Abstract
Objective
The papillary thyroid cancer (PTC) incidence is on the increase. We explored the diagnostic value of microRNA (miR)-129-5p & serologic indicator thyroid-stimulating hormone (TSH) test in PTC with cervical lymph node metastasis (LNM).
Methods
According to the pathological “gold standard,” 198 PTC patients were assigned into the LNM (n = 93)/non-LNM (n = 105) groups, with their medical records collected. The serum free-triiodothyronine (FT3)/free-thyroxine (FT4)/TSH/thyroglobulin (Tg)/thyroglobulin antibody levels were assessed using an electrochemiluminescence immunoassay device. Serum miR-129-5p expression was determined by reverse transcription quantitative polymerase chain reaction. Correlations between serum miR-129-5p/TSH levels with pathological indicators were analyzed by Spearman correlation coefficient. Independent influencing factors for cervical LNM in PTC patients was analyzed by logistic multivariate regression analysis. Diagnostic value of miR-129-5p combined with serologic indicator TSH test in PTC patients with cervical LNM and lateral cervical LNM was analyzed by the receiver operating characteristic curve.
Results
The two groups varied obviously in primary tumor size/Tg level. Serum miR-129-5p expression in the LNM group was reduced, and negatively correlated with Tg and primary tumor size, while the serologic indicator TSH level showed positive correlations with Tg and primary tumor size. Independent influencing factors for PTC with cervical LNM were miR-129-5p/TSH/Tg levels. miR-129-5p and serologic indicator TSH levels had high diagnostic value for PTC patients with cervical LNM and lateral cervical LNM, with their combination showing higher diagnostic value.
Conclusion
miR-129-5p and serologic indicator TSH had high diagnostic value for diagnosing PTC patients with cervical LNM, providing high reference value for the formulation of thyroid tumor resection.
Keywords
Introduction
Papillary thyroid cancer (PTC) is the most prevalent thyroid malignant tumor in thyroid cancer (TC) with high incidence and recurrence rates, and at present, lymph node metastasis (LNM) is proven to be an essential risk factor for the high recurrence rate in patients with PTC 1 Generally, LNM in PTC patients firstly metastasizes to the central lymph nodes (VI) and then involves the lateral neck (II–V), ultimately metastasizing to distant organs along the lymphatic drainage pathway in the neck. 2 Accordingly, cervical LNM is the most common LNM in PTC patients, which is also the location where PTC patients are most prone to recurrence. A study on 9904 PTC cases in the Surveillance, Epidemiology and End Results database of the National Cancer Institute has found that cervical LNM represents a critical predictor of poor prognosis. 3 Central LNM is recognized as the main area of cervical LNM, while lateral cervical LNM results in poor disease-specific survival in PTC patients; however, cervical central LNM has been proven to be one of the risk factors affecting lateral cervical LNM. 4 Furthermore, apart from the previously mentioned cervical LNM, cervical skip LNM is also present in TC patients, which primarily means the situation where metastasis occurs in the ipsilateral cervical lymph nodes or mediastinal lymph nodes except the central LNM; however, the TC patients with skip metastasis have a relatively small incidence rate (approximately 8.7%). 5 Along with the advancement of ultrasound diagnostic technology, the detection rate of thyroid nodules has been constantly increased, and preoperative ultrasound is also the predominant modality for lateral neck LNM evaluation; however, the preoperative ultrasound has only a 20–31% detection rate for central cervical LNM, and the detection rate is even lower in lateral cervical LNM. 6 Consequently, there is an urgent need for a more efficient method to guide the clinical diagnosis and treatment of PTC patients with LNM.
microRNAs (miRNAs) are a sort of highly conserved short-stranded non-coding RNAs participating in a variety of pathological and physiological processes in the organism, including differentiation, virus infection, proliferation, and tumor occurrence in various types of cancers. Accumulating evidence manifests that aberrantly-expressed miRNAs have high relevance to many human diseases, including cancers. 7 For instance, downregulation of miR-129-5p expression is discovered in nasopharyngeal carcinoma accompanied by LNM. 8 In particular, miRNAs have an influential role in PTC. miR-129-5p upregulation is capable of effectively suppressing the migration, proliferation, and LNM of PTC cells. 9 However, there is inadequate research regarding the specific role of miR-129-5p in PTC accompanied by LNM, as well as the potential of miR-129-5p as a diagnostic marker. It is noteworthy that serologic indicators including thyroid stimulating hormone (TSH) and thyroglobulin (Tg) are routinely used to detect thyroid-related disorders. PTC patients with cervical LNM have higher Tg expression in the serum than that of those without LNM, and Tg can serve as a molecular marker for the diagnosis of cervical LNM in PTC patients.10,11 Despite the fact that there is no clear research claiming that TSH is capable of being adopted as a molecular marker to diagnose the presence of cervical LNM in PTC patients, serum TSH level in PTC patients with LNM is markedly higher than that in PTC patients without LNM, 12 suggesting the possibility that TSH has the potential to be a molecular marker for diagnosing cervical LNM in PTC patients. However, most of the clinical studies have demonstrated that the detection of a single serum indicator has certain errors, and its sensitivity and specificity may be limited, causing interference to the prediction results. A previous study has elicited that miR-129-5p may serve as a pivotal index to assist in the monitoring of lymph node metastasis in colorectal cancer. 13 Given that the potential role of miR-129-5p in cancer and the significance of TSH in the assessment of thyroid function, a combined test of the two may help improve diagnostic accuracy and sensitivity. Nevertheless, the diagnostic value of miR-129-5p combined with serologic indicator TSH on cervical LNM in PTC remains largely unknown. Consequently, this study is to evaluate the diagnostic value of miR-129-5p combined with the serologic indicator TSH on assessing cervical LNM in PTC.
Materials and methods
Ethics statement
This study was approved by the academic ethics committee of RuiJin Hospital Lu Wan Branch, Shanghai Jiaotong University School of Medicine (Approval No. LWEC2022009). All procedures were implemented strictly in line with the Declaration of Helsinki. All subjects involved were fully informed of the study objective and signed the informed consents before sampling.
Study subjects
This study consecutively enrolled 244 PTC patients who underwent thyroid tumor resection and visited for review from January 2022 to August 2023. After excluding 21 patients who did not meet inclusion criteria and 25 patients who conformed to exclusion criteria, 198 patients were finally included as the subjects of this study. Pathological findings were regarded as “gold standard,” and the diagnosis of LNM was as follows: PTC metastasis was confirmed by preoperative fine-needle aspiration, intraoperative frozen section analysis, or postoperative pathology. PTC patients were assigned into the LNM group (n = 93), and the non-lymph node metastasis group (NLNM) (n = 105). Medical records were collected for all subjects, such as age, sex, body mass index (BMI), primary tumor size, free-thyroxine (FT4), free-triiodothyronine (FT3), miR-129-5p, Tg, TSH, and the presence of Graves’ disease (GD) or Hashimoto's thyroiditis (HT).
Inclusion and exclusion criteria
Inclusion criteria included (a) PTC diagnosed by preoperative fine-needle aspiration of the thyroid; (b) age 18–70 years old; (c) underwent thyroid tumor resection for the first time; and (d) complete clinical and pathological data.
Exclusion criteria were as below: (a) underwent biopsy but did not undergo thyroidectomy; (b) had undergone thyroidectomy previously; (c) did not accept lymph node examination; (d) distant metastasis; (e) a history or coexistence of other head and neck cancers; (f) severe infection, liver failure, or renal failure in the past 3 months; and (g) incomplete or lost medical records data.
Thyroid surgery
All PTC patients were routinely treated with partial thyroidectomy with unilateral central-compartment neck dissection or total thyroidectomy with bilateral central-compartment neck dissection. When metastatic cervical lymph nodes were doubted according to clear imaging and/or clinical evidence, modified or lateral neck dissection was performed. Lateral and central LNM were diagnosed as per pathological findings. All surgical procedures were accomplished by the same group of surgeons.
Serum indicator assessment
Before surgery, fasting venous blood (4 mL) was acquired from all subjects on admission in the morning without anticoagulation treatment. Within 30 min, the blood was centrifuged at 3000 r/min and 4°C for 5 min, with the supernatant collected for detecting serum FT3, miR-129-5p, FT4, Tg, and TSH levels. The serum levels of FT4, FT3, TSH, Tg, and thyroglobulin antibody (Tg-Ab) in the patients were examined using an electrochemiluminescence immunoassay (ECLIA) analyzer Cobas Eless 601 (Roche Diagnostics, Basel, Switzerland). The expression of miR-129-5p in patients’ serum was assayed by reverse transcription quantitative polymerase chain reaction (RT-qPCR).
RT-qPCR
RT-qPCR was implemented to determine the expression pattern of serum miR-129-5p in all groups of patients. As per the manufacturer's instructions, total RNA was extracted from the samples using TRIzol regent (Invitrogen, Carlsbad, CA, USA), and then transcribed into cDNA utilizing PrimeScript RT kit (Takara Biotechnology, Kyoto, Japan). Then, qPCR was conducted on the ABI 7900HT rapid PCR real-time system (Applied Biosystems, Foster City, CA, USA) using SYBR®Premix Ex Taq™ II (Takara Biotechnology). Reaction conditions were as follows: pre-denaturation at 95°C for 10 min, and 40 cycles of denaturation at 95°C for 10 s, annealing at 60°C for 20 s, and extending at 72°C for 34 s. U6 and GAPDH served as internal references. Primers synthesized by Sangon Biotech (Shanghai, China) were: miR-129-5p, Forward, 5′-GATCCGCAAGCCCAGACCGCAAAAAGTTTTTA-3′ and Reverse, 5′-AGCTTAAAAACTTTTTGCGGTCTGGGCTTGCG-3′; U6 Forward, 5′-CGCTTCGGCAGCACATATAC-3′ and Reverse, 5′-AACGCTTCACGAATTTGCGT-3′; GAPDH, Forward, 5′-CACATGGCCTCCAAGGAGTAA-3′ and Reverse, 5′-TGAGGGTCTCTCTCTTCCTCTTGT-3′. The 2−ΔΔCt method was employed for calculating the relative expression level of miR-129-5p.
LNM grouping
In accordance with the American Joint Committee on Cancer Classification (8th edition), 14 cervical lymph nodes were assigned into: (a) Area I: submental and submandibular area; (b) Area II: upper jugular lymph nodes; (c) Area III: middle jugular lymph nodes; (d) Area IV: lower jugular lymph nodes; (e) Area V: posterior triangle lymph nodes; (f) Area VI: anterior compartment lymph nodes; and (g) Area VII: superior mediastinal lymph nodes, which is called the central region together with Area VI. In accordance with the situation, we allocated patients into central area (Area VI–VII), lateral cervical area (Area I–V) and skip metastasis. Among them, skip metastasis meant no metastasis occurring in the central lymph nodes but occurring in the ipsilateral lateral cervical lymph nodes or mediastinal lymph nodes.
Statistical analysis
Statistical analysis and plotting were conducted using SPSS 21.0 (IBM, Armonk, NY, USA) and GraphPad Prism 8.01 software (GraphPad Software Inc., San Diego, CA, USA). The Shapiro-Wilk test was used for verifying the normal distribution of data. Normally distributed measurement data were expressed as mean ± SD, and analyzed by the independent sample t-test between two groups. The non-normally distributed measurement data were represented by the median (minimum, maximum) or interquartile range (IQR) (25–75 percentiles), and tested by the Mann–Whitney test between two groups, followed by the Spearman correlation coefficient analysis of the correlation among various indicators. Categorical variables were expressed as the number of examples (%) and tested by the Chi-square test. Multivariate logistic regression analysis was used to identify independent influencing factors for cervical LNM occurrence in PTC patients. The receiver operating characteristic (ROC) curve was plotted to analyze the diagnostic value of miR-129-5p combined with serologic indicator test for PTC patients with cervical LNM and lateral LNM. The P-value was calculated using a two-tailed test, where the value of P < 0.05 indicated statistical significance.
Results
Comparisons of clinical baseline characteristics
PTC patients were allocated into the LNM and NLNM groups as per the pathological findings. The clinical baseline characteristics of all patients were displayed in Table 1. There were no significant differences in age, sex, BMI, FT4, Tg-Ab, FT3 and the presence of GD or HT between the LNM and NLNM groups (all P > 0.05), but significant differences were noticed in tumor size, Tg, and degree of differentiation (all P < 0.001). In the LNM group, there were 23 cases with single LNM, 70 cases with multiple LNM, 58 cases with only central LNM, 31 cases with lateral LNM, and 4 cases with skip LNM.
Comparisons of clinical baseline characteristics.
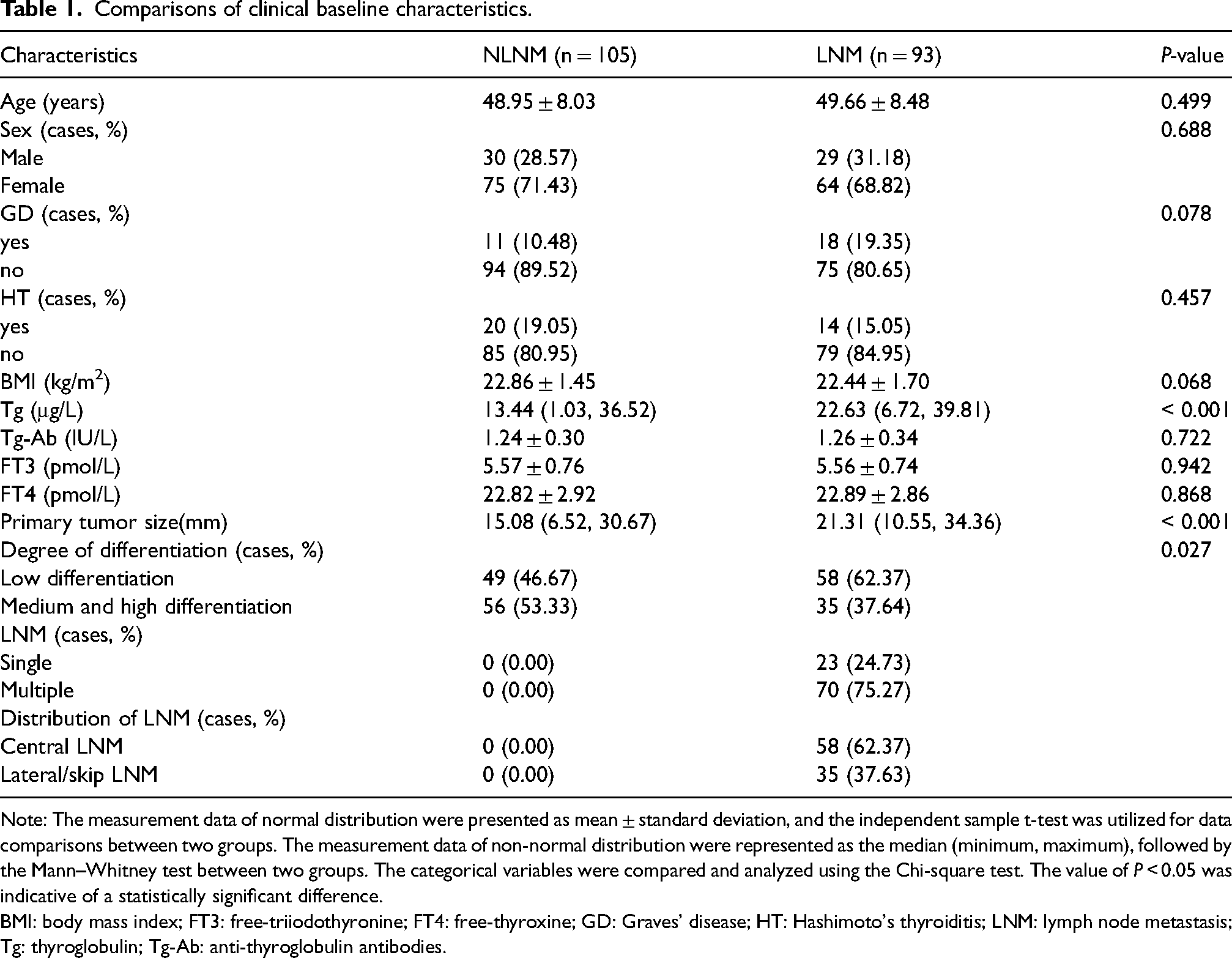
Note: The measurement data of normal distribution were presented as mean ± standard deviation, and the independent sample t-test was utilized for data comparisons between two groups. The measurement data of non-normal distribution were represented as the median (minimum, maximum), followed by the Mann–Whitney test between two groups. The categorical variables were compared and analyzed using the Chi-square test. The value of P < 0.05 was indicative of a statistically significant difference.
BMI: body mass index; FT3: free-triiodothyronine; FT4: free-thyroxine; GD: Graves’ disease; HT: Hashimoto's thyroiditis; LNM: lymph node metastasis; Tg: thyroglobulin; Tg-Ab: anti-thyroglobulin antibodies.
Comparisons of levels of serum mir-129-5p and serologic indicator TSH between two groups of patients
The differences in levels of serum miR-129-5p and serologic indicator TSH were compared between the NLNM and LNM groups. RT-qPCR manifested that TSH and miR-129-5p expression levels were (median 4.05 (3.32, 6.67)) and (median 1.33 (0.34, 4.25)) in LNM patients, and were (median 3.23 (1.49, 4.75) and median 2.32 (0.80, 5.30)) in the NLNM patients, respectively. The expression of miR-129-5p in the serum of LNM patients was prominently lower than that in the NLNM patients (Figure 1(A), all P < 0.001). Additionally, the ECLIA analyzer demonstrated that in comparison with the NLNM group, the TSH level in the serum of LNM patients was remarkedly elevated (Figure 1(B), all P < 0.001).
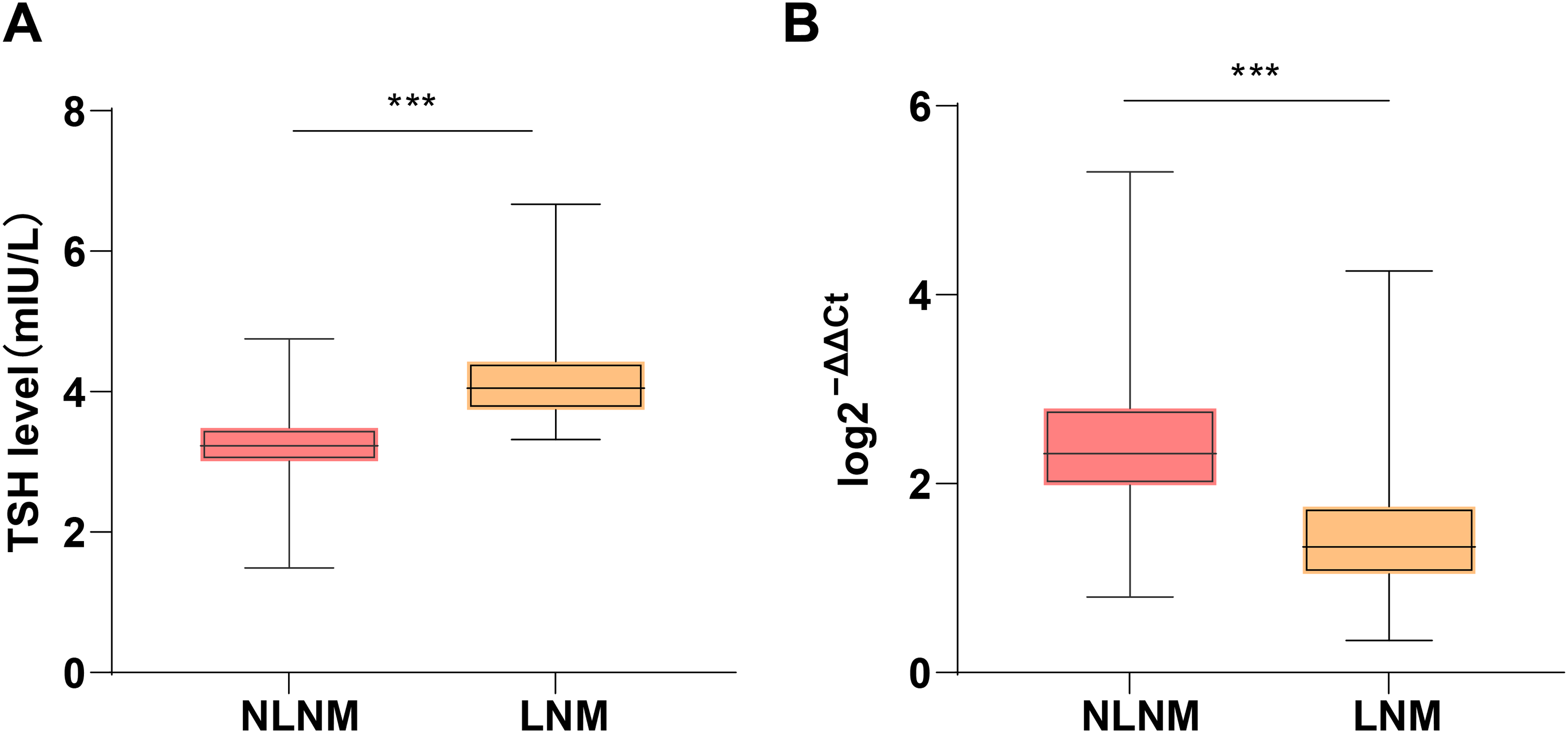
Comparisons of serum miR-129-5p and serological indicator TSH levels between the two groups. (A)–(B) Box plots of the medians of serum miR-129-5p and TSH levels of the NLNM and LNM groups. The box refers to the median (horizontal line in the box) and the IQR (25–75 percentiles), and the extension line refers to the minimum and maximum values. Comparisons were carried out between two groups using the Mann–Whitney test. *** P < 0.001.
Correlation analyses of serum mir-129-5p and serologic indicator TSH with pathological indicators in subjects
To clarify the correlations between miR-129-5p and TSH levels with pathological indicators of the subjects (primary tumor size, Tg, Tg-Ab, FT4, and FT3), we utilized Spearman correlation coefficients for analysis. The results uncovered that serum miR-129-5p level had noticeably negative correlations with primary tumor size and Tg (all |r| > 0.5), but had no significant correlations with Tg-Ab, FT3, and FT4. Serum TSH level had significantly positive correlations with Tg and primary tumor size (all |r| > 0.5), but had no obvious correlations with Tg-Ab, FT3, and FT4 (Table 2, all P < 0.001).
Correlation analysis of serum miR-129-5p and serological indicator TSH with pathological indicators in subjects.
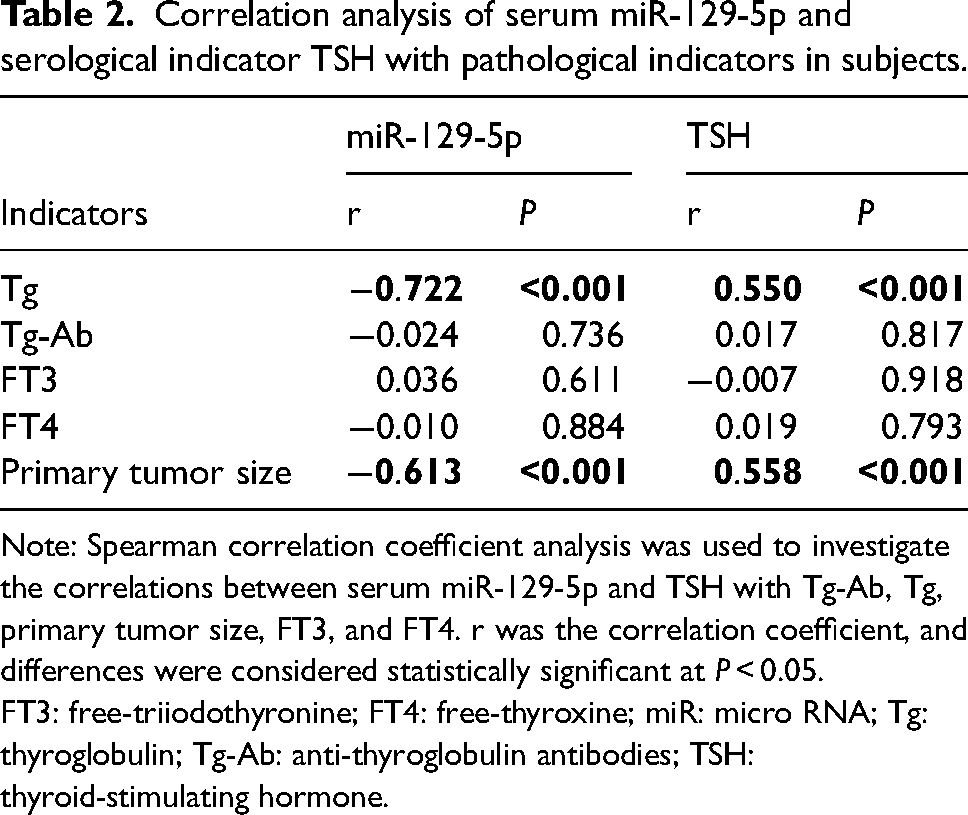
Note: Spearman correlation coefficient analysis was used to investigate the correlations between serum miR-129-5p and TSH with Tg-Ab, Tg, primary tumor size, FT3, and FT4. r was the correlation coefficient, and differences were considered statistically significant at P < 0.05.
FT3: free-triiodothyronine; FT4: free-thyroxine; miR: micro RNA; Tg: thyroglobulin; Tg-Ab: anti-thyroglobulin antibodies; TSH: thyroid-stimulating hormone.
miR-129-5p and serologic indicator TSH levels were independent influencing factors for cervical LNM in PTC patients
We included the indicators with P < 0.05 in Table 1 and Figure 1 in a multivariate logistic regression analysis, with the occurrence of cervical LNM as the independent variable. The analyses suggested that TSH, miR-129-5p, and Tg levels were independent influencing factors for cervical LNM in PTC patients (Table 3).
Serological indicator TSH and miR-129-5p were independent influencing factors for cervical LNM occurrence in PTC patients.

Note: A value of P < 0.05 was indicative of statistically significant.
CI: confidence interval; OR: odds ratio; LNM: lymph node metastasis; miR: micro RNA; PTC: papillary thyroid cancer; Tg: thyroglobulin; TSH: thyroid-stimulating hormone.
The diagnostic value of miR-129-5p combined with serologic indicator TSH detection for patients with PTC accompanied by cervical LNM
To explore the diagnostic value of the combination of miR-129-5p with TSH assessment on PTC complicated with cervical LNM, we performed a ROC curve analysis, with LNM as the state variable and serum miR-129-5p and TSH levels as test variables. The ROC curve analysis showed that both miR-129-5p < 1.91 and TSH > 3.53 had high diagnostic value for the diagnosis of cervical LNM in PTC patients (Supplementary Table 1, Figure 2(A)). The combination of serum miR-129-5p and TSH levels had high diagnostic value for the diagnosis of cervical LNM in PTC patients (the area under the curve (AUC) was 0.954, and the cutoff value was 0.64, with 82.80% sensitivity and 97.14% specificity) (Supplementary Table 1, Figure 2(A), all P < 0.05), which had amplified diagnostic value against them alone. These results indicated that serum miR-129-5p and serologic indicator TSH levels had high diagnostic value for diagnosing cervical LNM in PTC patients, with their combination showing higher diagnostic value. Subsequently, we allocated LNM patients into central LNM patients (n = 58) and lateral LNM patients (n = 31). The ROC curve indicated (Supplementary Table 1, Figure 2(B)) that the combined test of serum miR-129-5p and TSH levels exhibited high value for diagnosing the occurrence of lateral LNM in PTC patients (the AUC was 0.990, the cut-off value was 0.12, with a sensitivity of 96.77% and a specificity of 93.41%), which was observed to surpass the two alone in diagnosing whether or not PTC patients had lateral LNM (all P < 0.05).
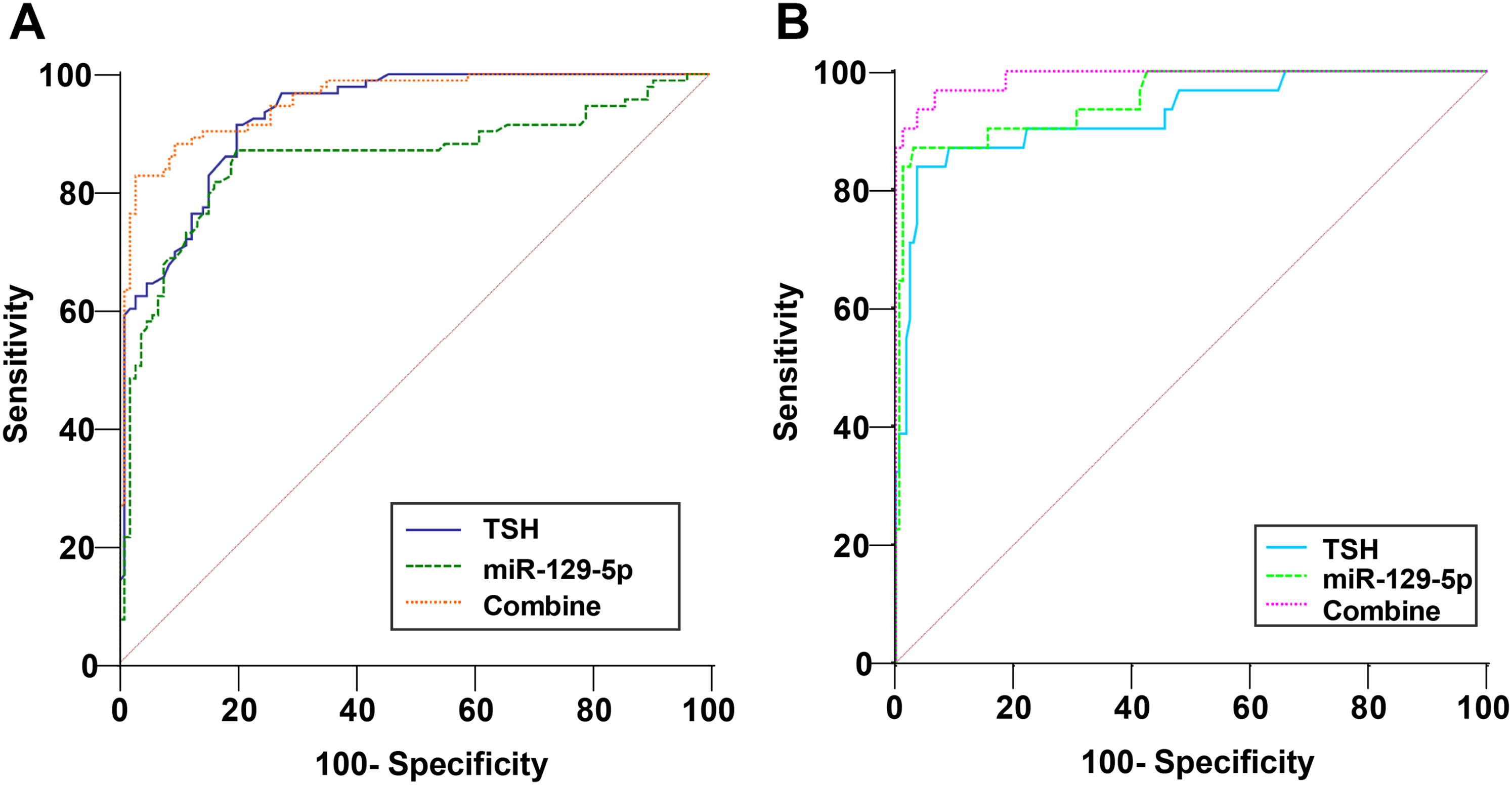
Diagnostic value of miR-129-5p combined with serological indicator TSH detection in PTC patients with cervical LNM and lateral cervical LNM. ROC curve was applied for analyzing the diagnostic value of miR-129-5p and serological indicator TSH combination in PTC patients with LNM and lateral LNM.
Discussion
TC, the most frequent cancer of the endocrine system, continues to maintain the momentum of accelerated development, against the social backdrop of stagnant or declining incidences of a variety of tumors. 15 As an indispensable indicator of PTC prognosis, surgery scope and way, LNM is a significant risk factor for low survival rate and high recurrence rate of patients as well. 16 Notably, miR-129-5p not only modulates a broad scope of biological function, but also plays a crucial role in the tumorigenesis process and functions via either advancing or repressing tumors. 17 In the present study, our findings elicited that serum miR-129-5p and serologic indicator TSH were independent influencing factors for the occurrence of LNM in PTC patients, and their combination had high diagnostic value for predicting LNM in PTC patients.
GD occurs because the activation of TSH receptor antibodies facilitates the TSH receptor activation, and thus induces carcinogenesis, cell proliferation, and angiogenesis.18,19 Notably, a previous study has elicited that in contrast to PTC patients without GD, PTC patients with GD have similar risk of structural recurrence at the time of diagnosis. 20 Specifically, some scholars have reported that the risk of recurrence and distant metastasis in GD-PTC patients is 1–2 times higher than that in PTC patients without GD, suggesting that GD may increase the risk of lymph node metastasis in PTC patients. 21 Moreover, another study has demonstrated that the occurrence of GD does not affect the prognosis of PTC, and has a negligible effect on the disease aggressiveness. 22 In a similar light, the present study also revealed no significant difference in the occurrence of GD between NLNM and LNM patients. Notably, the expression of miR-129-5p is markedly down-regulated in gastric cancer tissues relative to adjacent normal tissues. 23 TSH binding to receptors on thyrocytes facilitates a number of the growth stimulating pathways both in malignant and normal thyroid cells, and repression of TSH with levothyroxine at high doses is applied routinely after thyroidectomy to prevent disease recurrence, especially in the high-risk tumors. 24 Long noncoding RNA PVT1 is capable of regulating cell proliferation of TC via recruiting enhancer of zeste homolog 2 and modulating TSH receptors, while TSH levels in serum are involved in postoperative recurrence and the LNM of PTC. 25 Consistent with previous research, our study demonstrated that serum miR-129-5p was lowly expressed in PTC patients with LNM, while the serologic indicator TSH level was elevated in PTC patients with LNM compared with those without LNM.
Tg is one of the markers of TC, and its concentration may be increased in patients with malignant thyroid tumors. 26 TSH, as the main growth factor of thyroid cells, can stimulate thyroid hormone secretion; as the size of the primary tumor increases, the body's “cancer load” increases, which promotes further increases in TSH and Tg levels. 27 Notably, it is suggested that Tg plays a vital role in transporting and storing thyroid hormones, and its preoperative level is adopted for assessing the tumor size and degree of differentiation in patients with a thyroid tumor. 28 Meanwhile, miR-129-5p gene promoter CpG methylation and overexpression of miR-129-5p leads to apoptosis of radioresistant cells. 9 In a recent study, miR-129-5p has been revealed to be a novel therapeutic target for carcinoma treatment. 17 miR-129-5p induces the repression of cell proliferation, migration, and invasion in clear cell renal cell cancer via the target of SPN. 29 Moreover, miR-129-5p limits cell proliferation and migration in bladder cancer, and induces cell cycle arrest by targeting PROM2. 30 Nevertheless, we confirmed for the first time that the level of serum miR-129-5p level in PTC patients was obviously negatively correlated with primary tumor size and Tg, while TSH exhibited obviously positive correlations with these indicators.
Many molecules are reported to participate in modulating tumor cell invasion and metastasis. For instance, TSH is well-known to be involved in enhanced aggressiveness and proliferation of tumor. 31 Moreover, TSH possesses the ability to activate the thyroid malignant tumor's development or growth, and its elevation in sera is related to morbidity of thyroid carcinoma and advanced disorder. 32 Increased serum TSH level is closely related to DTC with cervical lymph node metastasis. 33 Furthermore, tumor size is considered as an independent negative prognostic factor for event-free survival in differentiated children with TC. 34 From these, TSH, Tg, and miR-129-5p levels were independent influencing factors for the occurrence of cervical LNM in PTC patients. Further, we disclosed for the first time that serum miR-129-5p and serologic indicator TSH levels had high diagnostic value in diagnosing cervical LNM and lateral cervical LNM in PTC patients, with their combinative diagnostic value higher than them alone. In summary, our data revealed the diagnostic value of the combination of miR-129-5p and serologic indicator TSH test on cervical LNM in PTC, suggesting that miR-129-5p and serologic indicator TSH had high diagnostic value for predicting LNM in PTC patients. However, a notable weakness of our research lies in the small number of cases and events included in the analysis and the single-center retrospective study, highlighting the need of multicenter studies, further expansion of the sample size, and analyses of short- and long-term prognosis of PTC patients receiving thyroidectomy. Moreover, proliferation of the thyroid cells requires a synergistic cycle related to TSH, CREB1 (a TSH-activated transcription factor) and miRNAs. 35 As reflected by an miRNA microarray analysis, TSH reduces the expression of 47 miRNAs in thyroid cells; among these, TSH stimulates cAMP/protein kinase A and PI3 K cascades to diminish miR-195 and miR-16, which stimulates cell proliferation by triggering Cdc6, Ccne1, and Mapk8. 36 TSH plays pivotal roles in negatively and positively regulating miRNA expression, which is vital for the thyrotropin stimulatory effect. Of note, miR-129-5p may play a role by impacting gene expression in PTC. For instance, miR-129-5p affects the invasion, migration, and proliferation of papillary thyroid microcarcinoma via its targeted regulation on FOXE1. 37 Furthermore, the novel anti-cancer drug histone deacetylase inhibitor can cause apoptosis in PTC cells by inducing miR-129-5p overexpression. 38 Circ_0027446 stimulates the expression of claudin 1 to facilitate PTC cell malignancy through binding with miR-129-5p. 39 As a consequence, the potential relationship between miR-129-5p and TSH mainly reflects in that TSH may influence the level of miR-129-5p by modulating relevant genes or signaling pathways, thereby affecting thyroid function. In future studies, we will conduct large-scale, multi-center clinical research and laboratory basic research to validate these preliminary conclusions. In addition, the lateral cervical lymph node metastasis was divided into regions II–V in this study, but we did not analyze the specific partitions, which limited the comprehensiveness of the analysis of lymph node metastasis regions. Consequently, these are the directions of our future studies. At present, there is no consensus on whether treatment is needed on older people with subclinical hypothyroidism, along with the treatment goals, but what is quite clear is that treatment is not advocated for patients with subclinical hypothyroidism who are aged over 85 years. Studies have suggested that the distribution of serum TSH level tends to be higher with aging, leading to an overestimation of the incidence of subclinical hypothyroidism in the older age group. 40 In contrast, even if the TSH level is over 4.5 mIU/L, the population > 70 years old with hypothyroidism cannot be diagnosed in the absence of antithyroid antibodies. We believe that thyroid hormone replacement therapy for such patients will bring cardiac burden and side effects. In the Chinese guideline for the diagnosis and treatment of thyroid diseases, the target TSH for subclinical hypothyroidism treatment in older people is set at 0.5–3 mIU/L, which is lower than that of the general young population, and we believe that it is supposed to be set at the upper limit of the normal range of TSH. 41 At the same time, the individualization should be taken into account, and it should not increase cardiac burdens on older people due to the need of lowering TSH. As a result, it is an interesting direction to explore the connection of age and TSH expression with cervical LNM in PTC patients. Accordingly, we will carry out further studies on PTC patients at different ages and investigate in more detail to analyze the influence of age, thyroid function, and TSH in PTC patients at different ages.
Supplemental Material
sj-doc-1-jbm-10.1177_03936155241303763 - Supplemental material for Diagnostic value of microRNA-129-5p and TSH combination for papillary thyroid cancer with cervical lymph node metastasis
Supplemental material, sj-doc-1-jbm-10.1177_03936155241303763 for Diagnostic value of microRNA-129-5p and TSH combination for papillary thyroid cancer with cervical lymph node metastasis by Yi Shen, Rongli Xie, Yupan Chen, Xujie Han and Xiao-en Li in The International Journal of Biological Markers
Footnotes
Acknowledgments
Not applicable.
Author contributions
Conceptualization: Yi Shen and Xiao-en Li; Methodology: Yi Shen and Rongli Xie; Software: Yupan Chen; Validation: Yupan Chen and Xujie Han; Formal analysis: Yi Shen and Rongli Xie; Investigation: Xiao-en Li; Resources: Xujie Han; Data Curation, Xiao-en Li; Writing—Original Draft Preparation: Yi Shen; Writing—Review & Editing: Rongli Xie and Xiao-en Li; Visualization: Rongli Xie; Supervision: Rongli Xie; Project Administration: Yi Shen and Xiao-en Li; Funding acquisition, Yi Shen and Rongli Xie.
Availability of data and materials
The findings of this study are available from the corresponding author upon reasonable request.
Consent for publication
Not applicable.
Declarations
Ethics approval and consent to participate
This study was approved by the academic ethics committee of RuiJin Hospital Lu Wan Branch, Shanghai Jiaotong University School of Medicine (Approval No. LWEC2022009). All procedures were implemented strictly in line with the Declaration of Helsinki. All subjects involved were fully informed of the study objective and signed the informed consents before sampling.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants from Shanghai Municipal Health Commission: A Study on the Risk Assessment and Prognostic Comprehensive Model of Lymph Node Metastasis in Thyroid Papillary Microcancer with Multi Model/Multi Feature Fusion (Grant No. 20214Y0223).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
