Abstract
MicroRNAs are a class of small non-coding RNA molecules that play crucial roles in the initiation and progression of lung cancer. This study was undertaken to investigate the expression and roles of miR-186 in lung cancer. Ectopic expression experiments demonstrated that miR-186 functions as a tumor suppressor. Bioinformatic predictions, a luciferase reporter assay, and protein expression analysis suggested that miR-186 could inhibit the protein levels of SIRT6, a purported tumor suppressor gene. Collectively, our results indicated that miR-186 could inhibit lung cancer progression through targeting SIRT6 and that miR-186 may be a therapeutic target for lung cancer.
Introduction
In China and many developed countries, lung cancer has become the most frequent cause of cancer-related death. Non-small cell lung carcinoma (NSCLC) accounts for 70–80% of all lung cancers and is generally diagnosed at an advanced stage [1]. Despite advances made in clinical treatment, the prognosis of NSCLC patients, especially those with advanced disease, is still very poor. The initiation and progression of NSCLC are part of a multistep process, which is attributed to the activation of oncogenes and inactivation of tumor suppressor genes [2]. Currently, the molecular mechanism underlying the development of NSCLC remains poorly understood.
MicroRNAs (miRNAs/miRs) are a class of short (22–24 nucleotides in length), single-stranded RNA molecules. MicroRNAs silence protein expression by either inhibiting translation elongation or inducing mRNA decay [3]. The expression and role of cancer-related miRNAs have been extensively explored in lung cancer [4]. A number of miRNAs were found to be deregulated in lung cancer; furthermore, their expression levels were associated with poor outcome [5]. Notably, upregulation of let-7 inhibits lung cancer cell cycle arrest by suppressing expression of RAS [6] or HMGA2 [7]. Generally, multiple miRNAs can repress one gene or one miRNA may repress multiple target genes, resulting in complex regulatory feedback networks. In different pathological and cellular environments, miRNAs have been reported to be involved in multiple cellular processes, such as differentiation, morphogenesis, and angiogenesis [8, 9, 10]. Many reports have indicated a role for miRNAs in the etiology and pathogenesis of cancer; microRNAs may fully or partially base pair with the 3’-UTR of oncogenes or tumor suppressors [11, 12]. Although the global alteration of miRNA expression is a hallmark of tumorigenesis, the studies addressing the role of miRNAs in cellular transformation and tumorigenesis remain unclear [13, 14]. Previous studies have shown that miR-186 acted as a tumor suppressor in different cancers [15, 16, 17]. Accordingly, miR-186 could inhibit proliferation, cell cycle progression, migration, and invasion of NSCLC cells by targeting different genes. In clinical samples, the downregulation of miR-186 correlated with poorer survival of NSCLC patients. Thus, identification of the miRNA targets would be critical to understanding the miRNA pathway and underlying molecular signaling associated with NSCLC progression and development.
Here, we identify the expression of miR-186 in specimens from patients with NSCLC. Furthermore, we investigated the function of miR-186 in NSCLC cells. Our findings suggest that SIRT6 translation is directly inhibited by miR-186, and miR-186 potentially acts as a growth suppressor in A549 cells.
Materials and methods
Tissue samples
A total of 4 patients, who were diagnosed with primary NSCLC in the Department of Cardiothoracic Surgery at the First People’s Hospital of Yunnan Province from 2012 to 2016, were included in this study. None of these patients received chemotherapy or radiotherapy before surgery. Tumor and matched non-tumor lung tissue samples were collected, rapidly frozen in liquid nitrogen, and stored at
Cell culture, RNA oligonucleotides, and transfection
The NSCLC cell line (A549) was cultured in RPMI1640 (GIBCO-BRL) medium supplemented with 10% fetal bovine serum, penicillin (100 U/ml), and streptomycin (100 U/ml) at 37
Cell viability assay
Cell proliferation was determined by using the CellTiter96 (Promega, Madison, WI) and Cell Counting Kit-8 assay kit (Dojindo, Kumamoto, Japan) assays according to the manufacturer’s instructions.
Flow cytometric analysis of the cell cycle
The A549 cells (4
Flow cytometric analysis of apoptosis
An annexin V-fluorescein isothiocyanate (FITC) apoptosis detection kit (Oncogene Research Products, Boston, MA, USA) was used to detect apoptosis, as previously described. All of the samples were assayed in triplicate.
RNA extraction and qRT-PCR
Total RNA, including miRNA, was extracted from cell lines and tissue samples using TRIzol reagent (Invitrogen, USA). The RNA was synthesized into cDNA using M-MLV reverse transcriptase (Promega, USA) in a 25
Luciferase reporter assay
To create a luciferase reporter plasmid, the 3’-UTR fragment containing putative binding sites for miR-186 was PCR-amplified from the genomic DNA of A549 cells. The PCR products were gel-purified, digested, and inserted into the digested psiCheck-2 plasmid (Promega, USA) between the Xho I and Not I sites. SIRT6 3’-UTR-targeted site mutations were generated using the KOD-plus mutagenesis kit (Toyobo, Japan), according to the manufacturer’s protocol.
A549 cells cultured in a 96-well plate were co-transfected with 200 ng/
In vivo studies
Animal studies were performed according to the institutional guidelines. The miR-NC-, miR-186-, SIRT6-, and SIRT6+miR-186-transfected A549 cells (5
Statistical analysis
The SPSS 10.0 program was used for general statistical and survival analysis. Experimental data were expressed as the mean
Results
Identification of aberrant miRNA expression in NSCLC tissue samples
The expression level of miR-186 was determined by quantitative real-time PCR of NSCLC tissue from 8 patients who underwent surgical resection in our department. The clinical characteristics of these patients are listed in Table 1. Our results confirmed that miR-186 was downregulated in all 8 lung cancer tissues compared with that in adjacent normal tissues (Fig. 1).
The clinical characteristics of patients in this study
The clinical characteristics of patients in this study
miR-186 expression was analyzed by real-time PCR and standardized against the expression of small nuclear RNA U6 (snU6), which was used as an endogenous control. The data are shown as Log10 of relative quantification (2
To test if miR-186 functions as a tumor suppressor, in vitro experiments were performed to determine the effect of miR-186 upregulation on NSCLC cell proliferation. The CCK-8 assays were performed to detect cell viability. A549 cells transfected with the miR-186 mimic grew more slowly than cells transfected with the control (NC) (Fig. 2A). The inhibitory rate of miR-186 mimic-transfected cells was higher than that of NC-transfected cells (Fig. 2B).
miR-186 inhibits cell growth and induces senescence in A549 and 95D cells. (A) A549 was transfected with 50 nM of either the miRNA mimic (open circles) or the negative control (NC, filled circles) in 96-well plates on day 0. Cell viability assays were performed on the days indicated. (B) The effect of miR-186 in A549 cells was examined by using a CCK-8 assay. Data represent the average of triplicate values. Error bars, SEM. 
To further ascertain the mechanism by which miR-186 expression mediated cell proliferation, the cell cycle of A549 cells was detected using flow cytometric analysis after transfection of either the miR-186 mimic or NC. As seen in Fig. 3, miR-186 overexpression caused significant G
Cell cycle profile changes were assessed by flow cytometry using propidium iodide (PI) staining to measure the DNA content.
To investigate if the introduction of miR-186 contributed to apoptosis, flow cytometric analysis was performed after transfection of A549 cells with either the miR-186 mimic or miR-NC. A549 cells underwent apoptosis 48 h after transfection with the miR-186 mimic. In miR-186 mimic-transfected A549 cells, the rates of early (UR quadrant) and late apoptosis/ necrosis (UR quadrant) were 28.80% and 53.69%, respectively. In contrast, the rates of early and late apoptosis/necrosis in miR-NC-transfected A549 cells were 3.88% and 0.8%, respectively (Fig. 4). These results suggest that ectopic expression of miR-186 could induce enhanced apoptosis in A549 cells.
Ectopic miR-186 expression enhances apoptosis of A549 cells. Approximately 48 h after transfection with either pre-miR-186 or miR-NC, A549 cells were collected for apoptosis analysis. The apoptotic rates of the cells were detected by flow cytometry.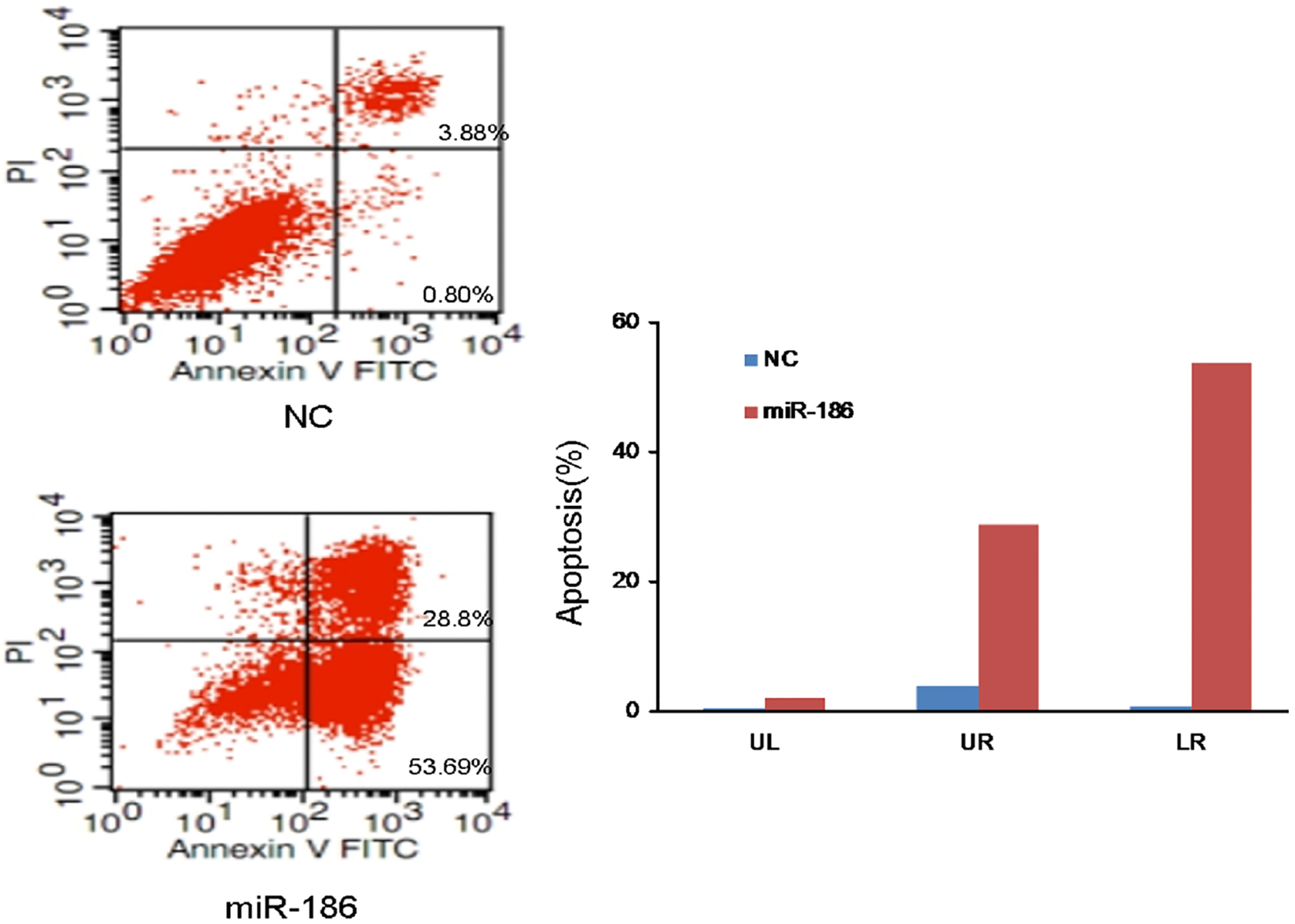
Using in silico miRNA target identification software, including TargetScan, miRBase, and PicTarget, SIRT6 was identified as a putative target of miR-186 (Fig. 5A). Consequently, we subcloned WT-3’-UTR or its mutant, MUT-3’-UTR, which lacks the miR-186 binding sequences, downstream of the firefly luciferase reporter gene in the pLUC vector. Then, each construct was co-transfected with either the miR-186 mimic, miR-NC, or miR-186 inhibitor into A549 cells. The relative luciferase activity of the reporter construct containing the wild-type 3’-UTR decreased by 70% when the miR-186 mimic was co-transfected. However, the relative luciferase activity of the reporter construct increased by 27.4% when the miR-186 inhibitor was co-transfected. The inhibitory effect of the miR-186 mimic was abolished in the mutated construct (Fig. 5B).
SIRT6 is a direct target of miR-186. (A) Sequence alignment between miR-186 and the 3’-UTR of human SIRT6 mRNA, seed-matching or seed-mutated regions. (B) Relative luciferase activity was calculated according to (Rluc
SIRT6 induces cell proliferation and inhibits cell apoptosis. (A) Effect of miR-186, SIRT6, or SIRT6+miR-186 on the growth of A549 cells, as determined by the CCK-8 assay. (B) The inhibitory rate of miR-186, SIRT6, or SIRT6+miR-186 on the growth of A549 cells, as determined by CCK-8 assay. (C) Summary of cell cycle profile changes. Effect of transient transfection of miR-186, SIRT6, or SIRT6+miR-186 on the cell cycle of A549, as determined by flow cytometry. 
miR-186 suppresses tumor growth in vivo. (A) Tumor formation in nude mice. The group contained ten mice (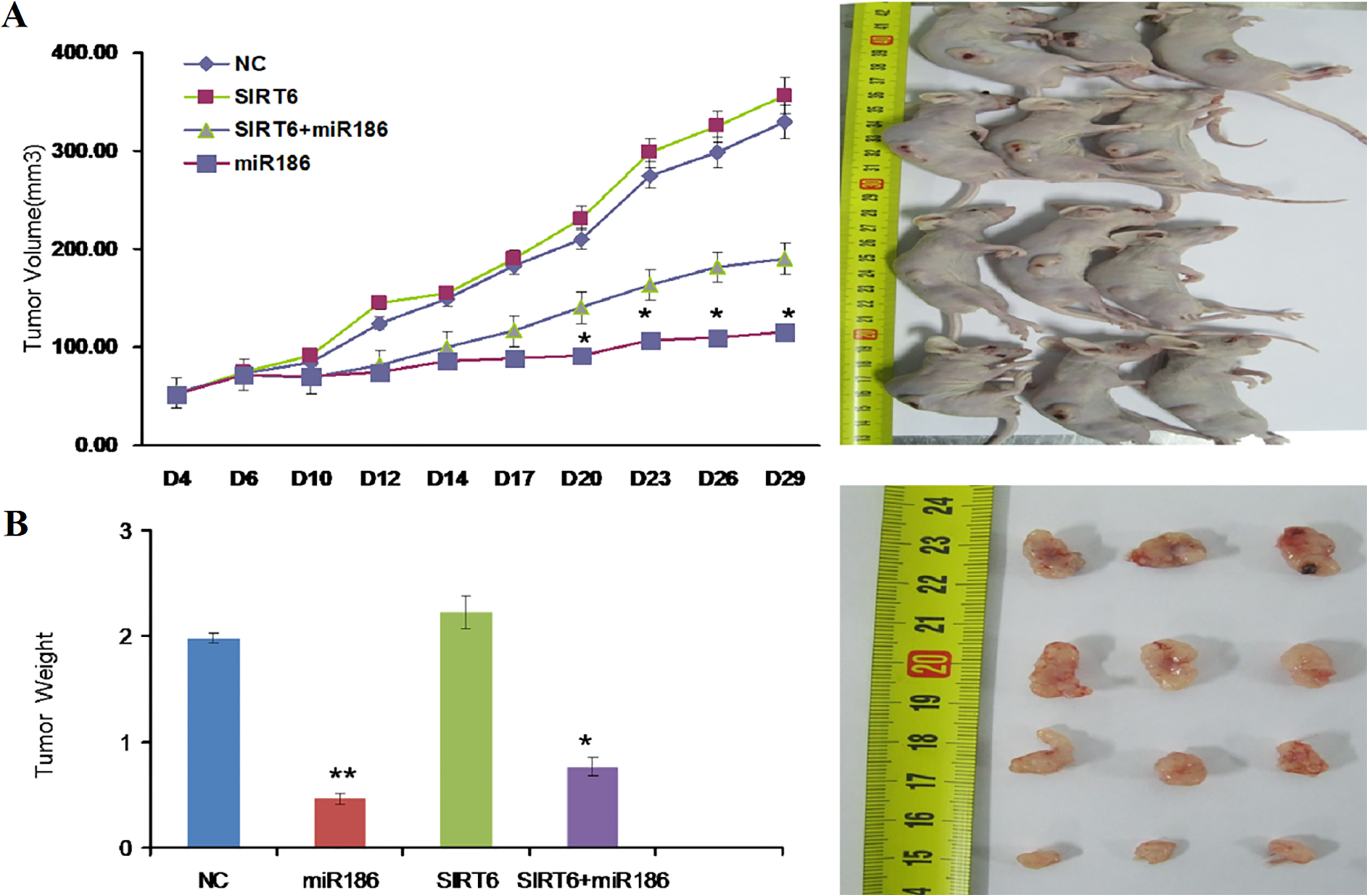
Next, we determined whether miR-186 expression affects endogenous SIRT6 expression at both the transcriptional and translational levels. As shown in Fig. 5C, the level of SIRT6 mRNA in miR-186 mimic-transfected A549 cells was significantly inhibited by 61% compared with that in miR-NC-transfected cells. Western blot analysis showed that the level of SIRT6 protein expression was also inhibited by treatment with the miR-186 mimic in A549 cells (Fig. 5D). Thus, these data suggest that miR-186 reduces SIRT6 expression by inhibiting translation and/or causing mRNA instability.
To investigate whether downregulation of SIRT6 was involved in miR-186-induced growth suppression and apoptosis enhancement, we examined whether SIRT6 reversed the inhibition of cell growth caused by miR-186. A549 cells were co-transfected with miR-186 mimic and pEZ-M02-SIRT6, which contained the entire SIRT6 coding sequence but lacked the 3’UTR of SIRT6 mRNA. The resulting SIRT6 overexpression rescued the suppression of cell viability caused by miR-186 (Fig. 6A and B). At the same time, SIRT6 significantly inhibited cell cycle arrest and apoptosis in A549 cells (Fig. 6C and D). Moreover, co-transfection of SIRT6 and miR-186 partially rescued miR-186-induced growth promotion and apoptosis inhibition in A549 cells. These results suggest that targeting SIRT6 may be a mechanism of miR-186-mediated tumor suppression in lung cancer cells.
At 28 days, the average volume of the tumors formed from miR-186- and miR-186+SIRT6- transfected A549 cells was significantly lower than that of tumors formed from miR-NC- and SIRT6-transfected cells. Notably, the tumor suppressor effect of SIRT6 knockdown mediated by miR-186 was similar to that of ectopic miR-186 expression.
Effect of miR-186 expression on in vivo proliferation of NSCLC cells
To explore whether the level of miR-186 expression affects tumorigenesis, A549 cells transfected with either the miR-186 mimic, miR-NC, SIRT6, or SIRT6+miR-186 were inoculated into male nude mice. During the whole-tumor growth period, tumors from the miR-186 mimic-transfected A549 cells grew slower, but the SIRT6-transfected cells grew faster in compassion with the negative control (Fig. 7A). Five weeks after inoculation, the average weight of the tumors from miR-186 mimic-transfected A549 cells was less than that of the control mice. The tumors from SIRT6-transfected cells, however, were larger than those of the control mice (Fig. 7B). These results suggest that the level of miR-186 expression was significantly associated with the in vivo proliferation capacity of NSCLC cells.
Discussion
In addition to protein-encoding genes, there is sufficient evidence that miRNAs have more than a cursory role in the initiation and development of human cancers by functioning as agents of the RNA interference pathway. The present study elucidated the biological roles of only a small fraction of identified miRNAs. These miRNAs participate in cancer-related biological processes, such as cell cycle arrest, proliferation, apoptosis, and chemoresistance [18, 19]. Until recently, the effects of miRNAs in NSCLC and the molecular mechanism by which miRNAs modulate the behavior of NSCLC cells remained unclear.
In the present study, we showed that miR-186 is significantly downregulated in NSCLC. Although very few studies explored the role of miR-186 in human cancers, miR-186 was one of the 8 best-studied miRNAs upregulated in pancreatic cancer [20]. Previous studies have shown that miR-186 acted as a tumor suppressor in different cancers [21]. By targeting different genes, miR-186 could inhibit proliferation, cell cycle progression, migration, and invasion of NSCLC. Myatt et al. showed that overexpression of miR-186 contributed to endometrial tumorigenesis, which suggests that miR-186 may play an oncogenic role in other cancer types [22]. On the other hand, Lv et al. reported that deletion of miR-186 occurred in 15% (
Our study is the first to show that miR-186 functions as an inhibitor of tumor growth in NSCLC cells. Moreover, this is the first report of direct interaction between the oncogene, SIRT6, and miR-186; miR-186 targets SIRT6 via binding to the 3’-UTR of SIRT6 mRNA in NSCLC cells in vitro and in vivo. SIRT6 is a highly specific NAD
Footnotes
Acknowledgments
This study was supported by the Applied Basic Research Project of Joint Special Fund of Kunming Medical University from Yunnan Province (No. 2017FE467 (-117) and 2017FE468(-109) and Scientific Research Fund from Yunnan Provincial Department of Education (2016ZDX009).
