Abstract
BACKGROUND:
Male breast cancer (MBC) is a rare and aggressive disease. Thus, identification of new therapeutic targets is crucial.
OBJECTIVE:
Our objective was to evaluate the protein expression of MARCKS (Myristoylated Alanine-Rich C-Kinase Substrate) in MBC and to investigate its prognostic value.
MATERIALS AND METHODS:
MARCKS protein expression in tumor and stromal cells was analyzed by immunohistochemistry (IHC) in a retrospective series of 96 pre-chemotherapy MBC samples and 80 normal breast samples, from Tunisian patients treated at Salah Azaiez Institute. Correlations were searched between MARCKS expression and clinicopathological features including overall survival (OS).
RESULTS:
MARCKS was overexpressed in epithelial tumor cells in 66% of the MBC samples versus 26% of normal samples (
CONCLUSION:
MARCKS tumor cell overexpression might in part explain the aggressiveness and the poor prognosis of MBC. MARCKS can represent a potential therapeutic target for MBC.
Introduction
Male breast cancer (MBC) is a rare disease and represents less than 1% of all cancers in males and 1,6% of breast cancers [1, 2]. Over the last four decades the incidence of MBC has increased with approximately 1 in 100,000 [3] but remains still very low compared to breast cancer in women [4]. Despite the rarity of MBC, its case-fatality rate is similar to that of breast cancer in women [4]. The prognosis of MBC remains poor due to the higher rate of advanced stages at presentation because of the late diagnosis and at present there are no treatment strategies specific for male breast cancer.
Myristoylated Alanine-Rich C-Kinase Substrate (MARCKS), is a substrate for protein kinase C, is located in the plasma membrane and is an actin filament cross-linking protein. MARCKS is phosphorylated by protein kinase C or by binding to the complex calcium-calmodulin, these effects lead to its translocation in the cytoplasm. MARCKS appears to play an essential role in the regulation of cytoskeletal plasticity and especially actin filaments [5, 6]. It is ubiquitously expressed in various tissues [7], and is associated to phagocytosis, cell adhesion, cytokines secretion membrane trafficking and mitogenesis [8, 9, 10, 11]. Different studies have demonstrated the implication of MARCKS in cancer aggressiveness explaining its association with high grade and stage at diagnosis, notably proliferation and metastatic process and therapeutic resistance [12, 13, 14, 15, 16, 17, 18, 19]. Besides, many studies have showed the efficiency of therapeutic inhibition of MARCKS [20, 21].
In this study, we sought to investigate the MARCKS expression level in male breast cancer and to evaluate its prognostic impact in male patients with breast cancer.
Patients and methods
Patients and samples
We collected retrospectively 96 pre-therapeutic diagnostic tumor samples from male patients with breast cancer collected and treated at Salah Azaiez Institute of Tunis, Tunisia between 2004 and 2013. Main inclusion criteria were male patient, with clinically-defined and pathologically-confirmed invasive breast carcinoma, written informed patient’s consent, available formaldehyde-fixed and paraffin-embedded pre-therapeutic diagnostic tumor sample and clinicopathological annotations. As control group; we collected 80 normal breast tissues from Tunisian men operated for non-tumor breast lesions. Clinicopathological annotations included patients’ age at diagnosis, TNM stage, pathological features (histological type, grade, tumor size and axillary lymph node status), IHC statutes of ER, PR, HER2 and Ki67 proliferation index used as IHC markers for breast cancer intrinsic subtype’s definition according to the St.Gallen criteria [22]. Our study was approved at Salah Azaiez Institute by our institutional ethics committee.
Immunohistochemical analysis
MARCKS protein expression was analyzed on standard slides for the 96 MBC and 80 normal tissues using standard IHC protocols. IHC was performed on 4-
Western blot analysis
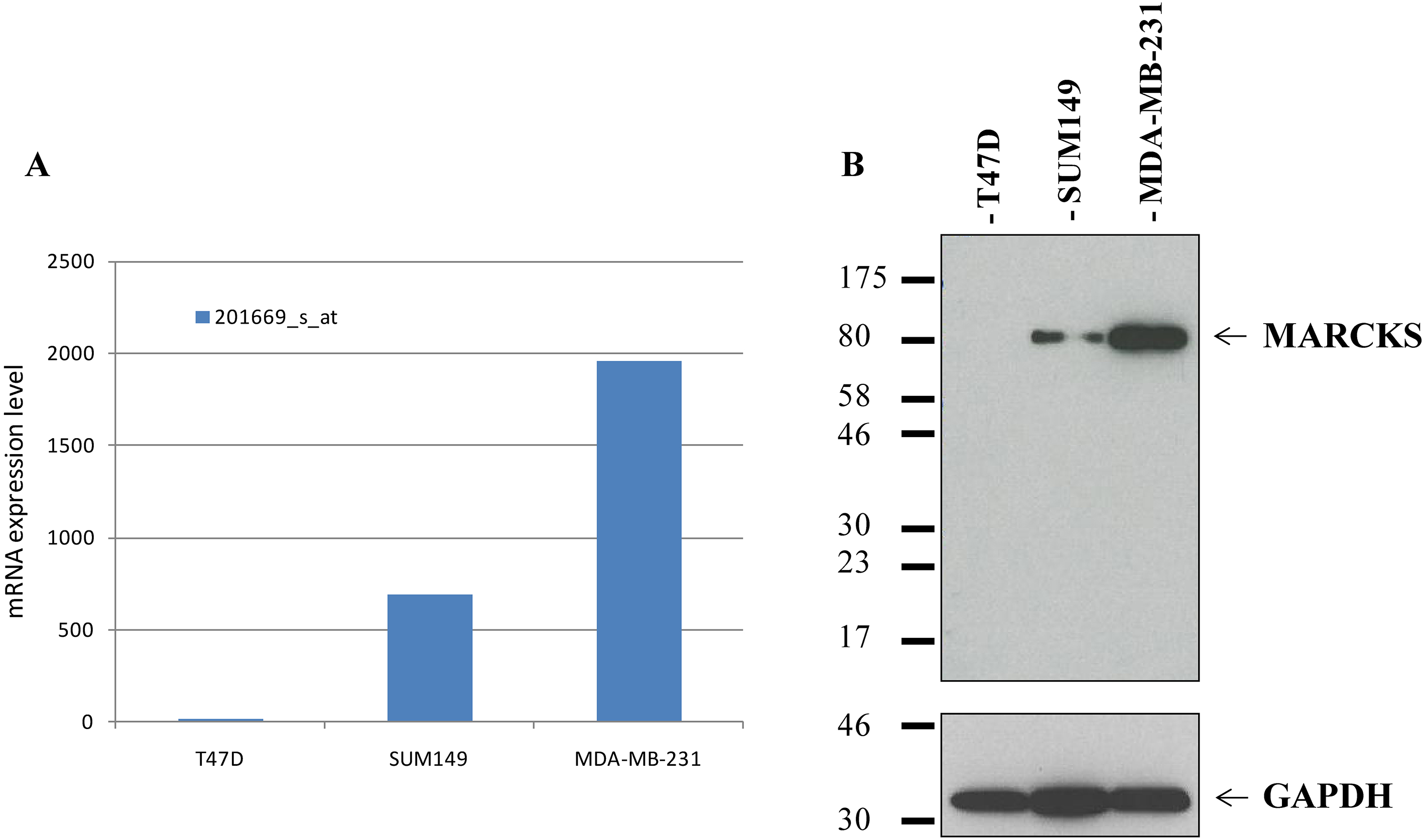
Firstly, we validated our MARCKS antibody before IHC analysis by using western blot analysis with breast cancer cell lines. The expression was analyzed in three breast cancer cell lines (T47D, SUM149, MDA-MB-231) previously profiled using Affymetrix (Thermo Fisher Scientific, Inc., Rockford, IL, USA) DNA microarrays and for which MARCKS mRNA expression was documented as very low (T47D), moderate (SUM149), and very high (MDA-MB-231) (Supplementary Fig. 1) [13, 23]. Cells were washed three times with ice-cold PBS and then resuspended for 30 min in 750
Statistical analysis
Data were summarized by numbers and percentages for categorical variables, and median and range for continuous variables. Correlations between MARCKS expression and clinicopathological features were analyzed using the
Results
Male breast cancer population and clinicopathological features
The clinicopathological features of tumor samples (
Clinicopathological characteristics of patients
Clinicopathological characteristics of patients
IDC
MARCKS (myristoylated alanine-rich C-kinase substrate) immunostaining in male breast cancer samples. (A) (a–b) Representative images (200) of negative MARCKS immunohistochemistry staining, (c–d) Representative images (200) of MBC samples with weak expression of MARCKS in tumor cells: 10% (c) and 20% (d); (e–g) Representative images (200) of MBC with strong expression in tumor cells: 60% (e), 80% (f) and 100% (g). (c) Box plots showing the percentage of MARCKS-positive (black) samples and MARCKS-negative (white) samples (normal, primary tumors) for the epithelial staining (left) and stromal staining (right). The p-values are for the Fischer’s exact test. (B) Box plots showing the percentage of positive MARCKS expression (black) and negative MARCKS expression (white) in normal (left) and primary tumor (right) samples for epithelial staining. The p-values are available for the Fisher’s exact test.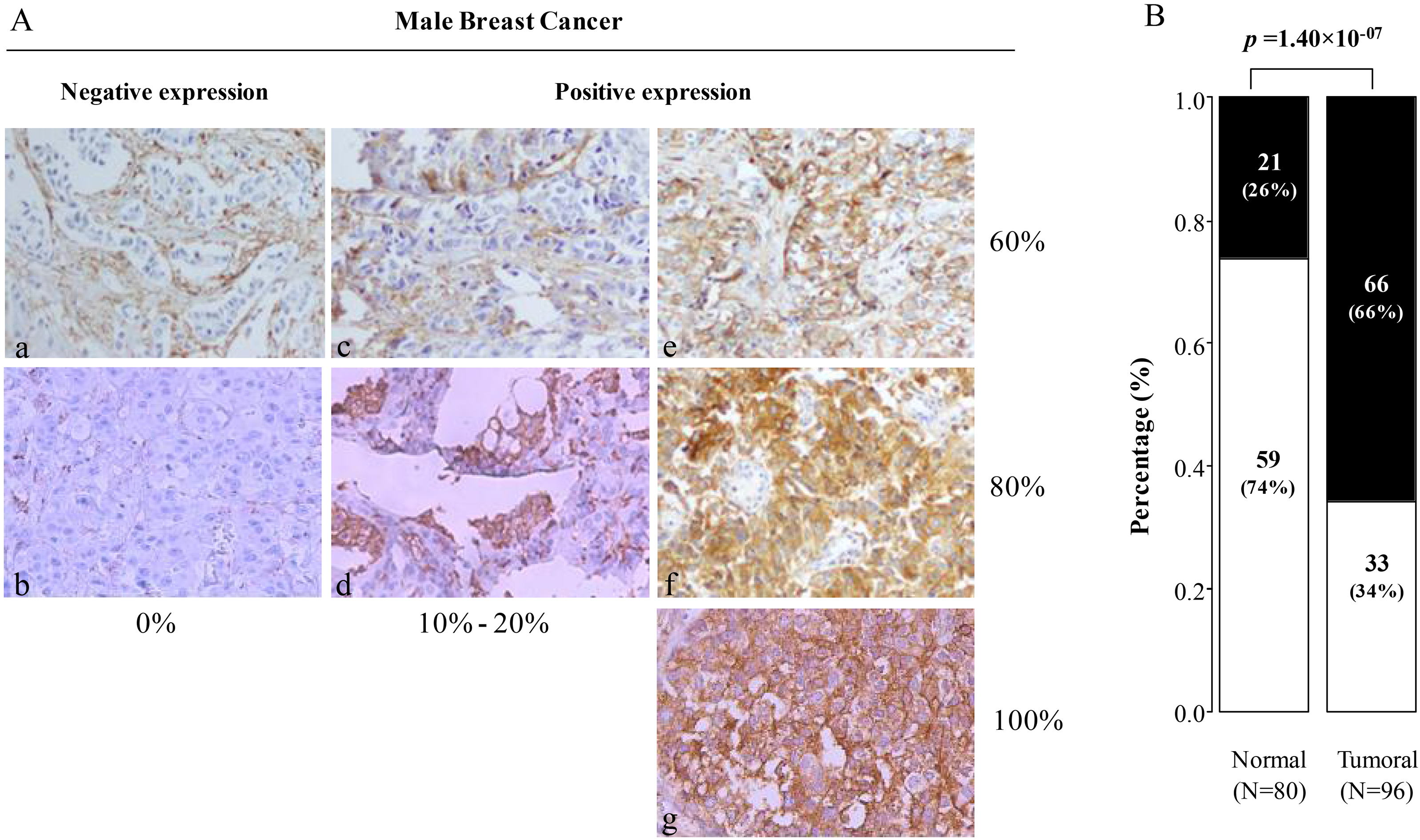
Before IHC analysis, we validated the MARCKS antibody using western blot analysis on three breast cancer cell lines with known MARCKS mRNA expression. As shown in Supplementary Fig. 1, the antibody specifically recognized MARCKS protein with a good correlation between protein and mRNA expression levels.
Using IHC analysis; MARCKS expression was then measured on the 96 tumor samples and compared to 80 normal samples. Examples of staining are shown in Fig. 1. A and results are summarized in Fig. 2. The staining was observed mainly in tumor cells and very weakly in stromal cells. The percentage of positively stained tumor cells ranged from 0% to 100%; using 1% of positive staining cut-off as used in our previous studies [13, 14], we found that MARCKS was expressed in 66% (66) of samples, whereas 34% [33] were MARCKS-negative (
Overall Survival according to MARCKS expression in MBC patients in epithelial and stromal cells. (A) Kaplan Meier analysis of OS curves in MBC patients with positive and negative MARCKS expression in tumor cells. (B) Similar to (A), but in stromal cells. The 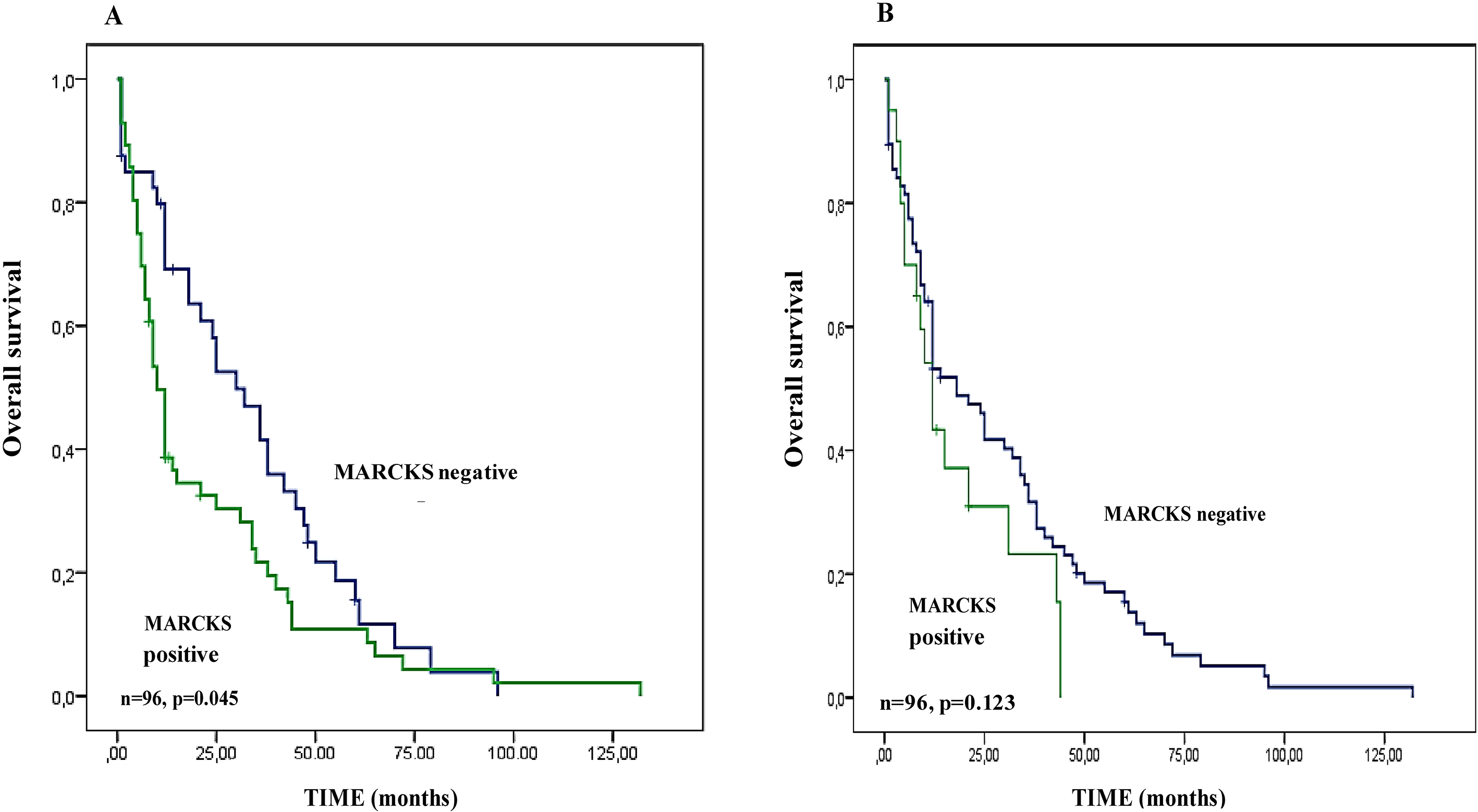
We searched for correlations between the MARCKS IHC status in tumor cells and in stroma cells and the prognostic clinicopathological features of samples including patient’s age at diagnosis, TNM stage, pathological features (type, tumor size, grade, axillary lymph node status, stage and distant metastasis), ER, PR and HER2 IHC statutes. As shown in Table 2, we found only a significant association between MARCKS tumor cell expression and HER2-positivity (
Epithelial and stromal MARCKS expression in tumor tissues and clinicopathological characteristics in male breast cancer
Epithelial and stromal MARCKS expression in tumor tissues and clinicopathological characteristics in male breast cancer
IDC
We assessed the prognostic value of MARCKS epithelial expression and MARCKS stromal expression for OS in the population of 96 patients. The 5-year OS rate was 20% [95 CI, 0.04–0.52]. In univariate analysis (Table 3), the patient’s age (
Univariate and multivariate analyses for overall survival in male breast cancer patients
Univariate and multivariate analyses for overall survival in male breast cancer patients
HR
Regarding the MARCKS stromal expression, we did not find a correlation between positive expression and OS (
Male Breast cancer is a rare entity for which biological studies are limited. Despite the introduction of multimodality treatment, the 5-year survival remains poor. Some studies indicate that the prognosis for breast cancer is more pejorative in men than in women, in general, is due to later diagnosis with an advanced stage of the disease [24]. More recently, several studies conducted on MBC outlined some notable biological differences compared to Female Breast Cancer (FBC) in prognosis and survival [25], genomic profiling and prognostic biomarkers [26, 27, 28, 29, 30]. Thus, management of MBC should not be extrapolated from research studies carried out on FBC patients. Given its aggressiveness, the identification of new therapeutic targets is crucial in MBC.
Here, our objective was to evaluate the protein expression of MARCKS in an extensive retrospective series of 96 MBC clinical samples and 80 normal samples collected from Tunisian patients and to search for correlations with tumor features. Our MBC series was coherent with literature in term of clinicopathological characteristics such as presence of papillary carcinomas as second most frequent pathological type [31]. We showed that MARCKS expression in tumor cells was associated with HER2 status and with shorter OS independently from other prognostic clinicopathological features.
We focused on MARCKS protein expression for many reasons: i) the role of MARCKS in cancer progression including metastatic process and therapeutic resistance; ii) commercial availability of a corresponding monoclonal antibody performing sufficiently well in IHC on paraffin-embedded tissues as previously reported [19, 32]; iii) availability of MARCKS inhibitors [33]. Before analysis of tissue samples, we revalidated our antibody on breast cancer cell lines by using western blot analysis. As expected, given the extreme rarity [34], there was no case of bilateral male breast cancer in our series.
MARCKS expression was heterogeneous in our MBC series. We found that 66% of cases showed a MARCKS-positive tumor cell staining and 34% were MARCKS-negative. In the literature, only two studies analyzed MARCKS protein expression using IHC in FBC but none in MBC. The first study was about 250 breast cancers [32], using IHC analysis with the same antibody and positivity cut-off (1% positive staining) as us in our present study, Browne et al. reported that and 40% of tumor samples had MARCKS-positive staining. Thus, higher frequency of positivity that we found (66%) could explain the aggressiveness of MBC versus FBC. The same study found a significant association of MARCKS expression with poor-prognosis features: metastatic stage, high grade, ER-negativity, PR-negativity, HER2-positivity, triple-negative and HER2+ statutes and with poor specific OS in all cases. HER2-positive tumors are rare in men with breast cancer, the frequency of this subtype ranges from 1 to 2% of MBC cases. Interestingly, in our study, we showed a positive correlation between epithelial MARCKS expression and HER2 status. But, giving the small number of the HER2 positive population, these results needs to be validated in a larger series. Moreover, stromal MARCKS expression was only significantly correlated to tumor grade. In our prior study [13], on an extensive series of 502 FBC cases (133 inflammatory breast cancer (IBC) and 369 non-IBC), we found MARCKS positivity in 11% of non-IBC samples, 36% of IBC, and 18% of all samples versus 66% in our present MBC series. We also showed that MARCKS expression was correlated with poor-prognosis features in the whole series of samples; MARCKS-positivity was more frequently associated with clinical axillary lymph node positive status, metastatic stage, high grade, ER-negativity, PR-negativity, HER2-positivity, and triple-negative and HER2 statutes. In the same study of IBC samples, MARCKS expression was associated with poor metastasis free survival (MFS) in a univariate and multivariate analysis [13]. Here, we confirmed the association of epithelial MARCKS-expression and HER2 status, and interestingly in term of OS we showed a correlation between the epithelial cells MARCKS-positivity and shorter OS in univariate and multivariate analysis. In a recent study, Chen et al. demonstrated frequent high IHC staining for phospho-MARCKS in breast cancer vs. normal breast tissue and showed a positive correlation with unfavorable histo-prognostic parameters and metastatic status. Using both in-vitro and in vivo models of breast cancer, they then demonstrated the implication of p-MARCKS in resistance to paclitaxel treatment and showed that the reduction of p-MARCKS by knockdown or by treatment with MANS (inhibitor peptide targeting the N-terminal myristoylation site of p-MARCKS) promoted paclitaxel sensitivity [35]. In this study, several mitotic inhibitors currently used in breast cancer such as paclitaxel, and eribulin, promoted phospho-MARCKS accumulation in multiple breast cancer cells, Altogether, these observations suggest the clinical potential of MARCKS inhibitors in breast cancer resistant to chemotherapy. All these studies suggested pivotal roles of MARCKS in the regulation of breast cancer aggressiveness, and therapeutic resistance, notably to hormone therapy and chemotherapy. In this context, the overexpression of MARCKS in MBC seems coherent especially since it was associated with unfavorable prognostic value for OS.
Different studies in other types of cancers showed; involvement of tumor MARCKS expression in cancer progression, chemoresistance, and also suggested that MARCKS inhibition could be a novel therapeutic approach. In colorectal cancer, MARCKS has a complex and vital role in the progression of colorectal cancer and its implication in the cell motility, invasion and proliferation, whereas inhibition of MARCKS clearly affects these cancer features and reduces metastases event [16]. In cholangiocarcinoma, MARCKS overexpression correlated with poor survival. Then, in vivo, the authors highlighted the crucial role of MARCKS in the migration of cholangiocarcinoma cells [17]. In melanoma, Chen and Rotenberg showed the contribution of p-MARCKS in the metastatic potential [12]. In squamous cell lung carcinoma, a significant correlation was reported between positive IHC expression and poor survival [15]. Still, in lung cancer studies, MARCKS, specifically its phosphorylated form, was reported as one of the key players in potentiating lung cancer cell migration/metastasis and the study suggested a potential use of MARCKS-inhibition peptides in the treatment of lung cancer metastasis [21]. The authors also demonstrated in another study that overexpression of p-MARCKS was associated with unfavorable survival and targeting MARCKS phosphorylation site domain (PSD) with MPS peptide (MARCKS phosphorylation site domain)suppressed tumor growth, metastasis and increased the sensibility to erlotinib treatment in vivo and in vitro [20]. A recent study in lung cancer demonstrated that a peptide mimetic of MARCKS ED inhibited MARCKS phosphorylation and leading to an increase in sensitivity to radiation [33]. In prostate cancer, MARCKS promotes migration and invasion [36]. In kidney cancer, MARCKS inhibition with MPS peptide synergistically interacted with regorafenib treatment and decreased survival of kidney cancer cells through inactivation of AKT and mTOR [37]. Finally, in a recent study about 118 epithelial ovarian carcinomas, authors showed that MARCKS expression was more frequent in stromal cells than in epithelial cells. Stromal MARCKS expression was associated with poor OS. Here we found an association of epithelial MARCKS expression with a poorer OS [14], but it is also possible that the stromal MARCKS overexpression in tumors observed in 21% of cases contributes to cancer-associated fibroblasts activation and to the poor prognosis.
Conclusion
In conclusion, we demonstrated that MARCKS expression was more frequently positive in Male Breast Cancer compared to its female counterpart. Moreover, the association of MARCKS overexpression with shorter OS in MBC was in accordance with several studies reported in female breast cancer and other solid tumors (colorectal, ovarian, lung, cholangiocarcinoma, melanoma, prostate, and kidney), its overexpression might in part explain the poor prognosis of MBC. Despite the rarity of MBC disease, the strengths of our study include the relatively important number of cases tested with a total of 96 male breast tumors, which represent rare but aggressive tumors. To our knowledge, this is the first study analyzing specifically MARCKS protein expression in MBC. The main limitations of our study consist in its retrospective nature and the number of 96 samples, clearly warranting analysis of a larger series. Taken together, our results suggest that MARCKS overexpression might in part explain the unfavorable prognosis of MBC in term of OS. This finding might represent a new potential target for a therapeutic approach in MBC.
Footnotes
Acknowledgments
Maroua MANAI received a grant (grant no. 15G08 05) from the Joint Committee of University Cooperation PHC Utique in a Franco-Tunisian mixed research project, the National League Against Cancer (label DB, R16009AA), SIRIC (INCa-DGOS-Inserm 6038 grant) and the Pink Rubbon (R16024AA).The authors would like to thank the teams at the Department of anatopathology, Salah Azaiez Institute of Tunis-Tunisia, Inserm and Paoli-Calmettes Institute, Marseille, France. The authors would like to thank the Ministère d’Enseignement Supérieur de la Recherche Scientifique de Tunisie and University of Tunis El Manar.
Conflict of interest
The authors declare no conflict of interest.
Supplementary data
MARCKS mRNA and protein expression in breast cancer cell lines. (A) mRNA expression level measured using Affymetrix DNA microarrays in three breast cancer cell lines with different expression levels. (B) Western blot analysis using the anti-MARCKS (D88D11) XP