Unlike antibiotics, which have been shown to regulate the gut microbiome, which in turn modulates response to ICIs, the effect of PPIs on gut microbiome alterations and on the response to ICIs have been only scarcely researched. 11 These observations prompted us to question whether the concomitant use of PPIs affects the treatment efficacy of ICIs. The contradictory survival results recently obtained from patients concomitantly treated with ICIs and PPIs lent credence to the need for our investigation.12,13 We, therefore, performed a systematic review and meta-analysis to better define the effect of PPIs on cancer patients treated with ICIs.
Methods
Protocol and reporting guidelines
The study protocol was registered on the International Prospective Register of Systematic Reviews (PROSPERO CRD42020181618) and INPLASY (Protocol 2020100088) and followed the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-analyses) 2020 checklist. 14
Information sources and search strategy
Studies reporting the impact of PPI use on the response to ICIs, including survival data of ICI-treated patients from inception to 22 March 2022 were identified by comprehensively searching the EMBASE, MEDLINE (via PubMed), Cochrane Library databases, and major oncology conference proceedings by two investigators (C.Y. and B.Q.C.) independently. Furthermore, emails requesting additional data were sent to the corresponding authors of the studies of interest. Moreover, reference lists of initially detected articles were hand-searched.
The search queries were built using the following broadly defined medical terms: (‘pembrolizumab’ OR ‘nivolumab’ OR ‘durvalumab’ OR ‘ticilimumab’ OR ‘toripalimab’ OR ‘sintilimab’ OR ‘camrelizumab’ OR ‘ipilimumab’ OR ‘immune checkpoint inhibitor*’ OR ‘ici*’ OR ‘anti-PD-1*’ OR ‘anti-PD-L1*’ OR ‘anti-CTLA-4*’) AND (‘omeprazole’ OR ‘lansoprazole’ OR ‘dexlansoprazole’ OR ‘esomeprazole’ OR ‘pantoprazole’ OR ‘rabeprazole’ OR ‘proton pump inhibitor*’ OR ‘ppi*’), without any filters and limits. The detailed queries of the search strategies for different data sources are shown in Supplemental Table 1.
Inclusion criteria
All available studies (publications, abstracts, posters, and clinical trials) comparing survival between concomitant use of ICIs and PPIs (intervention group) and ICI use alone (comparator group) in cancer patients were preliminarily included. The studies endpoints were defined as hazard ratios (HRs) with 95% confidence intervals (CIs) for overall survival (OS) and/or progression-free survival (PFS). Only studies reporting HRs with 95% CIs for OS or PFS were included in the meta-analysis; these data were extracted directly from original publications, requested and obtained from the researchers, or calculated using Kaplan–Meier curves based on a recognized method. 15
In case of duplicate data from overlapping cohorts, only the largest and the most up-to-date studies were included. Two investigators (C.Y. and B.Q.C.) independently screened the titles and abstracts and then assessed the full-text articles for eligibility. When discrepancies occurred, a third investigator (D.M.C.) reviewed the study and decided on its eligibility.
Data collection process and the risk of bias
The data from all included studies were extracted and summarized by one investigator (C.Y.) using a standardized spreadsheet, whereas the selection and coding of data were reviewed by two other investigators (B.Q.C. and M.P.D.) independently. When results of univariate and multivariate analyses were both available, the data from the multivariate analysis were included. The following data were collected:
– Source: Author, year and type of publication, type of study.
– Patient and cancer characteristics: Region and number of patients included, type and stage of cancer.
– ICI treatment: Type and treatment line of ICIs (i.e. first-line therapy, non-first-line therapy).
– PPI treatment: Number and percentage of patients receiving PPIs and ICIs, types and time window of PPI use.
– Results: Type of analysis, HRs with 95% CIs for OS and PFS, and statistical significance of the comparison.
The risk of bias in individual studies was assessed using the modified Newcastle–Ottawa Quality Assessment Scale (NOS), 16 which includes three aspects: selection of cohorts, comparability of cohorts, and assessment of outcomes. Given that cohorts differ in population size and treatment options, we further researched the studies’ comparability by assessing and comparing additional variables such as age, gender, race, tumor type, tumor stage, ICI type, Eastern Cooperative Oncology Group-Performance Status, treatment line of immunotherapy, and other concomitant treatments (e.g. antibiotics and corticosteroids). Two investigators (C.Y. and B.Q.C.) independently evaluated the risk of bias and discussed to reach a consensus when disagreements arose.
Statistical analysis
Data synthesis was performed on the effect measure HR with 95% CIs for OS and PFS for each analysis: HR > 1.0 indicated a worse outcome in the PPI-treated arm and two-sided p < 0.05 was considered statistically significant. The Cochrane Q test and the I2 index were used to evaluate heterogeneity; p < 0.1 for Q test or I² > 50% indicated significant heterogeneity. 17 When these two methods yielded heterogeneous results, the random effects model was adopted, which was more conservative and reliable for making conclusions 18 because it accounts for differences in population and treatment characteristics. All statistical analyses were performed using Stata (version 16.0, Stata Corp, College Station, TX, USA), and forest plots were used to display the results. Subgroup analyses and meta-regression were performed to investigate possible causes of heterogeneity among study results. Funnel plots with Egger’s regression tests were used to examine publication bias across studies. Sensitivity analyses were performed using the leaving-one-out approach.
Results
Study selection and study characteristics
The preliminary search yielded 826 records. After duplicate studies were removed, titles and abstracts of 613 studies were screened. Reviews, case reports, animal experiments, and studies unrelated to PPI or ICI were discarded (n = 566), and the remaining 47 publications were assessed. Six studies (two commentaries, two repeat studies, and two studies on antacids but not PPI alone) were excluded, and 41 were included in the qualitative synthesis. Eight additional studies were excluded because no HR data were available and the corresponding authors did not respond to our inquiry emails. Finally, 33 studies, of which 24 were peer-reviewed journal publications and 9 were conference abstracts, were included in the systematic review and meta-analysis.12,13,19–49 The flow diagram of identifying the eligible studies is shown in Figure 1.

Flow diagram of included studies.
A total of 33 studies were included, comprising 15,957 patients for OS and 7577 patients for PFS analyses. Characteristics of the studies included in the meta-analysis are presented in Table 1. These 33 studies were subjected to retrospective or, for randomized clinical trials, pooled post hoc analyses. The patient sample sizes of all studies ranged from small to medium (n = 49–3634). The prevalence of PPI use ranged from 21.2 to 84.3%, and there were 7383 PPI-treated patients and 8574 PPI-free patients. In 13 studies, patients with different cancer types were enrolled. Among all included studies, the following ICI regimens were used: anti-PD-1 monoclonal antibodies (mAbs) alone (toripalimab, sintilimab, camrelizumab, pembrolizumab, and nivolumab; 10 studies), anti-PD-L1 mAbs alone (atezolizumab; three studies), anti-PD-1 or anti-PD-L1 (anti-PD-[L]1) mAbs (9), anti-CTLA-4 mAb alone (ipilimumab; one study), or anti-PD-(L)1 combined with anti-CTLA-4 mAbs (one study). The use of PPI ranged from 60 days before ICI initiation to 60 days after ICI initiation in most studies, although other studies reported concomitant or baseline use.
Studies included in the meta-analysis.

Studies in which a statistically significant association with shorter OS and/or PFS was observed.
Studies in which a statistically significant association with longer OS and/or PFS was observed; in all other studies no statistical difference was obtained.
CI, confidence interval; CTLA-4, cytotoxic T lymphocyte-associated antigen 4; GC, gastric cancer; HCC, hepatocellular carcinoma; HNC, head and neck cancer; HR, hazard ratio; ICI, immune checkpoint inhibitor; MVA, multivariate analysis; NA, not available; NSCLC, non-small cell lung cancer; OS, overall survival; PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; PFS, progression-free survival; PPI, proton pump inhibitor; RCC, renal cell carcinoma; RP, retrospective design, prospective data collection; UC, urothelial cancer; UVA, univariate analysis.
Of the 33 publications, 25 received an NOS assessment score of 7 or higher, and 8 received a score of 5 or 6, suggesting the studies were of high or moderate methodological quality (Supplemental Table 2).
Meta-analysis of OS and PFS
Kulkarni et al. provided HR data of survival for renal cell carcinoma (RCC) and non-small cell lung cancer (NSCLC) separately, and Husain et al. and Mollica et al. provided HR data for subgroups categorized by ICI treatment line and ICI type, respectively, instead of the whole population; therefore, we pooled the HR data of these subgroups separately. Thus, there were 35 cohorts providing HR data for OS and 26 cohorts for PFS. HRs for PPI-treated versus PPI-free subgroups ranged from 0.30 (95% CI, 0.10–0.70) to 4.12 (95% CI, 2.28–7.46). In 17 cohorts, a statically significant association was observed between PPI and ICI use and shorter OS,13,20–23,25,26,29–31,33,37,43,45,48 and in 11 a significant association was observed between PPI and ICI use and shorter PFS.13,20,25,26,29–31,35,37,43,48 In 18 cohorts no impact on OS,12,19,24,27,28,32,34,36,38–42,44,46,47,49 and in 14 no impact on PFS,12,23,24,27,32,34,38,40–42,47,49 was observed in patients treated with ICI and PPI versus ICI without PPI. However, in one cohort significantly longer PFS was observed in patients treated with PPI and ICI. 19 Additionally, we observed that especially (but not exclusively) in larger studies, a significant association between ICI and PPI use versus ICI without PPI and shorter OS and/or PFS was likely to be detected. Together, these data proved once more the importance of our study question and that a method capable of combining multiple studies together is ideal for answering it.
The between-study heterogeneities were moderate (I2 of 72% and 63% for OS and PFS, respectively). Pooled HRs with 95% CIs were calculated using random effects models. For OS a pooled HR of 1.31 (95% CI, 1.19–1.44; p < 0.001) was obtained, which indicated that significantly shorter OS was found in patients treated with ICIs and PPIs (Figure 2(a)). In accordance with OS data, pooled HRs for PFS indicated that PPI use in patients treated with ICIs resulted in shorter PFS (HR, 1.30; 95% CI, 1.17–1.46; p < 0.001; Figure 2(b)).
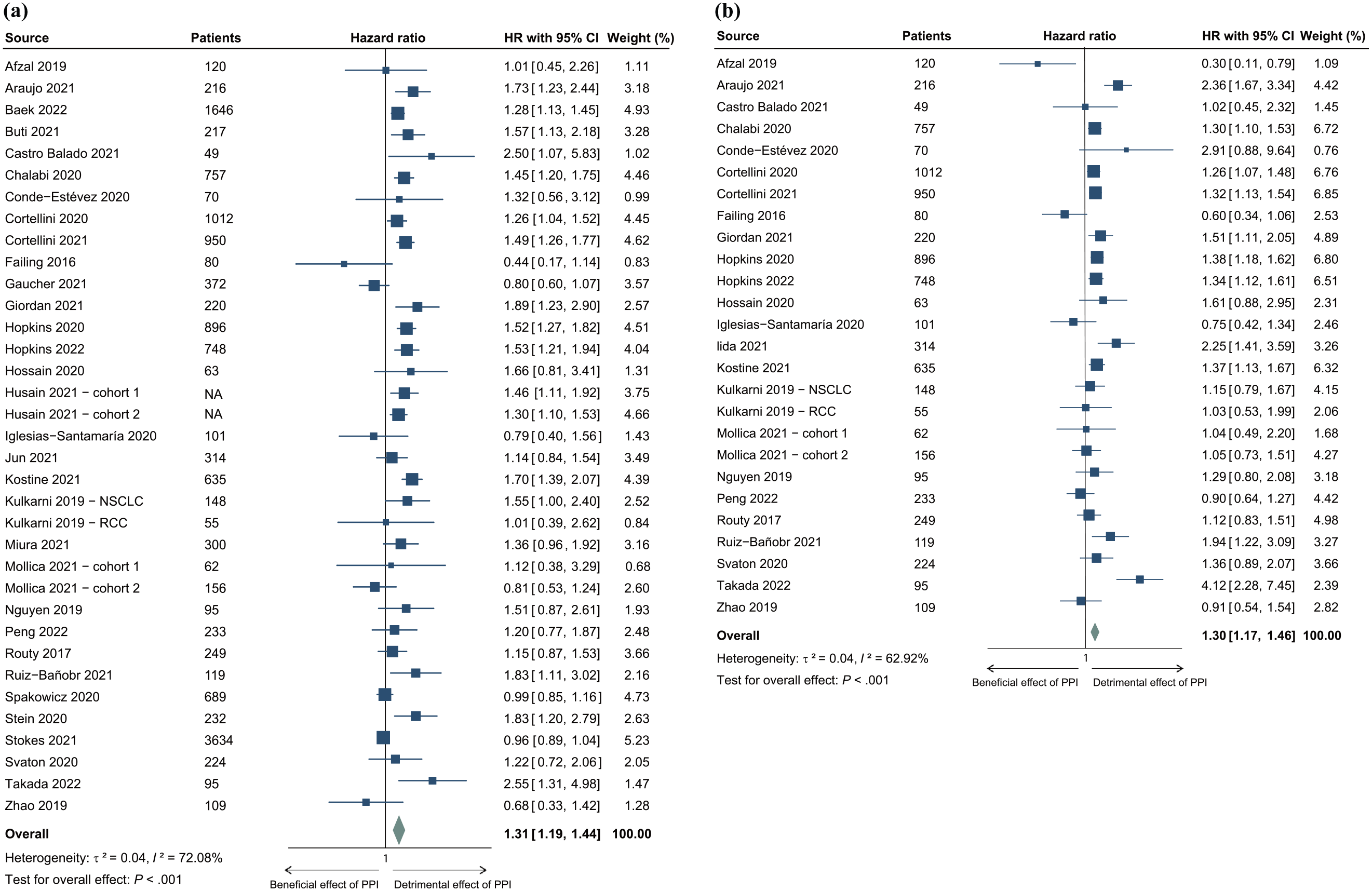
Forest plots comparing the OS and PFS of PPI-treated versus PPI-free patients treated with ICIs for cancer. Results for (a) OS and (b) PFS are shown.
Meta-analysis of subgroup factors
To explore possible factors of heterogeneity among study results, subgroup analyses were conducted by multiple factors, including time window of PPI use, cancer type, ICI type, treatment line of ICIs, and patients’ region of residence.
Patient populations were divided into three subgroups according to the time window of PPI use: group A, PPI therapy as baseline treatment; group B, PPI therapy begin within 60 days before ICI treatment initiation (−60, NA); group C, concomitant treatment (0, NA; Supplemental Figure 1). The pooled HRs for OS and PFS in these subgroups are displayed in Figures 3(a) and 4(a). In our comparison of PPI-treated versus PPI-free subgroups, shorter OS and PFS were found in group A (OS: HR, 1.43; 95% CI, 1.21–1.69; p < 0.001; PFS: HR, 1.29; 95% CI, 1.15–1.44; p < 0.001) and group B (OS: HR, 1.35; 95% CI, 1.22–1.51; p < 0.001; PFS: HR, 1.33; 95% CI, 1.20–1.48; p < 0.001). However, there was no significant impact of PPI therapy on survival when the PPI therapy was concomitantly started with ICIs (group C; OS: HR, 1.18; 95% CI, 0.98–1.41; p = 0.09; PFS: HR, 1.19; 95% CI, 0.65–2.17; p = 0.58).

Forest plots for OS of subgroup analyses. Results for OS by (a) time window of PPI use, (b) cancer type, and (c) ICIs type. Group A, PPI therapy as baseline treatment; group B (−60, NA), PPI therapy beginning within 60 days before ICI treatment initiation; group C (0, NA), PPI therapy beginning concomitantly with initiation of ICI treatment.

Forest plots for PFS of subgroup analyses. Results for PFS by (a) time window of PPI use, (b) cancer type, and (c) ICIs type. Group A, PPI therapy as baseline treatment; group B (−60, NA), PPI therapy beginning within 60 days before ICI treatment initiation; group C (0, NA), PPI therapy beginning concomitantly with initiation of ICI treatment.
Analyses of NSCLC, RCC, melanoma, and urothelial cancer (UC) were all reported in more than two studies and were included in the subgroup analyses. The HRs for OS of NSCLC and UC patients, respectively, in PPI-treated versus PPI-free groups were 1.33 (95% CI, 1.15–1.54; p < 0.001) and 1.54 (95% CI, 1.30–1.82; p < 0.001). The impact of PPI on the OS of ICI-treated patients with RCC (HR, 1.01; 95% CI, 0.77–1.33; p = 0.92) or melanoma (HR, 1.39; 95% CI, 0.87–2.22; p = 0.16) was not significant (Figure 3(b)). Similar to our OS findings, the use of PPIs resulted in shorter PFS in ICI-treated patients with NSCLC (HR, 1.33; 95% CI, 1.17–1.51; p < 0.001) and UC (HR, 1.63; 95% CI, 1.23–2.15; p = 0.001) but not RCC (HR, 1.11; 95% CI, 0.89–1.38; p = 0.37) and melanoma (HR, 0.89; 95% CI, 0.44–1.81; p = 0.75; Figure 4(b)).
For our analysis based on the type of ICIs, five subgroups (anti-PD-1 alone, anti-PD-L1 alone, anti-PD-[L]1 [anti-PD-1 or anti-PD-L1], anti-CTLA-4, and anti-PD-[L]1 combined with anti-CTLA-4) were identified, and their pooled HRs for OS and PFS are presented in Figures 3(c) and 4(c). In the subgroups treated with anti-PD-1, anti-PD-L1, and anti-PD-(L)1, a negative impact of PPI use was noted on both OS (anti-PD-1: HR, 1.36; 95% CI, 1.12–1.64; p < 0.01; anti-PD-L1: HR, 1.50; 95% CI, 1.33–1.68; p < 0.001; anti-PD-(L)1: HR, 1.28; 95% CI, 1.07–1.52; p < 0.01) and PFS (anti-PD-1: HR, 1.32; 95% CI, 1.16–1.51; p < 0.001; anti-PD-L1: HR, 1.34; 95% CI, 1.22–1.48; p < 0.001; anti-PD-(L)1: HR, 1.52; 95% CI, 1.13–2.03; p < 0.001). Unlike the broadly researched impact of PPI on anti-PD(L)1, the subgroups treated with anti-CTLA-4 and anti-PD-(L)1 combined with anti-CTLA-4 subgroups contained only one study each, and no association was noted between PPI treatment and OS or PFS. Therefore, the impact of PPI on anti-CTLA-4 and anti-PD-(L)1 combined with anti-CTLA-4 subgroups will need to be further investigated if additional studies appear.
PPI use shortened the OS and PFS when ICIs were used in non-first-line therapy rather than in first-line ICI therapy (Supplemental Figure 2A and 2B). Subgroup analysis based on the patients’ geographic region [Europe, America (USA and Brazil), Asia, and Australia] was also performed. PPI use showed a detrimental effect on PFS and OS in studies from Europe and Australia, but no effect on survival was noted in studies from America (USA and Brazil). For patients from Asia treated with ICIs, PPI use shortened OS but had no impact on PFS (Supplemental Figure 3A and 3B).
Test of subgroup differences and meta-regression analysis
Results of our test of subgroup differences demonstrated that subgroup factors including time window of PPI use, cancer type, ICI type, ICI treatment line, and geographic region were not statistically significant factors contributing to studies’ heterogeneity, although ICI type and cancer type showed a non-significant trend as heterogeneity contributors (Figure 5). Besides the above factors, sample size and percentage of patients receiving PPIs were also analyzed in meta-regression, but no factors were identified as heterogeneity contributors (Supplemental Table 3).

Analyses of subgroup factors and test for subgroup differences. Results for (a) OS and (b) PFS are shown. The subgroups of the time window of PPI use analysis: group A, PPI therapy as baseline treatment; group B (−60, NA), PPI therapy beginning within 60 days before ICI treatment initiation; group C (0, NA), PPI therapy beginning concomitantly with initiation of ICI treatment.
Publication bias and sensitivity analysis
Funnel plots with Egger’s test indicated no publication bias affecting OS or PFS in any study (Supplemental Figure 4). Sensitivity analyses examined by the leave-one-out approach showed that no individual study significantly affected the pooled HRs of OS or PFS, indicating results were stable (Supplemental Figure 5).
Discussion
It is well-known that PPIs can alter the microbiome, 11 and it is well-known that the microbiome can affect the survival of cancer patients treated with ICIs.7,12 The mechanism of this effect on survival is still debated and is probably mediated by the effect of commensal bacteria on the crosstalk between the host immune cells and cancer cells. 50 In this report, we present new data supporting a negative correlation of PPI therapy and survival of cancer patients treated with ICIs. Our findings reveal some subtle details that may need to be considered in clinical practice when simultaneously treating cancer patients with PPIs and ICIs.
First, we observed that therapy with PPIs shortens the OS and PFS of cancer patients treated with ICIs. Our results contradict the data from some previous meta-analyses performed on this topic. In a first meta-analysis of seven studies, Li et al. 51 observed that PPIs had no impact on survival of cancer patients treated with ICI. In a subsequent meta-analysis including only five studies, Li et al. 52 found that PPI therapy had no effect on the survival of cancer patients treated with ICIs. Later, in a more recent meta-analysis, Qin et al. 53 using data from seven studies reported that PPI therapy shortened the PFS and OS of cancer patients treated with ICIs. Finally, Liu et al. 54 in a meta-analysis including 17 studies, observed that PPI therapy for cancer patients receiving ICI shortens the OS, but not PFS. Therefore, despite these meta-analysis, the impact of PPIs on ICI therapy still remains controversial. In contrast to these studies, our meta-analysis was based on the largest curated list of studies to date, comprising data from 33 studies and including articles and abstracts collected from conference proceedings. Additionally, we obtained part of the data by directly contacting authors of several studies, some of whom provided additional unpublished data. Overall, we included almost 10,000 more patients compared to the other meta-analyses on this matter. Moreover, our large list of enrolled studies mitigated the risk of publication bias. Confirming this, our publication bias analysis showed symmetrical funnel plots for OS and PFS, and the Egger’s test was not significant for bias toward PFS and borderline non-significant for bias toward OS.
Second, by including 33 different studies, we were able to perform subgroup analyses and better understand the subtle mechanisms of PPI and ICI interplay. We observed that only baseline PPI therapy and PPI therapy preceding ICI treatment shortened OS and PFS. This may be explained by previous findings showing that long-term, but not short-term, use of PPIs decreased the gut microbiome’s bacterial richness and that this decrease was negatively correlated to ICIs’ effect.11,55,56 Our observation also indirectly implies that PPIs might affect the survival of cancer patients treated with ICIs by altering the microbiome. Moreover, our observation is of clinical value; many patients receiving ICIs develop upper gastrointestinal tract symptoms and need to be treated with PPIs. 57 Our data underscore that the use of PPI therapy is acceptable if started simultaneously with ICI treatment. One question though remains to be answered: how long can PPIs and ICIs be concomitantly prescribed before PPIs start affecting survival?
Third, the subgroup analysis enabled us to observe that PPI use affected the survival of NSCLC and UC patients, but not melanoma and RCC patients, treated with ICIs. These data are partially in accordance with those of Li et al. 51 who observed in the subgroup analysis that PPI shortened the survival of NSCLC patients. In patients with NSCLC who responded to ICIs, abundant Akkermansia muciniphila and Ruminococcus sp. have been detected in fecal samples. 12 In contrast, A. muciniphila and the family of Ruminococcaceae were found to be decreased in the gut microbiome of patients treated with PPIs.58,59 Therefore, PPIs may decrease the taxa of A. muciniphila and Ruminococcaceae that are beneficial for response to ICIs in NSCLC patients. Similarly, the Ruminococcus family was also enriched in melanoma patients who responded to ICI therapy. 7 Thus, the use of PPI is believed to weaken the efficacy of ICIs in melanoma patients. However, another taxon, the Bifidobacteriaceae family, were increased after PPI treatment and were found to positively correlate with response to anti-PD-L1 treatment in melanoma patients.59,60 These findings partially explain the controversy regarding the effect of PPI use on ICI efficacy in melanoma patients and are in accordance with our finding.
Fourth, our large list of studies made it possible to compare the PPI-ICI crosstalk for different types of ICIs and for different ICI treatment regimens (first line versus non-first line). Previous meta-analyses have proven that ICIs yield more robust OS and PFS outcomes compared with chemotherapy in pretreated cancer patients, although, importantly, heterogeneity between the included studies was noted.61,62 Our analysis sheds some light on this heterogeneity. We observed that PPI shortened the survival of patients treated with anti-PD-(L)1 and of patients treated with ICIs as non-first line therapy. A few studies have shown that the efficacy of CTLA-4 blockade was influenced by certain microbiota, including Bacteroides fragilis, B. thetaiotaomicron, and Burkholderiales.63,64 So far, no correlation between these three taxa and PPI treatment has been found. Once more, we believe that these observations could have important clinical impact. A subgroup analysis performed by Li et al. 52 showed that PPI had no impact on survival in subgroups treated with different ICIs; however, detailed information about the ICI subgroups was not provided in their study. Another meta-analysis showed that the PD-L1 status of tumor cells had an important role regarding the survival benefit of ICI treatment. 61 Unfortunately, only a few of the studies included in our analysis provided detailed information regarding PD-L1 status, and therefore such an analysis was not possible.
Additionally, our study shows that a regional difference existed for the PPI and ICI interaction. One potential explanation for this observation is the complex interaction between diet and the microbiome, which has an important impact on ICIs. 65
Limitations
This meta-analysis has some important limitations. For some subgroup analyses, we were not able to include the desired number of patients, and these data should be interpreted with caution. Additionally, we would have added additional subgroup analyses, but data were not available for characteristics such as type of PPIs prescribed, patient ethnicity, patient diet, and tumor cells/immune cells PD-L1 status on immunohistochemistry. Hence, we point out that future studies on this topic should report as many clinical and demographic details as possible, including diet, ethnicity, type of PPIs, dosage of PPIs, and indication for PPIs. Furthermore, definitive answers would likely come from comparative studies reporting the composition of the microbiome during ICI treatment with versus without PPI therapy.
Conclusions
This large-cohort meta-analysis revealed a negative correlation of PPI therapy with survival in cancer patients treated with ICIs. The association was observed in several specific subgroups of cancer patients: those who had NSCLC or UC, started PPI use long before ICI, were treated with anti-PD-(L)1 agents, were treated with ICI(s) as non-first-line therapy, and resided in Europe or Australia. PPIs should be strictly controlled and appear not to impact survival if given briefly and after ICI initiation. These observations could provide the basis for future clinical guidelines for the concomitant use of PPIs and ICIs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221111703 – Supplemental material for Association of proton pump inhibitor use with survival outcomes in cancer patients treated with immune checkpoint inhibitors: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359221111703 for Association of proton pump inhibitor use with survival outcomes in cancer patients treated with immune checkpoint inhibitors: a systematic review and meta-analysis by Baoqing Chen, Chen Yang, Mihnea P. Dragomir, Dongmei Chi, Wenyan Chen, David Horst, George A. Calin and Qiaoqiao Li in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors specially thank Dr. Spakowicz (Division of Medical Oncology, The Ohio State University Medical Center), Dr. Kulkarni (University of Minnesota, Minneapolis, MN, USA), and Dr. Nguyen (Department of Medical Oncology, Kyoto University) and their teams for generously sharing with us the data, which are not available in the online version of the publications or abstracts. Editorial support was provided by Bryan Tutt, Scientific Editor, Research Medical Library, MD Anderson Cancer Center.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
