Abstract
BACKGROUND:
Long noncoding RNA HOTAIR has been detected in the serum of patients with various malignances and may be served as novel biomarker for diagnosis and prognosis prediction of breast cancer. However, the value of circulating HOTAIR to predict the response to neoadjuvant chemotherapy (NAC) remains unclear.
OBJECTIVE:
In the present study, we analyzed whether pretreatment circulating HOTAIR levels predict the response to NAC and investigated prognostic impact of circulating HOTAIR on disease-free survival (DFS) in breast cancer patients treated with NAC.
METHODS:
Circulating HOTAIR levels in the serum of 112 breast cancer patients before NAC were measured using quantitative real-time PCR. The correlation of circulating HOTAIR with the clinicopathologic status and the response to NAC were analyzed. Kaplan-Meier survival analysis and log-rank test were used to estimate the DFS.
RESULTS:
In 112 serum samples obtained before NAC, high circulating HOTAIR was associated with larger tumor size, more positive lymph nodes as well as more distant metastasis. However, there was no significant correlation between the circulating HOTAIR levels and age, Ki67 status or hormone receptor. Furthermore, patients with high circulating HOTAIR achieved less clinical response as well as pathologic complete response than those with low circulating HOTAIR (
CONCLUSIONS:
High circulating HOTAIR level correlates with less response to neoadjuvant chemotherapy as well as a worse prognosis in breast cancer patients. Therefore, the present study provides a favorable basis to use circulating HOTAIR as a predictor of neoadjuvant chemotherapy response.
Introduction
Neoadjuvant chemotherapy (NAC) before surgery has been initially used as the standard treatment in inoperable locally advanced breast cancer [1]. In nowadays, it is also frequently adopted in patients with early-stage operable breast cancer to obtain primary tumor or lymph nodes shrinkage, improve the chance of breast-conserving surgery and provide an opportunity to observe the response to chemotherapy in vivo. Since the combined anthracycline and taxane regimes are widely used in clinical practice, the pathologic complete response (pCR) rate has been elevated leading to a better disease-free survival [2]. However, drug resistance to chemotherapeutic agents is still the most common reason for poor response and prognosis because of heterogeneity of the disease. Unfortunately, the molecular basis underlies drug resistance is largely undefined. Therefore, exploring novel biomarkers to predict the response to NAC will facilitate the selection of patients that will benefit from NAC.
Long noncoding RNA (lncRNA) is a kind of endogenous RNAs, which are more than 200 nucleotides in length and cannot be coded to protein [3]. Aberrant expression of lncRNAs has been reported in various malignancies, such as breast cancer [4], nasopharyngeal cancer [5] and ovarian cancer [6] and can be served as useful prognostic biomarkers. A large body of evidence shows that dysregulated lncRNA expression levels are involved in various malignant biological processes, including carcinogenesis, cell proliferation, drug resistance, and metastasis [7, 8, 9]. LncRNAs may be served as biomarkers for diagnosis and treatment of breast cancer. Among various lncRNAs previous identified, HOTAIR (HOX Transcript Antisense Intergenic RNA) attracted much interest due to its consistent and significant overexpression in breast cancer and has been proved to be a predictor for diagnosis and prognosis [10, 11]. Moreover, HOTAIR has been demonstrated to mediate radiosensitivity, endocrine resistance and chemoresistance in a variety of ways [12, 13], suggesting that HOTAIR may serve as a useful biomarker to predict the response to chemotherapy.
Recently, lncRNAs has been detected in the blood of cancer patients and demonstrated to be associated with a poor prognosis [14, 9, 15]. More specifically, elevated circulating HOTAIR level is correlated with disease progression and poor prognosis of various malignances [11, 16, 17]. Furthermore, some circulating lncRNAs, such as MALAT1 and MEG3, have been proved to be associated with chemotherapy response and survival [18, 19]. However, whether circulating HOTAIR can be used as a valid predictor for NAC response remains unclear. In the present study, we investigated the association of circulating HOTAIR with NAC response and the predictive value for prognosis in patients with breast cancer.
Material and methods
Patients and chemotherapy regimens
We identified 112 female breast cancer patients without metastasis who received NAC at the first affiliated hospital of Jinan university between January 2012 and December 2016. The present study was given ethical approval with ethical committee of the first affiliated hospital of Jinan university and informed consent was obtained from all participants. The median age was 45 years (27–72 years), the clinical stage was II or III, and all patients were diagnosed with primary invasive breast cancer according to pathological examinations by core needle biopsies before treatment. All patients received 3 or more than 3 cycles of NAC after diagnosis and underwent breast-conserving surgery or modified radical mastectomy. All patients presented with normal hepatic, renal, and bone marrow function. Patients with ductal or lobular carcinoma in situ with or without microinvasion, patients with stage IV breast cancer or inflammatory breast cancer, patients with missing information on pathology results and patients who were lost during follow-up were excluded.
Each patient’s medical history, age, chemotherapy regimen, chemotherapy cycles, surgical method, histopathologic results (such as histology grade), lymph nodes status (such as number of positive lymph nodes), hormonal status and Her-2 status were reviewed. The T stage, N stage and M stage before and after surgery are obtained according to the American Joint Committee on Cancer (AJCC).
Taxane-based and/or anthracyclin-based chemotherapy regimens in NAC settings every 3 weeks are as follows: epirubicin and cyclophosphamide (EC, E: 90 mg/m
Sample collection
Serum samples from female invasive breast can- cer patients and healthy women were obtained before treatment. A total of 4.0 ml peripheral blood samples were collected. Each sample was then incubated for 60 mins, and centrifuged at 2000 rpm for 15 mins at room temperature. The upper-layer supernatant was immediately transferred to an RNase-free Eppendorf tube and was stored at
RNA extraction and quantitative real-time PCR analysis (qRT-PCR)
Total RNA was extracted from the serum samples using TRIzol (Invitrogen, USA). The first-strand cDNA was obtained using reverse transcription with Invitrogen reverse transcription reagent kit according to the manufacturer’s instructions. The qRT-PCR was performed in a LightCycler 480 (Roche, Switzerland) using the SYBR (TaKaRa, Japan) according to the manufacturer’s instructions. Following an initial denaturation at 95
Evaluation of response to chemotherapy
The same method of assessment and the same technique should be used to characterize the tumor at baseline and during follow-up. The response to NAC was assessed according to tumors (RECIST) 1.1: (a) Complete Response (CR) was defined as a complete disappearance of the tumor mass, (b) Partial Response (PR) was defined as at least a 30% reduction in the sum of diameters of tumor mass, taking as reference the baseline sum diameters, (c) Progressive Disease (PD) was defined as at least a 20% increase in the sum of diameters of the tumor, the appearance of one or more new lesions is also considered progression and (d) Stable Disease (SD) was defined as a change that neither sufficient shrinkage to qualify for PR nor sufficient increase to qualify for PD, taking as reference the smallest sum diameters. PCR was defined as no invasive disease in the breast and no residual tumor in axillary lymph nodes.
Statistical analysis
All statistical analyses were done using SPSS for Windows version 19.0 (SPSS, Chicago). Student’s
Circulating HOTAIR expression in the serum of breast cancer patients and normal control. Circulating HOTAIR expression was significantly higher in breast cancer patients than that in normal control (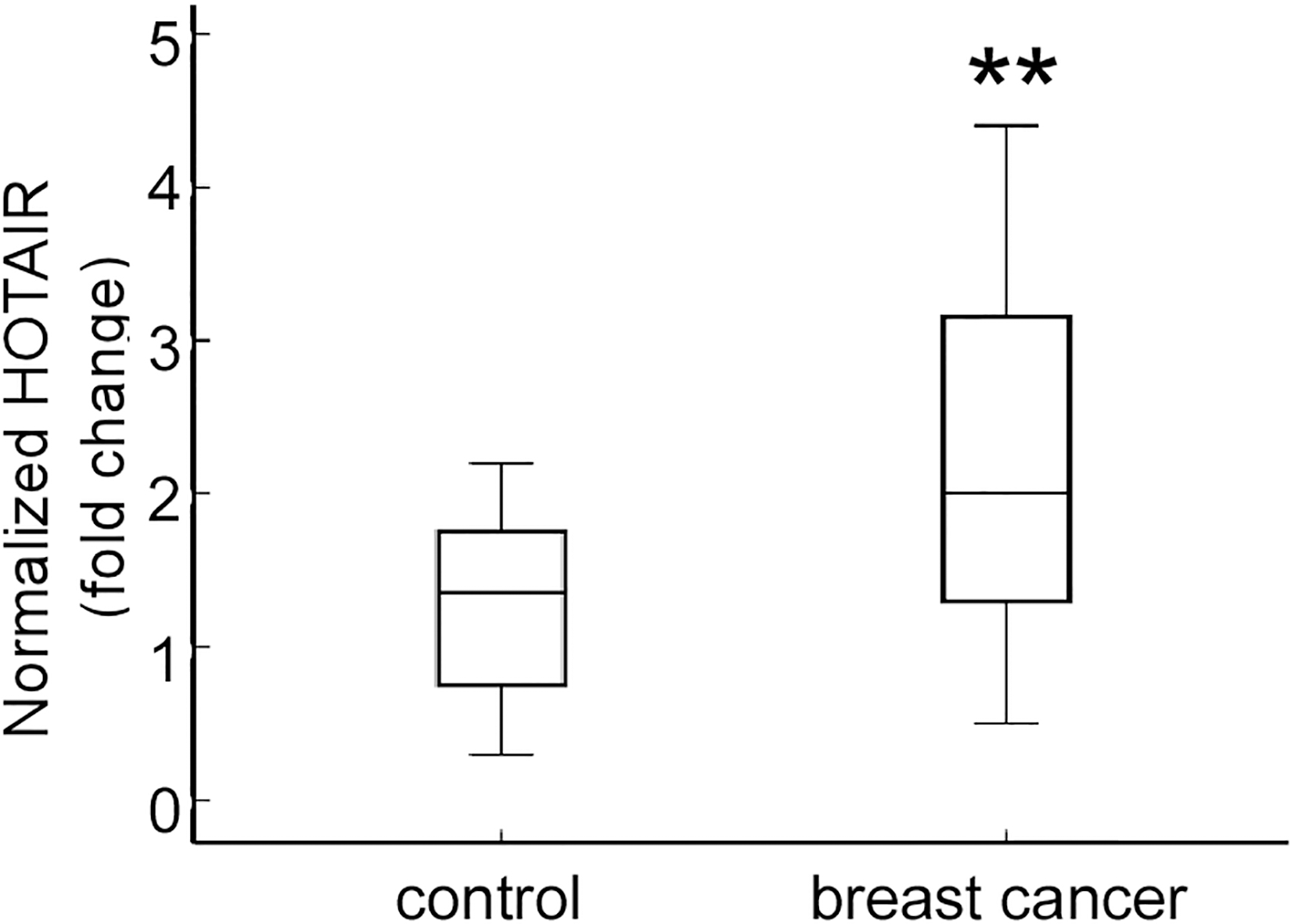
Association between circulating HOTAIR expression and clinicopathologic features of breast cancer patients No. (%)
The association between circulating HOTAIR expression and the clinicopathological characteristics of 112 breast cancer patients treated with neoadjuvant chemotherapy
To validate whether HOTAIR is detectable in the serum of breast cancer patients, quantitative real-time PCR (qRT-PCR) was performed to determine the expression levels of HOTAIR in patients with clinical stage II/III breast cancer or healthy women as normal control (
Treatment paradigms between patients with high and low circulating HOTAIR expression No. (%)
Treatment paradigms between patients with high and low circulating HOTAIR expression No. (%)
EC: epirubicin and cyclophosphamide, TC: docetaxel and cyclophosphamide, ECT: epirubicin, cyclophosphamide and docetaxel, TCH: docetaxel, carboplatin and trastuzumab.
To investigate whether circulating HOTAIR level is related to clinicopathological features of invasive ductal breast cancer, we measured relative circulating HOTAIR expression in the serum of 112 breast cancer patients before NAC at different clinical stages using qRT-PCR. The expression levels of circulating HOTAIR were categorized into high HOTAIR group (above the cut-off value) and low HOTAIR group (below the cut-off value) using the median value as the cut-off value (1.5). Next, we correlated circulating HOTAIR levels with the clinicopathological status of 112 breast cancer patients. The expression levels of HOTAIR were associated with larger tumor size (
Since anthracycline and taxane are widely used in NAC, we further analyzed the correlation of circulating HOTAIR levels with the response to anthracycline and/or taxane containing neoadjuvant chemotherapy. The chemotherapy regimens and cycles are as shown in Table 2.
The clinical responses were evaluated according to the Response Evaluation Criteria In Solid Tumors (RECIST) 1.1. In the present study, patients with CR and PR were categorized as responders, and patients with SD and PD as non-responders. An elevated response rate was observed in patients with lower pretreatment circulating HOTAIR. As is shown in Table 3, 14 patients (12.5%) achieved a CR and 30 patients (26.8%) achieved a PR in low circulating HOTAIR group. The objective clinical response rate is 39.3%. On the contrary, only 10 patients (8.9%) achieved a CR and 18 patients (16.1%) achieved a PR in high circulating HOTAIR group. The objective clinical response rate is 25%. Meanwhile, SD and PD rate were higher in high circulating HOTAIR group than that in low circulating HOTAIR group (
Clinical response to neoadjuvant chemotherapy in patients with high or low circulating HOTAIR expression No. (%)
Clinical response to neoadjuvant chemotherapy in patients with high or low circulating HOTAIR expression No. (%)
CR: complete response, PR: partial response, SD: stable disease, PD: progressive disease.
Percentage of pCR in patients stratified by circulating HOTAIR expression. Patients with low circulating HOTAIR expression showed relatively higher pCR rate than those with high circulating HOTAIR (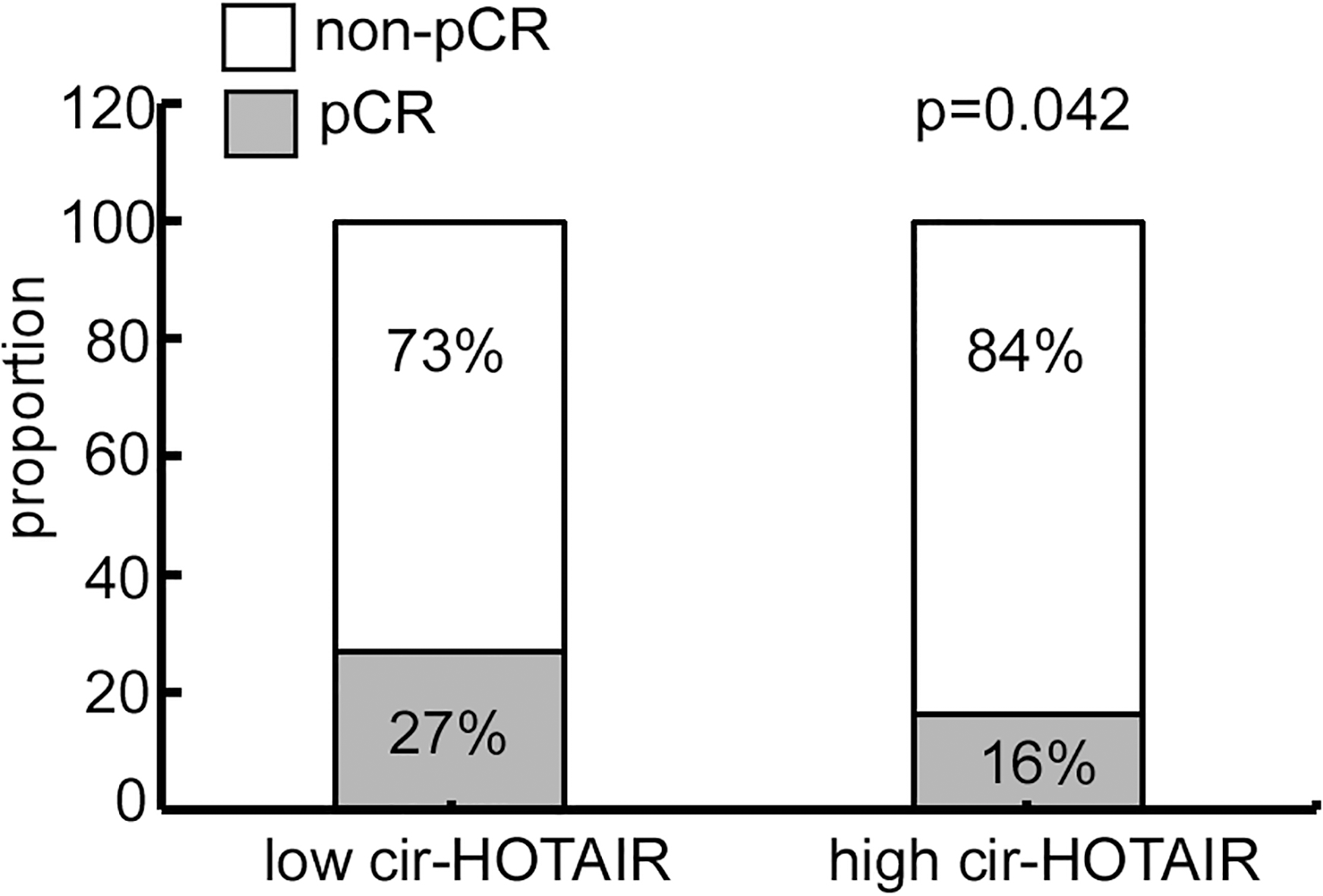
Chemotherapy resistance and tumor recurrence are responsible for the poor survival of breast cancer patients. To further investigated whether circulating HOTAIR levels were associated with tumor metastasis, qRT-PCR analysis was performed to determine the expression level of serum HOTAIR in breast cancer patients with or without metastasis. The results showed that circulating HOTAIR was much higher in patients with distant metastasis (bone, liver or lung) compared with those without metastasis (
Circulating HOTAIR expression correlated to metastasis events and worse DFS. A. Circulating HOTAIR was much higher in patients with distant metastasis (bone, liver or lung) compared with those without metastasis (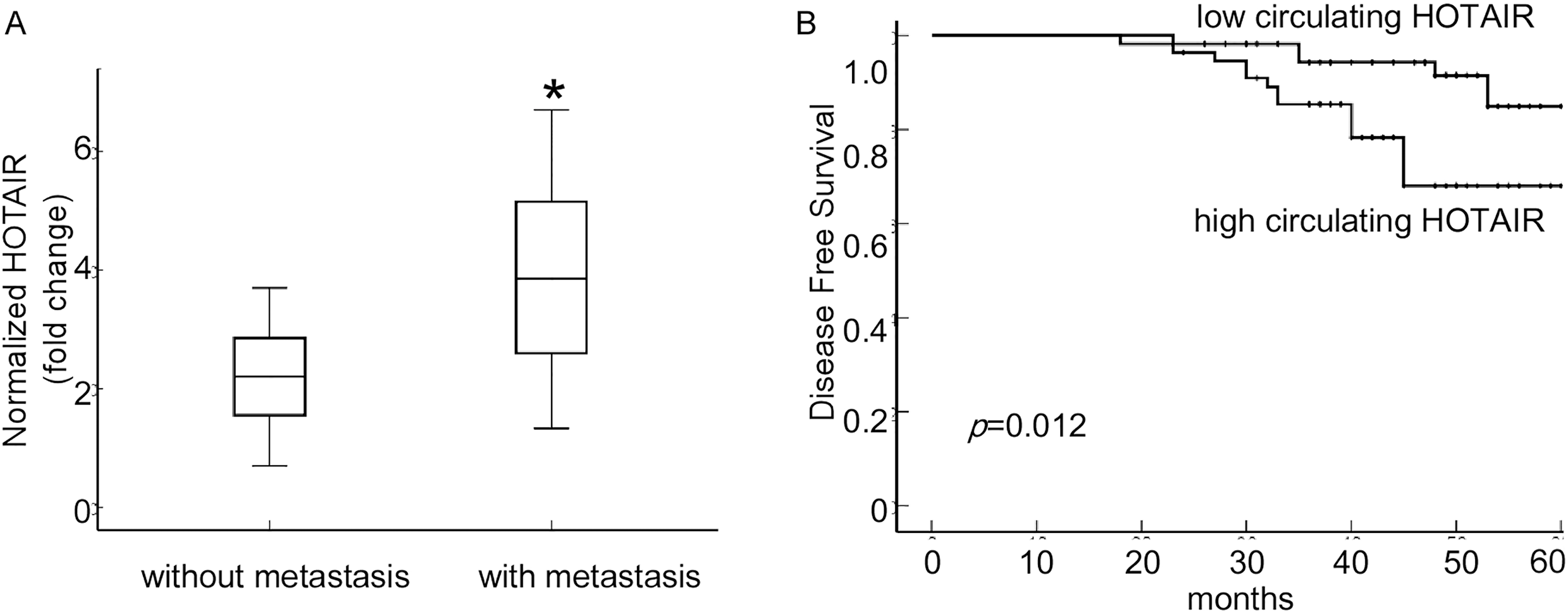
Neoadjuvant chemotherapy has become a reasonable choice for patients with breast cancer. Identifying novel biomarkers that distinguish patients likely to benefit from NAC will help to improve prognosis. In the present study, we found that elevated circulating HOTAIR expression was associated with advanced disease parameters. In particular, the circulating HOTAIR was significantly higher in patients with more positive lymph nodes, larger tumor size and more metastasis. Meanwhile, patients with high circulating HOTAIR level had a worse prognosis than that with low circulating HOTAIR levels. Furthermore, we correlated the circulating HOTAIR levels with NAC response and found that patients with lower pretreatment circulating HOTAIR achieved more response than those with higher circulating HOTAIR. Therefore, our study provides a favorable basis to use circulating HOTAIR as a potential predictor of the response to NAC.
HOTAIR is located in HOXC gene cluster on chromosome 12q13.13 and mediates the epigenetic repression by facilitating polycomb repressive complex (PRC2) recruitments to the specific genomic positions of target genes. In breast cancer, increased HOTAIR levels are observed in both primary tumors and metastases, which consequently enhance tumor epithelial-mesenchymal transition (EMT), treatment resistance and metastasis. There are increasing evidence showed that HOTAIR induces chemoresistance by activating the Wnt/
To date, more and more evidence showed that lncRNAs exist in the circulation of cancer patients and may be served as useful biomarkers. For instance, LncRNA HULC has been reported as a novel serum tumor marker for diagnosis and prognosis prediction of gastric cancer [14]. The expression of circulating H19 was significantly increased in breast cancer tissues and plasma compared with healthy controls and significantly correlated with lymph node metastasis [15]. LncRNA POU3F3 in circulation could be used as a novel biomarker for diagnosis of esophageal squamous cell carcinoma [9]. Recently, circulating lncRNAs have been demonstrated to be correlated to chemotherapy response. For instance, high serum MALAT1 expression level was associated with poor chemotherapy response and survival in glioblastoma multiforme patients receiving temozolomide treatment [18]. Decreased serum MEG3 expression was associated with poor chemotherapy response and low survival rate in colorectal cancer patients receiving oxaliplatin treatment [19]. Circulating GAS5 may serve as possible novel biomarkers to predict chemotherapy response [16]. Similarly, circulating lncRNA HOTAIR has been reported to be detectable in serum and may serve as a novel biomarker for breast cancer. The expression of HOTAIR level in breast cancer patients is much higher than that in normal control and elevated circulating HOTAIR expression was associated with disease progression and poor prognosis in various cancers [10, 11, 22, 23]. However, there are few studies about whether pretreatment circulating HOTAIR predicts chemosensitivity in breast cancer patients. Although high HOTAIR level in tissues has been demonstrated to inhibit chemosensitivity in cancer patients [24], this is the first time that the association between pretreatment circulating HOTAIR and the NAC response is described in breast cancer.
Although lncRNAs are stable in circulation, the origins of circulating lncRNAs of breast cancer patients and the mechanism of how they enter blood are currently unclear. A popular explanation is that lncRNAs enter into circulation through extracellular vesicles, such as exosomes, microvesicles and apoptotic body released from host cancer cells [25, 26]. Huang et al reported that lncRNAs account for 3.36% of total exosomal RNA content [27]. Accumulating studies have showed that extracellular vesicle lncRNA may also serve as potential biomarkers for diagnosis [28] as well as response to chemotherapy of various cancers [29]. As a matter of fact, the origin of circulating lncRNAs is not a prerequisite for its being qualified as biomarkers of breast cancer. Since HOTAIR is stable in serum, the circulating lncRNA alone or combination with other circulating nucleic acids might be used as novel predictor of the response to NAC.
Previous predictors of the response to NAC include estrogen receptor (ER), progesterone receptor (PR), human epidermal growth factor receptor 2 (HER2) and Ki67 as well as tumor size and pathology subtypes [30]. However, most of these factors are measured by core needle biopsy before NAC. Because of the heterogeneity of breast cancer, tissues obtained by core needle biopsy could not reflect the entirety situation of the tumor. Due to the relatively non-invasive sampling procedure, serum samples may be served as ideal biomarkers, not only providing non-invasions, stability and convenience, but also avoiding sampling errors. Therefore, circulating lncRNAs examination may be served as a more direct and useful method for predicting chemosensitivity compared with biopsy samples.
Elevated circulating HOTAIR expression was associated with advanced disease parameters. Pretreatment circulating HOTAIR could be used as a simpler and more convenient predictive biomarker for NAC response compared with other tissue histopathological indictors. The use of circulating HOTAIR may facilitate the administration of NAC in patients with low circulating HOTAIR to achieve a better response and prognosis.
Conclusions
Our findings demonstrate that circulating HOTAIR is a potential factor predicting the response to NAC in patients with breast cancer. Patients with high level of circulating HOTAIR showed a relatively low response rate to NAC. Meanwhile, high circulating HOTAIR may be served as a predictor of low DFS. However, large-scale sample collection, long-term follow up and multicenter studies are needed to confirm this clinical application.
Footnotes
Acknowledgments
This work was supported by grants from the Natural Science Foundation of China (81702598), the Natural Science Foundation of Guangdong Province (2017A030313803), the Fundamental Research Funds for the Central Universities (21617315), Science and Technology Program of Guangzhou (201804010011) and the Science Foundation of the First Affiliated Hospital of Jinan University (2016316).
Conflict of interest
The authors declare that they have no competing interests.
