Abstract
Background:
Male breast cancer (MBC) is a rare disease entity globally with poor prognosis compared with female breast cancer; however, studies reporting on MBC are rare especially in resource-limited settings.
Objectives:
We aimed to study the expression of Ki67 and its association with clinicopathological factors and intrinsic subtypes among male patients with breast cancer (BC) from a resource-limited setting.
Design:
This was a cross-sectional study which included retrospective data.
Methods:
The study included data of 54 males with BC who were diagnosed from January 2014 to December 2024. The study was conducted at the Department of Pathology, Uganda Cancer Institute (UCI) in Kampala Uganda from February to June 2025. Data were extracted from the electronic dataset and patients’ clinical files and laboratory forms. One-way analysis of variance statistical test was used to assess the association of Ki67 absolute value (mean) with clinical and pathological factors and intrinsic subtypes of BC, followed by performing multivariable linear regression analysis for adjusting for confounding factors.
Results:
The mean age of the patients was 56.4 ± 15.1 years, and the youngest patient had 25 years. Majority 68.5% (38/54) of the patients had advanced disease (stage III and IV). Also, majority 68.5% (37/54) of the cases comprised of ER+, PR+, and HER2− intrinsic subtype. Only intrinsic subtypes of BC (95% confidence interval [CI] = 3.397-16.503, P = .032) and PR status (95% CI = 5.693-24.397, P = .042) remained the predictors of Ki67 expression after performing multivariable linear regression analysis.
Conclusion:
The findings of this study have shown high expression in cases of MBC which are triple negative and negative PR status. This indicates that high Ki67 expression in MBC may be used in stratification of male patients with BC based on disease aggressiveness.
Plain Language Summary
Male breast cancer is a rare encounter in the management of patients with breast cancer globally which usually comprises of less than 1% of all breast cancers. Assessment of prognostic biomarkers such as Ki67 has been found valuable despite its challenges of lack of universal consensus of lack of cut-off point. Analysis of Ki67 expression and its association with clinicopathological factors in a cohort of males with breast cancer especially in low-and middle-income countries are scarce. Therefore, this study investigated the association of Ki67 expression and clinicopathological factors and also molecular subtypes of breast cancer. The expression of Ki67 in our study was associated with high tumor grade, advanced tumor stage, and lymph node involvement. Also, high expression of Ki67 was associated with negative estrogen receptor, positive HER2, and triple negative disease. Therefore, Ki67 should be regarded as a potential prognostic marker for patients with breast cancer even male breast cancer. It can help to stratify patients and also predict prognosis of the male breast cancer.
Introduction
Male breast cancer (MBC) still remains the rarest cancer compared with female breast cancer (FBC) which is the most common cancer among females globally. 1 Male breast cancer accounts for less than 1% of all breast cancer (BC) worldwide.2,3 Globally, the incidence rate is estimated to be 0.5 to 1.0 per 100 000. 4 While MBC is less common than FBC, its incidence and mortality rates have recently been observed to be increasing. 5 In Europe, the incidence rates are generally estimated to be less than 3 new cases per 100 000 men, and mortality rates are below 1 death per 100 000 men, with some geographical variations across various countries. 4 The incidence of MBC and FBC has been found to be higher in the African American and African populations, which stands at 1.4% and 4.2%, respectively. 6 In Uganda, a study which was conducted in Northern region reported that MBC accounted for 6.2% of BCs in that region. Another study which was conducted in the Central part of country reported that MBC accounted for 1.4% of all BC cases. 7 This suggests that the prevalence of MBC may vary within different regions of country. 8
Studies reporting on characterization of the intrinsic subtypes of MBC especially in low-and middle-income countries (LMICs) like Uganda are significantly rare. A few available studies regarding MBC conducted in LMICs have mainly described clinicopathological characteristics. Studies that analyzed the human receptors (HRs) and human epidermal receptor 2 (HER2) for MBC in LMICs have shown that most MBCs express estrogen receptor (ER) and progesterone receptor (PR) compared with FBCs.9,10 This is because men with higher than normal estrogen levels as well as lower testosterone levels in their blood are more susceptible to MBC. 11 ER+, PR+, and HER2− (luminal A) intrinsic subtype are considered the most prevalent type of BC among the 4 intrinsic subtypes of MBC, and it accounts for more than 75% followed by ER+, PR+/−, and HER2+/− (luminal B).6,12 -14 Luminal B also contributes for a significant number of MBC cases, and in some studies, ER+, PR+/−, and HER2+/− have even been reported to be the most prevalent intrinsic subtype of BC. 15 HER2-enriched (HER2-E) and triple-negative breast cancer (TNBC) or basal-like represent an even smaller fraction of MBCs compared with FBCs.16 -18
Determination of biomarkers such as Ki67 in patients with BC helps to stratify the patients based on the level of expression of such marker, which can determine aggressiveness of the disease and possibility of progression. Ki67 is one of the potential prognostic markers in patients with cancer including BC. This marker assesses tumor cell proliferation, and more importantly, the proliferation of Ki67 has also been found valuable in determining different survival analyses of MBC. High Ki67 has been associated with reduced overall survival in MBC.19,20 Low proliferation of Ki67 indicates low tumor growth rate and less aggressiveness. Studies have shown that ER+, PR+, and HER2− phenotypes of BC have low proliferation rate of Ki67 compared with other subtypes. Moreover, there is evidence that HER2-E and TNBC, which are the most aggressive intrinsic subtypes of BC, are associated with high expression of Ki67 compared with the luminal subtypes.21,22
In Uganda, there are limited data regarding characterization of the intrinsic subtypes of MBC, including their association with Ki67 expression. In addition, in Uganda, there is shortage of diagnostic services for patients with cancer, and usually patients are diagnosed at advanced stage. There is delay of diagnosis of patients with cancer as well as challenges related to treatment such as high cost of the chemotherapy drugs and availability. This is even worse for MBCs due to lack of knowledge among people in the country. Therefore, this contributes to limited studies on MBC despite fewer studies that have been reported regarding characterization of intrinsic subtypes of MBC in LMICs, including Uganda. Also, there is paucity of data regarding assessment of prognostic factors including conventional factors in MBCs in Uganda. Therefore, this study was undertaken so as to characterize the intrinsic subtypes of MBC using immunohistochemistry (IHC). Second, the study investigated the expression of Ki67 and its association with the intrinsic subtypes of BC and other clinicopathological factors in a cohort of MBC.
Methods
Study design and setting
This was a cross-sectional study which included retrospective data of male patients who were diagnosed with BC from January 2014 to December 2024. The study was conducted from February-November 2024 to March 2025-June 2025 at the Department of Pathology, Uganda Cancer Institute (UCI) in Kampala, Uganda. The department receives specimens mainly from Mulago National Hospital (MNH), which is the largest national hospital. However, other health facilities within Kampala and outside areas of the city as well as neighboring countries such as South Sudan and Kenya also submit specimens. Uganda Cancer Institute is the largest cancer center in the East African region.
Study population
The study included male patients who were diagnosed with BC from January 2014 to December 2024. The inclusion criteria included being male with confirmed diagnosis of BC and having available formalin-fixed paraffin-embedded (FFPE) tissue block with good quality. All female BC cases, cases with missing tissue blocks, spoilt FFPE blocks with insects, and MBC cases from other neighboring countries such as South Sudan and Kenya were excluded from the analysis.
Sample size estimation and sampling procedure
Sampling was based on convenience sampling method due to rarity nature of the disease which could not allow any sort of probability sampling. Therefore, all cases that met the inclusion criteria were selected consecutively. In total, 54 cases finally met the inclusion criteria and were included in the analysis.
Histopathological re-evaluation of the selected cases
The selected cases were histologically re-evaluated. The FFPE tissue blocks were sectioned at thickness of 4 μm thickness, followed by de-waxing them by placing them in the microwave at 60°C for 50 minutes, and then cleared in 2 changes of xylene using 10 dips in each. Hydration was done by dipping the sections 10 times in the following changes of alcohol: 100%, 95%, 80%, and 70% ethanol concentration followed by rinsing in running tap water and then stained with routine hematoxylin and eosin (H&E) stains. The slides were reviewed using a light microscope (Olympus Corporation, CX31RBSF Model, Tokyo, Japan) by 2 independent pathologists (JJY and TO), and for cases with discrepancy of the score, consensus was reached through a discussion to agree on the score. Histopathological classification of BC in this study was based on the study of Rakha et al. 23 Also, tumor grading and staging were based on the protocols from the College of American Pathologists (CAP). 24
Immunohistochemical staining of Ki67
The IHC-stained FFPE tissue sections were used for the assessment of Ki-67. MIB-1 antibody staining for Ki67 was evaluated at a magnification of ×400 in “hot spot” areas. The calculation of the percentage of positivity of Ki67 was performed manually by counting at least 1000 tumor cells at high-power fields at magnification of ×400 as it was done previously. 25 All brown-stained nuclei, regardless of staining intensity, were counted as positive. The cut-off value of Ki67 was ⩽20% based on the St. Gallen Consensus Guidelines 26 and the work by Dokcu et al. 22 Ki67 expression was associated with the intrinsic subtypes of BC as well as clinical and pathological factors so as to determine aggressiveness of the disease. This is because expression of Ki67 indicates tumor cell proliferation and aggressiveness. The expression of Ki67 was categorized as low (<20%) and high (⩾20%) due to the reason that such a threshold value could predict survival of the patients with BC and is also associated with other adverse prognostic factors despite lack of consensus for its clinical utility. 27
Immunohistochemical staining of ER, PR, and HER2
The tissue blocks were sectioned at thickness of 4 μm and placed on charged glass slides (FrostStat, DAKO-Denmark), and then deparaffinized at 50°C for 30 minutes on a hot plate. The slides were then heated at 750 watts in a microwave for 10 minutes using 10 mmol/L of tris-buffered solution of pH 7.0 as done previously. 28 Thereafter, the FFPE tissue sections were dipped in 3% of H2O2 solution for 10 minutes to block endogenous peroxidase activity to prevent background staining. The tissue sections were rinsed in phosphate-buffered solution followed by pretreating with a secondary antibody amplifier horseradish peroxidase, and then followed by rinsing in buffered water. We incubated the tissue slides with monoclonal mouse anti-ER, anti-PR, and anti-HER2 antibodies (DAKO Company, Denmark) for 30 minutes at room temperature. Diaminobenzidine tetrahydrochloride (DAKO LSAB2, Denmark) solution was added onto the tissue sections for 10 seconds for detection purpose. Harris hematoxylin was used to counterstain the tissue slides for 30 seconds followed by dipping the slides in 10 dips of 2 changes of xylene for clearing. The slides were finally mounted using DPX and cover-slipped. Reporting of the IHC-stained tissue slides was done as previously. 28
Molecular subtyping of BC and assessment of PR-negative status
The intrinsic subtypes of BC in our study were categorized into 4 types including ER+, PR+, and HER2− (luminal A), ER+, PR+/−, and HER2+/− (luminal B), HER2-enriched (HER2 positive), and TNBC as previously done. 29 In addition, emphasis on the assessment of the level of PR negative status in the study was also made.
Data collection procedure
Data were collected using a self-designed data sheet. The sheet was consisting of 3 sections (independent variables) which included demographic and clinical characteristics (age of the patients, disease duration, laterality, tumor stage, and treatment approaches), pathological characteristics (histological subtypes of BC, tumor grades, and lymph node involvement [LNI]), and IHC characteristics (ER, PR, HER2, and intrinsic subtypes of BC). Another immunohistochemical factor was Ki67 expression (outcome). Data were extracted from both electronic dataset and patients’ files and laboratory requisition forms and entered in the prepared data sheet. However, the collected data were checked for possibility of double entry after both sources of the data were compiled.
Statistical analysis
Statistical analysis was performed using Statistical Package for Social Sciences software version 22.0 (IBM Corp., Armonk, New York). Data cleaning for missing, duplicate, and wrongly entered data were cross-checked by running frequencies and percentages or crosstabs. All descriptive statistics regarding patients’ clinical, pathological and immunohistochemical characteristics were presented in frequencies and percentages. Continuous variables were summarized in mean ± standard deviation (SD). The association of absolute Ki67 values with the other clinical and pathological variables was assessed using 1-way analysis of variance (ANOVA) statistical test. Testing of the normal distribution of the Ki67 expression values was done using Shapiro-Wilk test, and normality was assumed when P > .05. All independent variables which had P < .2 in 1-way ANOVA was fitted into multivariable linear regression analysis so as to control for confounding factors. For categorical variables, we checked homogeneity of variance assumption by running Levene’s test. A 2-tailed P < .05 was considered statistically significant. The reporting of this study conforms to the STROBE guidelines for observational studies 30 (Supplementary file).
Results
Selection process of the cases for analysis
Figure 1 presents the flow chart of selection process of the analyzed cases. Of all the histological diagnoses for the period of 10 years, 28.7% (1403/4895) were breast conditions, among which 5.6% (78/1403) consisted of benign breast diagnoses. Majority 95.4% (1264/1325) of cases with BC comprised FBC, and MBC consisted of 4.6% (61/1325). A total of (54/61) of MBC cases were included in the analysis.
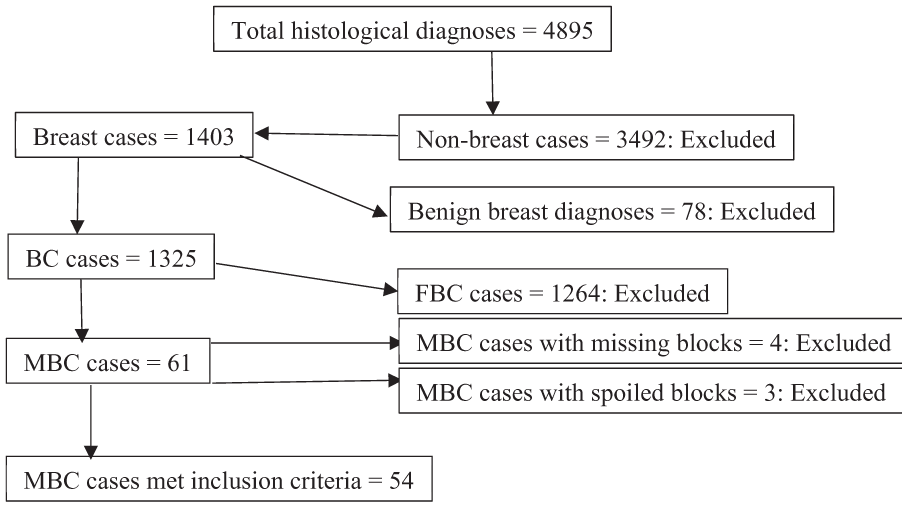
Flow chart indicating selection process of the MBC cases in the study.
Clinical and pathological characteristics of the patients
The mean age of the patients was 56.4 ± 15.1 years, and most of the patients 40.7% (22/54) were aged 60 years or above. Half of the patients, 50.0% (27/54), had BC involving the left breasts, and most of the patients, 42.6% (23/54), took more than 5 months with the condition before seeking medical intervention, with average disease duration of 7.0 ± 3.0 months. Majority 68.5% (38/54) of the patients had advanced disease. Also, majority, 85.2% (46/54), of the tumors consisted of infiltrating ductal carcinoma. In addition, almost half, 48.1% (26/54), of the patients had moderately differentiated disease (grade 2) (Table 1).
Demographic, clinical, and pathological characteristics of the patients (N = 54).

Clinical features of the patients
The clinical features of the patients included in this study are shown in Figure 2. Majority, 74.1% (40/56), of the patients were presenting clinically with painless subareolar masses. The second most common clinical feature was lymphadenopathy which accounted for 22.2% (12/56) of the patients. Only 3.7% (2/54) of the patients were presenting with gynecomastia.

Clinical features of males with BC.
ER, PR, HER2 and Ki67 expression, and intrinsic subtypes of BC
The expression of ER, PR, and HER2 was observed in 92.6% (50/54), 74.1% (40/54), and 5.6% (3/54), respectively. The ER+, PR+, and HER2− subtype consisted of majority, 68.5% (37/54), of the intrinsic subtypes followed by ER+, PR+/−, and HER2+/− subtype which was found in 16.7% (9/54) of all the cases. HER2+-enriched subtypes consisted of 5.6% (3/54) of all the cases. Triple-negative breast cancer was present in 9.2% (5/54) of all the cases. The mean level of Ki67 expression was 35.2 ± 18.0% with range of 10% to 70%. More than one-third, 46.3% (25/54), of the cases had low category of Ki67 expression (⩽20%), and more than half, 53.7% (29/54), of the cases which had high Ki67 expression (>20%) (Table 2). The distribution of Ki67 positivity for the groups of tumor grades is shown in the box plot (Figure 3).
ER, PR, HER2 and Ki67 expression, and intrinsic subtypes of BC (N = 54).


Distribution pattern of Ki67 positivity according to the groups of tumor grades.
Association of Ki67 expression with patients’ clinical, pathological, and immunohistochemical characteristics
We found that negative PR expression was significantly associated with high Ki67 expression (F-statistic = 8.937, P = .004). Triple-negative breast cancer cases were associated with high Ki67 expression (F-statistic = 3.157, P = .033). Also, the group means for ER expression (F-statistic = 3.014, P = .088) and those of age (F-statistic = 2.885, P = .085) were of marginal statistical difference. However, there was no association of age (P = .734) (Table 3). After adjusting for age, tumor stage, ER status, PR status, and intrinsic subtypes of BC in multivariable linear regression analysis (Table 4), only intrinsic subtypes of BC (95% CI = 3.397-16.503, P = .032) and PR status (95% CI = 5.693-24.397, P = .042) remained the predictors of Ki67 expression. This shows that there was a statistical difference for the group means regarding PR status and intrinsic subtypes of BC on expression of Ki67.
The association between absolute Ki67 values and clinical, pathological, and immunohistochemical characteristics.

Multivariable linear regression analysis for assessing the predictors of Ki67 expression.

Discussion
The primary aim of this study was to assess the correlation of Ki67 proliferation index with conventional clinical and pathological factors in a cohort of male patients with BC in a resource-limited setting. The key findings in this study was high expression of Ki67 in the known conventional indicators of diseases aggressiveness such as TNBC and negative PR status.
Despite lack of universal cut-off point for Ki67 in determining prognosis of various malignancies, the biomarker has been found to be a potential prognostic marker. This biomarker which is detected using MIB-1 antibody shows proliferation activity of the cancer cells for a particular tumor, and its proliferation increases with aggressiveness of the tumor. Although studies determine the cut-off points while studying the association of Ki67 expression with other prognostic factors, still such studies have reported positive association of Ki67 expression with conventional clinical and pathological factors even various survival analyses, including overall survival, event-free survival, progression-free survival, among many others.19,31 -33
Regarding the association of Ki67 expression with HRs, in this study, the vast majority of cases were expressing ER similar to the findings in the previous studies.34 -36 It has been found that MBCs as well as BC among postmenopausal women tend show higher level of ER expression compared with the expression of ER among males without BC. 37 In our study, there was no association of ER expression with high Ki67 expression, although cases with negative ER status had higher mean Ki67 expression compared with cases that had positive ER. In a study of Kilickap et al, 38 it was found that there was an association between high Ki67 expression and negative ER status, which indicates poor prognosis for both FBC and MBC. Estrogen receptor (ER)–positive patients with BC usually tend to benefit from hormonal therapies (HTs) such as tamoxifen and aromatase inhibitors such as anastrozole and letrozole,39,40 unlike for ER-negative BC cases which do not benefit from using estrogen inhibitors, which contributes to disease progression and poor prognosis.41,42 However, MBCs that are PR negative, particularly those that are also ER and HER2 negative usually tend to be more aggressive and most of them tend to be TNBCs.43,44 Breast cancers with PR negative status normally have reduced survival far beyond compared with those with ER negative status, 14 and also such patients have high Ki67 expression. 45 In the present study, PR negative status was significantly associated with high Ki67 expression, indicating more aggressive and fastest-growing tumors. This is similar to the observation in the study of André et al. 46 Also, André et al 46 reported that PR negative among males with BC was associated with high Ki67 expression and BRCA2 gene mutation. Regarding the comparison of the association of PR negative status between MBC and FBC, studies have shown that MBCs with negative PR expression usually tend to have shorter survival and disease progression compared with FBCs which are also PR negative.16,47,48 Therefore, this shows that lack of PR expression in MBC provides more insights on determining clinical outcomes among males with BC as it is for females.
Triple-negative breast cancer has been found to show high Ki67 expression in both MBC and FBC. This is because TNBC is associated with high progression rate, metastasis, and resistance to chemotherapy drugs, which in one way or another is reflected by increased proliferation of the tumor cells. In this study, high expression of Ki67 was significantly associated with TNBC. This is similar to the finding in the study of Arafah et al 49 in which TNBC cases showed statistically significant expression of Ki67. In another study of Hashmi et al 50 reported that TNBCs showed highest Ki67 labeling index compared with other intrinsic subtypes. Abubakr et al 51 also reported a significant association between high Ki67 expression and TNBC in a study which included both males and females. All studies that have shown a positive association of Ki67 expression with TNBC intrinsic subtype of BC indicate that such subtype of BC also have high grade, indicating that Ki67 expression may be used as a surrogate biomarker in identification of high-risk subpopulation of patients with BC.
Studies have reported that the HER2-enriched (HER2-E) subtype of BC is associated with adverse clinical outcome even in its localized stages (stages I and II)21,52,53 and reduced progression-free survival with HTs. 54 HER2-E BCs usually show a high Ki67 proliferation index, indicating aggressiveness, rapid growth rate, and potentially poorer prognosis.55 -57 In this study, the mean value for the absolute Ki67 expression was significantly higher than other subtypes of BC except TNBC, which is similar to what was reported elsewhere.55,56 However, in one study, it was found that the expression of Ki67 among patients with HER2-E BC was influenced by clinical factors including postmenopausal status of the patients. In that study, it was observed that women who were in postmenopausal states had higher odds of having HER2-E than women who were in premenopausal status. 58 In addition, cases with BC that are BRCA1 positive are more likely to be TNBC unlike luminal types of BC. 59 Therefore, this highlights the need to take into account the clinical context, including hormone expression, menopausal status, and intrinsic subtypes when considering the expression of Ki67.
In this study, tumor stage was not associated with high Ki67 expression similar to the findings in in the studies of Erices-Leclercq et al 31 and Wang-Rodriguez et al, 33 in which advanced tumor stages were not associated with high Ki67 expression. In another systematic review and meta-analysis study, it was found that there was no association between Ki67 expression and tumor stage among males with BC. 60 However, André et al 46 reported positive association between high tumor stages of MBC and high expression of Ki67. Variation in sociodemographic indicators such as high family income and high level of education across various studies may contribute to the discrepancy in awareness and knowledge regarding ability to seek medical attention among individuals.61,62 This helps early detection of diseases such as BC especially among males who are more likely to delay from seeking medical intervention; as a result, this contributes to detection of the disease when it is already advanced.
Breast cancer subtypes with ER+, PR+, and HER2− tend to grow slowly as compared with ER+, PR+/−, and HER2+/−; such tumors have also been found to have lower Ki67 expression than ER+, PR+/−, and HER2+/− type of BC, making them to have poorer prognosis than subtypes with ER+, PR+, and HER2− BCs. 63 In our study, it was also found that ER+, PR+/−, and HER2+/− subtype of BC had significantly higher Ki67 expression than subtypes with ER+, PR+, and HER2− cases of BC. In the study of Cheang et al, 56 it was suggested that Ki67 expression could be used to differentiate between ER+, PR+, and HER2− type and B subtypes of BC, in which it was found that ER+, PR+, and HER2− subtype had <14% level of Ki67, whereas ER+, PR+/−, and HER2+/− had ⩾ 14% Ki67 expression. In the study of Inic et al, 64 it was also reported that high Ki67 expression was associated with ER+, PR+/−, HER2+/−, LNI, and advanced disease.
Strengths and limitations of the study
To our understanding, this is the first study in Uganda which analyzed intrinsic subtypes of BC among male patients. In addition, this is the only study from Uganda which has highlighted on the association of a known prognostic proliferation biomarker (Ki67) with conventional clinicopathological factors in MBC. The study also encountered some methodological limitations including lack of follow-up data which could have given insights regarding the correlation of Ki67 expression with survival of the analyzed patients. Also, due to rarity of the disease, the study included a small sample size which in one way or another affected strength of the drawn conclusions including lack of generalizability of the results and selection bias due to hospital-based nature of the analyzed data. Another study limitation was potential biases such missing information of tumor stage and tumor size for a significant number of cases and convenience sampling, which was not possible to avoid due to retrospective nature of the data and poor quality of data in LMICs which is a well-known challenge.
Conclusion
This study has shown that majority of MBC express ER, and ER+, PR+, HER2− (luminal A) subtype was the most common intrinsic subtype. High Ki67 was associated with PR negative status and TNBC intrinsic subtype. Our findings highlight the importance of Ki67 expression in determining aggressiveness of MBC based on the threshold value that was used.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234251392693 – Supplemental material for Ki67 Expression and Its Association With Clinicopathological Factors and Intrinsic Subtypes of Male Breast Cancer in Uganda: A Cross-sectional Study
Supplemental material, sj-docx-1-bcb-10.1177_11782234251392693 for Ki67 Expression and Its Association With Clinicopathological Factors and Intrinsic Subtypes of Male Breast Cancer in Uganda: A Cross-sectional Study by Tonny Okecha, James J Yahaya, Ali Waiswa, Veronica Nyakato, Anatoli Mawanda, Naghib Bogere, Linda A Lutada and Nixon Niyonzima in Breast Cancer: Basic and Clinical Research
Footnotes
ORCID iDs
Ethical considerations
Ethical approval was obtained from the research ethical committee (REC) of the UCI (reference: UCI-2023-85, date: 18.04.2024). We also confirm that waiver of individual consent for retrospective use of the FFPE analyses was obtained from the chairperson of the REC. This research was conducted according to the local and Helsinki declaration guidelines for research involving human subjects.
Consent to participate
Written informed consent was obtained from the patients and a copy has been kept for review of the Editor-In-Chief of the journal.
Consent for publication
Not applicable.
Author contributions
Tonny Okecha: Conceptualization, Data curation, Investigation, Methodology, Writing—original draft, Writing—review & editing.
James J Yahaya: Formal analysis, Writing—original draft, Writing—review & editing.
Ali Waiswa: Investigation, Methodology, Writing—original draft, Writing—review & editing
Veronica Nyakato: Formal analysis, Project administration, Writing—original draft, Writing—review & editing.
Anatoli Mawanda: Conceptualization, Data curation, Formal analysis, Writing—original draft, Writing—review & editing.
Naghib Bogere: Conceptualization, Data curation, Project administration, Software, Visualization.
Linda A Lutada: Methodology, Validation, Visualization, Writing—original draft, Writing—review & editing
Nixon Niyonzima: Project administration, Supervision, Validation, Visualization, Writing—original draft.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The dataset that support the findings of this study is available upon reasonable request from the corresponding author.
Declaration of generative AI and AI-Assisted Technologies in the writing process
The authors declare that the process of preparing this work did not involve any use of AI for designing of the study as well as improving grammar and style of the English language.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
