Abstract
Breast cancer is the most common malignancies worldwide. LncRNA HOX transcript antisense intergenic RNA (HOTAIR) has been shown to promote progression and metastasis of various cancers, including breast cancer. This reasearch aimed to investigate the downstream regulatory pathways of HOTAIR in breast cancer. The levels of HOTAIR and miR-129-5p were examined in breast cancer tissues and SKBR3 and MCF7 cells by quantitative real-time PCR (qRT-PCR). Cell proliferation was examined by Cell Counting Kit-8 (CCK-8) assay. Cell migration and invasion were estimated by transwell assay. Epithelial-to-mesenchymal transition (EMT)-related markers (E-cadherin, N-cadherin and Vimentin) were measured by Western blot assay. The expression of Frizzled 7 (FZD7) was detected using qRT-PCR or Western blot assay. Bioinformatics analysis, luciferase reporter assay or RNA Immunoprecipitation (RIP) assay was performed to explore the molecular mechanism of HOTAIR in breast cancer. Xenograft analysis was utilized to evaluate the tumor growth in vivo. HOTAIR and FZD7 were upregulated, while miR-129-5p was down-regulated in breast cancer tissues and cells. Knockdown of miR-129-5p reversed the effect of HOTAIR knockdown on cell proliferation, migration, invasion and EMT. FZD7 restored the inhibition of miR-129-5p on breast cancer progression. Furthermore, HOTAIR was a sponge of miR-129-5p and FZD7 was a target of miR-129-5p. Knockdown of HOTAIR inhibited the tumor growth in vivo. HOTAIR facilitated breast cancer progression by regulating the miR-129-5p/FZD7 axis, indicating that HOTAIR may be a potential biomarker and therapeutic target for breast cancer.
Introduction
Breast cancer is the most common malignancies in the world and usually occurs in female [1]. The most important treatment for breast cancer is the early diagnosis, in which noncoding RNAs and exosomes can be used as new diagnostic and therapeutic biomarkers for monitoring breast cancer patients [2]. Therefore, finding new biomarkers for breast cancer will contribute to the diagnosis and treatment of breast cancer.
Long noncoding RNAs (lncRNAs) are more than 200 nucleotides in length and are a class of transcripts that do not encode proteins [3]. In recent years, increasing evidence has shown that lncRNAs affect a variety of biological processes through sponging miRNA [4]. LncRNA HOX transcript antisense intergenic RNA (HOTAIR) plays a critical role in various cancers, correlating with tumorigenesis, progression and growth [5]. Several studies have reported that high HOTAIR expression is associated with poor prognosis in cancer patients [6, 7, 8]. The previous study has demonstrated that HOTAIR functioned as a tumor promoter and induced tumor metastasis by regulating chondroitin sulfotransferase CHST15 in breast cancer [9]. However, the molecular mechanism of lncRNA HOTAIR in breast cancer requires further investigation.
MicroRNAs (miRNAs) are a group of highly conserved RNAs composed of 18–25 nucleotides. The miRNAs regulate the expression of genes at the post-transcriptional level and serve as biomarkers for many diseases [10]. Recent studies have suggested that miR-129-5p dysregulation inhibited tumorigenesis and progression in a variety of cancers, such as ovarian cancer [11], gastric cancer [12] and glioma [13]. In breast cancer, miR-129-5p blocked cell proliferation and metastasis by targeting CBX4 [14]. Many studies have elucidated that lncRNAs functioned as a sponge of microRNAs in breast cancer. Epithelial-to-mesenchymal transition (EMT) is considered to be the source of tumor invasion and metastasis in various cancers [15]. LncRNAs and miRNAs could regulate E-cadherin function through direct or by multiple factors controlling transcription of its gene [16]. However, the effect of the correlation between HOTAIR and miR-129-5p in breast cancer cell EMT has not been reported.
Frizzled 7 (FZD7) is the only evolutionary conserved in 10 Frizzled family members and acts as a therapeutic target for cancers [17]. Wnt signaling pathways are activated by Wnt ligand binding to Frizzled (FZD) receptors. FZD7 was upregulated as a Wnt receptor in various cancers, such as gastric cancer [18], triple negative breast cancer [19] and colorectal cancer [20]. In the present study, the expression levels of HOTAIR and miR-129-5p were detected in breast cancer tissues and cells. We also explored the underlying molecular mechanism of HOTAIR and miR-129-5p in breast cancer.
Materials and methods
Tissue samples
Breast cancer tissues (
Cell culture
Human normal mammary epithelial cell line (MCF-10A) and two breast cancer cell lines (SKBR3 and MCF7) were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were maintained in Dulbecco’s Modified Eagle Medium (DMEM; Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS; Gibco). All cells were cultured at 37
Cell transfection
Small interference RNA (siRNA) of HOTAIR (si-HOTAIR), shRNA of HOTAIR (sh-HOTAIR) and their corresponding negative controls (si-NC, sh-NC), miR-129-5p mimic (miR-129-5p) and the control mimic (miR-NC), miR-129-5p inhibitor (in-miR-129-5p) and the control inhibitor (in-miR-NC) were purchased from HanBio Technology (Shanghai, China). The pcDNA-HOTAIR (HOTAIR), pcDNA-FZD7 and the matched control (pcDNA) were purchased from RiboBio (Guangzhou, China). All plasmids and oligonucleotides were transfected by Lipofectamine 3000 reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions.
Quantitative real-time PCR (qRT-PCR)
TRIzol reagent (Invitrogen) was used to extract total RNA from tissues and cells following the manufacturer’s protocol. Total RNA was reversely transcribed into cDNA using Reverse Transcription System (Promega, Madison, WI, USA). RT-qPCR was carried out using SYBR Green PCR Master Mix (Thermo Fisher Scientific, Waltham, MA, USA) or TaqMan MicroRNA Assays (Applied Biosystems, Carlsbad, CA, USA). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and U6 were used as endogenous controls. Primers were as follows: HOTAIR forward, 5
Luciferase reporter assay
The full-length of HOTAIR containing miR-129-5p binding sites or the mutant was cloned into pGL3 plasmid (Promega) to establish pGL3-HOTAIR-WT and pGL3-HOTAIR-MUT vectors, respectively. Then, the corresponding vector was cotransfected with miR-129-5p mimic (miR-129-5p) or the control mimic (miR-NC) into SKBR3 and MCF7 cells. In addition, pGL3 plasmid harboring the wild-type or mutant FZD7 3’-UTR was cotransfected with miR-129-5p mimic (miR-129-5p) or the control mimic (miR-NC) into SKBR3 and MCF7 cells using Lipofectamine 3000 (Invitrogen). Luciferase activity was detected by Dual-Luciferase Reporter Assay System (Promega) at 48 h after transfection following the manufacturer’s protocol.
RNA immunoprecipitation (RIP) assay
EZ-Magna RIP Kit (Millipore, Billerica, MA, USA) was applied to perform RIP assay according to the manufacturer’s instructions. Briefly, transfected cells were lysed using RIP lysis buffer. The cell lysates were collected and incubated with magnetic beads conjugated with anti-Ago2 or the control anti-IgG for 4 h at 4
Cell counting Kit-8 (CCK-8) assay
Cell proliferation was assessed using Cell Counting Kit-8 (CCK-8; Dojindo, Kumamoto, Japan). Cells (2.0
Transwell assay
Transwell chambers were used to evaluate cell migration ability. However, transwell chambers for cell invasion were adhered with Matrigel (BD Biosciences, San Diego, CA, USA). Firstly, cells were suspended in serum-free medium, then seeded into the upper chamber containing 8
Western blot assay
Total protein was extracted with RIPA buffer (Thermo Fisher Scientific). Protein concentration was detected using the Easy II Protein Quantitative Kit (BCA) (TransGen Biotech, Beijing, China). Then proteins were separated by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore). The membranes were blocked by 5% non-fat milk for 2 h at room temperature and then incubated at 4
The expression levels of HOTAIR and miR-129-5p in breast cancer tissues and cells. (A and C) The expression levels of HOTAIR and miR-129-5p in breast cancer tissues (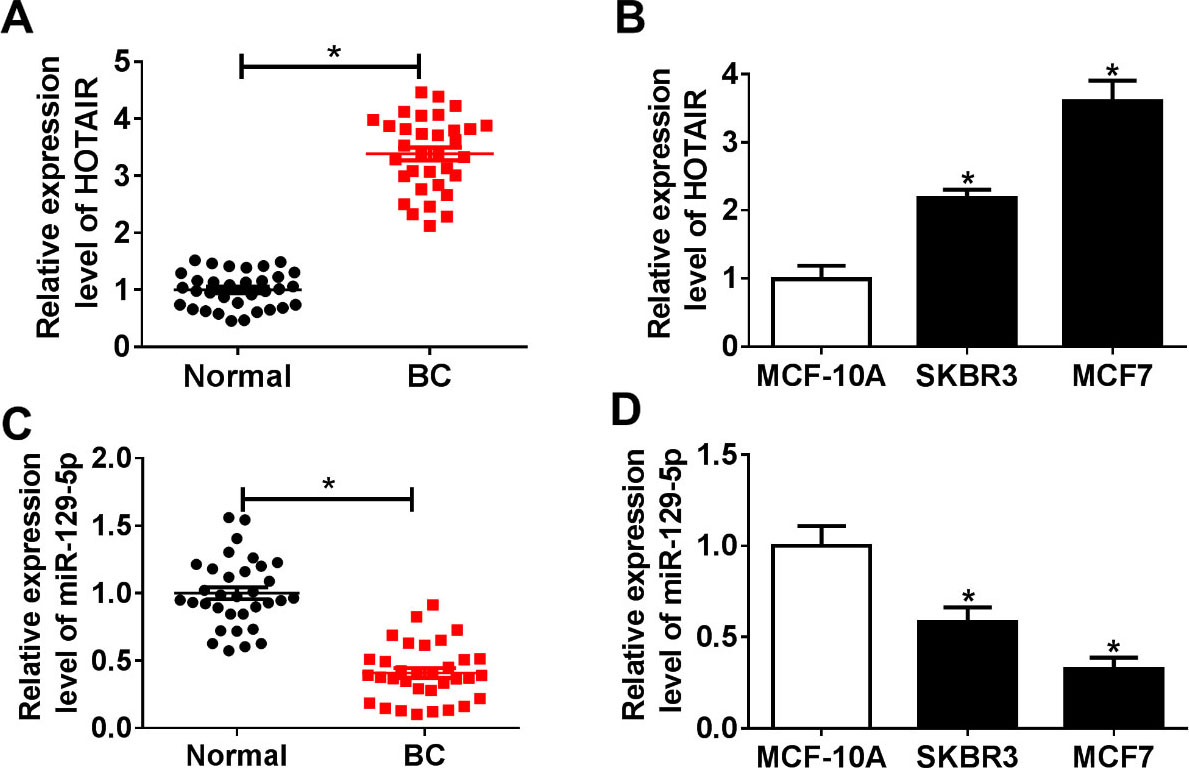
Xenograft nude mouse model was established with BALB/c nude male mice (6 weeks old). SKBR3 cells transfected with sh-NC or sh-HOTAIR were subcutaneously injected into nude mice. Tumor volume was monitored every 7 d. After mice were sacrificed, tumor weight was detected, and partial breast tissues were snap-frozen for RNA and protein extraction. The experiment was approved by the Animal Ethics Committee of The First Affiliated Hospital of Nanchang University.
Statistical analysis
All data were expressed as mean
Results
LncRNA HOTAIR was upregulated while miR-129-5p was down-regulated in breast cancer tissues and cells
Firstly, we examined the expression levels of HOTAIR and miR-129-5p in breast cancer tissues and found that HOTAIR expression in breast cancer (BC) tissues was dramatically higher than that in adjacent normal tissues (Fig. 1A), while miR-129-5p expression was significantly lower in BC tissues than that in adjacent normal tissue (Fig. 1C). Next, the levels of HOTAIR and miR-129-5p in different breast cancer cell lines (SKBR3 and MCF7) were detected. The results showed that HOTAIR expression was markedly increased and miR-129-5p expression was remarkably decreased in breast cancer cell lines (SKBR3 and MCF7) compared to normal mammary epithelial cells (MCF-10A) (Fig. 1B and D). These data speculated that lncRNA HOTAIR might be a tumor promotor in breast cancer.
LncRNA HOTAIR directly regulated miR-129-5p expression
Previous studies have reported that lncRNAs act as competing endogenous RNAs (ceRNAs) to competitively bind with miRNAs [21]. In this study, LncBase Predicted v.2 predicted that HOTAIR directly targeted miR-129-5p (Fig. 2A). In order to investigate the targeting relationship, luciferase reporter assay was performed. The results exhibited that luciferase activity was evidently decreased in SKBR3 and MCF7 cells co-transfected with HOTAIR-WT and miR-129-5p mimic (Fig. 2B and C). RIP assay was performed to further validate that HOTAIR targeted miR-129-5p. The results demonstrated that HOTAIR was obviously pulled down in miR-129-5p mimic group compared with the control group (Fig. 2D). Next, the expression levels of HOTAIR and miR-129-5p were negatively correlated in breast cancer tissues (Fig. 2E). Furthermore, we examined the expression levels of HOTAIR and miR-129-5p in SKBR3 and MCF7 cells transfected with pcDNA, pcDNA-HOTAIR (HOTAIR), si-NC or si-HOTAIR. The results showed that HOTAIR overexpression remarkedly increased HOTAIR expression and decreased miR-129-5p expression. However, HOTAIR was strikingly down-regulated and miR-129-5p was greatly upregulated in si-HOTAIR group compared to si-NC group (Fig. 2F and G). These results demonstrated that HOTAIR negatively regulated miR-129-5p expression.
HOTAIR directly targeted miR-129-5p. (A) The putative binding sites of miR-129-5p in HOTAIR were exhibited. (B and C) Luciferase activity was examined in SKBR3 and MCF7 cells cotransfected with pGL3-HOTAIR-WT or pGL3-HOTAIR-MUT and miR-NC or miR-129-5p mimic (miR-129-5p). (D) The enrichment of HOTAIR was detected in SKBR3 and MCF7 cells transfected with miR-NC or miR-129-5p. (E) The correlation between expression of HOTAIR and miR-129-5p in breast cancer tissues was analyzed. (F and G) The expression levels of HOTAIR and miR-129-5p were detected in SKBR3 and MCF7 cells transfected with pcDNA, pcDNA-HOTAIR (HOTAIR), si-NC or si-HOTAIR. *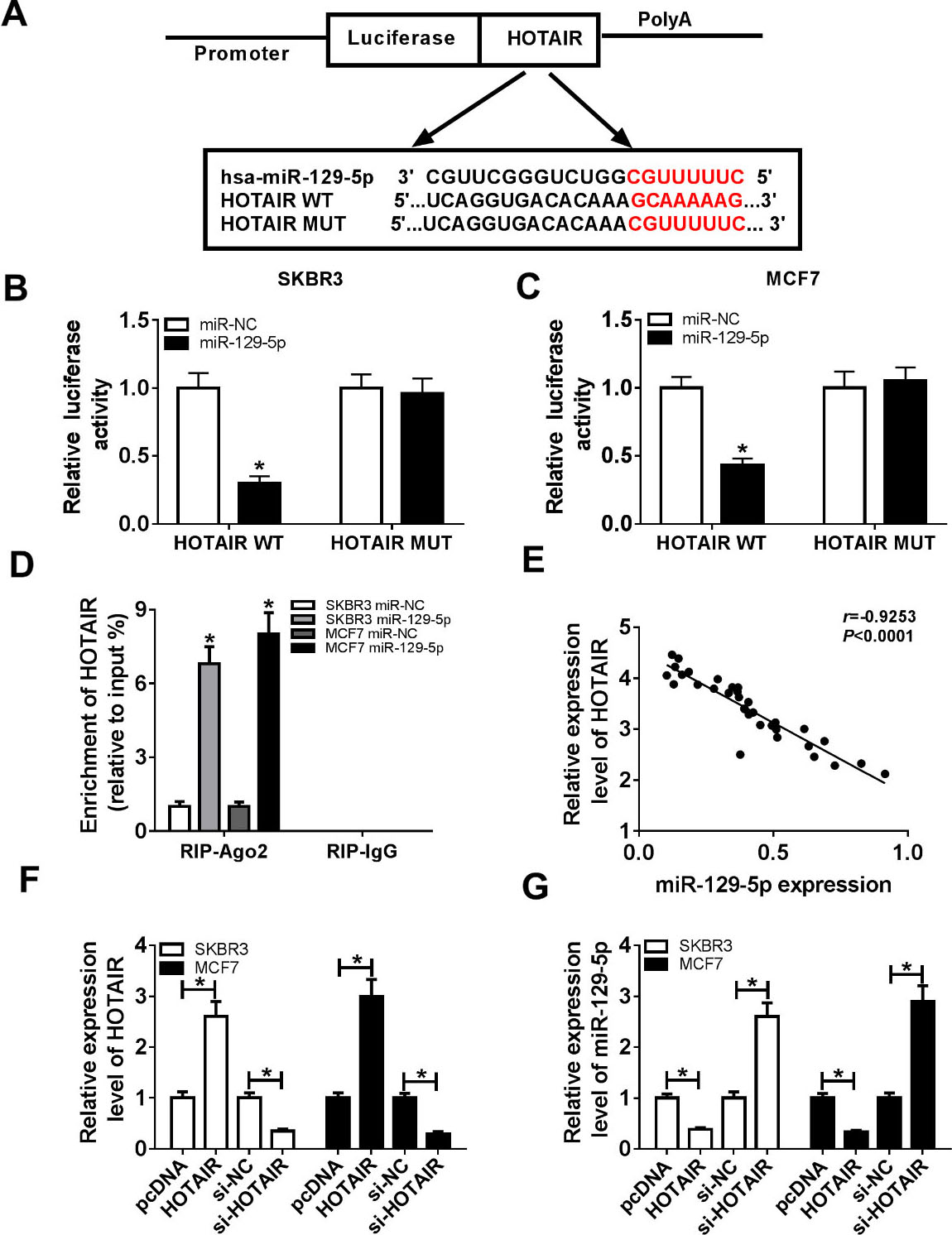
MiR-129-5p knockdown reversed the effect of HOTAIR on proliferation, migration, invasion and EMT of breast cancer cells. (A-E) SKBR3 and MCF7 cells were transfected with si-NC, si-HOTAIR, si-HOTAIR+in-miR-NC or si-HOTAIR+in-miR-129-5p. (A and B) Cell proliferation was examined by CCK-8 assay. (C and D) Cell migration and invasion were evaluated using transwell assay at 24 h after transfection. (E) The protein levels of EMT-related proteins (E-cadherin, N-Cadherin, and Vimentin) were measured by Western blot. *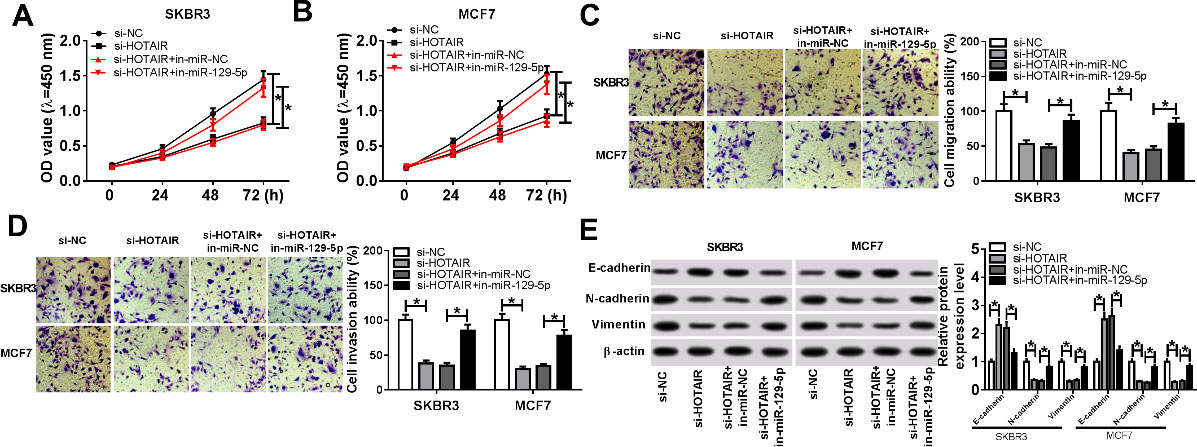
To determine the functional effect of HOTAIR on breast cancer progression, rescue experiments were performed. First, SKBR3 and MCF7 cells were transfected with si-NC, si-HOTAIR, si-HOTAIR+in-miR-NC or si-HOTAIR+in-miR-129-5p. CCK-8 assay revealed that cell proliferation was evidently restrained in si-HOTAIR group, whereas miR-129-5p inhibitor returned cell proliferation ability (Fig. 3A and B). Transwell assay indicated that HOTAIR knockdown obviously restricted cell migration and invasion, whereas miR-129-5p inhibitor reversed the inhibition of HOTAIR knockdown on cell migration and invasion abilities (Fig. 3C and D). In addition, the protein level of E-cadherin was strikingly increased and the protein levels of N-cadherin and Vimentin were specially decreased in si-HOTAIR group compared to si-HOTAIR+in-miR-129-5p group (Fig. 3E). In summary, these data revealed that miR-129-5p knockdown could restored the inhibition of HOTAIR knockdown on breast cancer progression.
FZD7 was a target of miR-129-5p
To explore the mechanism of miR-129-5p in breast cancer, we predicted the target genes of miR-129-5p using bioinformatics software. According to previous research, FZD7 predicted by microT-CDS was selected as the research target (Fig. 4A). Firstly, luciferase reporter assay was performed to validate whether FZD7 was a target of miR-129-5p. The results suggested that mature miR-129-5p dramatically decreased luciferase activity in FZD7 3’-UTR-WT group, but not in FZD7 3’-UTR-MUT group (Fig. 4B and C). In addition, qRT-PCR results showed that FZD7 expression was markedly increased in breast cancer (BC) tissues and cell lines (SKBR3 and MCF7) compared to adjacent normal tissues and normal mammary epithelial cells (MCF-10A) (Fig. 4D and E). The expression levels of miR-129-5p and FZD7 were negatively correlated in breast cancer tissues (Fig. 4F). Furthermore, SKBR3 and MCF7 cells were transfected with miR-NC or miR-129-5p mimic (miR-129-5p) and Western blot showed that FZD7 expression was apparently down-regulated in miR-129-5p mimic group (Fig. 4G). These data indicated that FZD7 was a direct target of miR-129-5p in breast cancer.
FZD7 was a target of miR-129-5p. (A) The predicted binding sites for miR-129-5p and FZD7 3’-UTR were shown. (B and C) Luciferase activity was examined in SKBR3 and MCF7 cotransfected with FZD7 3’-UTR-WT or FZD7 3’-UTR-MUT plasmids and miR-NC or miR-129-5p. (D) Relative expression of FZD7 was examined in breast cancer tissues and adjacent normal tissues. (E) FZD7 expression was detected in normal mammary epithelial cells (MCF-10A) and breast cancer cell lines (SKBR3 and MCF7) by qRT-PCR. (F) The correlation between miR-129-5p and FZD7 in breast cancer tissues were examined. (G) FZD7 expression was detected in SKBR3 and MCF7 cells transfected with miR-NC or miR-129-5p by Western blot. *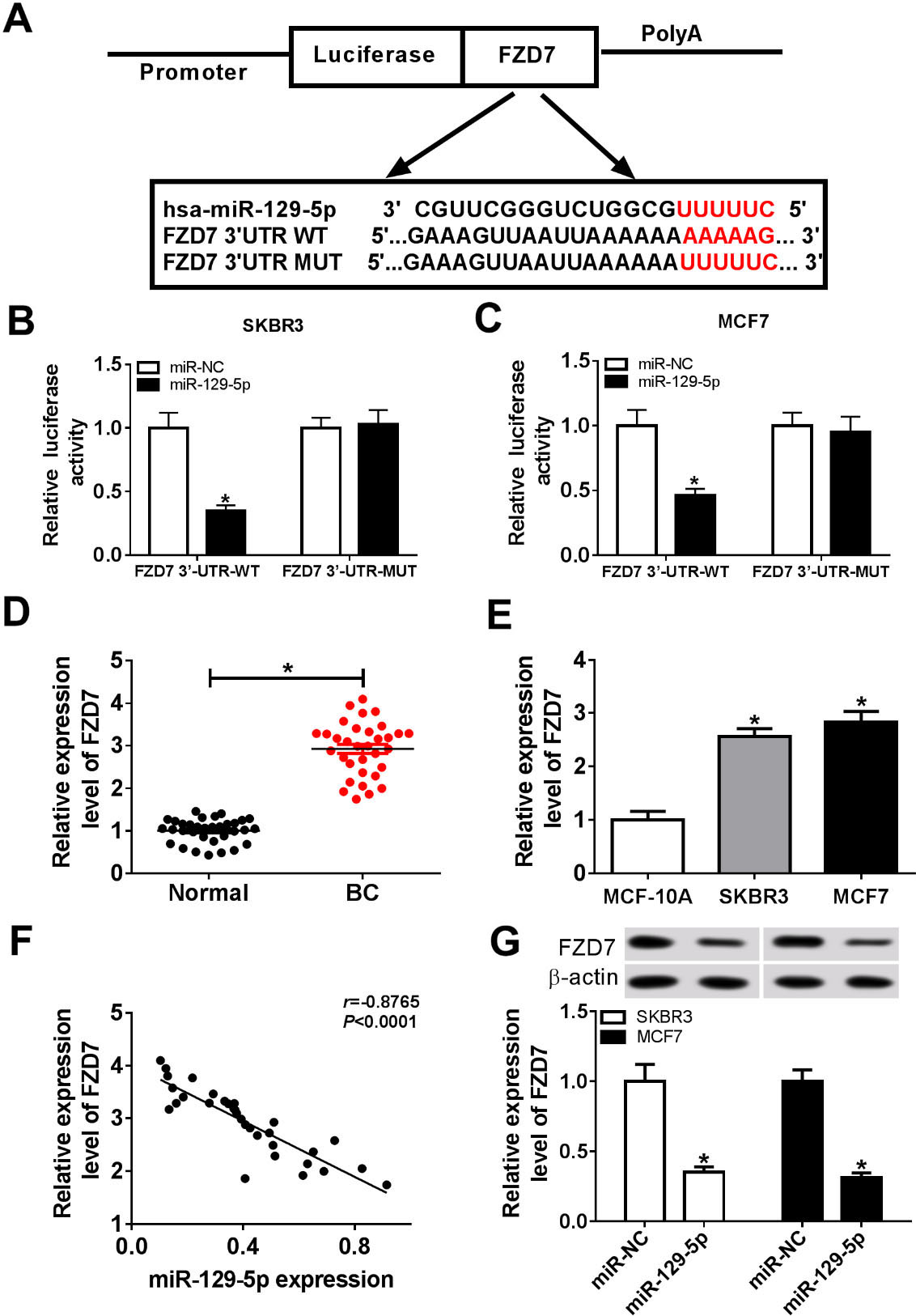
To investigate the effect of miR-129-5p on breast cancer progression, SKBR3 and MCF7 cells were transfected with miR-NC, miR-129-5p, miR-129-5p+pcDNA or miR-129-5p+FZD7. CCK-8 analysis showed that cell proliferation was drastically inhibited in miR-129-5p group, whereas returned in miR-129-5p+FZD7 group (Fig. 5A and B). Transwell assay revealed that cell migration and invasion of cells transfected with miR-129-5p were markably restrained, while cells transfected with miR-129-5p+FZD7 reversed the inhibition (Fig. 5C and D). To evaluate the effect of miR-129-5p on EMT, Western blot assay was performed. The results exhibited that miR-129-5p mimic evidently enhanced the protein level of E-cadherin and weakened the protein levels of N-cadherin and Vimentin, while overexpression of miR-129-5p and FZD7 restored EMT-related protein levels (Fig. 5E). In a word, these data demonstrated that FZD7 reversed the inhibition of miR-129-5p on proliferation, migration, invasion and EMT of breast cancer cells.
FZD7 reversed the effect of miR-129-5p on breast cancer progression. (A–E) SKBR3 and MCF7 cells were transfected with miR-NC, miR-129-5p, miR-129-5p+pcDNA or miR-129-5p+FZD7. (A and B) Cell proliferation was examined by CCK-8 assay. (C and D) Transwell assay was performed to evaluate cell migration and invasion at 24 h after transfection. (E) Western blot analysis was used to detect the protein levels of EMT-related proteins (E-cadherin, N-Cadherin, and Vimentin). *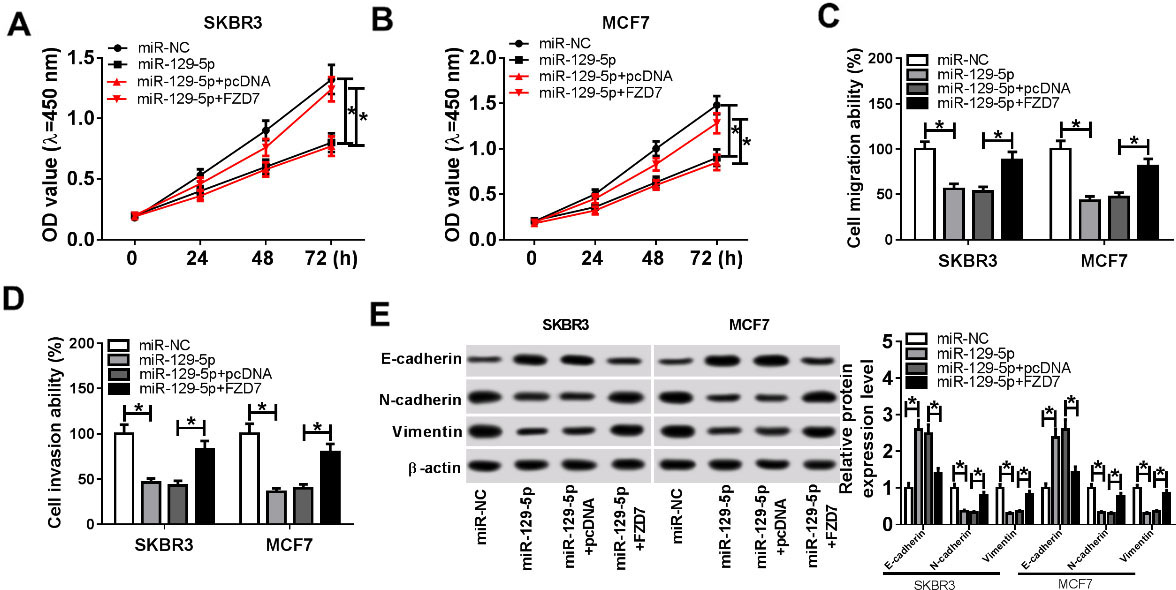
To investigate the effect of HOTAIR and miR-129-5p on FZD7 expression, qRT-PCR and Western blot assays were performed. Firstly, SKBR3 and MCF7 cells were transfected with si-NC, si-HOTAIR, si-HOTAIR+in-miR-NC or si-HOTAIR+in-miR-129-5p. The results showed that the mRNA and protein levels of FZD7 in si-HOTAIR group were dramatically lower than those in si-NC group, whereas the mRNA and protein levels of FZD7 in si-HOTAIR+in-miR-129-5p group recovered (Fig. 6A and B). All these data indicated that HOTAIR targeted miR-129-5p to elevate the expression of FZD7.
FZD7 expression was modulated through HOTAIR and miR-129-5p in breast cancer cells. (A–B) SKBR3 and MCF7 cells were transfected with si-NC, si-HOTAIR, si-HOTAIR+in-miR-NC or si-HOTAIR+in-miR-129-5p. (A) Relative mRNA expression of FZD7 was detected by qRT-PCR. (B) Relative protein expression of FZD7 was measured by Western blot. *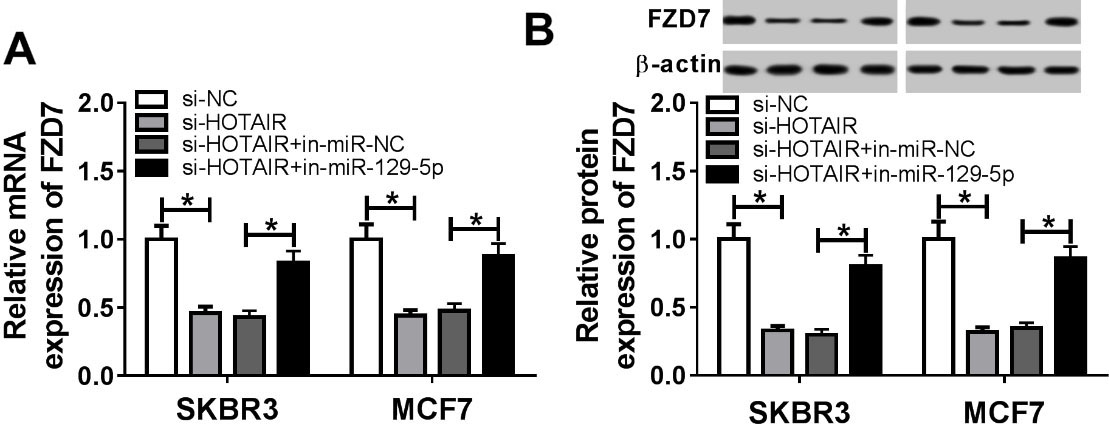
We established xenograft nude mouse model to evaluate the effect of HOTAIR on tumor growth of breast cancer. SKBR3 cells were transfected with sh-NC or sh-HOTAIR. Next, transfected cells were injected subcutaneously into nude mice. The results suggested that tumor volume was significantly decreased in sh-HOTAIR group compared to sh-NC group (Fig. 7A). After mice were sacrificed, sh-HOTAIR markedly reduced tumor weight (Fig. 7B). The results of qRT-PCR or Western blot assay showed that the expression of HOTAIR and FZD7 was obviously lower, and miR-129-5p expression was notably higher in sh-HOTAIR group than that in sh-NC group (Fig. 7C–E). Thus, these data indicated that HOTAIR knockdown inhibited the tumor growth in vivo.
LncRNA HOTAIR regulated tumor growth in vivo. (A-E) SKBR3 cells transfected with sh-NC or sh-HOTAIR were subcutaneously injected into nude mice. (A) Tumor volume was measured every 7 d after injection. (B) After mice were sacrificed, tumors were weighed. (C and D) The expression levels of HOTAIR and miR-129-5p were measured by qRT-PCR. (E) The protein level of FZD7 was examined by Western blot. *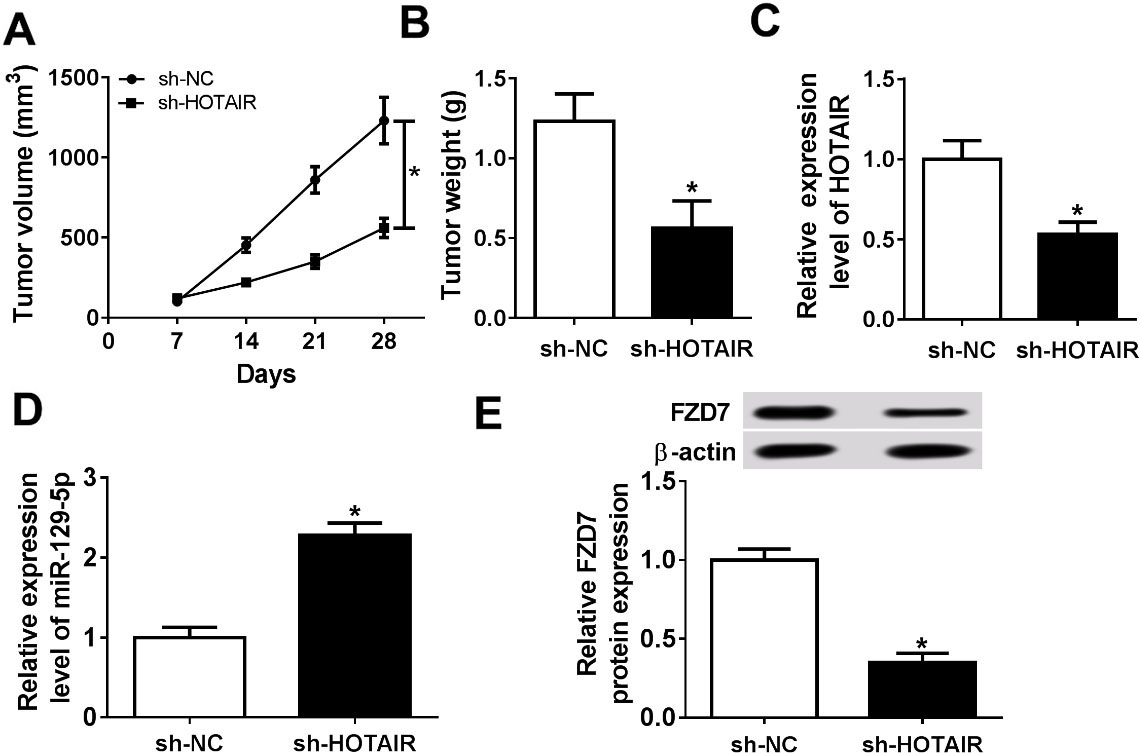
Breast cancer is a common serious disease in the world. Recent studies have shown that the dysregulation of lncRNAs plays a vital role in various human cancers, including breast cancer [22]. Recent studies have demonstrated that lncRNA HOTAIR was upregulated in various cancers and was associated with metastasis and poor prognosis [23]. Han et al. demonstrated that HOTAIR suppressed PTEN expression by recruiting EZH2 and induced cell proliferation, migration and invasion in breast cancer [24]. Zhao et al. revealed that HOTAIR facilitated cell proliferation and metastasis and inhibited cell apoptosis via regulating the HOTAIR/miR-20a-5p/HMGA2 axis in breast cancer [25]. In addition, HOTAIR regulated breast cancer cell proliferation, apoptosis, migration and invasion via modulating the P53/Akt/JNK signaling pathway [26]. Previous study has shown that HOTAIR knockdown could promote the sensitivity of breast cancer cells to ionizing radiation, indicating that HOTAIR was a novel radiosensitizing target [27]. Consistent with previous studies, the present research demonstrated that HOTAIR was markedly upregulated in breast cancer tissues and cells. HOTAIR directly targeted miR-129-5p, and knockdown of HOTAIR blocked cell proliferation, migration, invasion and EMT in breast cancer.
In this study, HOTAIR directly targeted miR-129-5p as a competing endogenous RNA. Accumulating evidence suggested that miR-129-5p functioned as a tumor inhibitor in various cancers, including triple-negative breast cancer [28]. In hepatocellular carcinoma, lncRNA PCAT-1 induced cell metastasis through sponging miR-129-5p to elevate the expression of high mobility group box 1 (HMGB1) [29]. MiR-129-5p played an important role in regulating EMT and multi-drug resistance (MDR) in breast cancer cells [30]. Luo et al. showed that miR-129-5p inhibited autophagy and heightened radiosensitivity of breast cancer cells via targeting HMGB1 [31]. The present study confirmed that HOTAIR negatively regulated the expression of miR-129-5p. It was then shown that miR-129-5p knockdown could abolish the inhibition of HOTAIR knockdown on proliferation, migration, invasion and EMT of breast cancer cells.
In addition, the targets of miR-129-5p were predicted by online prediction software. FZD7 was selected as a putative target because FZD7 was reported to act as a tumor promoter in various cancers [18, 19, 20]. FZD7, as Wnt pathway receptor, triggered cell proliferation and invasion by activating the Wnt signaling pathway in triple negative breast cancer [32]. Xie et al. found that recombinant soluble peptide fragment (rhFzd7) could suppress cell proliferation and invasion through antagonizing FZD7 by competitively binding with Wnt ligands, indicating that rhFzd7 could be used to screen anti-FZD7 antibody in triple negative breast cancer [33]. Consistent with this study, FZD7 expression was distinctly increased in breast cancer tissues and cells, and FZD7 reversed the inhibition of miR-129-5p on breast cancer progression.
In conclusion, lncRNA HOTAIR expression was remarkably upregulated in breast cancer tissues and cells. HOTAIR elevated the expression of FZD7 through sponging miR-129-5p to induce cell proliferation, migration, invasion and EMT in breast cancer cells. HOTAIR knockdown blocked the tumor growth in vivo. Therefore, these results indicated that the HOTAIR/miR-129-5p/FZD7 axis might provide a novel therapeutic target for breast cancer treatment. Nevertheless, there are some limitations in this study. The association between HOTAIR expression and clinical features of breast cancer is lacking for some reasons.
