Abstract
BACKGROUND:
Downregulation of PCDH20 is frequently involved in tumorigenesis of many cancers, but the role of PCDH20 protein in hypopharyngeal squamous cell carcinoma (HSCC) is still unknown.
OBJECTIVE:
The aim of this study was to investigate the role of PCDH20 in hypopharyngeal squamous cell carcinoma (HSCC).
METHODS:
Immunohistochemistry (IHC) and qRT-PCR was carried out to estimate the expressions of PCDH20 protein and mRNA in HSCC tissues and adjacent non-tumor tissues. Correlation between the PCDH20 expression and clinicopathological characteristics was evaluated using chi-square test. Meanwhile, Kaplan-Meier method and log-rank test were applied to analyze the overall survival. After transfection of PCDH20, the CCK8 assay, Cell migration assay and invasion assay were used to investigate the changes in the viability, migration and invasion of Fuda cells. The mechanisms by which reduced PCDH20 promote migration and invasion of Fuda cells were examined using western blotting.
RESULTS:
PCDH20 protein showed in tumor tissue low expression rates of 67.5% (54/80). The mRNA of PCDH20 indicated the consistent trend (80%, 8/10). Reduced PCDH20 expression was positively related to T stage and lymph node metastasis (
CONCLUSION:
Our data highlight that the downregulated PCDH20 may serve as reliable diagnostic biomarker in HSCC.
Keywords
Introduction
Hypopharyngeal squamous cell carcinoma (HSCC), considered as one of the common malignant tumors of head and neck cancer, accounts for about 5% [19]. Owning to the anatomical structure and submucosal infiltration, the early clinical symptoms of the patients are not greatly apparent. Conseqently, when diagnosed, most of the patients are in the advanced stage or metastasis which is the main cause of treatment failure. Recent studies showed that the 5-year survival rate of patients is only 15–30% [12, 13, 16]. Hence, it’s quite urgent to explore the pathogenesis and expound special diagnostic biomarkers for HSCC.
Cadherins, acted as a key role in the cell-cell communication, which contains at least 80 members. Among these, more than 60 protoc adherins have been identified and are currently a major subfamily of the cadherin superfamily [15, 23]. The protocadherins (PCDHs), divided into two subtypes: “clustered” and “non-clustered” groups, is a kind of calcium-dependent molecule [5, 20]. Several members of the PCDHs family, such as PCDH8 in nasopharyngeal carcinoma [4], PCDH10 in gastric cancer and cervical cancer [27, 14], and PCDH17 in esophageal squamous cell carcinoma,gastric and colorectal cancers [3, 7], have recently been reported to be involved in the disruption of carcinogenesis. Regrettably, the relationship between PCDHs and the pathogenesis of the other cancers remains unclear. PCDH20, belonging to PCDHs family, contains 6 extracellular cadherin regions, a transmembrane region and a spore tail region [18]. Recent studies have shown that PCDH20, acted as tumor suppressor, frequently inhibited proliferation, repressed migration and invasion, blocked cell cycle, and promoted apoptosis in variety of cancers, such as non-small-cell lung cancers [8], nasopharyngeal carcinoma [21], and hepatocellular carcinoma [9]. Nevertheless, the relationship between PCDH20 expression and HSCC has never been reported in the previous studies.
In the present study, we clarified the expression of PCDH20 in HSCC tissues and non-tumor tissues. In addition, we estimated the correlation between the expression of PCDH20 and prognosis of HSCC. According to the above data, we further evaluated the biological function and molecular mechanism of PCDH20 in Fadu cells.
Materials and methods
Patients and specimens
Here in this study, a total of 80 patients with HSCC and corresponding non-tumor tissues were collected from the department of Head and Neck Surgery, First Affiliated Hospital of Chongqing Medical University, China, between 2007 and 2012. Absolutely, the specimens were diagnosed by biopsy at pathology department of Chongqing Medical University according to the admitted standard. All these samples were saved with snap-frozen in liquid nitrogen, then transmitted into
RNA extracting and quantitative real-time polymerase chain reaction (qRT-PCR)
Total RNA of 10 coupled HSCC and matched non-tumor samples were extracted with Trizol reagent (Takala, Dalian, China) based on the manufacturer’s instruction. RNA intensity and purity ranged from 1.8 to 2.0 inspected by spectrophotometry. Briefly, the prepared RNA were reversely transcribed into cDNA using PrimeScript RT Reagent Kit (Takala, Dalian, China) according to the manufacturer’s specification. Then 0.5
PCDH20-forward 5’-TCTACATCGTCCCAGGA GCA-3’ PCDH20-reverse 5’-GCGGGTAGTCCCTCGTTT AG-3’ GAPDH-forward 5’-GAAGGTGAAGGTCGGAG TC-3’ GAPDH-reverse 5’-GAAGATGGTGATGGGATT TC-3’
The qRT-PCR was carried out for 37
The relative expression levels of mRNA were calculated by 2
Immunohistochemistry (IHC)
The expression of PCDH20 protein of 80 pairs HSCC patients and matched non-tumor tissues were tested by immunohistochemistry (IHC). Shortly, on the first day, histologic sections were dried at 60
The expression level of PCDH20 in tumor tissues and non-tumor tissues was measured by qRT-PCR and IHC assay, respectively. (A). Relative mRNA expression of PCDH20 in HSCC tissues displayed significantly lower than that in non-tumor tissues by qRT-PCR. Paired T-test was for the statistical analysis (
HSCC cell line Fuda was obtained from Cell Bank of Chinese Academy of Science (Shanghai, China). The cells were cultured at 37
Cell counting kit-8 (CCK-8) assay
Fadu cells were inoculated into 96-well plates at a density of 1
Cell migration and invasion assay
After 48 h transfection, Fadu cells suspensions were prepared with serum-free medium. And the number of cells was counted by 1
Western blotting analysis
Lipofectamine 2000 was used to transfect the PCDH20 expression plasmid and empty vector into Fuda cells. Cell proteins were collected with a mixture of ice-cold lysis buffer (Applygen, Beijing, China) (RIPA) and 1% Phenylmethanesulfonyl fluoride (PMSF) (Beytime, Biotechnology, Shanghai, China) and examined with the standard BCA method (BCA
Statistical analysis
The results were expressed as mean
Results
Downregulated PCDH20 expression in HSCC
The expression level of PCDH20 in 10 paired HSCC samples and corresponding non-tumor tissues was detected by qRT-PCR, normalized with a control (GAPDH). The results turned out that PCDH20 expression was significantly low in 80% (8/10) of HSCC tissues compared that in the non-tumor tissues (
Relationship between PCDH20 expression and clinicopathological characteristics
A total of 80 pairs of HSCC and corresponding adjacent non-tumor tissues were examined by IHC with special antibody. As shown in Table 1, of all 80 HSCC cases, 67.5% (54/80) showed reduced PCDH20 expression. Moreover, PCDH20 was frequently located in the membrane and cytoplasm of adjacent normal tissues (Fig. 1). The result showed that the negative rate of PCDH20 expression in HSCC was significantly higher than that in the non-tumor tissues (chi-text,
Relationship between clinicopathological features and expression of PCDH20 protein
Relationship between clinicopathological features and expression of PCDH20 protein
Since the expression of PCDH20 was abnormal in HSCC, we explored the role of PCDH20 during tumor progression. Thus, the relationship between PCDH20 expression and clinicopathological characteristics was estimated in our study. The results indicated that no significant differences were found between expression of PCDH20 with sex, age, smoking (
To determine the prognostic value of PCDH20 expression in HSCC, Kaplan-Meier curves for overall survival are described in Fig. 2. The curves depicted that patients with low levels of PCDH20 had worse overall survival compared with those with high PCDH20 levels (log-rank
Univariate and multivariate Cox regression analysis of overall survival in 80 patients with HSCC
Univariate and multivariate Cox regression analysis of overall survival in 80 patients with HSCC
RR risk ratio, CI confidence interval.
The Kaplan-Meier survival curves constructed for HSCC patients with PCDH20 expression. Patients with low levels of PCDH20 had worse overall survival compared with those with high PCDH20 levels (log-rank 
In the beginning, the expression of PCDH20 in Fadu cells was evaluated after transfected PCDH20 gene. As shown in Fig. 3A, the result demonstrated that the expression of PCDH20 was significantly higher in Fadu cells than that in vector cells (
PCDH20 regulated cell viability, migration and invasion in Fadu cells. (A). The level of PCDH20 was significantly overexpressed after transfected PCDH20 gene in Fadu cells by qRT-PCR. (B). CCK-8 assays showed that PCDH20 inhibited cell viability in Fadu cells. (C). Migration assays showed that PCDH20 repressed the migration capacity of Fadu cells. (D). Invasion assays demonstrated that PCDH20 blocked the invasion of PCDH20 cells.* means 
Recent studies reported that several members of PCDHs,such as PCDH10, PCDH17, could antagonize the Wnt/
PCDH20 regulated Wnt/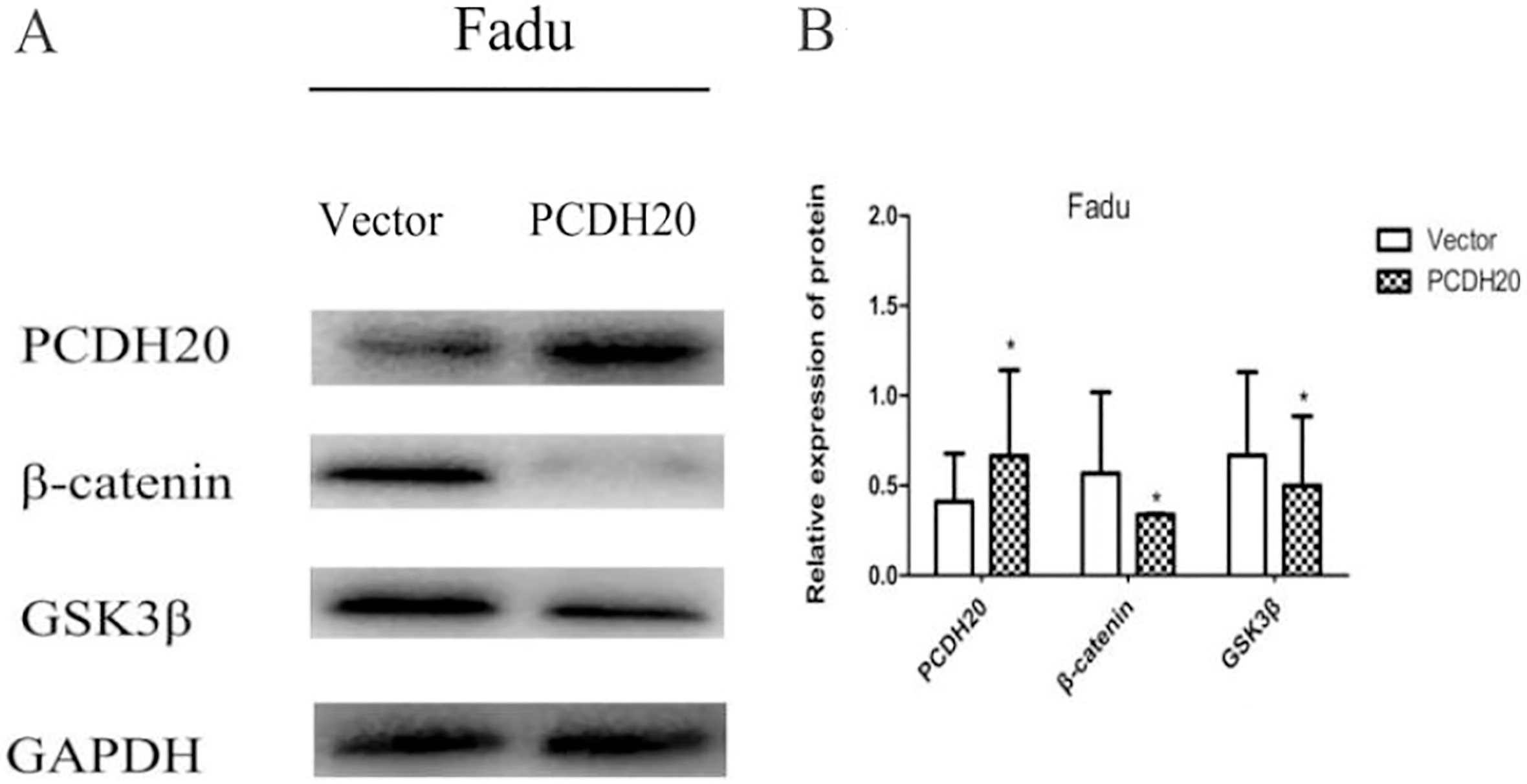
As is known to all, hypopharyngeal squamous cell carcinoma (HSCC) is one of the common head and neck malignant cancers with increasing rates of morbidity and mortality [10, 22]. Despite of advanced treatment to reverse this condition, the 5-year survival rate for patients with HSCC remains no ideal, due to recurrence and metastases [6]. Hence, it is highly urgent to identify novel biomarkers to illuminate this allround disease process and to exploit the mechanism.
The protocadherin (PCDH) family, a kind of tumor suppressor gene family, is relatively well investigated in various human cancers. Several members of PCDHs, have been discovered in varieties of cancers, such as PCDH8 [4], PCDH10 [27, 14], PCDH17 [3, 7], inhibiting tumor cell growth, invasion, and metastasis. PCDH20, also known as PCDH13, which belongs to the subgroup of non-clustered protocadherin gene family, plays a crucial role not only in growth control but also in cell-cell communication. As a novel protocadher, PCDH20 is expressed predominantly in nervous system [2, 11]. Till now, there are limited studies with respect to the expression of PCDH20 in HSCC and its relationships with clinical characteristics and clinical outcome.
In the present study, Our results definitely showed that mRNA level of PCDH20 was frequently reduced in tumor tissues 80% (8/10) compared to paired non-tumor tissues by qRT-PCR. Meanwhile, the immunohistochemical results displayed that PCDH20 protein was notably downregulated in HSCC tissues 67.5% (54/80), which is concordant with previous reports. Imoto et al. [8] discovered PCDH20 was silenced in 54.2% non-small cell lung cancer (NSCLS) tissues. A decreased expression of PCDH20 in nasopharyngeal carcinoma but not in normal nasopharygeal tissues was observed [21]. All these results suggest that reduced PCDH20 expression is significantly associated with tumor progression. Therefore, we hypothesize that PCDH20 might participate in the development of HSCC.
A study on hepatocellular carcinoma (HCC) suggested that the reduced PCDH20 expression was associated with poor tumor differentiation [25]. In our study, tumors with negtive PCDH20 protein expression were more frequently observed in advanced T stage, lymph node metastasisin in HSCC. These results again demonstrated that downregulated PCDH20 might be involved in promoting the poor progression of HSCC. Meanwhile, it was showed in our study the PCDH20 expression was unrelated to age, which was not accordance with previous study [9], we suggested that the role of PCDH20 on age in HSCC need to be confirmed in more cases and further study was needed.
In HCC [25], NSCLS [8], decreased expression of PCDH20 was related to poor prognosis of these patients. Furthermore, PCDH20 could be used to be an independent prognostic factor for HCC [25]. Our group found that there was a positive correlation between PCDH20 expression and prognosis of HSCC. Cox regression analysis suggested PCDH20 could be an independent prognostic factor for patients with HSCC. Meanwhile, the Kaplan-Meier curves indicated that patients with low PCDH20 expression was significantly short overall survival compared to those with high PCDH20 expression. Therefore, we suppose analysis of PCDH20 expression might provide a useful additional prognostic tool in case of HSCC.
Further, we found that restoration of PCDH20 expression induced inhibition of Fadu cells proliferation, migration and invasion. The same observations that ectopic expression of PCDH20 inhibited cell migration and invasion are also identified in NPC cells [21]. Meanwhile, in other protocadherins, various tumor suppression mechanisms have been reported: PCDH10 inhibits cell proliferation via downregulation of Wnt/
Wnt signalling pathway plays an important role in the embryonic development of animals, and abnormal alterations of Wnt signalling pathway are the most common events associated with human carcinogenesis [17]. Recent studies reported that certain members of PCDHs can regulate the expression of some downstream targets of Wnt signalling pathway [24, 26, 28]. In this study, we are the first to demonstrate that PCDH20 suppresses the Wnt/
In summary, we firstly validated that reduced PCDH20 expression can effectively promote the tumorigenesis of HSCC, and verified the relationship between clinicopathological features and PCDH20 expression. What’s more, we also futher demonstrated that PCDH20 can inhibit cell viability, migration and invasion through blocking Wnt/
Footnotes
Acknowledgments
This work was financed from the funds of the National Natural Science Foundation of China (81470676 and 81271061).
Conflict of interest
None.
