Abstract
BACKGROUND:
Low-density lipoprotein receptor-related protein 4 (LRP4) has been reported to be implicated in multiple types of cancers. However, the significance of LRP4 in gastric cancer (GC) remains poorly elucidated. Therefore, it’s urgent to investigate the importance and underlying mechanisms of LRP4 in GC.
OBJECTIVE:
To investigate the clinical roles of LRP4 in GC.
METHODS:
The LRP4 mRNA and miR-140-5p was measured by qRT-PCR. The protein expression was determined Western blot. Kaplan-Meier survival curves and Cox proportional hazard regression models were performed to evaluate prognosis.
RESULTS:
We demonstrated that LRP4 mRNA and protein was up-regulated in GC tissues for the first time. Its high expression was significantly correlated with malignant clinical features including TNM stage and lymph-node metastasis and poor prognosis for GC patients. LRP4 promotes migration, invasion and epithelial-mesenchymal transition (EMT) progress of GC cells. Mechanically, LRP4 regulated PI3K/AKT in GC cells. AKT inhibitors reversed the effects of LRP4. Finally, LRP4 was regulated by miR-140-5p in GC.
CONCLUSIONS:
Our findings showed that LRP4 has an important function in GC progression and promotes GC migration, invasion and EMT by regulating PI3K/AKT under regulation of miR-140-5p, providing a potential therapeutic target for GC.
Introduction
Gastric cancer (GC) is one of the most common malignant digestive tract tumors and the second most lethal cancer worldwide [1, 2]. Despite significant progress with the early intervention and treatment, the advanced GC patients still have poor prognosis mainly because of the frequent metastasis and recurrence after surgery [3, 4]. Metastasis is regulated by activation of multiple oncogenes and loss of tumor suppressors genes to activate corresponding signaling pathways [5, 6]. However, the underlying molecular mechanisms responsible for GC metastasis remains largely unelucidated. Therefore, it’s urgent to identify novel prognostic biomarkers for its diagnosis and develop anticancer targeted therapies.
Low-density lipoprotein receptor related protein 4 (LRP4), is a member of LDL family receptors and evolutionarily conserved transmembrane proteins, which contains a large extracellular region with EGF and LDLR repeats [7, 8, 9]. LRP4 promotes proliferation, migration and invasion in papillary thyroid cancer. Moreover, LRP4 knockdown decreased N-cadherin expression while enhances EZH2 and ZEB1 expression [10]. LRP4 induces extracellular matrix productions and facilitates chondrocyte differentiation by suppressing Wnt/
In this research, we confirmed that LRP4 expression was significantly up-regulated in GC tissues and cell lines. Its overexpression was correlated with malignant clinical features and poor survival. LRP4 promoted migration and invasion of GC cells using by gain- and loss-of-function experiments. Moreover, LRP4 regulated epithelial-to-mesenchymal transition (EMT) phenotype in downstream and the pathway between upstream was regulated by miR-140-5p in GC cells. In conclusion, we demonstrated that LRP4 plays critical oncogenic effects in GC progression and facilitates its potential biomarkers as a therapeutic target for GC treatment.
Materials and methods
Tissue specimens and cell lines
All the primary GC tissues and corresponding adjacent normal tissues (
A panel of GC cell lines (MKN45, MGC803, BGC823 and AGS) and normal gastric epithelial GES-1 cells were purchased from the Type Culture Collection of the Chinese Academy of Sciences (Shanghai, People’s Republic of China) and were cultured in RPMI1640 (Invitrogen, Carlsbad, USA) supplemented with 10% FBS (HyClone, Logan, UT, USA) and 1% penicillin/streptomycin (Thermo Fisher Scientific). All cell lines were propagated at 37
qRT-PCR
Total RNA from GC tissues or cells was extracted using the TRIzol reagent (Thermo Fisher Scientifc, CA, USA) and reverse transcribed to cDNA using the PrimeScript RT Reagent Kit (TaKaRa, Dalian, China) according to the manufacturer’s protocol. LRP4 mRNA was determined using the SYBR Green assay kit (Takara, Kusatsu, Japan) and miR-140-5p was quantified using TaqMan miRNA Assay Kit (TaKaRa, Dalian, China) on a 7500 RT-PCR system (Applied Biosystems, Waltham, MA, USA). The primers for LRP4 (HQP010873) and miR-140-5p (HmiRQP0182) were purchased from Genecopoeia (Guangzhou, China). GAPDH and U6 gene was used as internal control.
Western blot
Total protein was extracted by cell lysis in RIPA lysis (Solarbio, Beijing, China) containing 1% PMSF (P0100; Solarbio). Equal amounts of protein were separated by 10% SDS-PAGE and then transferred to PVDF (IPVH00010; Millipore, MA). After blocking, the membranes were incubated with a primary antibody LRP4 (ab85697, Abcam, San Francisco, USA), GAPDH (#5174, Cell Signaling Technology, Beverly, MA, USA) at 4
Transwell assay
Eight-
Immunohistochemical (IHC) staining
Briefly, the paraformaldehyde-fixed paraffin slides were deparaffinized, rehydrated and heat-treated for antigen retrieval. Then, blocked with hydrogen peroxide, serum and incubated with a primary antibody (E-cadherin #14472, N-cadherin #13116, Vimentin #5741, Cell Signaling Technology, Beverly, MA, USA) at 4
Luciferase reporter assay
The transcript of LRP4 3’-UTR (HPRM44248, NM_002334, GeneCopoeia) was constructed into pGL3 luciferase reporter vector (Promega, Madison, WI, USA). The potential binding sites were mutated by the Quick-change site-directed mutagenesis kit (Agilent Technologies, Santa Clara, CA, USA). The wild type (wt) LRP4 3’-UTR vector or mutant (mt) LRP4 3’-UTR vector and miR-140-5p mimics or miR-140-5p inhibitors were co-transfected into MGC803 cells by using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). The luciferase activity was measured using Dual-Luciferase Reporter Assay System (Promega, Madison, WI, USA) under luminometer (Berthold Detection System, Pforzheim, Germany), and luciferase activity was normalized to TK Renilla activity.
Statistical analyses
LRP4 expression and clinicopathological features were compared using the chi-squared test. The Kaplan-Meier survival curve was assessed by the log-rank test. Kaplan Meier and Cox regression analyses were utilized to assess the association between candidate genes and overall survival as well as the prognosis of GC. The PH assumption was tested graphically using a plot of the log cumulative hazard, where the logarithm of time is plotted against the estimated log cumulative hazard calculated as ln [-ln (S(t))]. If the curves for the two treatment groups were approximately parallel, the PH assumption was deemed reasonable. Other data are presented as mean
Correlation between LRP4 expression and clinicopathological features in GC patients (
100)
Correlation between LRP4 expression and clinicopathological features in GC patients (
LRP4 is overexpressed and correlates poor prognosis of GC patients
To explore the expression level of LRP4 in GC, qRT-PCR was used and revealed that LRP4 mRNA expression was significantly higher in tumor compared to corresponding adjacent normal tissues (Fig. 1A,
Multivariate analysis of 5-year overall survival of GC patients
Multivariate analysis of 5-year overall survival of GC patients
TNM, tumor-node-metastasis; HR, hazard ratio; CI, confidence interval;
Relative LRP4 expression and its clinical significance in human GC. (A) qRT-PCR detection of LRP4 mRNA expression levels in gastric cancer tissues and matched adjacent normal tissues. (B) Western blot analysis of LRP4 expression in the GC (T) and normal tissues (NT). (C) Kaplan-Meier overall survival cures of GC patients with high or low LRP4 expression. 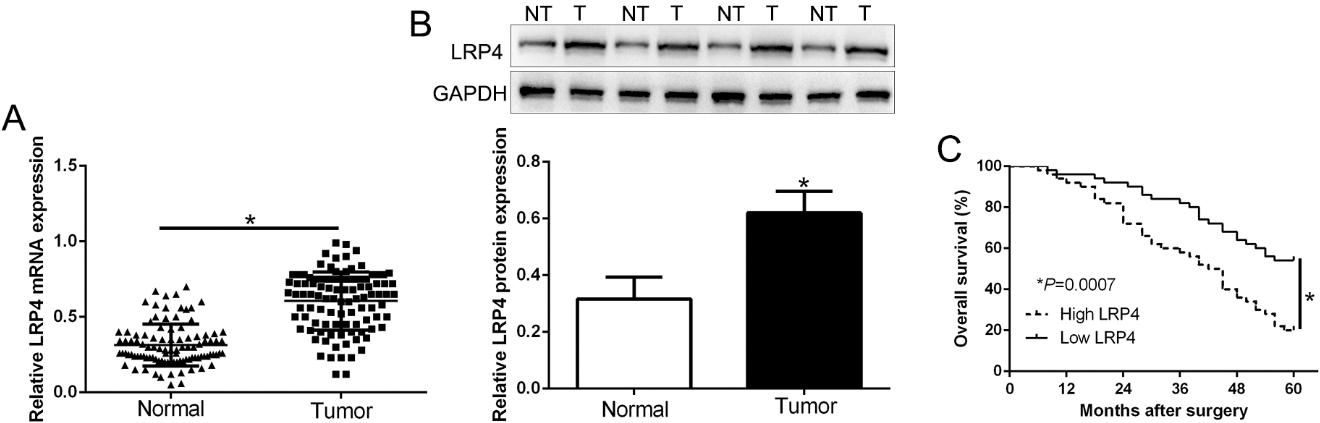
To detect the biological effects of LRP4 in GC, we measured the expression of LRP4 in GC cell lines. Compared to the normal gastric epithelial GES-1 cells, LRP4 expression was significantly elevated in GC cells (Fig. 2A,
LRP4 promotes GC cell migration and invasion. (A) Western blot analysis of LRP4 protein expression in GC cell lines and normal gastric epithelial GES-1 cells. (B) Western blot analysis of LRP4 expression in LRP4 knockdown or overexpression cells. (C) Effects of LRP4 knockdown or overexpression on the migratory capability of GC cells by transwell assay. (D) Effects of LRP4 knockdown or overexpression on the invasive capabilities of GC cells by transwell assay. 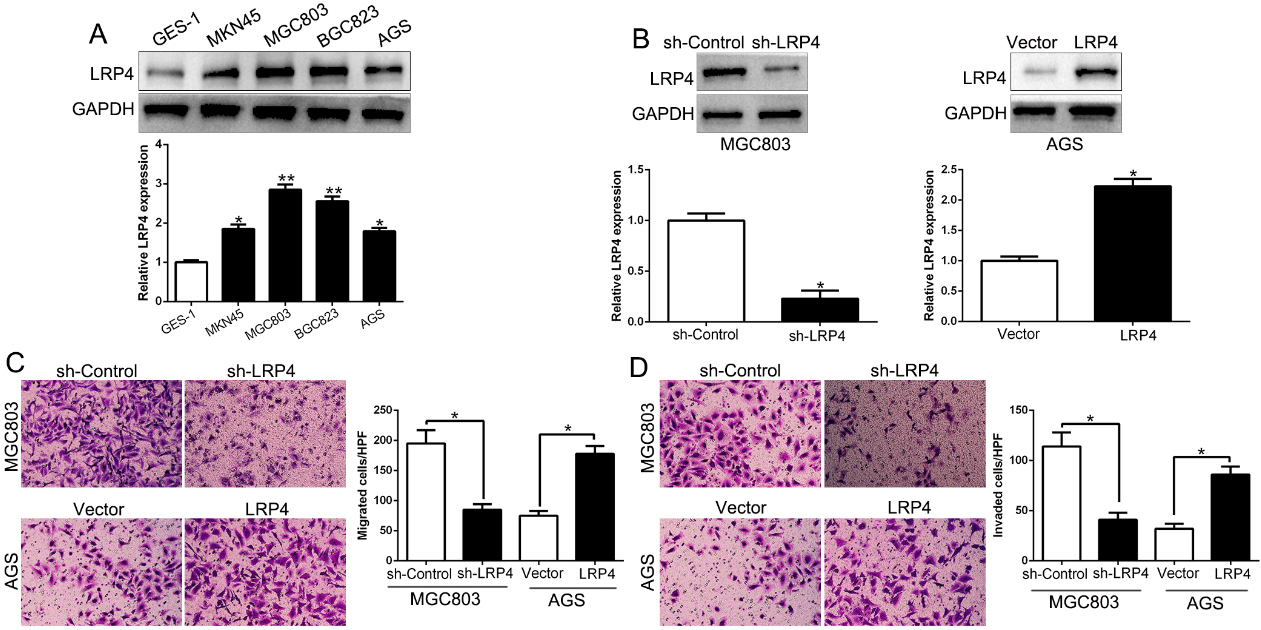
Since EMT activation facilitate tumor metastasis in cancers, including GC [17, 18]. We confirmed that LRP4 knockdown promoted epithelial marker E-cadherin and inhibited mesenchymal marker N-cadherin and Vimentin (Fig. 3A,
Detection of LRP4 and EMT-related marker expression in LRP4-alteration GC cells. (A) LRP4 knockdown increased E-cadherin expression and decreased N-cadherin and Vimentin expression in MGC803 cells, while (B) the overexpression of LRP4 in AGS cells inhibited the expression of the epithelial cell marker E-cadherin and promoted the expression of the mesenchymal cell marker N-cadherin and Vimentin. (C) Immunohistochemical analysis of E-cadherin, N-cadherin and Vimentin in GC tissues. In cases of high LRP4 expression; there was strong N-cadherin, Vimentin and no detectable E-cadherin protein expression in the same tissue section. In contrast, in the case of low LRP4 expression, there was no detectable N-cadherin, Vimentin and strong E-cadherin protein expression; 
LRP4 regulates GC cell migration and invasion via activating the PI3K/AKT signaling pathway. (A) Western blot analysis of the PI3K/AKT signaling pathway proteins in cells after LRP4 overexpression or knockdown in GC cells. (B and C) AGS-LRP4 cells were treated with or without AKT inhibitor MK2206, and cell migration and invasion abilities determined by Transwell assays, respectively. (D) Western blot analysis of the EMT-associated proteins afer treatment with AKT inhibitor. 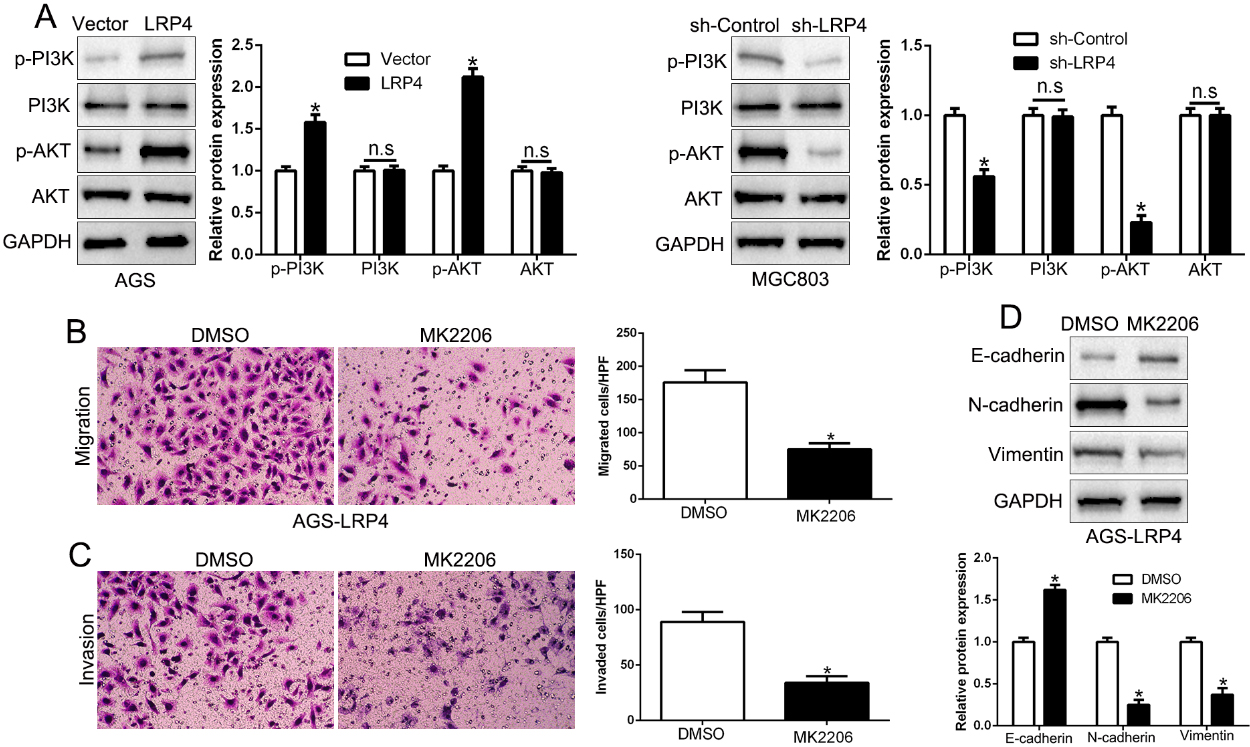
LRP4 is identified as a direct target of miR-140-5p in GC. (A) miR-140-5p and its putative binding sequence in the 3’-UTR of LRP4. The mutant binding site was generated in the complementary site for the seed region of miR-140-5p. (B) miR-140-5p significantly suppresses the luciferase activity that carried wild-type (wt) but not mutant (mt) 3’-UTR of LRP4. Anti-miR-140-5p led to a notable increase in the luciferase activity of wt 3’-UTR of LRP4. (C) qRT-PCR analysis of LRP4 mRNA expression in MGC803 cells with miR-140-5p or miR-control vector transfection and AGS cells with anti-miR-140-5p or anti-miR-NC vector transfection. (D) Overexpression of miR-140-5p reduced the expression of LRP4 protein in MGC803 cells and knockdown of miR-140-5p increases the level of LRP4 protein in AGS cells. GAPDH as the control. 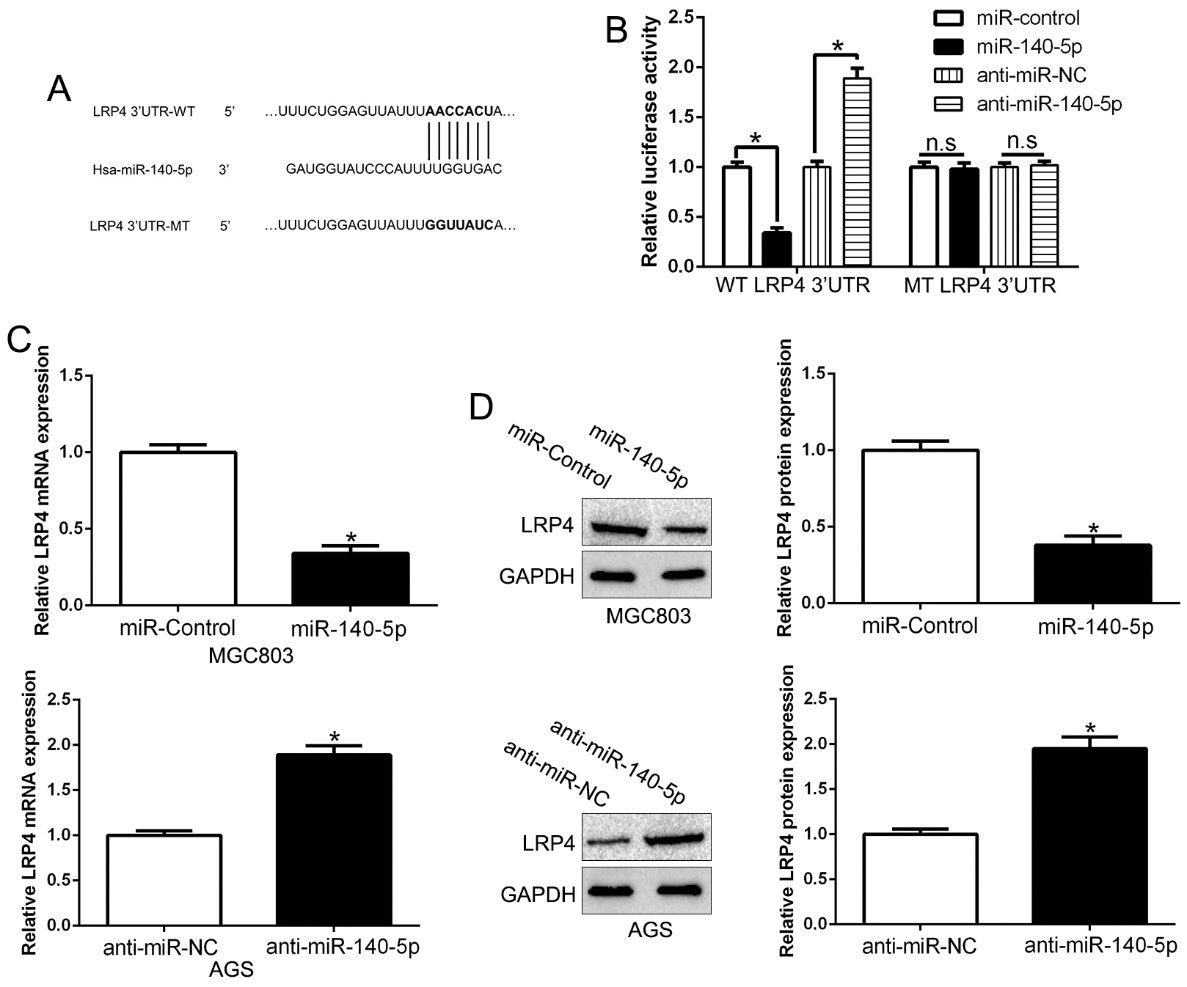
LRP4 inversely correlated with miR-140-5p expression in GC tissues. (A) The expression level of miR-140-5p in GC tissues and adjacent non-tumor tissues. (B) The expression of miR-140-5p in LRP4 high-expressing tumors was significantly lower than that in LRP4 low-expressing tumors. (C) A significant inverse correlation between the mRNA levels of LRP4 and miR-140-5p was observed in GC tissues. 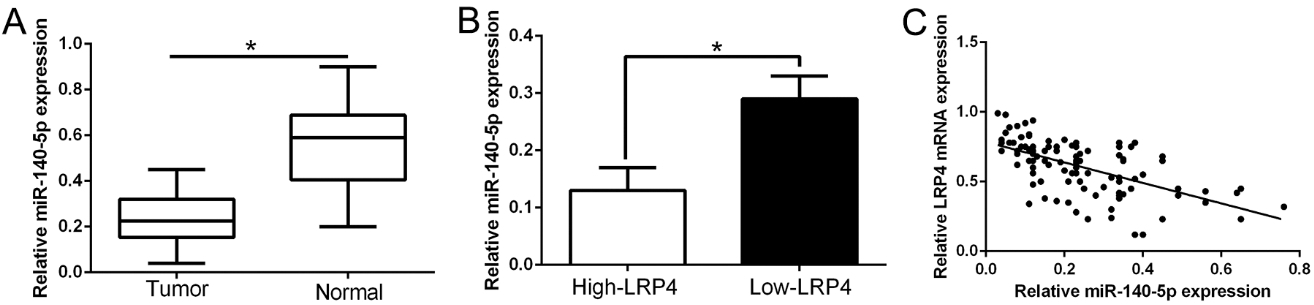
Previous studies reported that PI3K/AKT signaling pathway plays critical role in cancers, including GC [19]. Here, we demonstrated that LRP4 overexpression promoted phosphorylated PI3K and AKT expression while LRP4 knockdown reduced the phosphorylation of PI3K and AKT (Fig. 4A,
LRP4 expression was regulated by miR-140-5p
Previous studies reported that miRNAs were involved in GC and exercise functions by binding to the 3’UTR of target mRNAs [20]. We predicted that LRP4 was a putative target of miR-140-5p (Fig. 5A) by using the platform Targetscan and miRNAda. Previous studies confirmed that miR-140-5p functions as a tumor suppressor in GC [21]. Dual-luciferase reporter assays confirmed that LRP4 was a direct target of miR-140-5p (Fig. 5B).To further validate the association between miR-140-5p and LRP4, we performed qRT-PCR and Western blot assays to show that miR-140-5p overexpression inhibited LRP4 mRNA and protein (Fig. 5C and D,
miR-140-5p inversely correlated with LRP4 in GC tissues
To further validate the association of miR-140-5p and LRP4 in GC tissues, we firstly confirmed that miR-140-5p was down-regulated in GC tissues compared to normal tissues (Fig. 6A,
Discussion
The clinical outcomes of gastric cancer remain poor due to frequent tumor metastasis and tumor recurrence despite considerable improvement in cancer diagnosis and treatment [22]. Therefore, it’s urgently needed to identify cancer-specific biomarkers and understand the detailed underlying mechanisms to develop novel therapeutic strategies [5]. Recent reports revealed that LRP4 was involved in human malignancies and related to metastasis of tumors. However, the clinical significance and role of LRP4 in GC remains unknown.
In this research, we confirmed that LRP4 served as an oncogene to promote metastasis of GC patients. Firstly, we showed that LRP4 mRNA and protein was up-regulated in GC tissues compared to normal tissues. Moreover, the patients who has high LRP4 expression confers to poor survival and malignant clinical features, including TNM stage and lymph-node metastasis. These results showed that LRP4 was a critical player in cancer progression. LRP4 promotes tumor cell migration, invasion and EMT phenotype. Moreover, LRP4 was associated with EMT markers in GC tissues. EMT exerted key function in the cancer metastatic progression, which gives migratory and invasive properties to tumor cells [23]. EMT means the progress that the epithelial cells lose adhesion and owns the mesenchymal phenotype which promoted motility of cells and get an invasive phenotype [24]. The process contributes to the cancer cells spread to distant metastasis [25]. These results showed that LRP4 promoted migration and invasion of GC cells by regulating EMT. Moreover, the PI3K/AKT signaling pathway was activated by LRP4 overexpression. AKT inhibitors reversed the biological effects of LRP4. These data suggest that PI3K/AKT mediated the effects of LRP4.
Numerous studies reportedthat miRNAs have important significance and regulate multiple processed of vital activities [26]. Accumulating evidences demonstrated that miRNAs play a critical role in cancer development and progression. Previous studies confirmed that miR-140-5p was a tumor suppressor in GC [21, 27]. Here, we demonstrated that miR-140-5p binds 3’UTR of LRP4 and regulate its expression. In GC tissues, miR-140-5p was down-regulated in GC, which is consist with previous studies. Moreover, LRP4 expression showed an inverse relationship with miR-140-5p.
In conclusion, our results demonstrated that LRP4 was significantly up-regulated in GC tissues and correlated with poor pathological features and an unfavorable prognosis in GC patients. We also confirmed that LRP4 promoted GC cell migration and invasion via activation of EMT progress by regulating PT3K/AKT pathway. Moreover, we determined that LRP4 was regulated by miR-140-5p. These findings provided novel insight progression for GC and have potential value as a diagnostic and prognostic markerfor GC.
Footnotes
Acknowledgments
This study was supported by scientific and technological project of Xi’an (2017113SF/YX007(11)) and Shaanxi Provincal project of scientific and technological innovation team (2017KCT-28).
Conflict of interest
The authors declare no conflict of interest.
