Abstract
Background:
Infiltrative hepatocellular carcinoma (HCC) is often associated with an unfavorable prognosis, posing a challenge in determining the optimal therapeutic approach. Immunotherapy, employing immune checkpoint inhibitors (ICIs), has become a preferred first-line treatment for advanced HCC. However, the overall effectiveness of ICIs in patients with infiltrative HCC remains unclear. This study aims to compare the effect of ICI treatment on clinical outcomes between patients with infiltrative and non-infiltrative HCC.
Materials and methods:
A retrospective cohort consisting of unresectable HCC patients who underwent immunotherapy with ICIs, categorized into infiltrative and non-infiltrative groups was studied. Primary outcomes comprised treatment response according to Response Evaluation Criteria in Solid Tumors (RECIST) criteria, progression-free survival (PFS), and overall survival (OS).
Results:
Of 198 patients, 60 (30.3%) had infiltrative HCC, while 138 (69.7%) had non-infiltrative HCC. In the infiltrative group, the objective response rate (ORR) was 36.7% and the disease control rate (DCR) was 55.0%. For the non-infiltrative group, the ORR was 33.3% and the DCR was 56.5%, showing no significant difference between the two groups. However, patients in the infiltrative group had significantly shorter median of PFS and OS following immunotherapy, with a PFS of 4.1 months (95% CI: 2.5–6.7; p = 0.0409) and an OS of 10.4 months (95% CI: 6.7–14.4; p = 0.0268), compared with the non-infiltrative group, which had a PFS of 5.5 months (95% CI: 3.2–7.6) and an OS of 17.0 months (95% CI: 12.8–21.8).
Conclusion:
For immunotherapy, infiltrative HCC exhibits treatment responses similar to non-infiltrative HCC. Nonetheless, infiltrative HCC is associated with shorter survival outcomes, compared with non-infiltrative type. Our findings emphasize the essential of considering type discrepancies when developing management strategies for immunotherapy.
Introduction
Patients diagnosed with advanced hepatocellular carcinoma (HCC) have an unfavorable prognosis. 1 In Taiwan, around 24.4%–28.7% of HCC patients are categorized as stage C of Barcelona Clinic Liver Cancer (BCLC) at their initial diagnosis. 2 Systemic therapy is recommended as the primary approach for these patients. Sorafenib, a tyrosine kinase inhibitor (TKI), had been previously regarded as the only agent of systemic therapy for patients with advanced HCC with a median overall survival (OS) of 10.7 months. 3 However, the development of immune checkpoint inhibitors (ICIs) has challenged this concept. The combination of ICIs, such as atezolizumab, along with anti-vascular endothelial growth factors (VEGFs; bevacizumab), is currently suggested as the first-line treatment for advanced HCC in updated guidelines.4,5 Moreover, nivolumab demonstrates similar efficacy and safety as a first-line therapy when compared to sorafenib. 6 Pembrolizumab has been proven to extend OS as a second-line therapy following sorafenib- or oxaliplatin-based chemotherapy in Asians with advanced HCC. 7
Approximately 7%–20% of advanced HCC patients exhibit an infiltrative pattern in their primary hepatic tumors. 8 Infiltrative HCC is characterized by the lack of well-defined, fibrotic margins, often leading to a blurred boundary between the tumor and the surrounding cirrhotic liver tissue, resulting in a crucial diagnosis challenge. Furthermore, it frequently appears on imaging with heterogeneous or homogeneous signal intensities and displays variable washout patterns in the portal venous or delayed phases.9,10 This indistinct boundary increases the risk of delayed diagnosis and may result in suboptimal treatment outcomes. Consequently, patients with infiltrative HCC are more likely to have larger tumor sizes, experience symptoms, and face a higher risk of malignant venous thrombosis and extra-hepatic metastasis compared to those with non-infiltrative HCC.11,12 Accordingly, infiltrative HCC is associated with poorer prognoses and poses a significant treatment challenge due to its aggressive behavior and the difficulty in achieving effective local control. 13
In contrast, non-infiltrative HCC typically presents as nodular lesions with well-defined, distinct borders that clearly separate the tumor from the surrounding liver parenchyma. 9 This morphology is more easily identifiable on imaging, facilitating a clearer distinction between tumor and healthy tissue. As a result, non-infiltrative HCC is often diagnosed earlier and is more suitable for locoregional therapies, such as surgical resection or radiofrequency ablation (RFA).
Despite the distinct clinical manifestations and outcomes between infiltrative and non-infiltrative HCC, guideline recommendations detailing the management and treatment of patients with infiltrative HCC are limited.4,5 Given that ICIs serve as first-line therapeutic agents for advanced HCC, it remains uncertain whether the therapeutic effect of ICIs is consistent among patients with varying HCC morphology. Therefore, we conducted a retrospective real-world study to compare the efficacy of ICIs on clinical outcomes between patients with infiltrative HCC and those with non-infiltrative HCC.
Materials and methods
Study designs and patients
Between November 1, 2016, and September 30, 2022, we retrospectively included patients aged 18 years or older with unresectable HCC who underwent programmed cell death protein-1 (PD-1)/programmed cell death-ligand 1 (PD-L1)-targeting immunotherapy at a tertiary referral hospital in southern Taiwan. The diagnosis of HCC was established based on pathological or imaging findings in accordance with the criteria outlined by the American Association for the Study of Liver Diseases. 5 The cutoff date for observations was set as of August 30, 2023. The study was conducted in accordance with the Declaration of Helsinki. Informed consent was waived because of the retrospective nature of anonymous clinical data use, which was granted by the Institutional Review Board of the National Cheng Kung University Hospital (B-ER-112-376). The reporting of this study conforms to the STROBE guidelines, 14 and the completed checklist is provided as Supplemental Table 1.
The exclusion criteria encompassed patients diagnosed with combined HCC and cholangiocarcinoma, individuals who received only a single dose of ICI, those lacking follow-up imaging post-immunotherapy, participants enrolled in clinical trials, individuals treated with ICI solely for extra-hepatic metastatic lesions without measurable hepatic lesions, those with ruptured HCC hindering morphology interpretation, and those with early discontinuation of follow-up within 3 months of treatment initiation who also lacked sufficient imaging data prior to the observation cutoff date. Among eligible patients, experienced radiologists individually analyzed the most recent images obtained through computed tomography (CT) or magnetic resonance imaging (MRI) before ICI initiation for HCC image interpretation. Patients with HCC exhibiting infiltrative features were classified into the infiltrative group, while those without such features were assigned to the non-infiltrative group. In addition, comprehensive information on patient demographics, clinical characteristics, treatment modalities, and outcomes was systematically recorded.
Chronic hepatitis B was defined as the presence of hepatitis B surface antigen for more than 6 months, regardless of the presence or absence of detectable Hepatitis B Virus (HBV) DNA. 15 Chronic hepatitis C was diagnosed by the detection of hepatitis C virus RNA or anti-Hepatitis C Virus (HCV) antibodies, persisting for more than 6 months. 16 Alcoholic hepatitis was diagnosed in individuals with a history of daily alcohol consumption exceeding 40 g for females and 60 g for males over more than 6 months, accompanied by clinical or histological evidence of hepatitis. 17 Nonalcoholic steatohepatitis was diagnosed in patients without significant alcohol consumption, based on imaging findings and liver biopsy showing steatosis along with hepatocyte ballooning degeneration and lobular inflammation. 18
Tumor morphology
Patients diagnosed with unresectable HCC were classified into the infiltrative group through Eggel’s classification, the Liver Cancer Study Group of Japan classification, and by considering the following characteristics, as previously outlined9–11,19–21: (i) The absence of well-demarcated, fibrotic margins, resulting in blending into the background of the cirrhotic liver, and/or (ii) manifestation of heterogeneous or homogeneous abnormal signal intensity, accompanied by a variable washout appearance on portal venous or delayed phases, and/or (iii) often, though not necessarily, accompanied by portal venous thrombosis. Each cross-sectional image was blinded and individually reviewed by hepatologists (HY Kuo and TT Chang) as well as radiologist (YS Liu) to determine the tumor morphology.
Assessment of treatment efficacies and adverse events
This study examined the utilization of ICIs, specifically nivolumab, nivolumab plus ipilimumab, pembrolizumab, and atezolizumab plus bevacizumab. Nivolumab and pembrolizumab were administered intravenously at doses of 3 mg/kg every 2 weeks and 100–200 mg every 3 weeks, respectively. Atezolizumab and bevacizumab were administered intravenously at doses of 1200 mg and 15 mg/kg every 3 weeks, respectively. Ipilimumab was prescribed in combination with nivolumab at a dose of 3 mg/kg for a maximum of four doses.
MRI or tri-phase CT scans were performed at intervals of 8–12 weeks. Treatment responses were evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 and modified RECIST (mRECIST) criteria, classifying eligible individuals into categories such as complete response (CR), partial response (PR), stable disease, or progressive disease (PD).22,23 Information on adverse events (AEs) was methodically gathered and evaluated according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events (version 5.0). 24
Study outcomes
The primary study outcomes included the objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), and OS. ORR was defined as the proportion of patients achieving either CR or partial response (PR) as their best overall response, while the DCR indicated the proportion of patients with at least CR, PR, or stable disease as their best overall response. OS was calculated as the period from the initiation of immunotherapy treatment until the patient’s death from any cause. PFS was assessed as the period from the commencement of treatment to the occurrence of tumor progression or all-cause mortality, whichever occurred first. The Kaplan–Meier plots for PFS and OS were truncated at 36 months for the overall population and at 42 months for patients who achieved an objective response. This adjustment ensures that the number of patients analyzed does not drop below one-tenth of the total enrolled population, thereby maintaining the reliability of the survival curves and minimizing potential bias from a small sample size over longer periods.
Secondary outcomes focused on the incidence and severity of AEs. Safety assessments included the comprehensive documentation of AEs, clinical laboratory tests, including hematologic and biochemical analyses, and physical examinations.
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences for Windows (version 25.0; Chicago, IL, USA). Baseline patient characteristics are presented as the median (interquartile range, IQR) or number (percentage). Differences in variables between infiltrative HCC and non-infiltrative HCC were assessed using the Chi-square test or paired sample t test.
Survival outcomes, including OS and PFS, were evaluated using the Kaplan–Meier method and the median along with 95% confidence intervals (CIs) were determined using Cox regression for adjusting potential confounders. Variables with a p value <0.2 recognized from the univariate analyses were selected for inclusion in the multivariable analyses. For the multivariate analysis to identify prognostic factors of OS, variables selected for the univariate analysis included tumor morphology, Eastern Cooperative Oncology Group (ECOG) performance status, 25 Child-Pugh class, BCLC stage, Cancer of the Liver Italian Program (CLIP) score, presence of macrovascular invasion (MVI), and the line of immunotherapy as a systemic therapy. For PFS, the selected variables were tumor morphology, gender, ECOG performance status, Child-Pugh class, CLIP score, and the line of immunotherapy as a systemic therapy. Further subgroup analyses of survival outcomes were conducted, with the results presented in a forest plot to illustrate the adjusted hazard ratios (HRs) for each variable.
Therapeutic efficacies, as indicated by ORR and DCR, were compared among patients with different characteristics using Fisher’s exact test. All statistical tests were two-tailed, and significance was defined as a p value of <0.05.
Results
Differences in baseline characteristics between the infiltrative and non-infiltrative groups
Out of the initially included 356 patients, 198 individuals with unresectable HCC were deemed eligible, showcasing a prevalence of the non-infiltrative type (138 patients, 69.7%), as outlined by the exclusion criteria (Figure 1). Baseline characteristics between the infiltrative and non-infiltrative groups are presented in Table 1. The infiltrative group exhibited a significantly higher percentage of patients with an advanced tumor stage or MVI and a larger size of primary HCC. For patients with infiltrative-type HCC, 60 cases lacked well-demarcated, fibrotic margins. In addition, 54 of these cases simultaneously exhibited uneven signal intensity. The leading etiology of HCC are chronic hepatitis B (140 patients, 70.7%), followed by chronic hepatitis C (50, 25.3%), alcoholic hepatitis (14, 7.1%), nonalcoholic steatohepatitis (6, 3.0%), and unknown etiology (9, 4.5%).
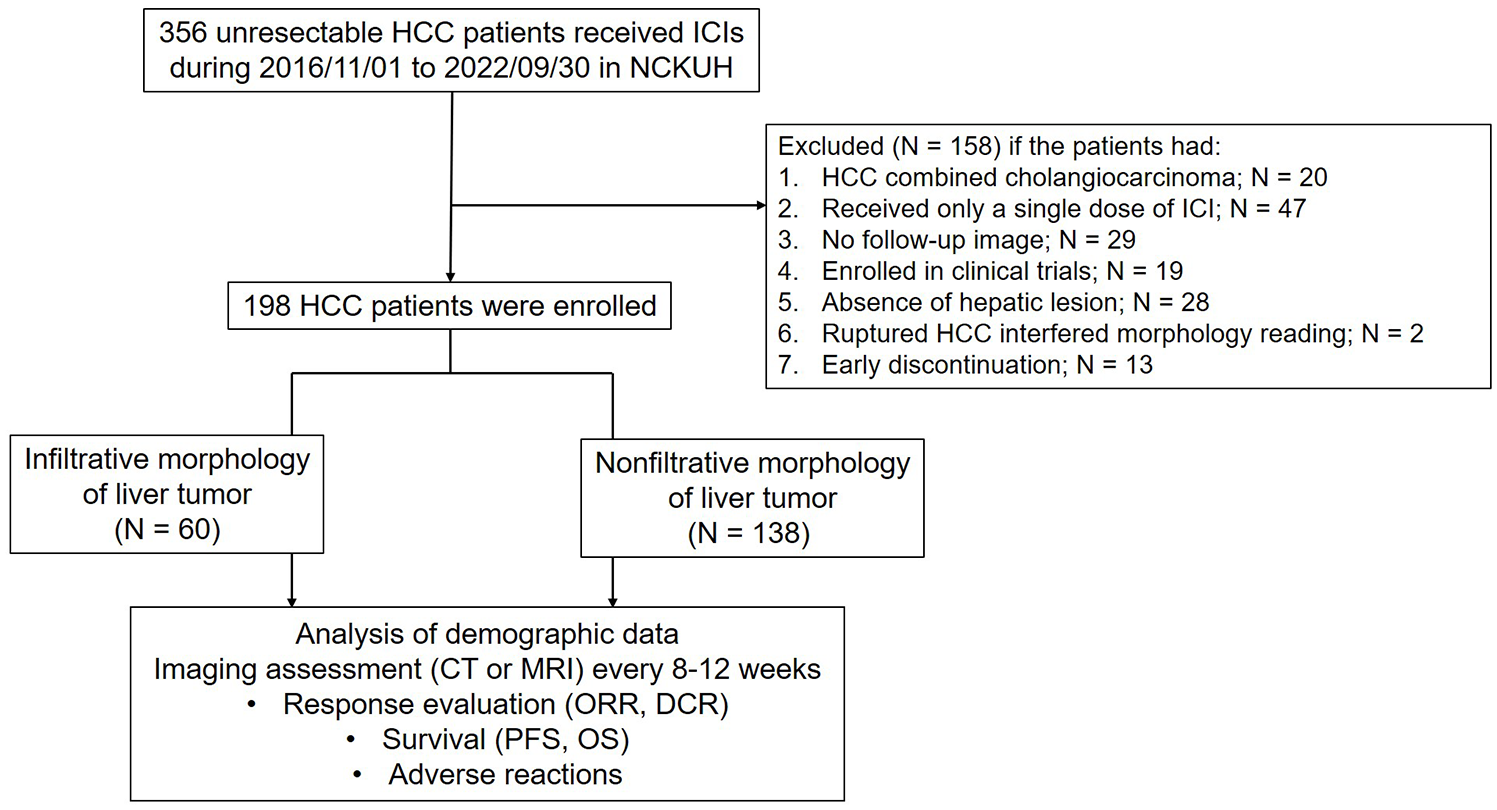
Flowchart of patient selection.
Baseline characteristics between infiltrative and non-infiltrative HCC patients who received immune checkpoint inhibitors.

Patients classified under “unknown etiology” are those without chronic viral hepatitis or other known liver diseases (e.g., hemochromatosis, Wilson disease, autoimmune hepatitis) but who may have other risk factors for HCC, such as environmental exposures, metabolic liver disease, or genetic predispositions.
Modified albumin-bilirubin grade and serum albumin were not available in six patients due to missing albumin data. The modified ALBI (Albumin-Bilirubin) grade was calculated using the following formula: ALBI score = (log10 bilirubin (μmol/L) × 0.66) + (albumin (g/L) × (−0.085)). Based on this score, patients were categorized into three grades: Grade 1 (ALBI score ⩽ −2.60), Grade 2 (ALBI score between −2.60 and −1.39), and Grade 3 (ALBI score ⩾ −1.39). Grade 2 was further subdivided into two subgrades, 2a and 2b, using a cutoff value of −2.270. The four resulting ALBI grades were referred to as mALBI grades.26,27
BCLC, Barcelona Clinic Liver Cancer; CLIP, Cancer of the Liver Italian Program; 5-FU, fluorouracil; HAIC, hepatic arterial infusion chemotherapy; HCC, hepatocellular carcinoma; IQR, interquartile range; IVC, inferior vena cava; MVI, macrovascular invasion; PEI, percutaneous ethanol injection; PV, portal vein; RFA, radiofrequency ablation; TACE, transarterial chemoembolization; TKI, tyrosine kinase inhibitor.
As shown in Table 1, systemic chemotherapy was administered to a small subset of patients. Specifically, 1.7% of infiltrative HCC patients and 1.5% of non-infiltrative HCC patients received systemic chemotherapy. The regimens included fluorouracil (2000 mg/m2) combined with oxaliplatin (85 mg/m2), which was given to one infiltrative and one non-infiltrative patient. Additionally, gemcitabine (800 mg/m2) combined with cisplatin (25 mg/m2) was administered to one non-infiltrative patient. Previous TKI therapy was defined as the administration of TKIs, such as sorafenib or lenvatinib, prior to the initiation of ICIs. The median dosages of sorafenib and lenvatinib administered orally were 400 mg (IQR, 400–800 mg) per day and 8 mg (IQR, 4–10 mg) per day, respectively, in the infiltrative group, and 700 mg (IQR, 400–800 mg) per day and 12 mg (IQR, 9–12 mg) per day, respectively, in the non-infiltrative group. In the infiltrative group, the median duration of sorafenib therapy was 98 days (IQR, 42–157 days), compared to 130 days (IQR, 63–245 days) in the non-infiltrative group. For lenvatinib therapy, the median duration was 14 days (IQR, 12–43 days) in the infiltrative group and 130 days (IQR, 65–239 days) in the non-infiltrative group. Additionally, one patient in the infiltrative group was treated with regorafenib at a dosage of 80 mg, with a treatment duration of 277 days. Among the sixteen infiltrative HCC cases that had received previous transarterial chemoembolization (TACE) treatment, 14 initially presented with nodular tumors, later progressing to an infiltrative pattern. The remaining two cases displayed an infiltrative morphology from the outset but were treated with TACE due to tumor rupture. Similarly, of the 12 infiltrative HCC cases treated with RFA, all had nodular-type HCC initially, which evolved into an infiltrative pattern over time.
Combination therapy involves the concurrent administration of ICIs alongside TKIs. In the infiltrative group, the median daily dosages were sorafenib 400 mg (IQR, 400–700 mg), lenvatinib 8 mg (IQR, 5–12 mg), and regorafenib 160 mg (IQR, 80–160 mg). In the non-infiltrative group, the median daily dosages were sorafenib 400 mg (IQR, 400–500 mg), lenvatinib 10 mg (IQR, 8–12 mg), and regorafenib 80 mg (IQR, 80–80 mg). In the infiltrative group, the median duration of sorafenib combination therapy was 121 days (IQR, 24–248 days), compared to 69 days (IQR, 45–174 days) in the non-infiltrative group. For lenvatinib combination therapy, the median duration was 100 days (IQR, 62–203 days) in the infiltrative group and 73 days (IQR, 42–153 days) in the non-infiltrative group. When combined with regorafenib, the median duration was 116 days (IQR, 69–154 days) in the infiltrative group and 176 days (IQR, 133–258 days) in the non-infiltrative group (Supplemental Table 2).
The median follow-up period was 12.8 months (IQR, 5.8–22.5). The categories of ICIs prescribed for patients in the infiltrative group and non-infiltrative group were similar (Supplemental Table 3).
Differences in the treatment response between the infiltrative and non-infiltrative groups
Table 2 illustrates the treatment responses to immunotherapy based on RECIST and mRECIST criteria. In total, 60 patients in the infiltrative group and 138 in the non-infiltrative group, each with at least one follow-up image, were assessable for evaluating treatment response. No statistically significant difference in treatment response was observed between the two groups. According to RECIST, 22 (36.7%) and 46 (33.3%) patients in the infiltrative and non-infiltrative groups, respectively, achieved a similar ORR (p = 0.771). Likewise, 33 (55.0%) and 78 (56.5%) patients in the infiltrative and non-infiltrative groups, respectively, achieved a comparable DCR (p = 0.966). Subsequent subgroup analysis was conducted based on the Child-Pugh class, the line of systemic therapy, and the use of combined therapy with or without TKI. The ORR and DCR did not significantly differ between the infiltrative and non-infiltrative groups across all subgroups (Supplemental Tables 4–6).
Treatment response to immunotherapy among infiltrative and non-infiltrative HCC patients.
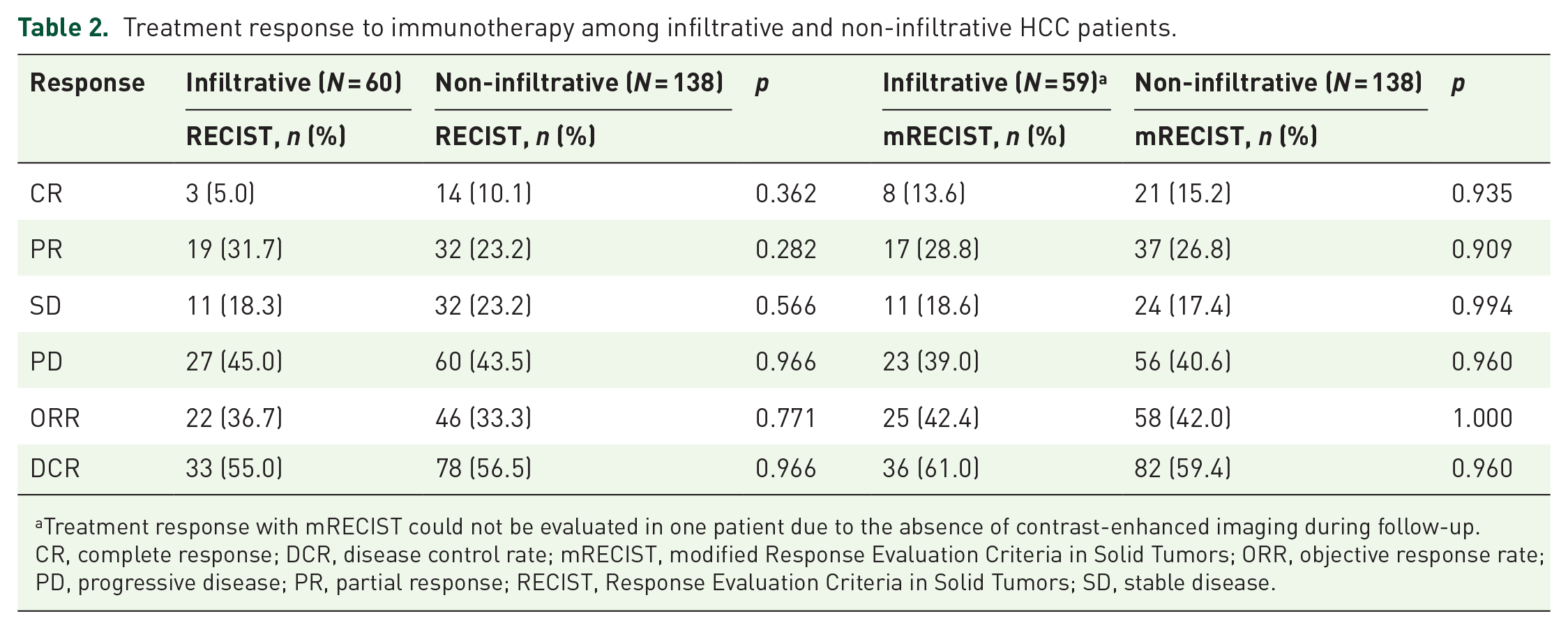
Treatment response with mRECIST could not be evaluated in one patient due to the absence of contrast-enhanced imaging during follow-up.
CR, complete response; DCR, disease control rate; mRECIST, modified Response Evaluation Criteria in Solid Tumors; ORR, objective response rate; PD, progressive disease; PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors; SD, stable disease.
A subgroup analysis of responders and non-responders within the infiltrative HCC cohort treated with ICIs identified significant differences in baseline characteristics (Supplemental Table 7). Responders demonstrated better ECOG performance status (p = 0.013), lower CLIP scores (p = 0.018), and were more likely to have received prior therapies, such as surgery (p = 0.003) or RFA/percutaneous ethanol injection (PEI) (p = 0.022), compared to non-responders. These findings suggest that patients with favorable performance status, lower disease severity, and prior curative interventions may experience a more favorable therapeutic response to immunotherapy.
At the conclusion of the follow-up period, 4 patients with infiltrative HCC (6.7%) and 17 (11.6%) with non-infiltrative HCC continued ICI treatment (p = 0.509; Supplemental Table 8). In the infiltrative HCC group, local therapies—including TACE, RFA, and surgical resection—were selectively utilized to control symptoms and potentially enhance the efficacy of subsequent treatments. Patients receiving these therapies exhibited varied clinical characteristics, including differences in tumor size, lesion number, and the presence of MVI. These factors, alongside physician assessment, guided the choice of treatment to optimize outcomes. Specifically, TACE was administered in four cases: three cases after significant tumor reduction post-immunotherapy to provide localized control and prevent disease progression, and one case based on physician preference. RFA and surgical resection were considered for cases in which successful downstaging enabled resection, offering a potential curative pathway. In the non-infiltrative HCC group, a similarly tailored approach was applied, guided by lesion characteristics and patient health profiles: TACE was performed in seven cases with varying objectives: one case following downstaging, one following tumor reduction post-immunotherapy, one as an emergency intervention due to HCC rupture, and four cases based on physician preference. Surgical resection was conducted in 11 cases with specific objectives: 5 cases after downstaging to a resectable status, 4 cases following significant tumor reduction post-immunotherapy, and 2 cases based on physician preference. RFA was administered in five cases: two following successful downstaging and three based on physician preference (Supplemental Table 9).
Survival outcomes in the infiltrative and non-infiltrative groups
As shown in Figure 2, the median PFS of the infiltrative and non-infiltrative groups was 4.1 (95% CI: 2.5–6.7) and 5.5 (95% CI: 3.2–7.6) months, respectively (p = 0.0409; Figure 2(a)). The median OS of the infiltrative HCC and non-infiltrative HCC groups was 10.4 (95% CI: 6.7–14.4) and 17.0 (95% CI: 12.8–21.8) months, respectively (p = 0.0268; Figure 2(b)). The median PFS was 14.5 (95% CI: 8.2–21.0) and 26.8 (95% CI: 17.3–not estimable) months in the infiltrative and non-infiltrative groups exhibited objective response, respectively (p = 0.0032; Figure 3(a)). The median OS was 26.1 (95% CI: 16.9–41.4) and 62.9 (95% CI: 30.2–not estimable) months in the infiltrative and non-infiltrative groups achieved objective response, respectively (p = 0.0499; Figure 3(b)).

Kaplan–Meier curves for (a) progression-free survival and (b) overall survival between infiltrative and non-infiltrative HCC patients.

Kaplan–Meier curves for (a) progression-free survival and (b) overall survival between infiltrative and non-infiltrative HCC patients who achieved objective responses after immunotherapy.
In the Cox-regression models, the adjusted HR for infiltrative HCC in contrast to non-infiltrative patients was 1.39 (95% CI: 1.01–1.93; p = 0.045) for PFS and 1.55 (95% CI: 1.05–2.29; p = 0.028) for OS, as presented in Table 3.
Multivariable analysis for progression-free survival and overall survival.

Variables included in the multivariable analysis were selected based on a p value threshold of <0.2 in the univariate analysis to ensure the inclusion of potentially relevant predictors.
BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; CLIP, Cancer of the Liver Italian Program; ECOG, Eastern Cooperative Oncology Group; HR, Hazard ratio; MVI, macrovascular invasion; OS, overall survival; PFS, progression-free survival; Ref., reference; TKI, tyrosine kinase inhibitor.
Subgroup analyses for survival outcomes in the infiltrative and non-infiltrative groups
In the subgroup analysis, patients in the infiltrative group with immunotherapy as the initial systemic therapy, combination therapy with TKIs, advanced BCLC stage, the presence of MVI, and tumors meeting up-to-seven criteria 28 exhibited significantly poorer PFS compared to those in the non-infiltrative group (Figure 4(a)). Additionally, patients in the infiltrative group with Child-Turcotte-Pugh class A, immunotherapy as the initial systemic therapy, and tumors meeting up-to-seven criteria experienced significantly shorter OS compared to those in the non-infiltrative group (Figure 4(b)).

Adjusted HRs of progression-free survival (a) and overall survival (b) for patients with infiltrative HCC, compared with those with non-infiltrative HCC. Squares represent subgroup-specific pooled HR. Horizontal lines indicate a 95% CI.
AEs between the Infiltrative and Non-infiltrative Groups
The common treatment-related AEs between the infiltrative and non-infiltrative groups included skin rash, hepatitis, diarrhea, fatigue, weakness, and thyroid dysfunction. The incidence of treatment-related AEs between the infiltrative and non-infiltrative groups was not significant (p = 0.108). Overall, 27 (45.0%) and 44 (31.9%) patients experienced AEs in the infiltrative and non-infiltrative groups, respectively. In the infiltrative group, 2 (7.4%) patients experienced grade 3 or 4 AEs, compared to 10 (22.7%) in the non-infiltrative group. The difference in the incidence of these AEs was not significant (p = 0.115; Supplemental Table 10).
Discussion
To the best of our knowledge, this study represents the inaugural exploration into the correlation between HCC morphology and patient outcomes following immunotherapy within a real-world context. Notably, individuals with infiltrative HCC, upon receiving immunotherapy, exhibited a therapeutic response akin to those with non-infiltrative HCC. Consequently, regardless of infiltrative status, prioritizing immunotherapy is warranted, and efforts should be made to prevent treatment delays attributed to the diagnostic challenges associated with infiltrative HCC. Crucially, diminished survival outcomes were observed in patients with infiltrative HCC in comparison to their non-infiltrative counterparts following immunotherapy. To substantiate these findings and assess the effectiveness of immunotherapy in conjunction with other strategies, additional prospective studies are imperative. These investigations will not only validate the current study’s results but also contribute to a more comprehensive understanding of the efficacy of immunotherapy in cases where HCC manifests as infiltrative-type morphology.
Given the poorer survival outcomes in infiltrative HCC, patients with this subtype may benefit from more intensive management, such as frequent imaging surveillance to detect early progression and potentially improve therapeutic outcomes. 9 Regular monitoring of tumor markers, namely alpha-fetoprotein (AFP) and protein induced by vitamin K absence or antagonist-II (PIVKA-II), along with imaging, can assist in the timely detection of recurrence, enabling earlier intervention and effective multidisciplinary management. 29
Systemic therapy combinations, particularly PD-1/PD-L1 inhibitors with multikinase inhibitors, VEGF, or cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) inhibitors, have demonstrated efficacies in HCC treatment. 30 While specific data on differential responses among HCC subtypes is limited, these combinations may provide the most reasonable approach for aggressive infiltrative HCC. Notably, the STRIDE regimen (tremelimumab and durvalumab) has shown significant survival benefits for unresectable HCC, suggesting its potential in managing infiltrative HCC, where aggressive disease behavior may require a more comprehensive treatment approach. 31 Conversely, non-infiltrative HCC, characterized by distinct tumor borders and generally smaller sizes, may allow better local control through resection or ablation. This subtype may require less frequent systemic therapy, as local treatments might effectively manage the disease. Thus, early administration of immunotherapy may provide favorable outcomes in infiltrative HCC, given its aggressive nature. Further research is warranted to refine management strategies tailored to specific HCC phenotypes. 32
The poorer OS and PFS demonstrated in infiltrative HCC, despite comparable ORRs to ICIs, may stem from the aggressive and diffuse characteristics of these tumors. Infiltrative HCC often presents with larger tumors, MVI, and a greater tendency for extra-hepatic spread, all factors that accelerate disease progression and restrict therapeutic options. 9 The extensive tumor burden and diffuse nodular spread throughout the liver frequently limit curative treatment options, even when PRs or CRs to immunotherapy are achieved. Moreover, patients with non-infiltrative HCC may be better candidates for curative treatments following immunotherapy, such as surgical resection or RFA, as opposed to infiltrative HCC patients who frequently have poorly defined tumor borders. 10 Limited access to potentially curative treatments following ICI therapy likely further exacerbates the survival disparity observed in infiltrative HCC cases.
In addition, infiltrative HCC may harbor unique molecular and genetic alterations that drive its aggressive behavior and treatment resistance. Studies such as those by Yan et al., utilizing whole-exome sequencing, have revealed distinct fibroblast growth factor receptor 3 (FGFR3) mutations in infiltrative HCC compared to non-infiltrative types, with FGFR3 overexpression associated with monocyte chemotactic protein 1-mediated angiogenesis and tumor invasiveness. 33 These FGFR3 mutations may play a significant role in promoting the invasive phenotype and poorer survival outcomes. Thus, the inferior survival outcomes in infiltrative HCC, even with initial treatment response, may result from dysregulated genetic signatures. This complexity underscores the need for further research to clarify the mechanisms driving poorer survival.
While prevailing guidelines have faced challenges in furnishing definitive recommendations, particularly concerning the management of infiltrative HCC, several retrospective studies have provided insightful findings, albeit constrained by limited sample sizes.12,34–37 Previous research consistently showcased that locoregional therapies such as TACE and transarterial radioembolization (TARE), along with systemic treatments such as sorafenib, conferred survival benefits upon patients with infiltrative HCC compared to those receiving best supportive care alone.12,34,35 An investigation by An et al. disclosed that hepatic arterial infusion chemotherapy (HAIC) yielded longer PFS and OS than TACE in a cohort of 160 treatment-naïve patients with advanced-stage infiltrative HCC, specifically those classified as Child-Turcotte-Pugh class A. 36 Similarly, Kim et al.’s analysis suggested that sorafenib correlated with superior survival outcomes compared to alternative modalities (TACE, radiotherapy, chemotherapy) in a study involving 190 patients with advanced-stage infiltrative HCC. 37 Nevertheless, reports on survival outcomes for infiltrative HCC patients undergoing systemic therapy have been infrequent and applicable only to specific patient subgroups. Chuma et al.’s study, encompassing patients with large tumor sizes (occupying more than 50% of the liver volume) or main portal venous thrombosis, indicated unsatisfactory outcomes for patients with infiltrative HCC following lenvatinib treatment, with a median PFS of 2.8 months and a median OS of 6.7 months. 38 In line with this, our study revealed that patients with infiltrative HCC undergoing immunotherapy exhibited a median PFS of 4.1 months and a median OS of 10.4 months. In addition, we observed comparable treatment responses between infiltrative and non-infiltrative groups. The findings from our study suggest that immunotherapy, given the therapeutic challenges posed by infiltrative tumor morphology, may represent a valuable treatment option.
The efficacy of diverse therapeutic modalities concerning tumor morphology remains inconclusive. When assessing the effectiveness of varied therapies in patients with infiltrative HCC versus non-infiltrative HCC, Kneuertz et al. reported no significant difference in survival between the two groups following intra-arterial treatments (TACE and TARE). 11 Conversely, Chuma et al. demonstrated that among patients with large tumor sizes or main portal venous thrombosis treated with lenvatinib, those with infiltrative HCC experienced significantly shorter PFS and OS compared to their non-infiltrative counterparts. 38 In line with our findings, patients with infiltrative HCC exhibited shorter PFS and OS when compared to those with non-infiltrative HCC following immunotherapy. Notably, patients with infiltrative HCC achieving an objective response after immunotherapy led to a remarkable improvement in median PFS and OS, with a median PFS of 14.5 months and a median OS of 26.1 months. Our study provides initial insights into the effectiveness of immunotherapy for patients with infiltrative HCC and compares treatment efficacy between the infiltrative HCC and non-infiltrative groups. However, future longitudinal and large-cohort studies will be essential to validate the present findings.
In our subgroup analysis, the infiltrative HCC group displayed inferior survival outcomes in patients with advanced BCLC stage, the presence of MVI, and tumors meeting criteria beyond seven. This aligns with the findings of Chuma et al., who noted significantly shorter PFS in patients with tumors occupying more than 50% of the liver volume and treated with lenvatinib compared to those with non-infiltrative HCC. 38 These factors denote an advanced stage and widespread cancer, resulting in unfavorable survival outcomes that may outweigh the potential benefits of combining immunotherapy with infiltrative HCC. Despite its limitations, this study offers valuable real-world insights and lays the groundwork for more targeted and effective treatments in the future.
Our subgroup analysis provides valuable insights into potential predictors of treatment response in infiltrative HCC patients undergoing immunotherapy. The significant differences observed between responders and nonresponders—in terms of ECOG performance status, CLIP score, and prior treatments such as surgery or RFA/PEI—suggest that patients with better health status, lower tumor burden, and history of curative therapies may derive greater benefit from ICIs. A detailed review of patient records revealed that among the 16 cases of infiltrative HCC that responded to ICIs and had a history of surgery, RFA, or PEI, 93.7% (15/16) initially presented with nodular tumors (ranging from single to multinodular patterns), which later progressed to an infiltrative morphology. This nodular pattern enabled these patients to undergo curative therapies at that time. Conversely, among nonresponders, 85.7% (36/42) had received prior treatments such as TACE, hepatic HAIC, TKIs, or systemic chemotherapy, with 63.8% (23/36) initially presenting with infiltrative-type tumors and only 36.1% (13/36) with nodular patterns. These findings suggest that in infiltrative HCC cases, patients whose liver tumor morphology was initially nodular before progressing to an infiltrative type may respond better to immunotherapy once the disease progresses to an infiltrative stage. These factors may offer preliminary guidance for clinicians in identifying patients who are more likely to benefit from immunotherapy, supporting a more personalized treatment approach for infiltrative HCC. Prospective studies are warranted to validate these predictive factors and explore the mechanisms influencing therapeutic outcomes.
Infiltrative HCC is widely regarded as resistant to local therapies such as TACE and RFA due to its diffuse growth pattern, poorly defined tumor margins, and frequent vascular invasion. Nevertheless, in this study, certain patients with infiltrative HCC had received local therapies prior to initiating immunotherapy. This decision was influenced by various clinical considerations, including symptom management, control of tumor burden, patient-specific tolerance, and treatment accessibility at the time. Furthermore, under Taiwan’s health insurance criteria, patients with BCLC B stage HCC are required to undergo a minimum of three unsuccessful local therapy rounds before becoming eligible for immunotherapy.
A detailed review of patient records revealed that among the 16 infiltrative HCC cases that had previously undergone TACE, 14 initially presented with nodular tumors (ranging from single to multinodular patterns) that progressed to an infiltrative morphology over time. The remaining two cases had an infiltrative morphology from the beginning but received TACE due to tumor rupture. Likewise, among the 12 infiltrative HCC cases treated with RFA, all initially presented as nodular-type HCC, later progressing to an infiltrative form. These findings illustrate the evolving morphology of HCC and underscore the clinical rationale for using local therapies to manage symptoms and tumor burden in the initial stages of HCC, even when tumors later transition to an infiltrative form.
This study presents several noteworthy limitations. The patient numbers in both the infiltrative and non-infiltrative groups were suboptimal, potentially compromising the statistical power of the subgroup analysis due to small sample sizes. The retrospective nature of the study introduces variability in diagnostic, treatment, and follow-up strategies, lacking standardization, and necessitating consideration of potential selection bias. This study also highlights that complex treatment histories may affect liver function and ICI effectiveness. Variability in therapeutic strategies, including repeated sessions of TACE or RFA, can influence the hepatic microenvironment and potentially impact ICI efficacy. These observations underscore the importance of considering previous treatment history and employing adaptive therapy strategies when managing HCC cases prior to immunotherapy. The diversity in prescription preferences for ICI, cross-over therapeutic strategies, and treatment sequences post-ICI further complicates the comparability of results. In addition, the absence of a universal definition for infiltrative HCC introduces potential information bias in the interpretation of morphology and measurement of tumor size. Patients with infiltrative HCC exhibited more advanced disease stage and vascular invasion at baseline, raising concerns about confounding factors. To address this, we employed the Cox regression model to minimize the potential effects of these confounders.
Conclusion
Our study highlights the significance of tumor morphology in the management of patients with unresectable HCC, underscoring the potential benefits of immunotherapy in the context of infiltrative HCC. In our ongoing efforts to enhance patient outcomes, there is a clear need for additional prospective studies. These studies are essential for gaining a deeper understanding of the intricacies associated with infiltrative HCC and for identifying the most efficacious treatment strategies tailored to this challenging clinical subset.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241312141 – Supplemental material for Comparing immunotherapy effectiveness for unresectable hepatocellular carcinoma: infiltrative versus non-infiltrative types in real-world settings
Supplemental material, sj-docx-1-tam-10.1177_17588359241312141 for Comparing immunotherapy effectiveness for unresectable hepatocellular carcinoma: infiltrative versus non-infiltrative types in real-world settings by Chien-Ming Chiang, Kuan-Kai Huang, Chun-Te Lee, Tzu-Chun Hong, Juei-Seng Wu, Hung-Tsung Wu, Ting-Tsung Chang, Yi-Sheng Liu, Wei-Ting Chen, Chung-Teng Wang, Chen Chang, Po-Jun Chen, Ming-Tsung Hsieh, Chiung-Yu Chen, Chiao-Hsiung Chuang, Ching-Chi Lee, Sheng-Hsiang Lin, Yih-Jyh Lin and Hsin-Yu Kuo in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank Prof. Sheng-Hsiang Lin and Chih-Hui Hsu from the Biostatistics Consulting Center, National Cheng Kung University Hospital, for providing statistical consulting services. We also thank Dr. Ching-Chi Lee in the Clinical Medicine Research Center, National Cheng Kung University Hospital, for the assistance in providing insightful comments regarding manuscript writing.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
