Abstract
BACKGROUND:
LncRNA MEG3 rs7158663 has been shown to confer cancer susceptibility, maybe through altering its gene expression level.
OBJECTIVE:
We aimed to weigh the effect of rs7158663 on MEG3 serum level and breast cancer susceptibility.
METHODS:
We genotyped rs7158663 G
RESULTS:
The presence of rs7158663 G
CONCLUSION:
The presence of both rs7158663 and low MEG3 are diagnostic and unfavorable prognostic factors for BC patients.
Introduction
Breast cancer (BC) is the second common cancer in females and is a leading cause of death between women [1]. Despite great advances in the early diagnostic and treatment regimens of BC cases, there is no significant improvement in the BC survival rate [2]. Breast cancer etiology is heterogeneous in which multiple genetic and epigenetic variations affect its incidence and prognosis. The exact mechanisms that contribute to the occurrence of BC are still obscure [3]. Numerous studies have attempted to identify molecular factors predispose to and underlying BC pathogenesis in order to introduce new effective therapeutic approaches [4].
Long non-coding RNAs (lncRNAs) are RNA polymerase II transcripts without protein-coding capacity. They are longer than 200 nucleotides in length. It has been approved that lncRNAs are implicated in carcinogenesis through regulating several biological activities, including proliferation, apoptosis, autophagy, angiogenesis, and metastasis by influencing histone modification, transcription, and posttranscriptional processes [5].
Maternally expressed gene 3 (MEG3) is lncRNA imprinted from the maternal allele. It is expressed in various normal tissues [6]. LncRNA MEG3 is a tumor suppressor that has been detected to be downregulated in many cancerous tissues such as gastric cancer [7] colorectal cancer [8], ovarian cancer [9] and hepatocellular carcinoma [10].
Several studies investigated the relationship between MEG3 expression level and breast cancer and demonstrated that: First, MEG3 is normally expressed in normal breast tissues [6]. Next, Loss of MEG3 expression has been identified in breast cancer tissues and cell lines. Moreover, the decreased lncRNA MEG3 expression was significantly associated with the lymph node metastasis, TNM stage, and molecular subtypes. Furthermore, Low lncRNA MEG3 expression level predicts a shorter overall survival and relapse-free survival in breast cancer Last, overexpressed MEG3 suppresses tumor growth, angiogenesis and induces apoptosis in breast cancer cells both invivo and invitro [11, 12, 13, 14]. Collectively the previous findings suggested that MEG3 may act as a possible novel diagnostic, therapeutic, and even prognostic target of breast cancer.
Recent studies reported that abnormal MEG3 expression levels identified in the serum samples from bladder cancer and colorectal cancer patients suggesting the potential use of MEG3 as a biomarker for cancer early diagnosis, prognosis, and prediction of recurrence of the tumor as well as drug resistance [15, 16, 17]. No study measures the expression level of MEG3 in the serum of BC patients and examines its utility as a non-invasive biomarker for BC.
Single nucleotide polymorphisms (SNPs) are one of the most common types of genetic variations in the human genome. SNPs in genes affect gene expression by different mechanisms and are associated with genetic susceptibility to different diseases, including cancer [18, 19]. MEG3 rs7158663 G
Fibroadenoma (FA) is an expression that refers to a wide range of solid, benign breast lesions that commonly affect women in their reproductive years. Moreover, there is not any risk of breast cancer development on top of fibroadenoma tissue. The standard treatment procedure of fibroadenomas by surgical excision has recently been challenged because if confident diagnosis and exclusion of malignancy is available with noninvasive investigations. Then the excision biopsy can be avoided with avoidance of surgical hazards, including disruption of shape and texture of the breast [23]. So we aimed to examine the utility of serum MEG3 in differentiation between BC patients and those with fibroadenomas.
By taking in consideration the preceding findings, including the pivotal role of dysregulated MEG3 in breast cancer, along with the potential impact of rs7158663 on MEG3 expression level and/or its structure, we suppose that rs7158663 SNP of MEG3 may modify the susceptibility to breast cancer through affecting its expression level. To test this supposition, we performed an association study between rs7158663 SNP in MEG3 and breast cancer risk as well as the correlation between this SNP and MEG3 serum expression level in a case-control study involving 150 BC patients, 95 fibroadenoma patients and 154 control subjects from Egypt.
Materials and methods
Subjects
This case-control study was done on 245 Egyptian patients. They were enrolled from the General Surgery Department at the Fayoum University Hospital at Fayoum University and the General Surgery Department at the General Fayoum Hospital. Initial diagnoses depended on history taking, clinical examination. However, final diagnoses were confirmed by mammography and surgical biopsies. Accordingly, patients were classified into two groups: Group I: 150 patients with breast cancer. All breast cancer patients were newly diagnosed postmenopausal women with no past history of breast cancer. All patients experienced previous oral contraception usage for variable periods throughout their lives, also all of them with parity status for more than two times. None of them received antihormonal, chemotherapy, or radiotherapy treatment before participation in the study. Since the age, tumor size, tumor type, TNM staging (tumor size-the number of lymph nodes involved-metastasis) [24] are essential issues in cancer, they were involved in this study. Also, estrogen receptors and progesterone receptors status (ER/PR status) were determined; ER/PR+ve means ER/PR-positive, while ER/PR-ve means ER/PR-negative. ER/PR+ve tumors are much more likely to respond to hormone therapy than tumors that are ER/PR-ve. Group II: 95 patients with fibroadenoma. Clinical information (Age, Parity status, Family history (FH), and/or past history, contraceptive usage, and the state of menstrual cycles) were obtained from patients reports.
While group III included 154 age-matched healthy females (controls) with no family history of breast cancer or fibroadenoma, with no palpable breast masses, received no contraceptives, and they do not suffer from hypertension or diabetes mellitus were recruited during a routine checkup.
The current study was permitted by the ethical committee of the Faculty of Medicine, Fayoum University. Consequently, all subjects signed a written consent after the declaration of the general aim and procedure of the study. The ethical principals of Helsinki were respected and followed in this study.
Blood sample collection and storage
Blood samples were obtained from all contributors and divided into two tubes. The first tube was used for serum separation and estimation of MEG3. The second tube contained EDTA and was used for DNA extraction and MEG3 genotyping analysis. The samples were frozen at
DNA extraction and genotyping
Genomic DNA was extracted from whole EDTA blood samples of all participants via using QIA-amplification extraction kit (Qiagen, Venlo, Limburg, Netherlands) as per the manufacturer’s instructions. Quantitation and assessment of DNA purity were measured using NanoDrop
Measurement of serum lncRNA MEG3 fold change
The MEG3 Expression level was detected in the serum. Recent studies measured MEG3 in sera of colorectal cancer and bladder cancer patients [12, 19].
Total RNA (with preserved lncRNAs) was extracted from Serum by miRNeasy extraction kit (Qiagen, Valencia, CA, USA) by following the manufacturer’s protocol. Quantitation and purity of RNA samples were evaluated by using the NanoDrop (ND)-1000 spectrophotometer (NanoDrop Technologies, Inc. Wilmington, USA).
Reverse transcription (RT) was achieved on 60 ng of total RNA in a final volume of 20
The serum expression level of the lncRNA MEG3 was determined by using GAPDH as an internal control [25] using customized primers and Maxima SYBR Green PCR kit (Thermo, USA) according to the manufacturer’s protocol. The primer sequences for GAPDH were as follows: 5
Baseline characteristics of studied groups (breast cancer, fibroadenoma, and control groups)
Baseline characteristics of studied groups (breast cancer, fibroadenoma, and control groups)
BC, Breast Cancer, FA, Fibroadenoma. DM, Diabetes Mellitus. HTN, Hypertension. FH, Family History. # One way ANOVA ## Chi squared-test
Real-time PCR was done on 20
The 2
Deviation from Hardy-Weinberg equilibrium (HWE) was tested for polymorphism using a Chi-square test by a specific calculator, available online at
Results
Characteristics of the study population
The baseline characteristics (demographic, clinical, and pathological) of the 150 BC, 95 FA cases, and 154 healthy controls are shown in (Tables 1 and 2).
Clinical and pathological characteristics of studied groups
Clinical and pathological characteristics of studied groups
BC, Breast Cancer, FA, Fibroadenoma. ER/PR, Estrogen Receptor/Progesterone Receptor. U/S, Ultrasound. All breast cancer patients were newly diagnosed postmenopausal women with no past history of breast cancer. All patients experienced previous oral contraceptive usage for variable periods throughout their lives, also all of them with parity status for more than two times. All contols were healthy postmenopausal women with no history of usage of contraception. Liver ultrasound is missing data for fibroadenoma patients and controls.
There was a significant difference between breast cancer and FA patients as well as between controls and FA group as regard age (
As for clinicopathological data of the breast cancer patients, Table 2 shows that patients with weight
As for clinicopathological data of the FA patients, 89.5% of FA patients were premenopausal (73.7% had irregular menstrual cycles, 15.8% had regular menstruation), while 10.5% were postmenopausal. 5.3% use contraceptive pills. 15.8% were with a past history of FA (recurrent fibroadenoma) and 31.6% of FA patients with parity more than two times.
Rs7158663 G
The genotype and allele distributions of MEG3 SNP (rs7158663) in cases (BC, FA) and controls are shown in (Table 3). A significant difference was determined in the frequency distribution of genotypes and alleles of rs7158663 between breast cancer patients and healthy controls with the following details; in all models having a mutant genotype AA and/or GA or A allele were risk factors for the development of BC.
Genotypes and allelic distribution of MEG3 rs7158663 G/A in breast cancer, fibroadenoma cases and control group
Genotypes and allelic distribution of MEG3 rs7158663 G/A in breast cancer, fibroadenoma cases and control group
BC, Breast Cancer, FA, Fibroadenoma.
MEG-3 ct values and MEG-3 fold change in different studied groups
In genotypic model AA vs. GG genotype (OR
Besides, a significant difference was observed in the frequency distribution of rs7158663 polymorphism between breast cancer patients and fibroadenoma patients. Again in all models the presence of mutant genotypes or allele is associated with increased risk of BC; Presence of GA or AA in genotypic model (
No significant differences were detected as regard MEG3 rs7158663 genotypes distribution between fibroadenomas and healthy controls.
On examination of the serum expression level of MEG3 in different studied groups (Table 4), Decreased serum MEG3 was observed in BC group when compared with FA and or controls [median (IQR)
Association between rs7158663 genotypes and MEG3 serum expression level
We further investigated the correlations between rs7158663 genotypes and MEG3 serum expression levels. As shown in Table 5, lncRNA MEG3 serum expression levels in BC group were significantly higher for the GG (median
No significant differences were found between rs715 8663 genotypes and alleles as regards the serum expression level of MEG3 in FA group.
Association between rs7158663 genotypes and MEG3 serum expression level
Association between rs7158663 genotypes and MEG3 serum expression level
FA, Fibroadenoma.
We further examined the association between MEG3 expression levels and clinicopathological characteristics of BC. As shown in Table 6, the results showed that lower MEG3 level was significantly associated with patients with hypertension (
Association between MEG3 fold change and clinical data of breast cancer group
Association between MEG3 fold change and clinical data of breast cancer group
DM, Diabetes Mellitus. HTN, Hypertension. FH, Family History. U/S, Ultrasound.
We evaluated the association between rs7158663 genotypes and alleles and clinical features of BC (Table 7).
Relation of genotypes and allelic frequency of MEG3 rs7158663 with different characteristics in breast cancer group
Relation of genotypes and allelic frequency of MEG3 rs7158663 with different characteristics in breast cancer group
DM, Diabetes Mellitus. HTN, Hypertension. FH, Family History. U/S, Ultrasound.
The presence of mutant GA+AA and/or A allele showed significant association with positive FH (
Also, the presence of mutant GA+AA and/or A allele were significantly associated with N1 of TNM staging when compared with N2 (
No significant correlation was found between rs715 8663 SNP and age, HTN, DM, weight, and tumor grade (all
The relationship between MEG3 expression levels and clinicopathological characteristics of FA are shown in (Table 8), lower MEG3 level was significantly associated with older age of patients (
Association between MEG3 fold change and clinical data of fibroadenoma group
Association between MEG3 fold change and clinical data of fibroadenoma group
DM, Diabetes Mellitus. HTN, Hypertension. FH, Family History. U/s, Ultra sound.
We evaluated the correlations between rs7158663 genotypes and alleles and clinical features of FA (Table 9).
Relation between genotypes and allelic frequency of MEG3 rs7158663 and different characteristics in fibroadenoma group
Relation between genotypes and allelic frequency of MEG3 rs7158663 and different characteristics in fibroadenoma group
DM, Diabetes Mellitus. HTN, Hypertension. FH, Family History.
The presence of mutant GA+AA and/or A allele showed a significant association with younger age of patients (
No significant correlation was found between rs715 8663 SNP and past history of fibroadenoma or parity (
In order to determine the MEG3 diagnostic potential, as a specific biomarker for diagnosis of breast cancer among FA patients and controls, a ROC curve was drawn. ROC analysis revealed that serum MEG3 discriminated BC from non-cancerous group (FA+ controls) with best cut-off
A-ROC curve of MEG-3 ct value revealed that serum MEG3 discriminated BC from non-cancerous group (FA+ controls) with best cut-off 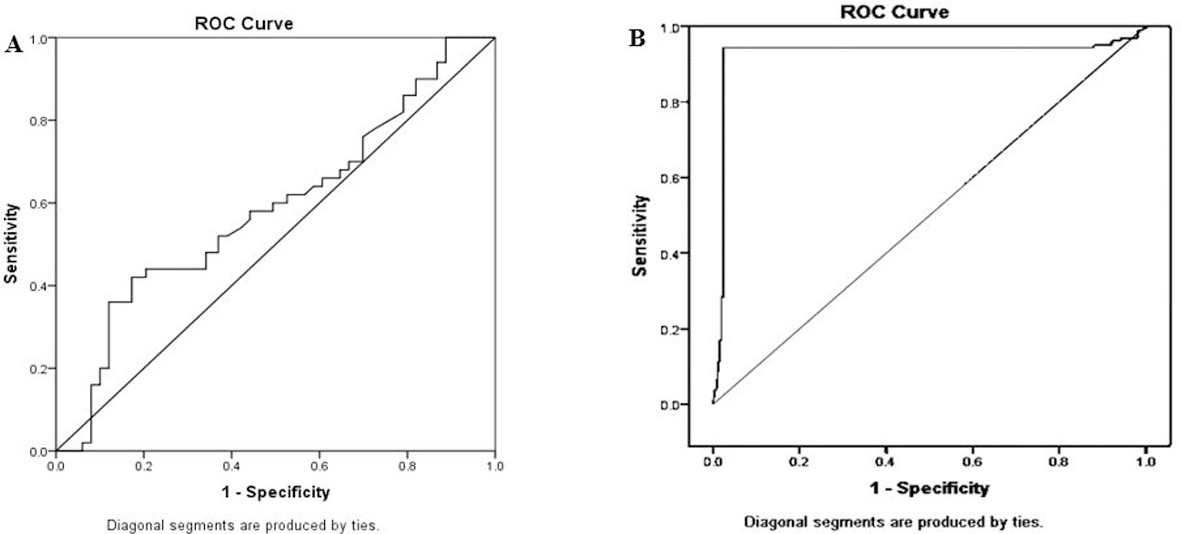
MEG3 fold change couldn’t differentiate grade III BC from lower grades (I
ROC curve of significance of MEG-3 ct value and MEG3 serum fold change differentiating grade III tumor from other grades (I 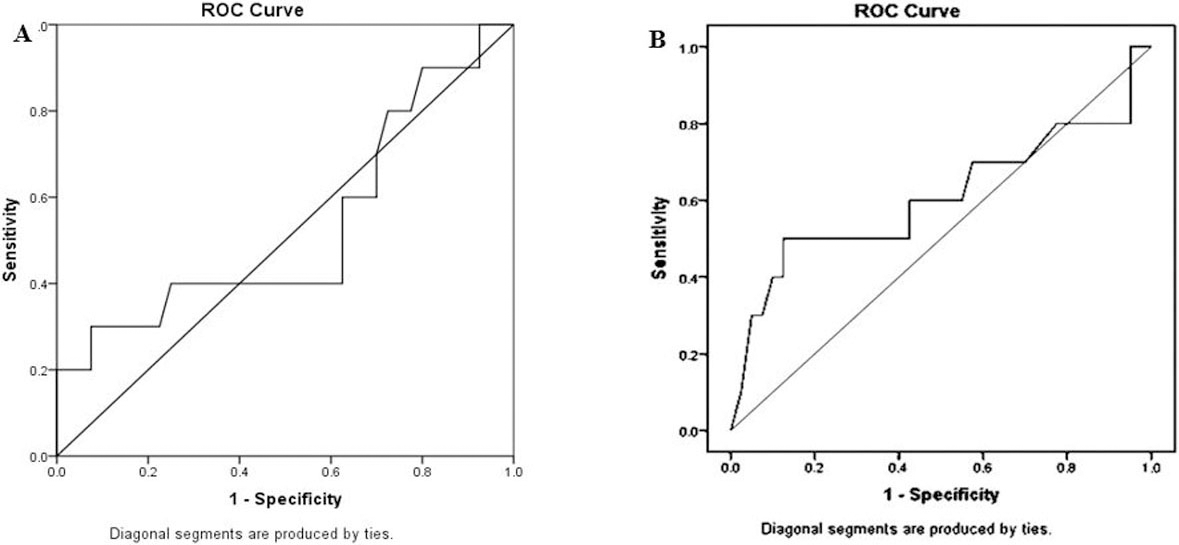
Breast cancer (BC) is considered one of the most prevalent cancers worldwide. In 2018, according to GLOBOCAN Statistics, there were 2.1 million newly diagnosed BC cases among females all over the world. That year, nearby 626,679 patients died of the disease, accounting for 6.6% of all cancer deaths, and ranking BC the second most common cause of death from cancer [27].
Long non-coding RNAs (lncRNAs) were defined as genomic noncoding transcripts longer than 200 nucleotides. Accumulating evidence suggested that lncRNAs have been involved in the initiation and propagation of BC. Furthermore, recent studies have indicated that SNPs in lncRNAs could potentially impact BC susceptibility by influencing gene expression level and biological pathways [5]. For example, Luo et al. reported that there is a robust relationship between rs4801078 and the risk of BC by altering the expression of Linc-ROR mRNA [28]. Also, Yan et al. identified an allelic regulation of rs10463297 on lncRNA SRA expression level and breast cancer susceptibility [29].
Science, lncRNA MEG3 was highlighted as a tumor suppressor gene in breast cancer tissues [11, 12, 13, 14], aberrantly expressed MEG3 can be detected in serum [15, 16], and SNP function prediction showed that rs7158663 might affect transcription factor binding sites [22]. So based on the evidence above, we proposed a hypothesis that the polymorphism of lncRNA MEG3 may be a regulatory SNP and contribute to the risk of BC through altering MEG3 serum expression level. We conducted a case-control study to weigh the serum expression level of MEG3 as a non-invasive biomarker in BC patients and to elucidate the effect of rs7158663 on this level and breast cancer susceptibility.
Our study was conducted on 150 BC, 95 FA patients, and 154 healthy subjects collected from the Egyptian population.
In our study, there were statistically significant differences between the age of FA (30.68
When we studied the distribution of rs7158663 G
Former studies explained the association between rs7158663 and risk of cancer or other diseases by performing silica analysis which predicted that SNP located in the binding sites of transcription factors and the mutant A allele of rs7158663 can bind to transcriptional factors [C/EBP
However, no study elucidate the correlation between MEG3 expression level and different rs7158663 genotypes and alleles.
To explore this point first, we measure serum MEG3 in studied groups, and we demonstrated that decreased serum MEG3 could be used as a non-invasive biomarker differentiating BC patients from FA and/or controls (
Regarding FA patients, significant upregulation of MEG3 was detected in the sera of FA patients when compared to controls (
Second, we performed a correlation study between rs7158663 genotypes, alleles, and serum MEG3 and elucidated that medians of MEG3 were 0.52, 0.41, and 0.37 for GG, GA and AA genotypes respectively (
On analyzing the relationship between MEG3 expression levels and clinicopathological characteristics of BC, a lower MEG3 level was significantly associated with patients with hypertension (
As well, lower MEG3 was significantly linked to higher T stage of TNM staging (
The current study is the first one that examined the relationship between rs7158663 SNP and clinical characters of BC and FA and by referral of these associations to MEG3 expression level which affected by the presence of different rs7158663 alleles, Our data revealed that rs7158663 was strongly associated with positive FH (in both BC and FA patients), younger age group (in FA), sequentially lower MEG3 was associated with positive FH and positive past history in the FA group. These findings were consistent with Hemminki et al., who reported that single nucleotide polymorphisms (SNPs) are inherited from parents, and they measure heritable events [45].
Also, we detected a significant correlation between A allele (and in turn lower MEG3) and positive ER/PR status and though this finding consented with their synergistic effects on BC cells proliferation, propagation and prognosis [46], it was against results demonstrated by Cui et al. who cited that higher MEG3 mRNA level was linked to the
Besides, A allele was significantly associated with the invasive lobular subtype of BC, higher T but with lower N. For interpretation of this result, we have to take into consideration that TNM is a complicated staging with different subcategories depending on multiple factories, including tumor size, ER, PR, HER2 status [24]. So the tumor may be N1or of lobular subtype but with higher stage according to other TNM factors. However, we failed to find significant differences between different tumor grades.
Moreover, there was a significant positive association between A allele (lower MEG3) and fatty liver in the BC group, which detected by ultrasound. It was demonstrated that high-fat diet treatment of mice (induced Non-Alcoholic Fatty Liver Disease model in mice) significantly increased serum lipid levels and reduced MEG3 expression [47].
Meanwhile, in the FA group, higher MEG3 was significantly associated with the younger age of patients, premenopausal patients having irregular menstruation, and with patients who do not use contraception. We noticed that these factors represent the most frequent risk factors for FA; however, for the definite explanation, larger scaled functional studies were needed [48].
Limitations faced the present study were: Firstly, these results were relied on genotyping of a single gene with a relatively small size of Breast cancer and Fibroadenoma patients, which need to be validated using an independent functional study. Secondly, our study was depended on a hospital-based study; therefore, potentially valuable sources of selection bias may be present. Thirdly, reproductive factors are major determinants of risk for breast cancer and fibroadenoma [48]; however, our study had incomplete data on these reproductive issues. Further studies are needed to distinguish the association between genetic variants in MEG3 and its expression level and reproductive period details (age at menarche, number of pregnancies, menopausal status and age,).
Conclusion
In summary, this study demonstrates that rs7158663 differentiate between BC patients from FA patients and/or healthy individuals. Also, MEG3 rs7158663 G
Higher serum MEG3 expression level was significantly associated with FA and with its risk factors.
Footnotes
Acknowledgments
All authors acknowledge help from all paramedical personals to accomplish and finalize the clinical research work.
Conflict of interest
The authors have no conflict of interest related to this study.
