Abstract
The objective of this study was to investigate the between autophagy activity and local immune response in hypopharyngeal squamous cell carcinoma (HSCC). Herein, we observed the expression of autophagy marker microtubule-associated protein light chain 3B (MAP1LC3B), CD8 cytotoxic T lymphocytes (CTLs), CD39 (regulatory T cells Tregs) and CD163 (tumor-associated macrophages TAMs) in HSCC, and determined the prognostic roles of CD8+/CD39+ and CD8+/CD163+ in patients with HSCC. The expression of light chain 3B (LC3B) and CD8+/CD39+ was found to be significantly lower in HSCC tissues than in adjacent non-tumor mucosa tissue samples; LC3B expression was positively correlated with the infiltration rate of CD8+/CD39+ in HSCC. Further studies revealed that the ratio of CD8+/CD39+ immune cells was negatively correlated with tumor lymph node metastasis and TNM classification, while the ratio of CD8+/CD163+ immune cells was negatively correlated with TNM classification. Moreover, the expression of LC3B was analyzed and the patients were grouped according to their immune infiltration characteristics. The 5-year cumulative survival rates of LC3B+, CD8+/CD39+, and CD8+/CD163+ patients were significantly higher than those of other group patients. Collectively, our studies indicated that the expression of LC3B in HSCC was correlated with the infiltration ratio of immune cells, and a change in autophagy activity may affect the cellular immunity in HSCC. The ratios of tCD8+/CD39+ and tCD8+/CD163+ may serve as prognostic factors for HSCC.
Introduction
Hypopharyngeal malignancies, primarily categorized as squamous cell carcinomas, account for approximately 5% of head and neck malignancies. 1,2 According to GLOBOCAN’s statistics in 2018 estimates, there were about 80,608 new confirmed cases of hypopharyngeal cancer and 34,984 deaths reported globally, accounting for approximately 0.4% of the total sites cancers. 3 Although the low incidence of hypopharyngeal squamous cell carcinoma (HSCC) is low, most patients present at advanced stages because early symptoms are non-specific. Approximately 70-85% of HSCCs are diagnosed at stage III or IV, with a 5-year overall survival rate of 15–45% for those patients. 4 Therefore, it is imperative to explore the molecular mechanisms regulating HSCC and discover new diagnostic and prognostic biomarkers.
Autophagy is a highly conserved and ubiquitous catabolic process characterized by the degradation of intracellular components in lysosomes, an evolutionarily conserved process of self-depletion that involves maintaining organelle function to remove cellular waste and provide metabolic substrates. 5 In normal cells, autophagy protects against excessive reactive oxygen species, DNA damage, and genomic instability. 6 In some cases, autophagy may play a role in inhibiting cancer in the early stage of tumorigenesis, 7 however, in the latter stages, autophagy maintains tumor metabolism and promotes tumor cell survival. 8,9 Microtubule-associated protein light chain 3(MAP1LC3), referred to as light chain 3(LC3), is currently recognized as a marker of autophagy. LC3 in mammals can be divided into 3 types: LC3A, LC3B and LC3C. LC3B, as a type of LC3, can also be used as a molecular marker of autophagy. 10
Immunogenicity plays an important role in the control of tumorigenesis. The interaction between the host’s immune response and tumor cells often results in immune cell infiltration. 11 Studies have shown that tumor infiltrating lymphocytes (TILs) are important prognostic factors for a variety of tumors, such as esophageal cancer, renal cancer, breast cancer, colon cancer, and so on. 12 -15 Therefore, immune factors may play a crucial role in assessing the prognosis of HSCC patients. The factors affecting the immune function of tumors are relatively complex. Recent studies have found that changes in autophagy activity can affect the function of immune cells. 16
Regulatory T cells (Tregs), an important subset of TILs, mediate self-antigen immune responses and inhibit abnormal immune responses. Tumor-infiltrating Tregs play an important role by suppressing antitumor immune responses, and promoting tumor immune escape. 17 Regulatory T cells express a variety of surface antigens, including CD25, CD103, and CTLA-4, which are commonly used Treg immunology markers. 18 Ectonucleoside triphosphate diphosphohydrolase-1 (ENTPD1 CD39) is an adenosine generated enzyme, expressed at the cell surface of Tregs. It is a novel surface marker of Tregs, 19 and plays a role in nucleotide hydrolysis promoting the secretion of adenosine, which affects both A2A receptors on the surface of T cells, inhibiting T-cell proliferation, migration, and antitumor effect. 20,21
Macrophages are important components of the body’s natural immune response and can be divided into different subtypes in different tissues. In the tumor microenvironment, macrophages are primarily divided into M1 classically activated macrophages and M2 substitutively activated macrophages. 22 The markers CD16/32, CD11b, and CD11c are highly expressed in M1-type macrophages, whereas markers CD163 and CD206 are highly expressed in M2-type macrophages. 23 Multiple studies have shown that M2 macrophage invasion of the tumor microenvironment could lead to tumor progression and is associated with angiogenesis, metastasis, and immunosuppression. This macrophage phenotype is called tumor-associated macrophage (TAM). 24,25
CD8+ cytotoxic effector cells (CTLS) and Tregs, as well as the balance between CTLS and TAM have been shown to be important in tumor progression and prognosis. Tumor invasive CD8+T lymphocytes play an important role in the anti-tumor immune response and have prognostic value for various tumor types. 26 At present, no such studies have been reported in HSCC. Thus, to discover a better prognostic indicator of HSCC, the relationship between autophagy and immune infiltration was investigated
This study observed the autophagy marker LC3B 27 and the expression of cytotoxic T lymphocytes (CTL, expression of CD8 28 ), immunosuppressive factors of regulatory T cells (Treg expression of CD39 19 ) and tumor-associated macrophage (TAM, expression of CD163 29 ) infiltration in HSCC and adjacent non-tumor mucosa tissue.
Materials and Methods
Patients and Tissue Specimens
Human tissue specimens were collected from the Department of Otolaryngology of The First Affiliated Hospital of Anhui Medical University from 2011 to 2014. A total of 70 surgical specimens of HSCC and 22 adjacent non-tumor mucosa tissue samples were collected. Sample use was approved by the Biomedical and written informed consent was provided by all patients. All selected cases were first identified and did not receive neoadjuvant chemotherapy or radiotherapy. Diagnosis and histology classification were performed according to the World Health Organization’s classification system, by 2 experienced pathologists. TNM staging was performed according to the American Joint Committee on Cancer (Eighth Edition AJCC). Formalin-fixed paraffin specimens with complete clinicopathological and follow-up data were selected.
Reagents
Rabbit anti-human LC3B (SAB5600101) was purchased from Sigma-Aldrich (St Louis, MO, USA). Mouse anti-human CD39 (TAB804559) and 3,3-diaminobenzidine (DAB) were purchased from Beijing Chinese Zhongshan Golden Bridge. Ready-to-use antibody CD8 (RMA-0514), CD163 (MAB-0206), sheep rabbit/rat secondary antibody (ab5878) were purchased from Fuzhou Maxim Biotech (China).
Immunohistochemistry
For immunohistochemistry, 4-µm-thick sections of 70 HSCC tissues and 22 adjacent normal mucosal tissues were deparaffinized and rehydrated. Antigen retrieval was carried out by microwave treatment of slides in 50 mM Tris-EDTA (pH 9.0) for 20 min. Endogenous peroxidase was quenched with 3% hydrogen peroxide for 10 min at 37°C. Slides were incubated with rabbit polyclonal anti-LC3B antibody (1:75), mouse anti-human CD39 (1:200), and ready-to-use antibody CD8, CD163 overnight at 4°C. Thereafter, sections were incubated with sheep rabbit/rat secondary antibody at 37°C for 15 min. The chromogenic reaction was carried out using DAB. Slides were counterstained with hematoxylin. For negative controls, the primary antibody was PBS.
Quantification Methods
Diagnoses were performed by viewing of radiographs by 2 senior double-blinded pathologists. All samples were anonymous and the pathologists had no knowledge of the clinicopathological data before diagnosis by double-blind radiographs by 2 senior pathologists. LC3B was expressed in the cytoplasm of tumor cells, showing a brown-yellow color as a positive marker. In at least 5 high-power microscope fields (200×), the experimental results were determined according to cell staining intensity. The LC3B proportion of positive cells were divided into 4 categories: 0 (5%), 1 (6%–25%), 2 (26%–50%), and 3 (>50%). A score of 0 to 1 was defined as negative expression, and a score >2 was defined as positive. 30 CD8 and CD39 were expressed in the cell membrane of lymphocytes with corresponding immunophenotypes, and CD163 was expressed in the cell membrane of antigen-presenting cells with corresponding immunophenotypes, all of which were brown. Positive staining of CD39 in 5 fields was detected under a microscope (200×) by the optical density method. 31 At low magnification, 10 independent fields were observed. The 5 regions with the highest numbers of CD8- and CD163-positive cells were selected, counted at high magnification, and averaged. 31 We selected the median intratumoral CD8/CD39, CD8/CD163 cell count as the cutoff for possible risk factors. 31
Overall Survival and Relapse-Free Survival
Overall survival (OS) rate and relapse-free survival (RFS) were calculated from data received telephonically and hospital follow-up in which patient status, time of death, immediate cause of death, and time of recurrence was recorded.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 7.0 (GraphPad Software Inc. USA) and SPSS 25.0 (SPSS Inc. USA). The chi-square test was used for enumeration data, the independent sample t-test was used for comparison between 2 groups, and 1-way analysis of variance was used for comparison between multiple groups. Spearman’s correlation analysis was performed on protein expression, and
Results
LC3B Expression in HSCC and Adjacent Non-Tumor Mucosa Tissues, and the Ratio of CD8+/CD39+ and CD8+/CD163+ Immune Cells
Immunohistochemical results showed that LC3B was expressed in HSCC tissues and adjacent non-tumor mucosa tissues to different degrees, and the positive rate was higher in adjacent non-tumor mucosa tissues (68.18%) than that in HSCC tissues (35.71%). The difference between both the 2 groups was statistically significant (
Expression Patterns of LC3B and CD8+/CD39+ CD8+/CD163+ in Hypopharyngeal Squamous Cell Carcinoma (HSCC) and Adjacent Non-Tumor Tissues.

*

Representative immunohistochemical staining patterns. (A) Hypopharyngeal squamous cell carcinoma (HSCC) specimen and para-cancer normal tissues were stained for the detection of LC3B (200×), (B) CD8+ cytotoxic T lymphocytes (400×), (C) CD163+ tumor-associated macrophages (400×), (D) CD39+ regulatory T cells (200×).
Correlation Between LC3B Expression, and the Ratio of CD8+/CD39+ and CD8+/CD163+ Immune Cells in HSCC
The 70 cases of HSCC were divided into LC3B positive and LC3B negative groups according to LC3B expression. The positive rate of infiltration of CD8+/CD39+ was (15/25, 60%) in the LC3B positive group and (16/45, 35.6%,
The Correlation Between LC3B and the Infiltration of CD8+/CD39+, CD8+/CD163+ Immune Cells in Hypopharyngeal Squamous Cell Carcinoma (HSCC).

*
The Relationship Between LC3B Expression, the Ratio of CD8+/CD39+ and CD8+/CD163+ Immune Cells, and the Clinicopathological Features of HSCC
No significant correlation was observed between LC3B expression and the clinicopathological features of HSCC; and the infiltration amount of CD8+/CD39+ negatively correlated with lymph node metastasis and clinical stage (
Association of LC3B Expression and Infiltration of CD8+/CD39+ and CD8+/CD163+ Immune Cells in hypopharyngeal Squamous Cell Carcinoma (HSCC) With Clinicopathological Parameters From HSCC Patients.
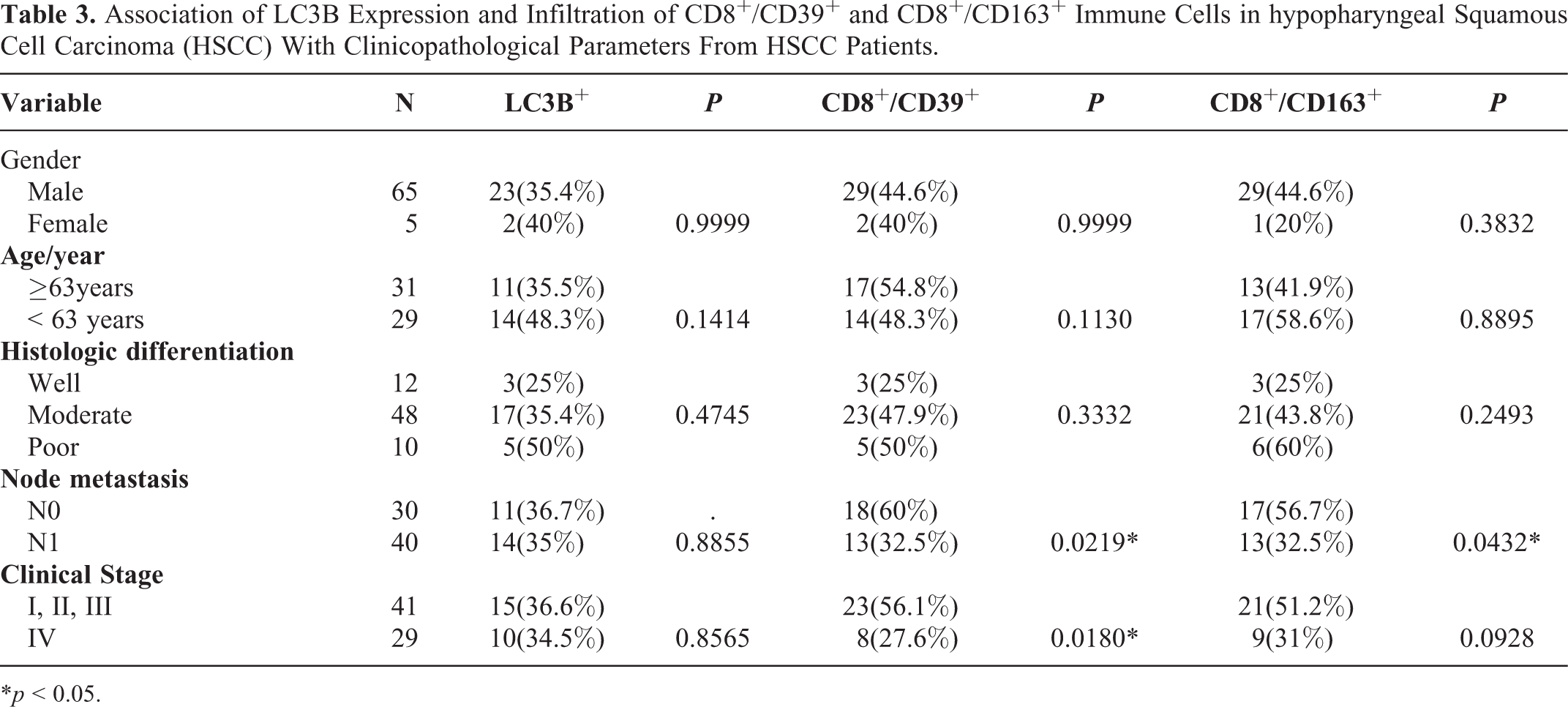
*
Relationship Between LC3B Expression, Immune Cell Ratio in HSCC, and Overall Survival Rate of HSCC Patients
According to Kaplan Meier curves and the Log-rank test, we found that patients with HSCC tissue LC3B+ overall survival rate is higher than that of negative patients (

Overall survival analyses based on LC3B and related immune cell ratio. Kaplan-Meier curves of overall survival in relation to LC3B (A and D), CD8+/CD39+(B and E) CD8+/CD163+ (C and F).

Kaplan-Meier curves of overall survival in relationship to the LC3B+ and CD8+/CD39+ co-positive, (A and C) LC3B+ and CD8+/CD163+ co-positive (B and D).
Relationship Between LC3B Expression, Immune Cell Ratio, and Relapse-Free Survival Rate in HSCC
Kaplan Meier curves and the Log-rank test showed that with LC3B+, CD8+/CD39+, and CD8+/CD163+ patients, with co-positive expression of LC3B and CD8+/CD39+, CD8+/CD163+, regardless of in HSCC tissues or adjacent non-tumor mucosa tissues all had a higher relapse-free survival rate than other patient groups; however, the difference was not statistically significant (

Relapse-free survival analyses based on LC3B and related immune cells ratio. Kaplan-Meier curves of overall survival in relationship to the LC3B (A and D), CD8+/CD39+ (B and E) CD8+/CD163+ (C and F).

Kaplan-Meier curves of relapse-free survival in relationship to the LC3B+ and CD8+/CD39+ copositive (A and C), LC3B+ and CD8+/CD163+ co-positive (B and D).
Discussion
HSCC is a highly malignant squamous cell carcinoma of the head and neck with poor clinical prognosis.
32
The molecular mechanism regulating the development of hypopharyngeal cancer remains unclear. Currently, surgery combined with radiotherapy is the most effective treatment, and its efficacy is markedly better than radiotherapy alone and surgery alone; however, immunotherapy has not been fully applied.
33
In adaptive immunity, the autophagy pathway has been shown necessary for antigen presentation, and downregulation of the major histocompatibility complex Ⅰ (MHC-Ⅰ) has been reported to be directly or indirectly involved in the mechanism of immune escape.
34
Autophagy is involved in cancer-related regulation, and its particular in cancer is complex and most likely associated with the internal environment.
35
In other types of tumors, CD8 infiltration is functionally considered cytotoxic/inhibits T lymphocytes and is typically associated with a good prognosis.
36
Distel et al.
37
reported 62 patients with low-risk head and neck squamous carcinoma (HNSCC) and high CD8 expression, who had a higher survival rate after first stage surgery and external radiotherapy. Additionally, Wang et al. reported that patients with high CD8+ T cell infiltration had a better prognosis than those with low CD8+ T cell infiltration. Moreover, CD8+ T cell infiltration was closely related to adenocarcinoma of the esophagogastric junction (AEG) patient TNM stage.
38
Recent studies have shown that CD39 is a member of the exo-nucleoside triphosphate bisphosphonolytic enzyme (e-ntpdase) family and plays an important role in regulating cancer progression in several human cancers. For example, Magis et al. showed that CD39+ Treg frequency and adenosine mediated inhibition were significantly increased (
Here, we detected the expression of LC3B, CD8, CD39 and CD163 in the tumors and para-cancer tissues of hypopharyngeal cancer patients. The expression levels of LC3B in para-cancer tissues were positive, and the ratios of CD8+/CD39+ and CD8+/CD163+ in tumor tissues was lower than that in para-cancer tissues. The ratio of CD8+/CD39+ was negatively correlated with the lymph node metastasis and clinical stage of patients, and the ratio of CD8+/CD163+ was negatively correlated with the lymph node metastasis in patients. Moreover, the ratios of CD8+/CD39+ and CD8+/CD163+ was associated with prognosis. We found that in patients with intra-tumor co-positive LC3B+ and CD8+/CD39+ and co-positive LC3B+ and CD8+/CD163+, the overall 5-year survival rate was higher than that of the other groups. Our study confirmed the influence of tLC3B+ and tCD8+/CD39+ ratio, tLC3B+ and tCD8+/CD163+ ratio, but not the pLC3B+ and pCD8+/CD39+ ratios and pLC3B+ and pCD8+/CD163+ ratio as prognostic factors in HSCC.
Furthermore, autophagy affected the relapse-free survival rate of patients with HSCC affected by autophagy and autophagy through immunity was also detected. We found that LC3B+, CD8+/CD39+, CD8+/CD163+, LC3B+, CD8+/CD39+, and LC3B+ and CD8+/CD163+ co-positive patients had higher RFS than other groupsin both intra and peritumor immune infiltrates, but the effect was not significant (
Several mechanisms by which LC3B induced autophagy can prevent cancer by influencing the immune microenvironment. Firstly, autophagy leads to the death of tumor cells. 49 Secondly, autophagy can eliminate damaged organelles, remove the source of genotoxic substances including damaged DNA products, and prevent oxidative stress from damaging organelles. 50 The tumor inhibitory effects of autophagy also stems from the negative regulation of cell proliferation ability to quench excessive activation of autophagy in cancer and may strengthen metabolism protein.51 The protein degradation process is likely to produce new and different molecular substances such as protein or antigen peptide conformations, including MHC-Ⅰ and MHC-Ⅱ molecules and the activity of these antigens affect different immune cell subsets. It is suggested that the change in autophagy level may be an important factor affecting the immune function of tumor cells.
In summary, the changes in autophagy activity of HSCC are closely associated with the degree of infiltration and prognosis of immune cells. The enhancement of autophagy activity of tumor cells is of great importance in improving the immune function of HSCC patients, and could potentially be a new implementation in research and development of tumor immunotherapy. However, the biological mechanism of autophagy is complex, and a variety of factors affect the immune function of tumors. In this study, the relationship between autophagy and known immune cells in HSCC was preliminarily discussed. However, the molecular biological mechanisms regarding the metabolism of autophagy proteins to produce new antigens, and changes in the corresponding immune response require further discussion.
Footnotes
Abbreviations
Acknowledgments
Author Contributions
C.G. and Z.W. conceived and designed the experiments, the data analysis; S.G., J.C, X.H, D.W, performed the experiments and the data analysis; S.G.wrote the paper; C.G. and Z.W.revised the paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statement
The use of the group was approved by the Biomedical Ethics Committee of Anhui Medical University with informed consent was signed by all relevant patients (approval No. 20200148.)
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by collective grants from Natural Science Foundation of Anhui Province, No.1608085MH189; Anhui Province Academic and Technical Leaders Reserve Candidates Grant, No.2018H160.
