Abstract
BACKGROUND:
Esophageal cancer is a common tumor with high mortality worldwide. In the present study, we aimed to investigate the prognostic significance and regulatory effects of miR-105 on cellular functions of esophageal cancer cells.
METHODS:
The expression level of miR-105 was analyzed in esophageal cancer tissues and cell lines by qRT-PCR. Survival analysis was carried out using the Kaplan-Meier and the prognostic significance of miR-105 was analyzed with Cox regression analysis. The effects of miR-105 on cell proliferation, migration, and invasion abilities were detected with cellular experiments.
RESULTS:
We found that miR-105 was significantly upregulated in esophageal cancer tissues and cell lines, compared with the control group, respectively. Moreover, overexpression of miR-105 was significantly associated with positive lymph node metastasis, advanced TNM stage, and poor overall survival. In addition, overexpression of miR-105 promoted cell proliferation, migration, and invasion in esophageal cancer cells, while downregulation of miR-105 suppressed these cellular behaviors.
CONCLUSION:
Our findings indicate that a higher level of miR-105 predicts poorer prognosis in esophageal cancer patients, and miR-105 can promote esophageal cancer cell proliferation, migration, and invasion.
Introduction
Esophageal cancer is one of the most common types of cancer in the digestive system worldwide [1]. In China, it is the sixth most common malignant tumor and the fourth leading cause of cancer-related death nationwide [2]. What’s more, more than half of the global incidence of esophageal cancer is in China [3]. Despite diagnostic and therapeutic advancements in the past years, including surgery, chemotherapy, and radiotherapy, the prognosis of esophageal cancer patients has shown little improvement with the 5-year survival rate of approximately 20–40% [4, 5]. Esophageal cancer patients at early stages usually have no obvious symptoms, and a large proportion of patients are initially diagnosed at advanced stages with a 5-year survival rate of less than 20%, which seriously affected the quality of life of patients [6]. Therefore, identifying more effective therapeutic methods and novel prognostic molecular biomarkers are necessary for improving the patients’ survival rate [7].
MicroRNAs (miRNAs or miRs) are a group of small non-coding RNA molecules (approximately 22 nucleotides), and can negatively regulate gene expression at the post-transcriptional level by binding to 3’-UTRs of target genes [8, 9]. In recent years, numerous microRNAs have been demonstrated aberrantly expressed or mutated in human cancers that they acted as oncogenes or tumor suppressor genes by regulating multiple cellular processes, including cell proliferation, migration, and invasion [10, 11, 12]. In addition, many miRNAs also can be used as a novel molecular diagnostic/prognostic biomarkers and therapeutic targets for cancer therapies [13, 14, 15]. A number of studies have indicated that miRNAs may be involved in esophageal cancer progression and associated with prognosis [16, 17, 18]. MicroRNA-105 (miR-105), located on human chromosome Xq28, has been reported to play diverse oncogenic and tumor suppressor roles in many cancers [19]. In esophageal cancer, a study identified differentially expressed miRNAs using TCGA data, in which miR-105 was one of the upregulated miRNAs [20]. However, the role of miR-105 in esophageal cancer has not been clarified.
In the present study, we explored the expression levels of miR-105 in esophageal cancer tissues and cell lines. In addition, its clinical prognostic significance and functional role were also evaluated.
Materials and methods
Patients and tissue specimens
In all, 115 esophageal cancer patients who had undergone routine surgery at Dongying People’s Hospital of Shandong Province from June 2011 to December 2013 were enrolled in the present study. Paired esophageal cancer tissue specimens and adjacent normal tissue specimens were collected from the 115 esophageal cancer patients. All tissue specimens were confirmed by experienced pathologists and immediately snap-frozen in liquid nitrogen and then stored at
Comparison of clinical characteristics between patients with low or high miR-105 expression
Comparison of clinical characteristics between patients with low or high miR-105 expression
Three esophageal cancer cell lines (Eca-109, TE-1, and KYSE-150) and human normal esophageal endothelial cell line Het-1A were purchased from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All esophageal cancer cells were cultured in RPMI-1640 medium (Gibco, Carlsbad, CA, USA) supplemented with 10% FBS (Gibco). The Het-1A cells were cultured in DMEM (Gibco) supplemented with 10% FBS (Gibco). All cells were incubated in a humidified 37
MiR-105 mimic, miR-105 inhibitor, and miRNA negative controls (NCs) (mimic NC and inhibitor NC) were purchased from GenePharma (Shanghai, China). Esophageal cancer cells were seeded in 6-well plates at a density of 3
The relative expression levels of miR-105 were determined by qRT-PCR in esophageal cancer tissues and cell lines. A. The relative expression levels of miR-105 were upregulated in tumor than that in matched adjacent normal tissue specimens. B. The relative expression levels of miR-105 were higher in esophageal cancer cells than that in normal Het-1A. 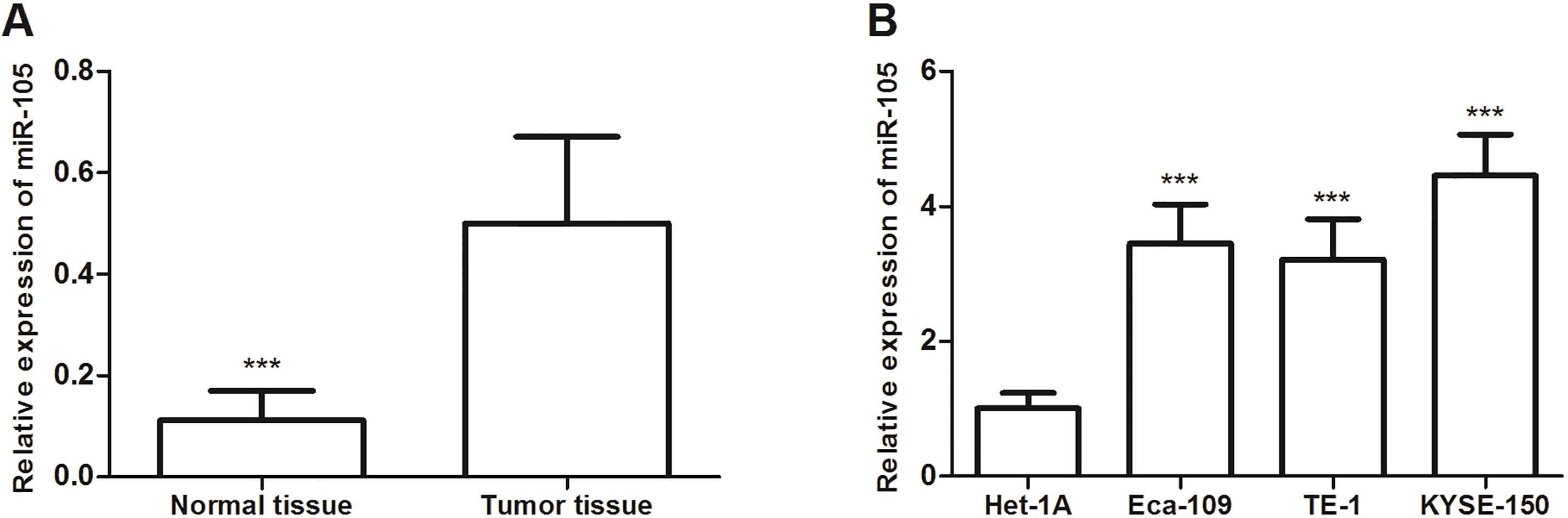
Total RNAs were extracted by TRIzol reagent (Thermo Fisher Scientific, Waltham, MA, USA) from esophageal cancer tissues and cell lines. RNAs were reverse transcribed to cDNA by PrimeScript II 1
Cell proliferation assay
Cell proliferation ability was detected with cell counting kit-8 (CCK-8; Dojindo, Kumamoto, Japan) following instructions. Transfected cells were seeded into 96-well plates and cultured at 37
Cell migration and invasion assays
Cell migration and invasion abilities were measured using 24-well transwell chambers with 8
Statistical analysis
Data are presented as the mean
Results
miR-105 was upregulated in esophageal cancer tissues and cell lines
The expression of miR-105 was detected using qRT-PCR assay. As shown in Fig. 1A, the miR-105 expression level was significantly upregulated in esophageal cancer tissue specimens compared with matched adjacent normal tissues (
According to the median expression level of miR-105 in esophageal cancer tissue specimens, the patients were divided into low miR-105 expression group (
The relationship between miR-105 expression and clinical characteristics in patients with esophageal cancer
The relationships between miR-105 expression levels and clinicopathological characteristics of eso- phageal cancer patients were analyzed using the
The prognostic significance of miR-105 in esophageal cancer
To explore the prognostic significance of miR-105 in esophageal cancer, survival curves were conducted by the Kaplan-Meier method and compared by the log-rank test. Esophageal cancer patients with low miR-105 expression levels exhibited longer overall survival than those with high miR-105 expression levels (log-rank test,
Multivariate Cox analysis of factors for the overall survival of esophageal cancer patients
Multivariate Cox analysis of factors for the overall survival of esophageal cancer patients
Esophageal cancer patients with high miR-105 expression levels had shorter overall survival than those with low miR-105 expression levels (log-rank test, 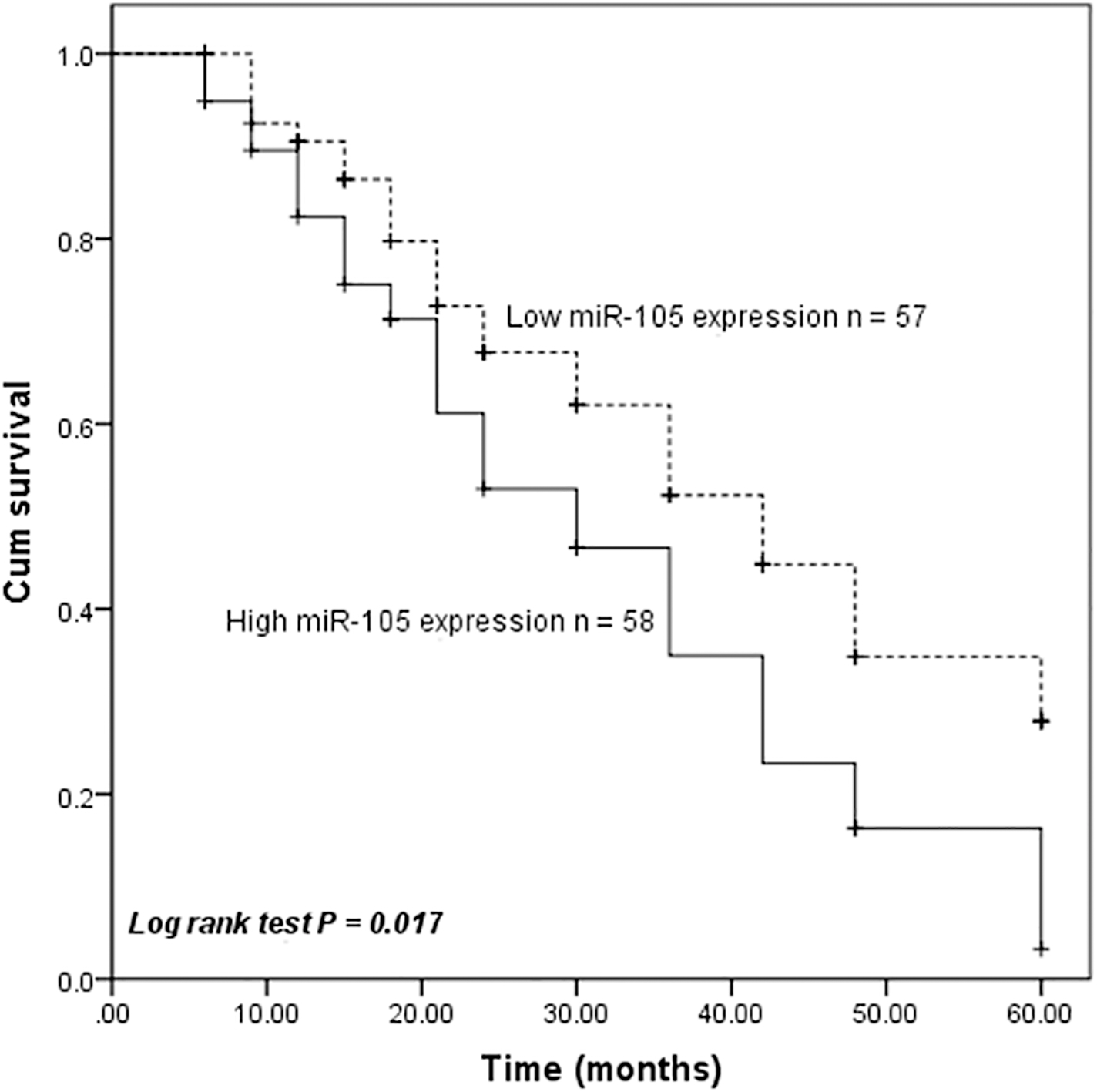
miR-105 overexpression and inhibitor affect the proliferation ability of human esophageal cancer Eca-109 and KYSE-150 cells. A. The expression levels of miR-105 were analyzed following transfection with miR-105 mimic, inhibitor, and negative controls. B. The Eca-109 and KYSE-150 cell proliferation were analyzed by CCK-8 assay. 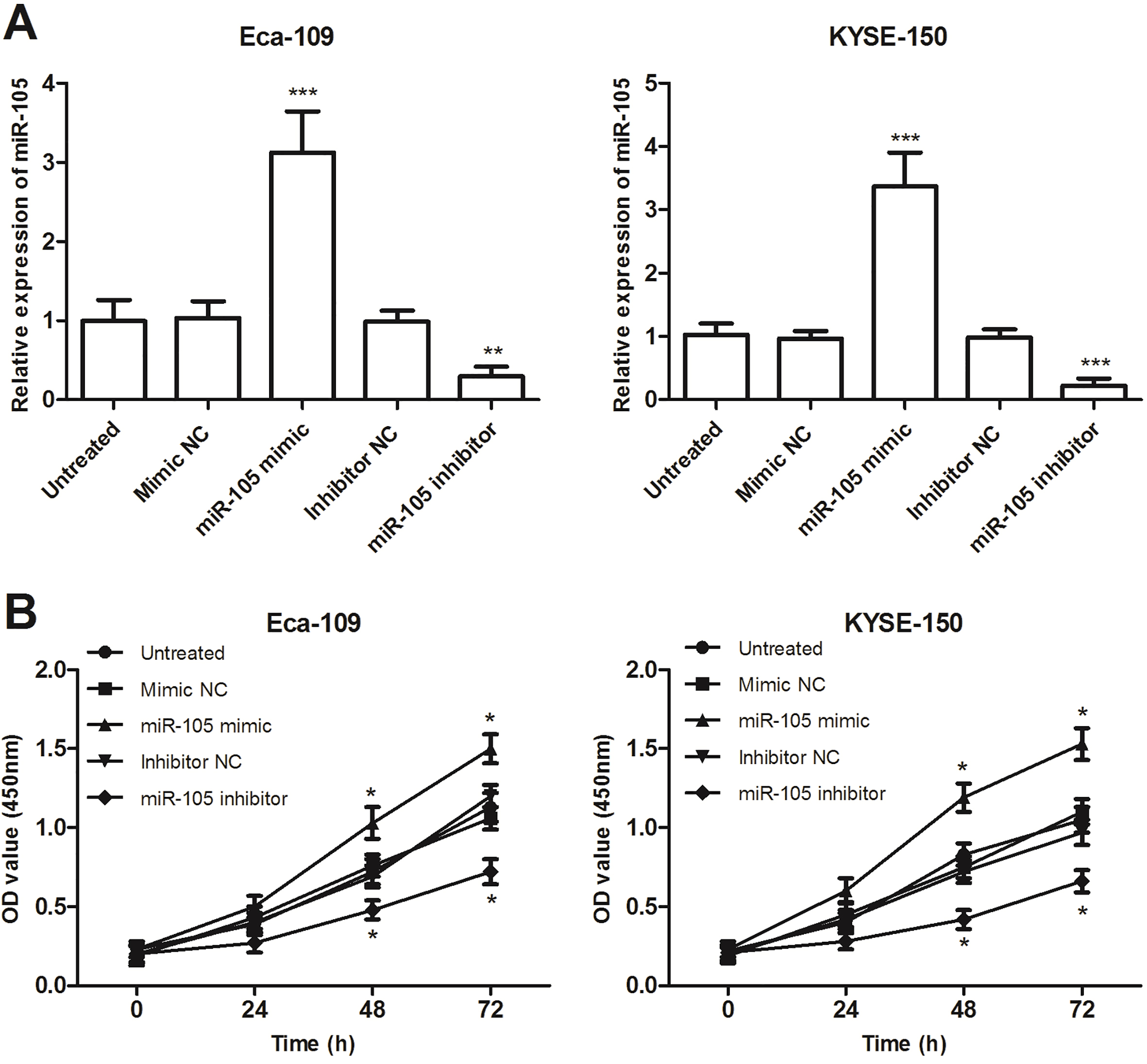
miR-105 promotes the migration and invasion of esophageal cancer Eca-109 and KYSE-150 cells. A. Transwell migration assays using Eca-109 and KYSE-150 cells transfected with miR-105 mimic, inhibitor, or negative controls. B. Transwell invasion assays using Eca-109 and KYSE-150 cells transfected with miR-105 mimic, inhibitor, or negative controls. 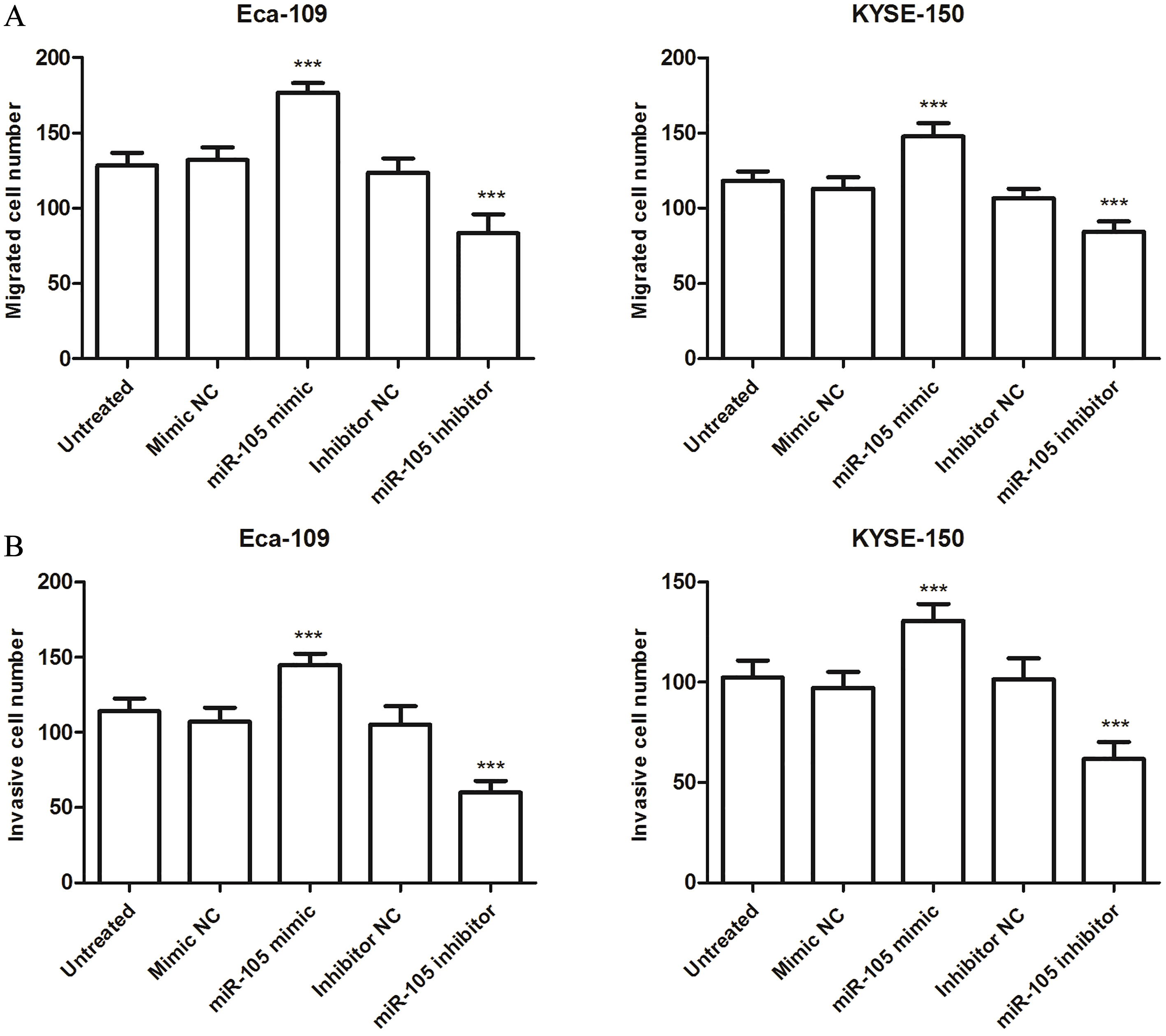
According to the above-mentioned results in cells, the expression of miR-105 in Eca-109 and KYSE-150 exhibited relatively higher in three esophageal cancer cells, thus both the cell lines were selected for the following experiments. We overexpressed/downregulated miR-105 in Eca-109 and KYSE-150 cell lines and examined the effects of miR-105 on proliferation, migration, and invasion. As shown in Fig. 3A, using qRT-PCR, we confirmed miR-105 mimic significantly increased miR-105 expression, while miR-105 inhibitor significantly decreased miR-105 expression in both Eca-109 and KYSE-150 cell lines, compared with that in untreated cells (
Discussion
In the present study, we indicated that miR-105 was significantly upregulated in esophageal cancer tissues and cell lines compared with that in adjacent normal tissues and normal Het-1A cells, respectively. In addition, we observed that high miR-105 expression was independently associated with positive lymph node metastasis, advanced TNM stages, and unfavorable overall survival. These findings suggest that miR-105 might be an independent prognostic biomarker and be an oncogene gene in esophageal cancer. What’s more, we explored the effects of miR-105 on cell proliferation, migration, and invasion in esophageal cancer cells and found that miR-105 overexpression significantly promoted these cellular activities, while downregulation of miR-105 significantly subdued these cellular activities.
Numerous miRNAs have been identified to play important roles in tumor progression in a variety of tissues and to serve as diagnostic/prognostic biomarkers [21, 22, 23]. For instance, lower levels of miR-4262 predict poorer prognosis in gastric cancer patients and inhibit cell proliferation and invasion in gastric cancer by targeting proto-oncogene CD163 [24]. In esophageal cancer, tissue and serum miR-1290 levels were both increased in esophageal cancer, and its expression level could reflect the progress of esophageal cancer, suggesting miR-1290 might be a useful diagnostic and prognostic biomarker of esophageal cancer [25]. Another study indicated that miR-145 expression is significantly decreased in esophageal squamous cell carcinoma (ESCC) tissues and miR-145 may be a prognostic marker for predicting the overall survival of patients with ESCC [3]. These studies suggested that identifying functional miRNAs in tumor progression is important for the improvement of cancer treatment.
In this study, we used qRT-PCR analysis to detect the expression of miR-105 in esophageal cancer tissues and cell lines. Our findings showed the expression of miR-105 was significantly increased in both esophageal cancer tissues and cell lines compared to respective controls. The expression pattern of miR-105 in esophageal cancer was consistent with that in many other cancers, such as colorectal cancer [26] and triple-negative breast cancer [27]. The further clinical-based analysis showed that miR-106 expression was significantly associated with lymph node metastasis and TNM stage. The above results suggested miR-105 expression may be associated with the development of esophageal cancer. Considering metastasis and TNM stage are usually associated with overall survival, we speculate miR-105 expression may correlate with clinical outcomes of esophageal cancer patients. Kaplan-Meier curve method and Cox regression analysis results showed a high expression of miR-105 correlated with poor clinical outcomes and maybe an independent prognostic biomarker for esophageal cancer. The results are consistent with those from previous studies on various types of tumors [27, 28]. For instance, a previous study also indicated that miR-105 was associated with overall survival and disease-free survival in non-small cell lung cancer [28].
A number of studies have demonstrated that miR-105 functions as an oncogene or a tumor suppressor depending on tumor contexts [19]. miR-105 was downregulated in gastric carcinoma tissues and cells and inhibited cell progression of gastric cancer by targeting SOX9 [29]. High expression of miR-105 positively correlates with the clinical prognosis of hepatocellular carcinoma by targeting NCOA1 [30]. In non-small cell lung cancer (NSCLC), overexpression of miR-105 significantly promotes cell viability and migration of A549 and Calu-3 cells and promotes the epithelial-mesenchymal transition (EMT) of NSCLC cells through upregulation of Mcl-1 [31]. By these findings, we speculate that the functional role of miR-105 may be tumor-specific. In this study, overexpression of miR-105 promoted cell proliferation, migration, and invasion of Eca-109 and KYSE-150 cells, while suppression expression of miR-105 inhibited these cellular behaviors, suggesting miR-105 may play an oncogene role in esophageal cancer. In colorectal cancer, overexpression of miR-105 functions as an oncogene role and promotes cell migration and EMT by directly targeting RAP2C [26]. We hypothesized miR-105 may also function as an oncogene in esophageal cancer to promote the progression of esophageal cancer by targeting RAP2C or other genes. Further studies will be carried out to figure out the detailed molecular mechanism of miR-105 in esophageal cancer progression.
In conclusion, we found that miR-105 is significantly upregulated in esophageal cancer tissues and cell lines. Overexpression of miR-105 promotes esophageal cancer proliferation, migration, and invasion of esophageal cancer cells. The findings provide evidence that miR-105 may serve as a prognostic biomarker and therapeutic target for esophageal cancer and be involved in the progression of esophageal cancer.
Footnotes
Conflict of interest
The authors declare that they have no conflict of interest.
