Abstract
BACKGROUND:
Foxhead box D1 (FOXD1) is validated to be over-expressed in a variety of human malignancies and promotes cancer progression. Nevertheless, the role of FOXD1 and the associated mechanism in nasopharyngeal carcinoma (NPC) remain largely unknown.
METHODS:
A total of seventy-five cases of NPC tissue samples were collected. FOXD1 expression in NPC tissues and cells (SUNE1, CNE1, CNE2, and HONE1) was detected using immunohistochemistry and Western blot, respectively. The relationship between FOXD1 expression and clinicopathological parameters of NPC patients was analyzed. FOXD1 mRNA and miR-186 expression in NPC tissues and cells was detected using quantitative polymerase chain reaction (qPCR). The cell viability of NPC cells was detected using CCK-8 assay. Colony survival of NPC cells exposed to different doses of radiation was detected using colony formation assay. Transwell assay was used to evaluate the migration and invasion of NPC cells. The dual-luciferase reporter gene assay was employed to verify the targeting relationship between miR-186 and FOXD1.
RESULTS:
FOXD1 was over-expressed in NPC tissues (average fold change on mRNA level
CONCLUSION:
FOXD1, which is negatively regulated by miR-186, acts as a novel oncogene in NPC and serves as potential biomarker and therapeutic target for NPC. The research will provide great theoretical basis for further clinical diagnosis and therapy.
Introduction
Nasopharyngeal carcinoma (NPC) is an epithelial malignancy with unique geographical distribution, which frequently occurs in East Asia and Southeast Asia, with an annual morbidity of approximate 30 cases per 100,000 people [1, 2]. Unfortunately, most of NPC patients were diagnosed with stage III or IV, accompanied by a five-year survival rate of 50%–60% [3]. Radiotherapy is an essential treatment for NPC. Nonetheless, radio-resistance is still the major cause of treatment failure as well as the occurrence of distant metastasis of cancer cells [4]. Hence, it is of great importance to further probe the underlying molecular mechanisms for NPC progression.
Forkhead box D1 (FOXD1) is a member of the FOX transcriptional factors family, located on chromosome 5q12 [5]. Recent studies have shown that FOXD1 is crucial to the tumorigenesis, tumor progression and metastasis in a variety of human cancers. For instance, FOXD1 facilitates colorectal cancer progression by activating the ERK1/2 signaling pathway; and its over-expression is significantly related to the poor prognosis of colorectal cancer patients [6]. In non-small cell lung cancer, FOXD1 presented oncogenic characteristics through activating Vimentin; furthermore, it was an independent prognostic factor for overall survival (OS) and disease-free survival (DFS) of the patients [7]. In breast cancer, FOXD1 inhibits p27 and thus promotes cancer progression [8]. Additionally, FOXD1 has been reported to promote the progression of cervical cancer, glioma, and osteosarcoma [9, 10, 11]. Nevertheless, the role of FOX transcriptional factor family, including FOXD1 in NPC has never been investigated before.
MicroRNAs (miRNAs) are non-coding, highly conserved small RNAs containing 19–25 nucleotides that bind with the 3’ untranslated regions (3’ UTR) of the target mRNA by base pairing to regulate the expression of human genes [12]. miRNAs participate in almost all physiological and pathological processes, including cancer progression [13, 14]. miRNAs act as tumor suppressors or oncogenic factors involved in the regulation of tumor cell proliferation, migration, metastasis and radio-sensitivity [15, 16]. It has been reported that Upregulation of miR-186-5p inhibits cell viability and promotes cell apoptosis and autophagy in hepatocellular carcinoma [17]. miR-186 inhibits hepatocellular carcinoma cell proliferation and metastasis by targeting MCRS1-mediated Wnt/
In this article, we found that FOXD1 was over-expressed in NPC tissues and its high expression was remarkably linked to lymph node metastasis of cancer cells and low degree of tissue differentiation. Additionally, the expression of FOXD1 is negatively modulated by miR-186, thus promoting NPC cell proliferation, migration, invasion, and radio-resistance. This result promises to supply potential novel therapeutic targets for NPC treatments in the future.
Materials and methods
Clinical data and ethical statements
The study was approved by the Clinical Research Ethics Committee of Luoyang Central Hospital Affiliated to Zhengzhou University. Written informed consent was acquired from each patient before the study. Seventy-five pairs of NPC tissues/non-cancerous tissues were obtained from patients who were subjected to nasopharyngeal biopsy in our hospital. None of the NPC patients enrolled in the study had a history of radiotherapy or chemotherapy previously.
Cell line and cell culture
Human normal nasopharyngeal epithelial cell line (NP69) and NPC cell lines (SUNE1, CNE1, CNE2, and HONE1) were available from China Center for Type Culture Collection (CCTCC, Wuhan, China). The above cell lines were cultured in Dulbeccos Modified Eagles medium/Hams Nutrient Mixture F12 (DMEM/F12, Gibco, Grand Island, NY, USA) containing 10% fetal bovine serum (FBS, Thermo Fisher Scientific, Shanghai, China), 100 U/mL penicillin and 100 U/mL streptomycin (Sigma, St. Louis, MO, USA).
Cell transfection
FOXD1 over-expressed plasmid, empty vector plasmid, siRNA negative control (si-NC), siRNA targeting FOXD1 (si-FOXD1), miRNA negative control (miR-NC), miR-186 mimics, miR-186 inhibitor were procured from RiboBio (Guangzhou, China). The transfection was conducted in the NPC cell lines using Lipofectamine
Immunohistochemistry (IHC)
The wax block containing NPC tissues and non-cancerous tissues was sliced, dewaxed and hydrated using xylene. Following that, it was incubated in 0.3% H
qPCR
Total RNA was extracted from NPC cells and seventy-five pairs of NPC tissues/non-cancerous tissues using TRIzol reagent (Invitrogen, Carlsbad, CA). Nanodrop-spectrophotometer was employed to detect the concentration and purity of the extracted RNA. One
Sequences used for qPCR
Sequences used for qPCR
Abbreviation: F stands for forward; R stands for reverse; RT stands for reverse transcription.
CNE2 and HONE1 cells were lysed using pre-cooled RIPA lysis buffer (Thermo Fisher Scientific, Inc.), and then the lysed cells were maintained in ice for 30 min. After centrifugation at 4
Cell counting kit-8 assay (CCK-8) experiment
CNE2 and HONE1cells were inoculated into a 96-well plate at a density of 3
Colony formation experiment
CNE2 and HONE1 cells in the logarithmic growth phase were collected and trypsinized, and the cell density was adjusted to 1
Transwell assay
Migration experiment: NPC cells were resuspended in serum-free medium and the obtained cell suspension solution was adjusted to a density of 1
FOXD1 was highly expressed in NPC. Immunohistochemistry was used to detect the expression of FOXD1 in NPC tissues and non-cancerous tissues. The expression of FOXD1 was strongly positive, weakly positive, and negative in NPC tissues and non-cancerous tissues. The expression of FOXD1 mRNA in NPC tissues and non-cancerous tissues was detected using qPCR (
The 3’-UTR fragment of FOXD1 mRNA containing the binding site for miR-186 was cloned into the pGL3 vector (Promega, Madison, WI, USA) to obtain pGL3-FOXD1-WT vector. The mutation was introduced into the binding site where the 3’-UTR fragment of FOXD1 mRNA binds with miR-186 using the QuikChange Site Oriented Mutagenesis Kit (Stratagene, LaJolla, CA), and the mutant vector was named pGL3-FOXD1-MUT. The miR-186 mimics or its negative control and pGL3-FOXD1-WT or pGL3-FOXD1-MUT were co-transfected into 293T cells, respectively. After 48 h of the transfection, the luciferase activity of each group was detected using the dual-luciferase gene reporter assay kit (Promega, Madison, WI, USA) with Renilla luciferase activity as internal control.
Statistical analysis
All data were analyzed using SPSS software version 17.0 (SPSS Inc., Chicago, IL, USA) and the results were expressed as mean
Clinicopathologic characteristics in NPC patients
Clinicopathologic characteristics in NPC patients
Over-expression of FOXD1 and its clinical significance in NPC
To investigate the expression characteristics of FOXD1 in NPC, IHC was adopted to detect FOXD1 expression in 75 pairs of NPC cancer tissues and non-cancerous tissues. As shown, FOXD1 was mainly expressed in nucleus, which was consistent with its role as a transcription factor; representative IHC images of the three staining levels of FOXD1 (strong positive, weak positive, and negative) were shown (Fig. 1A). The results of IHC suggested that the positive expression rate of FOXD1 in NPC tissues was 69.3% (52/75). Nevertheless, only 46.7% of the specimens (35/75) were positively expressed in the non-cancerous tissues. Statistical results indicated a significant increase in the positive rate of FOXD1 expression in NPC tissues in comparison with non-cancerous tissues (Fig. 1B). Additionally, qPCR results indicated significant up-regulation of FOXD1 mRNA in NPC tissues (fold-change
FOXD1 enhanced the proliferation, radio-resistance and metastasis of NPC cells
The FOXD1 over-expressed plasmid and empty vector plasmid were respectively transfected into CNE2 cells to construct FOXD1 over-expressed cell model. Besides, si-FOXD1 and si-NC were respectively transfected into HONE1 cells to build FOXD1 knocked-down cell model. qPCR and Western blot were performed certify successful transfections (Fig. 2A and B). To probe the effects of FOXD1 on the proliferation of NPC cells, CCK-8 assay was performed. Compared to the control group, FOXD1 over-expression significantly promoted the proliferation of CNE2 cells (Fig. 2C). Subsequently, to explore the effects of FOXD1 on radio-sensitiveness of NPC cells, colony formation assay confirmed that FOXD1 over-expression significantly enhanced the viability of CNE2 cells exposed to varied dose of radiation (Fig. 2D). Following that, through Transwell assays, we found that FOXD1 over-expression significantly promoted the migration and invasion of CNE2 cells (Fig. 2E and F). On the contrary, the knockdown of FOXD1 in HONE1 cells elicited inhibitory effects on the proliferation, radio-resistance and metastasis of the cells by comparison with the control group (Fig. 2C–H).
FOXD1 promoted the proliferation, migration, invasion and radio-resistance of NPCs. A. The FOXD1 plasmid and the empty vector plasmid were transfected into CNE2 cells, and si-FOXD1 and si-NC were transfected into HONE1 cells, and the expression of FOXD1 mRNA in cells was detected using qPCR. B. Western blot was used to detect the expression of FOXD1 protein in NPC cell after transfection. C. The proliferation of NPC cells was monitored using CCK-8 assay. D. NPC cell survival exposed to different doses of radiation was detected using colony formation experiments. E–H. Transwell assay was used to detect the migration and invasion of NPC cells. 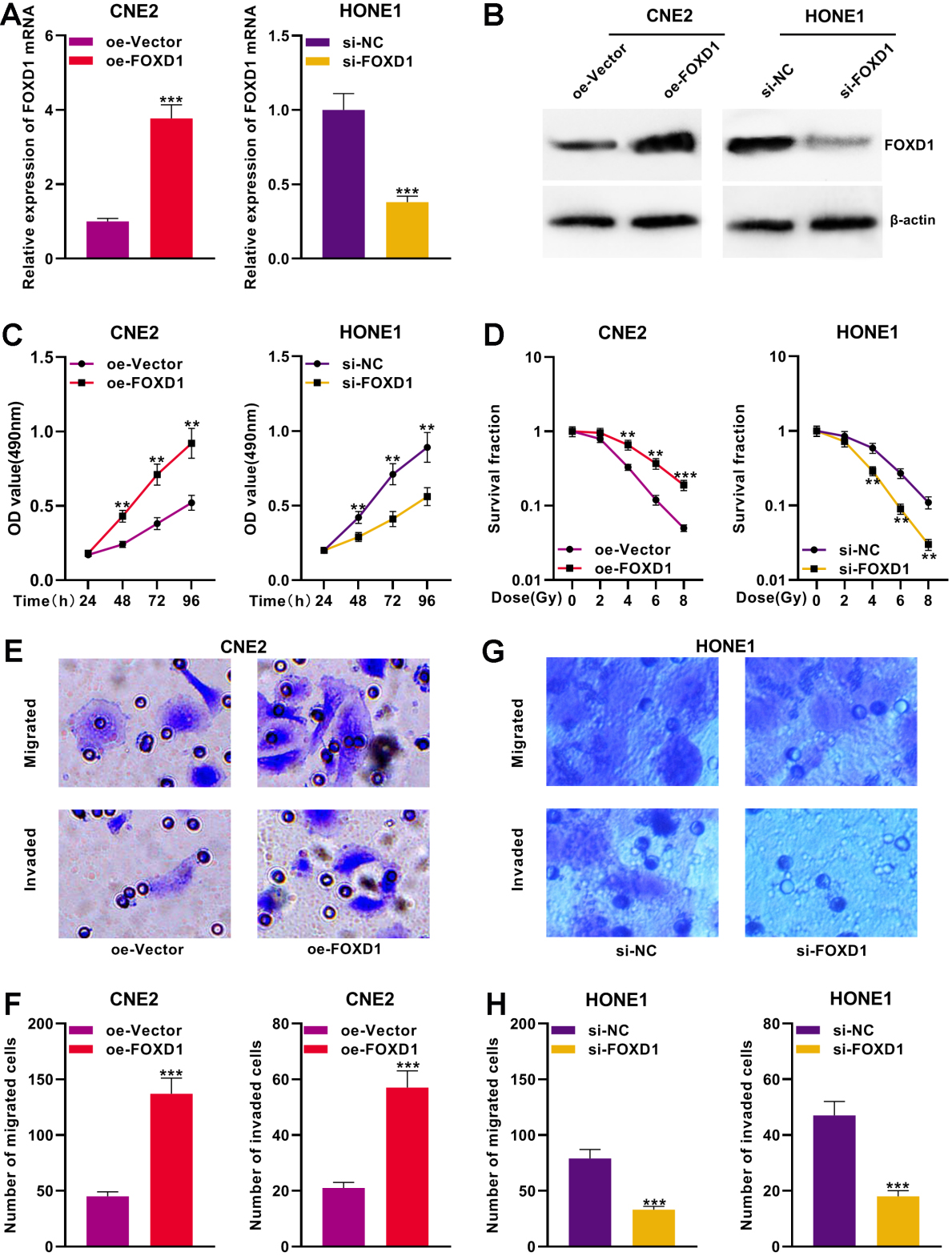
FOXD1 was a target of miR-186. Bioinformatics analysis indicated that there was a potential binding site between miR-186 and FOXD1. The miR-186 mimics were transfected into CNE2 cells and HONE1 cells, and the expression of FOXD1 mRNA in two types of cells was detected using qPCR. Western blot was used to detect the expression of FOXD1 protein in NPC cells. D. The results of Dual-luciferase reporter gene assay showed that miR-186 had a negative regulatory effect on FOXD1-WT luciferase activity, but had no significant effect on FOXD1-MUT luciferase activity. E. Person correlation analysis showed a significant negative correlation between FOXD1 and miR-186 expressions in NPC tissues. 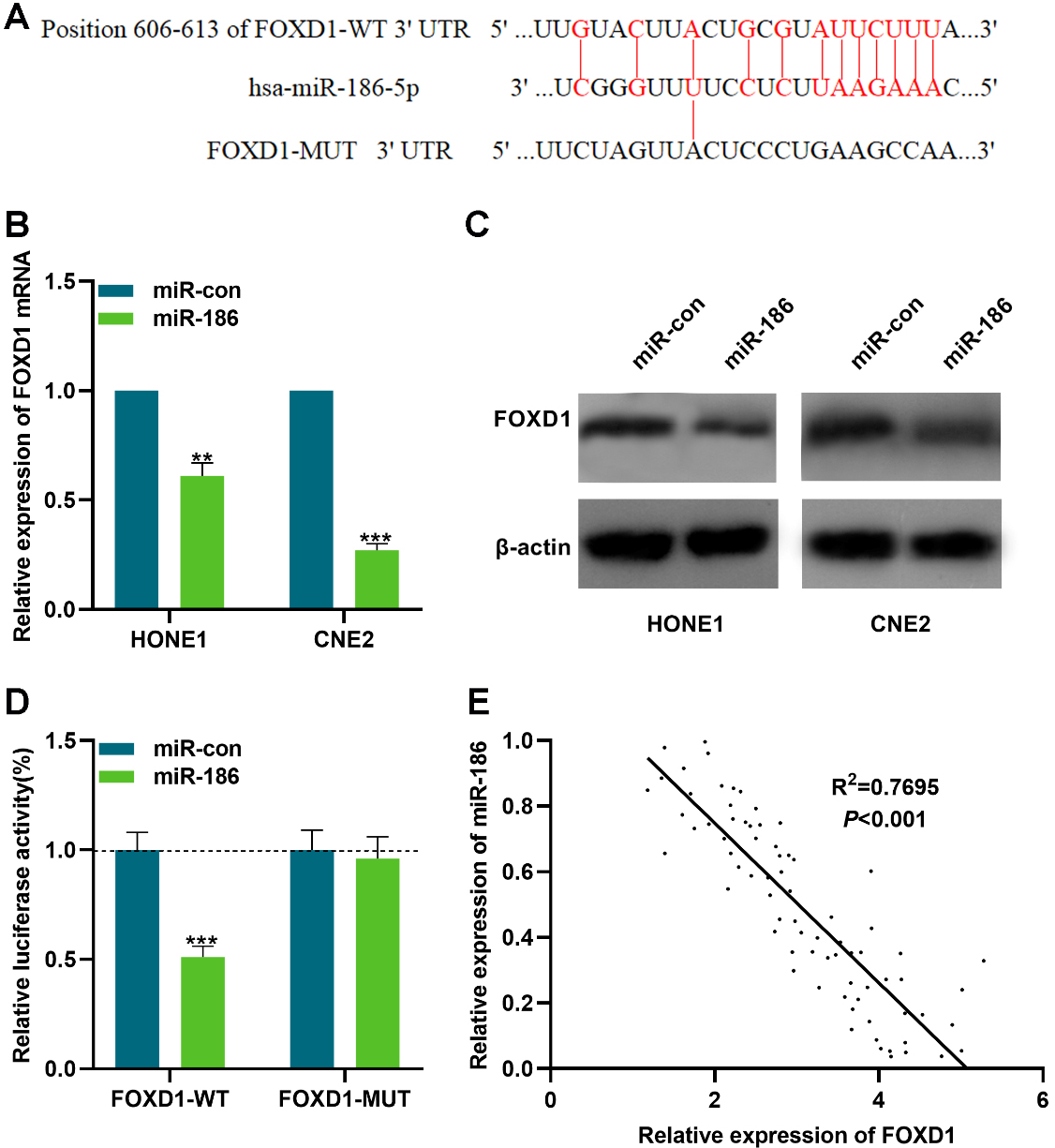
The role of highly-expressed FOXD1 in cancer tissues remained to be further investigated, and miRNA was crucial for the regulation of gene expression. StarBase Database 3.0 was used to screen the miRNA which may regulate FOXD1, and it was indicated that a binding site was found between miR-186 and FOXD1 (Fig. 3A). To investigate the regulatory function of miR-186 on FOXD1, FOXD1 expression at both mRNA and protein levels was detected using qPCR and Western blot, respectively after the over-expression of miR-186. It was found that miR-186 significantly inhibited FOXD1 expression in NPC cells (Fig. 3B and C). To verify whether miR-186 binds with the 3’UTR of FOXD1, Dual-luciferase reporter gene assay was performed. pGL3-FOXD1-WT or pGL3-FOXD1-MUT was co-transfected into 293T cells with miR-186 mimics or negative control, respectively. As demonstrated, compared to the control group, the miR-186 mimics remarkably decreased the luciferase activity of pGL3-FOXD1-WT group (Fig. 3D). Furthermore, based on Pearson analysis, a negative correlation between miR-186 expression and the expression of FOXD1 was found in NPC tissues (
The expression of miR-186 was reduced in NPC and it inhibits NPC cell proliferation, metastasis and radiosensitivity
To further probe the expression characteristics of miR-186 in NPC, qPCR was conducted to detect miR-186 expression in 75 pairs of NPC tissues/non-cancerous tissues. It was demonstrated that miR-186 expression was remarkably down-regulated in NPC tissues (Fig. 4A). Additionally, miR-186 expression in NPC cells was significantly down-regulated in comparison with NP69 cells (Fig. 4B). To further study the role of miR-186 in NPC, miR-186 mimics were transfected into HONE1 cells to establish miR-186 over-expressed model, and miR-186 inhibitors were transfected into CNE2 cells to establish miR-186 lowly-expressed model. Besides, qPCR was used to verify successful transfections (Fig. 4C). The proliferation, radio-resistance and metastasis of the cells were assessed using CCK-8 assay, Colony formation assay, and Transwell assay, respectively. The data indicated that the up-regulation of miR-186 suppressed the proliferation, migration, invasion, and radio-resistance of the cells (Fig. 4D–G). As expected, in CNE2 cells, the inhibition of miR-186 exerted the reverse effect (Fig. 4D–G). The findings revealed that miR-186 exerted tumor-suppressing effects in NPC.
miR-186 acted as a tumor suppressor in NPC. A. qPCR was used to detect the expression level of miR-186 in NPC tissues and non-cancerous tissues (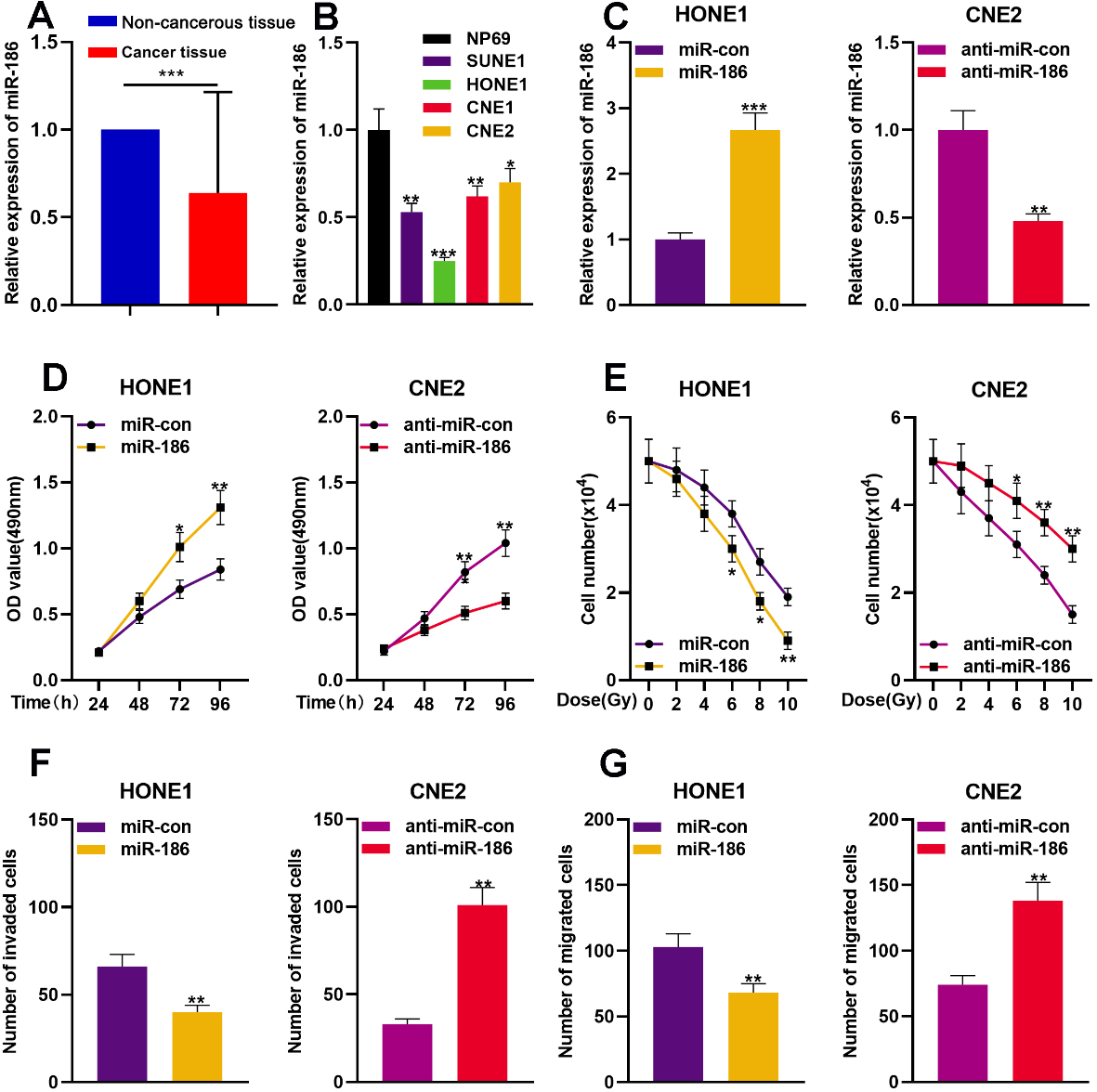
To further explore the function of the miR-186/ FOXD1 axis in NPC, the FOXD1 over-expression plasmid was transfected into HONE1 cells with over-expressed miR-186, and si-FOXD1 was transfected into CNE2 cells with inhibited miR-186. Transfection efficiency was determined using qPCR and Western blot (Fig. 5A and B). The role of miR-186/FOXD1 axis in regulating the proliferation, radio-resistance and metastasis of NPC cells was investigated using CCK-8 assay, colony formation assay and Transwell assay, respectively. We found that the over-expression of FOXD1 partially reversed the inhibitory effects on the proliferation, radio-resistance and metastasis of HONE1 cells induced by miR-186 over-expression (Fig. 5C–F). Conversely, the inhibitory effects on the enhancement of malignant phenotypes of CNE2 cells induced by miR-186 were partially attenuated by the knockdown of FOXD1 (Fig. 5C–F). These results indicated that the miR-186/FOXD1 axis was involved in the regulation of the proliferation, metastasis and radio-sensitivity of NPC cells.
miR-186/FOXD1 axis was involved in the regulation of the malignant phenotypes of NPC cells. The FOXD1 over-expressed plasmid was transfected into miR-186 over-expressed HONE1 cells, and si-FOXD1 was transfected into miR-186 down-regulated CNE2 cells. The expression level of FOXD1 mRNA in NPC cells was detected using qPCR. The expression of FOXD1 protein level in HONE1 cells and CNE2 cells was detected using Western blot. C–F. Up-regulation of FOXD1 weakened the inhibitory effects of the over-expression of miR-186 on proliferation, radio-resistance, migration and invasion of HONE1 cells, while knocking down FOXD1 reversed the enhancement of proliferation, radio-resistance, migration and invasion induced by the down-regulation of miR-186 in CNE2 cells. 
With the advancement of radiology technology, the therapeutic effect of radiotherapy in NPC has become increasingly prominent [20]. Radiotherapy can significantly improve the prognosis of NPC patients with a five-year overall survival of 70–80% [21]. Nevertheless, some patients have developed radio-resistance, resulting in local recurrence and distant metastasis of cancer cells after being subjected to radiotherapy [22, 23]. Merely increasing the radiation dose can lead to severe post-treatment adverse effects. Further exploration of key genes in NPC progression may provide novel targets for improving radiotherapy efficacy in NPC. In the present study, we observed that FOXD1 was significantly highly-expressed in both NPC tissues and cell lines, and its over-expression was strongly related to lymph node metastasis of cancer cells and low degree of tissue differentiation.
FOXD1, a member of the forkhead box superfamily, has a transcriptional regulatory function [24]. FOXD1 possesses a highly conserved DNA binding domain consisting of approximately 100 amino acids in a helix-turn-helix structure that binds with specific DNA sequences on the promoter region of a target gene, thereby promoting or inhibiting the gene expression [25]. Recent studies suggest that FOXD1 is also implicated in a wide range of human diseases, including cancer [26]. For instance, FOXD1 expression is significantly increased in colorectal cancer, which facilitates the proliferation of colorectal cancer cells and inhibits apoptosis by positively modulating Plk2, and its high expression indicates poor prognosis of patients [27]. In breast cancer, FOXD1 promotes cell proliferation by accelerating the transformation from G1 phase to S phase [9]. FOXD1 also functions as a regulator of VEGF-A, and silencing FOXD1 can suppress angiogenesis [28]. FOXD2, another member of FOX transcription factor family, has been found progressively over-expressed in uterine cervical precancerous lesions [29]. It is likely to be associated with cancer onset/progression. In addition, FOXD2 and FOXD1 present high sequence homology [30, 31]. These studies indicate that the inhibition of FOXD1 expression will probably provide a novel therapeutic approach for cancer. In this work, the results of functional experiments indicated that the over-expression of FOXD1 enhanced the proliferation, migration, invasion and radio-resistance of NPC cells. Conversely, FOXD1 knockdown inhibited the malignant phenotypes of NPC cells. It is well-known to us that this is the first work to investigate the oncogenic function of FOXD1 in NPC.
Studies have shown that miRNA plays pivotal roles during the carcinogenesis and progression of NPC. Aberrantly-expressed miRNA in tissues can be considered as a biomarker to evaluate tumor progression [32, 33]. For instance, miR-125b enhances NPC radio-resistance by targeting A20 to activate the NF-
However, this work still has several limitations. Foremost, data in vivo is necessary to further validate our conclusion in the following work; what’s more, other downstream mechanism by which FOXD1 promotes NPC progression remains to be clarified; furthermore, to explore the potential of FOXD1 and miR-186 as biomarkers, more samples from multiple centers should be recruited, and the survival analysis of NPC patients based on the expression of FOXD1 and miR-186 is necessitated.
In summary, we first found that FOXD1 was highly expressed in NPC and its high expression was significantly correlated to the positive lymph node metastasis and low degree of differentiation in NPC patients. The experiments in vitro confirmed that the miR-186/FOXD1 axis was involved in modulating the proliferation, migration, invasion, and radio-resistance of cells. FOXD1 may be a potential new biomarker for the diagnosis, treatment, and prognosis of NPC. The present study provided new insights for further NPC research and clinical therapy.
Footnotes
Acknowledgments
We thank the colleagues from the Department of Pathology, Luoyang Central Hospital, for their help to provide pathological diagnosis. Our study was approved by the ethics review board of Luoyang Central Hospital Affiliated to Zhengzhou University.
Conflict of interest
The authors declare that they have no competing interest.
