Abstract
BACKGROUND:
Micro(mi)RNAs are a series of 20–24 nt non-coding small-molecule single-stranded RNAs that are believed to be closely related to tumor occurrence, development and other biological processes. MicroRNA-125a modulates radiochemotherapy sensitivity. However, the mechanism by which miRNA-125a regulates radiation resistance by lung cancer cells is yet to be elucidated.
OBJECTIVE:
The present study was designed to explore the biological role of miR-125a in regulating radioresistance in non-small cell lung carcinoma (NSCLC).
METHODS AND RESULTS:
The expression of miR-125a was assessed by quantitative real-time PCR in the human lung cancer cell lines, A549 and LTEP-a2. Notably, we found that miRNA-125a-5p regulated lung cancer radiosensitivity. We found that miRNA-125a-5p was more highly expressed in LTEP-a2 cells, which showed radiosensitivity compared to A549 cells with lower expression of miRNA-125a-5p. In addition, we up-regulated or down-regulated miR-125a-5p expression using an miR-125a-5p mimic or inhibitor, respectively, to reverse radioresistance. Flow cytometry revealed that the mimic increased the apoptotic rate as well as the expression of the apoptosis-related protein, cleaved poly ADP-ribose polymerase (PARP). Gene detection by luciferase reporter showed that sirtuin (SIRT)7 is a direct target of miR-125a-5p. Inhibiting SIRT7 using a small interfering RNA (siSIRT) abrogated resistance to radiation. In addition, the overexpression of SIRT7 decreased radiation-induced cell apoptosis.
CONCLUSION:
Our results indicated that the miR-125a level varies in NSCLC cell lines with different radiosensitivities. We demonstrated that miR-125a-5p upregulated SIRT7 and further upregulated apoptosis in lung cancer cells to increase their radiosensitivity. Our findings provide new directions for improving radiosensitivity in malignant lung tumors.
Introduction
Lung cancer is one of the most common cancers and leading causes of cancer death in the world. Non-small cell lung cancer (NSCLC) accounts for about 80% of all cases of lung cancer. Because the opportunity for surgery is lost for most patients by the time of diagnosis, this makes radiotherapy one of the important treatments for lung cancer. The overall 5-year survival rate is still only 15% in spite of the development of radiotherapy. To improve the radiosensitivity of cancer cells thus becomes a challenging problem.
MicroRNAs are a series of non-coding small-mole-cule single-stranded RNAs that are widely involved in the regulation of gene expression: studies on miRNA expression, before and after radiation, and differences in miRNA expression in tumors with distinct radiosensitivity profiles have gradually became hot topics of investigation [1, 2, 3, 4]. Numerous studies have shown that miRNAs can regulate the radiosensitivity of tumor cells and improve the potential of radiotherapy [5, 6, 7, 8, 9, 10]. Wagner–Ecker [11] showed that radiotherapy can change the expression of miRNAs in human vascular endothelial cells; interestingly, the expression of miR-125a, -127, -189, and let-7g can significantly influence the radiosensitivity of human vascular endothelial cells. Later, Moskwa et al. [12] suggested that the expression of miR-125a is associated with a suppressive effect in radiotherapy for malignant glioma. However, the specific mechanism of miR-125a in regulating radiosensitivity is still unknown. The sirtuin family of proteins (the SIRT family) is a set of highly conserved nicotinamide adenine dinucleotide (NAD
Based on the above, we hypothesized that miR-125a-5p regulates tumor cell apoptosis via SIRT7 expression. In the present study, we found that miR-125a-5p, was highly expressed in LTEP-a2 cells that showed radiosensitivity compared with A549 cells. In addition, we up-regulated miR-125a-5p expression using a mimic in A549 cells so that radioresistance was reversed. Further, we determined the apoptotic rate and expression of the apoptosis-related protein, cleaved poly ADP-ribose polymerase (PARP). Finally, luciferase reporter gene expression showed that SIRT7 is a direct target of miR-125a-5p. Targeting SIRT7 abrogated the resistance to radiation after SIRT7 inhibition by siSIRT7. The overexpression of SIRT7 decreased radiation-induced cell apoptosis and further decreased the radiosensitivity of cells.
Materials and methods
Cell lines
The human lung cancer cell lines, A549 and LTEP-a2, were purchased from the Chinese Academy of Sciences Cell Bank and cultured in RPMI-1640 medium. All cell lines were maintained with 100 U/mL of penicillin and 100 mg/mL of streptomycin, 10% fetal bovine serum, and incubated at 37
Quantitative real-time PCR
Quantitative real-time polymerase chain reaction (qRT–PCR) was used to detect the expression of miR-125a-5p in A549 and LTEP-a2 lung cancer cell lines. Total RNA was extracted with an RNeasy Minireagent Kit (Axygen Biosciences, Union City, CA, USA) and cDNA was then synthesized using miScript II RT Kit (Qiagen, Hilden, Germany) according to the manufacturers’ instructions. AceQ qPCR SYBR Green Master Mix (Qiagen) was used to measure miRNA expression levels and DNA expression using an ABI Prism 7500 Detection System (Applied Biosystems, Inc, Waltham, MA, USA). U6 was used an internal reference. Relative miRNA expression was calculated by the 2
After transfection of an miR-125a-5p mimic or inhibitor in A549 and LTEP-a2 lung cancer lines for 24 h, qRT–PCR was used to detect the expression of miR-125a-5p. With the same method, SIRT7 expression was tested in the same group, while
Colony formation assays
Pre-treated cells that underwent different treatments were cultured on 6-well plates at 1000–2000 cells/mL. The cells were irradiated using a linear accelerator (Unique System; Varian, Palo Alto, CA, USA) with a dose rate of 300 cGy/min and doses of 0, 2, 4, 6 and 8 Gy. The cells were cultured for 10–14 days to form colonies of more than 50 cells. The data were analyzed with a classic multitarget single hit model:
Transfection of miRNA mimic and inhibitor
An miR-125a-5p mimic and inhibitor were purchased from RiboBio (Guangzhou, China) and assessed according to the manufacturer’s instructions. The negative controls (RiboBio) used for miR-125a-5p transfection experiments were not homologous to any human miRNA sequences. The transfection was performed using 5
Flow cytometry
Pre-treated cells were cultured on 6-well plates. Cells were divided into four treatment groups: control, miR-125a-5p mimic or miR-125a-5p inhibitor, irradiation (IR), and irradiation combined with mimic or inhibitor (IR
Relative expression of miR-125a-5p and radiosensitivity in cell lines. A: Relative expression of miR-125a-5p in A549 cell line and LTEP-a2. B: Radiosensitivity of A549 and LTEP-a2 cell lines. C: Relative expression of miR-125a-5p in A549 with mimic. D: Relative expression of miR-125a-5p in LTEP-a2 with inhibitor. E: Radiosensitivity of A549 with mimic. F: Radiosensitivity of LTEP-a2 with inhibitor (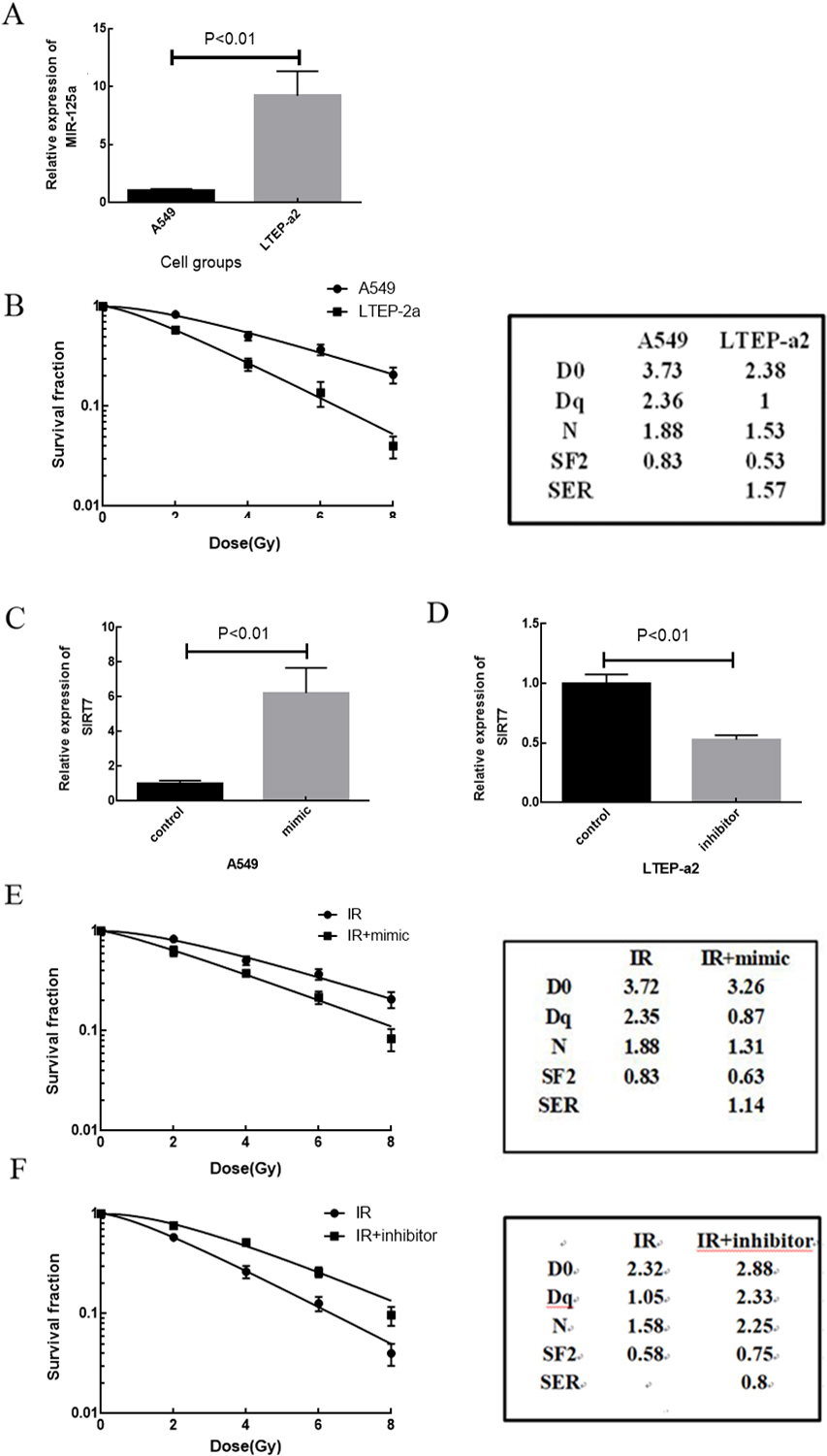
A. Apoptosis rate in the A549 cell line. B. Apoptosis rate in the LTEP-a2 cell line. C. Cleaved-PARP expression in the A549. D. Cleaved-PARP expression in the LTEP-a2 cell line (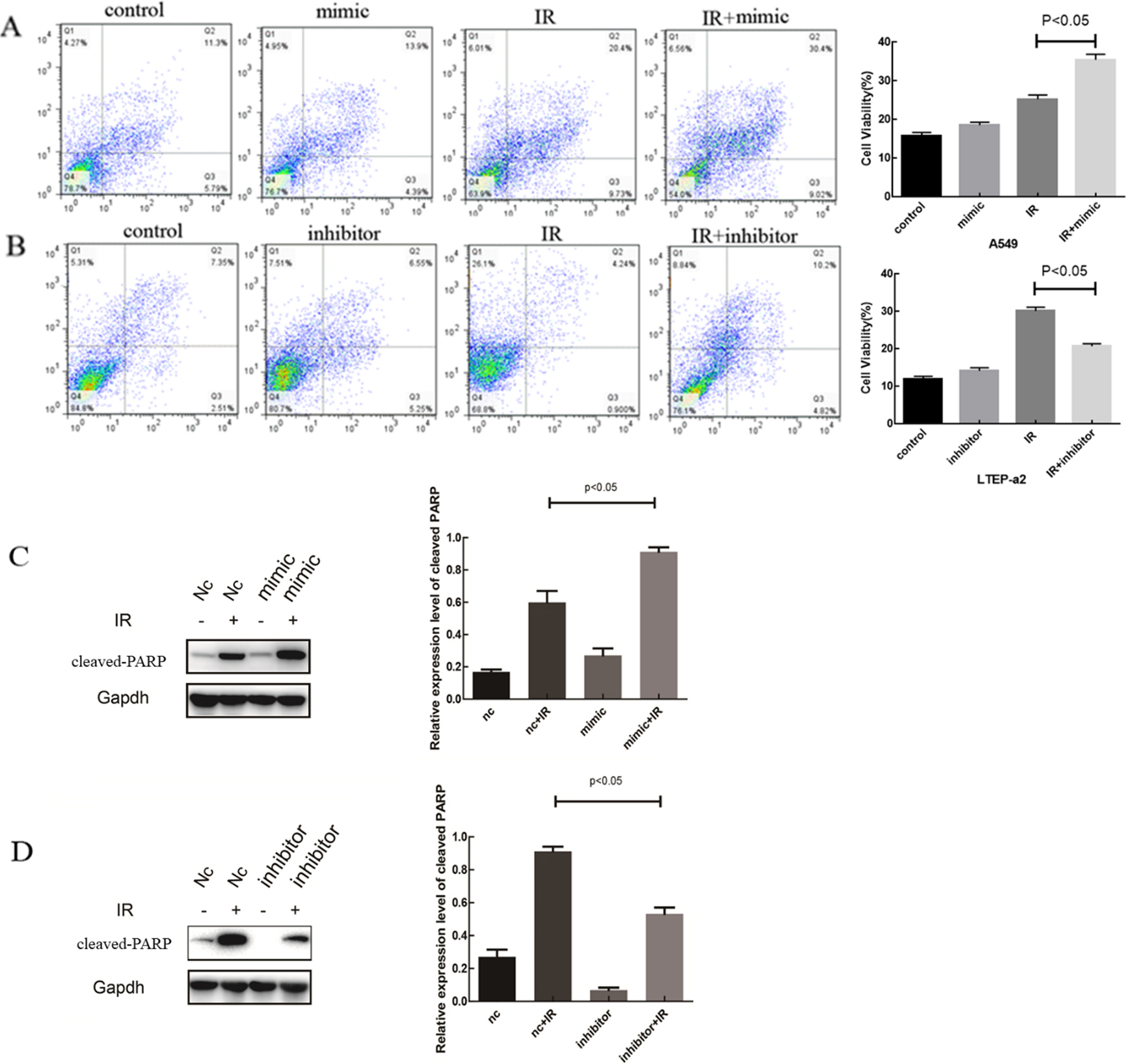
Cells were harvested and lysed using radioimmunoprecipitation buffer (Beyotime, Beijing, China). The total protein concentration was measured by a Bradford assay kit (Beyotime). Equal amounts of protein in samples were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis, and transferred onto polyvinylidene fluoride membranes. The membranes were blocked with 5% non-fat milk followed by incubation with primary antibodies overnight at 4
Dual luciferase assay
A luciferase reporter gene system was used to validate whether SIRT7 was the target gene of miR-125a-5p. We constructed a wild-type (WT) and mutation-type (MT) of the SIRT7 luciferase reporter plasmid, SIRT7 WT or SIRT7 MT, respectively. A miR-125a-5p mimic or negative control were transfected into A549 cells. After 24 h of transfection, cells were collected. A dual-luciferase reporter gene system (Promega, Madison, WI, USA) was used in this experiment. Renilla luciferase was used as the reference to detect firefly luciferase activity. Finally, a ratio of the firefly/Renilla was used as the relative expression value. Quantitative real-time (qRT)-PCR analysis was used to determine the effect of miR-125a-5p on the expression of SIRT7. After 24 h of A549 cell transfection with an miR-125a-5p mimic or negative control, and LTEP-a2 cell transfection with an miR-125a-5p inhibitor or negative control, the cells of each group were collected and assayed by qRT-PCR.
Tunel assay
Apoptosis of A549 cells was detected by terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL) using an In Situ Cell Death Detection kit (Roche, Mannheim, Germany) following the manufacturer’s instructions. The cells were plated on a 12-well plate and divided into groups: negative control, siSIRT7, radiation, siSIRT7 combined with radiation, expression, and expression combined with radiation. Slides with the cells were washed with PBS solution. The cells were then fixed with 4% paraformaldehyde for 15 min. After that, cells were perforated with 100
Statistical analysis
Data are presented as mean
Results
MiR-125a-5p expression in lung cancer cells
Quantitative RT-PCR analysis revealed significant differences in the expression level of miR-125a-5p in cells. The expression level of miR-125a-5p in LTEP-a2 cells was significantly higher than that in A549 cells (
A. Schematic representation of SIRT7 3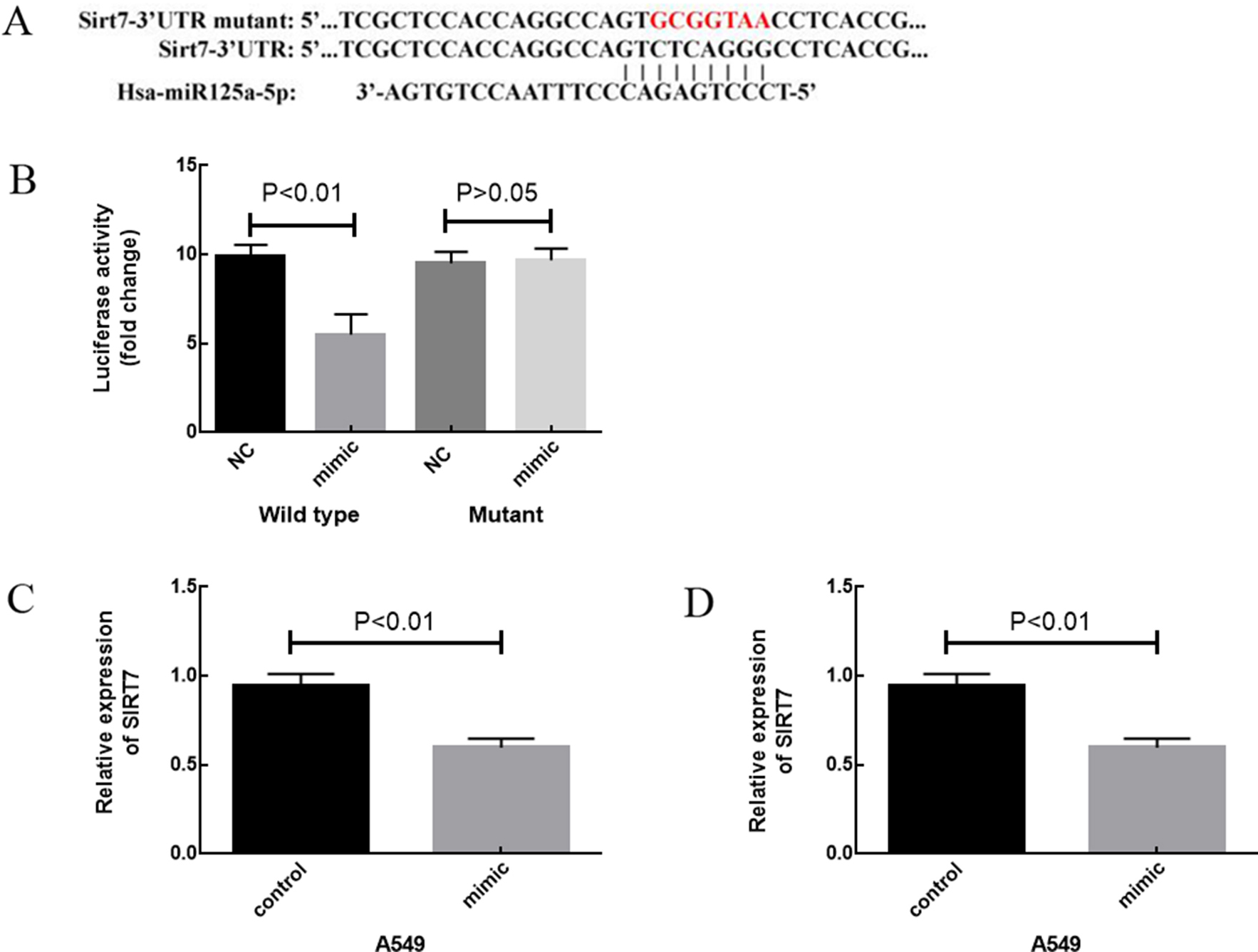
The radiosensitivity of the two cell lines was tested by colony formation assay. We found that the survival fraction of LTEP-a2 cells was significantly lower than that of A549 cells after irradiation with the same dose, with a radiation sensitization ratio of 1.57. This result suggested that LTEP-a2 was sensitive to radiation compared with A549 cells. Taken together, we found that cells in which miR-125a-5p was highly expressed were more sensitive to radiation than those in which MiR-125a-5p was expressed at a low level (Fig. 1B). To further investigate the association of the expression of miR-125a-5p with radiosensitivity, we transfected an miR-125a-5p inhibitor into LTEP-a2 cells that normally showed a high expression of miR-125a-5p. An miR-125a-5p mimic was transfected into A549 cells, which normally showed a low level of miR-125a-5p expression, to upregulate its expression level while a negative control sequence was transfected into the control group. The results suggested that the expression of miR-125a-5p in the two cell lines was significantly reversed after a mimic or inhibitor of miR-125a-5p was transfected; the difference is statistically significant (
A. Relative expression of SIRT7 in A549 cells with siSIRT7 1(si1) and siSIRT7 2(si2). B. Radiosensitivity of A549 cells with si1. C. Radiosensitivity of A549 cells with si2 (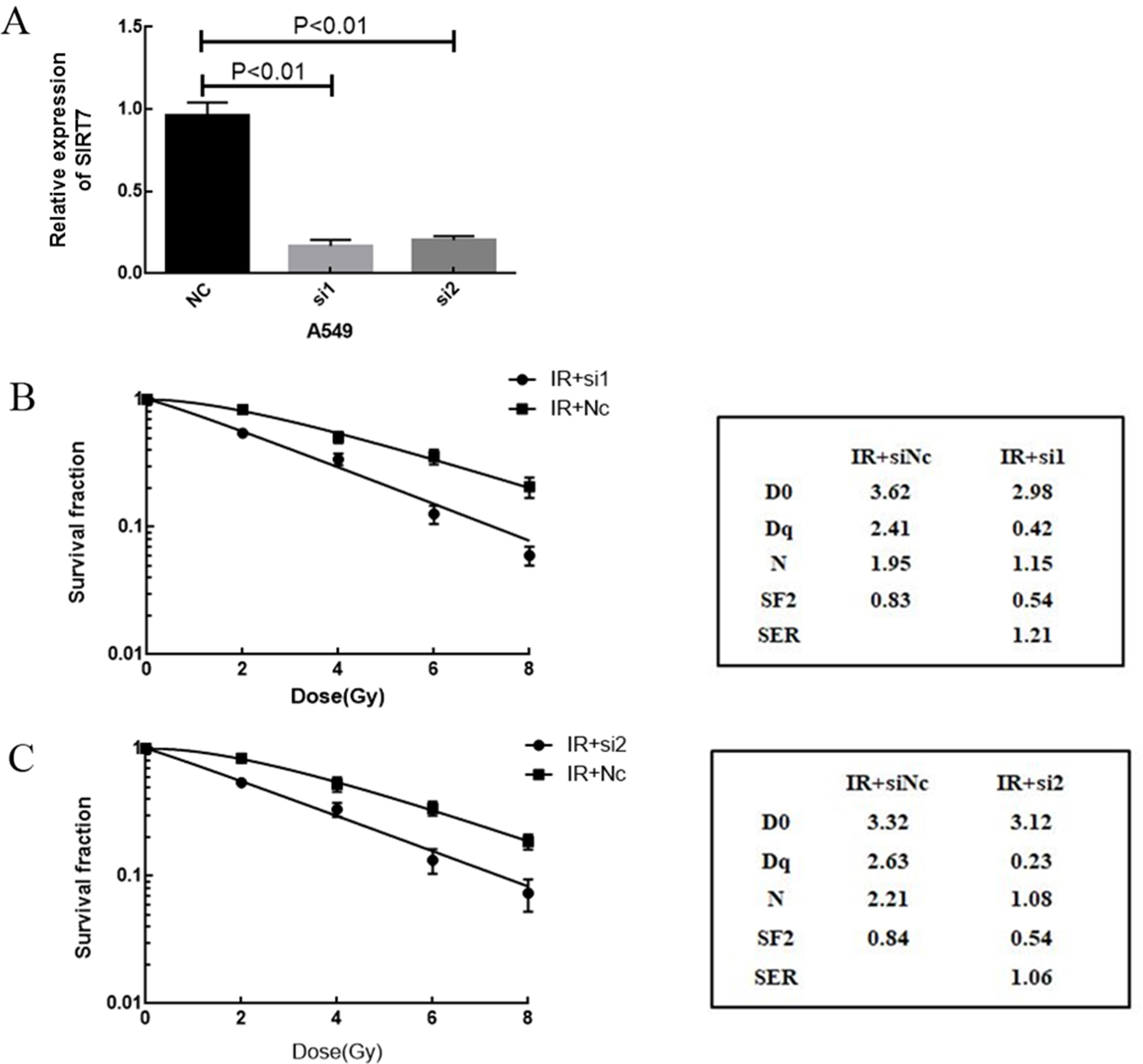
A: Apoptosis with siSIRT7 in A549 cell line. B: Apoptosis with si1 or si2 combined radiation in A549 cell line. C: Apoptosis with SIRT7 overexpression or SIRT7 overexpression combined radiation in A549 cell line.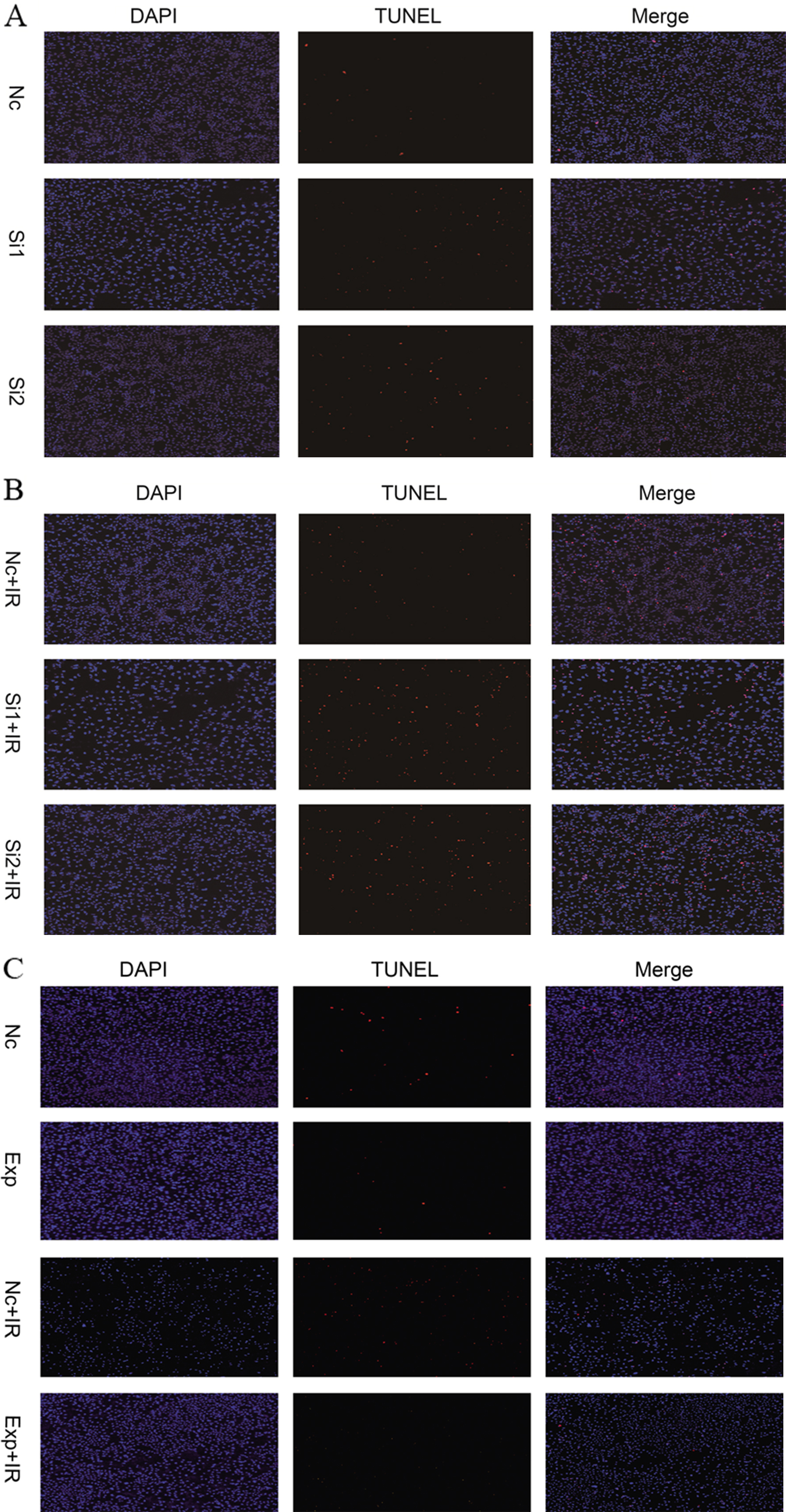
A. Expression of SIRT7 and cleaved-PARP in si1 or si2 group and si1 or si2 combined radiation group in A549 cell line. B. Expression of SIRT7 and cleaved-PARP in SIRT7 overexpression or SIRT7 overexpression combined radiation group in A549 cell line (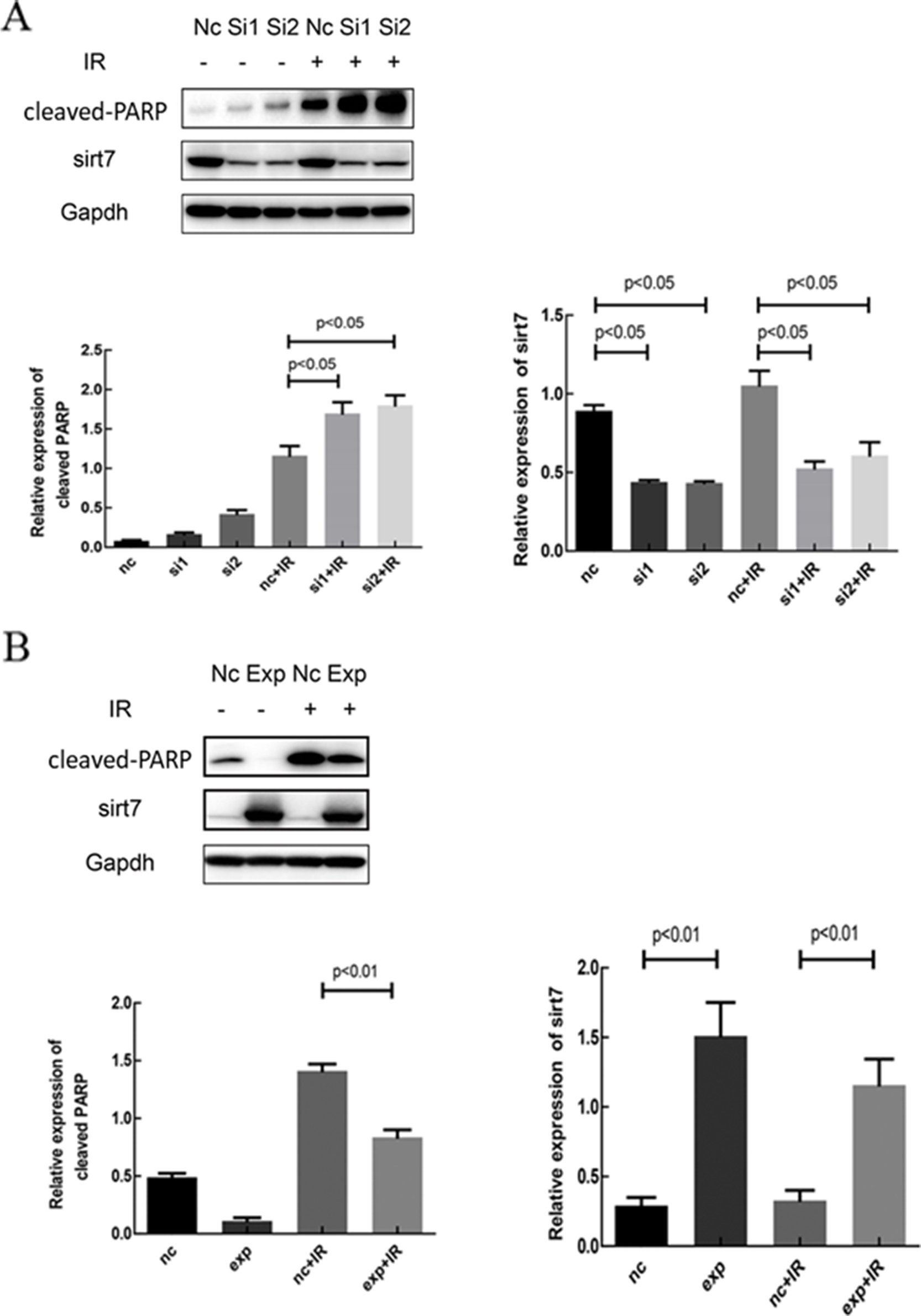
In accordance with the results of the cloning assays, we selected an ID50 irradiation dose to give the two cell lines: A549 was given a single irradiation of 4 Gy to induce apoptosis, and LTEP-a2 was given 2 Gy. We found that the apoptotic rate of the mi-125a-5p mimic combined with radiation group was increased in the A549 cell line compared with that of the simple irradiation group. In LTEP-a2, the apoptotic rate for the mi-125a-5p inhibitor combined with radiation group was lower than that of the simple irradiation group. This result clarified that miR-125a-5p increased radiosensitivity-induced apoptosis (Fig. 2A and B). To further investigate the regulation of cell apoptosis by miR-125a-5p, western blotting was used to estimate the expression of the apoptosis-related protein, cleaved-PARP. In the A549 cell line, the expression of cleaved-PARP was significantly increased in the mi-125a-5p mimic combined with radiation group compared with the simple irradiation group. While in LTEP-a2, the expression of cleaved-PARP was significantly decreased in the mi-125a-5p inhibitor combined with radiation group compared with the simple irradiation group; the difference was statistically significant (
SIRT7 is a direct target of miR-125a-5p
In accordance with bioinformatics analysis, SIRT7 was identified as a possible target of miR-125a-5p by three prediction algorithms (miRDB, miRWalk and TargetScan) (Fig. 3A). To confirm whether SIRT7 was a direct target of miR-125a-5p, a luciferase reporter vector containing the 3
Targeting SIRT7 knockdown abrogated resistance to radiation in A549 cells
To elucidate the role of SIRT7 in the regulation of radiosensitivity in A549 cells, SIRT7 was silenced in A549 cells by siRNA transfection. A qRT-PCR assay showed that siSIRT7 downregulated the expression of SIRT7; the difference between siSIRT7 treated cells and control was statistically different (
SIRT7 induced apoptosis and increased the radiosensitivity of A549 cells
To investigate the mechanism underlying the regulation of radiosensitivity of cells by SIRT7, cell apoptosis was measured by TUNEL assay. Compared with the negative control group, apoptosis in the siSIRT7 group was higher (Fig. 5A). In the siSIRT7 combined with radiation group, cell apoptosis was higher than that in the radiation group (Fig. 5B), which further suggested that the inhibition of SIRT7 increased radiation-induced apoptosis. In addition, in SIRT7 overexpression group, apoptosis was lower than in the negative control group. Apoptosis was lower in the SIRT7 overxepression combined with radiation group than in the radiation group (Fig. 5C). This suggested that upregulating the expression of SIRT7 decreased cell apoptosis. In a western blot, we detected cleaved-PARP and SIRT7 protein expression in the groups. As shown in Fig. 6A, the expression of the apoptotic protein, cleaved-PARP, was higher in the siSIRT7 combined radiation group, which suggested the effect of radiation after SIRT7 silencing was more marked than that induced by simple radiation (
Discussion
Lung cancer is one of the most malignant tumors according to the China Cancer Registration Annual Report [13]. NSCLC represents almost 85% of lung cancer cases [14]. As is well known, radiotherapy is one of the important treatment modalities for lung cancer. Unfortunately, the prognosis of most patients is weak mainly due to tumor radioresistance. The radiosensitivity of a tumor is affected by multiple factors such as the size of the tumor, the condition of the tumor bed, the proportion of the tumor cell population and the degree of differentiation of tumor cells; however, such factors are far from being adequately explained.
Evidence suggests that miRNA expression is associated with cancers, such as in lung, colon and breast [15, 16, 17, 18]. Therefore, further exploring the biological functions of miRNAs in cancer is important. MicroRNA may have several targets that regulate gene expression. This regulation mainly depends on the characteristics of their 3
Radiotherapy can induce cell apoptosis, which is the main route of cell death. It can enhance the apoptotic rate, leading to an increase in the radiosensitivity of cancer cells as shown in our previous study of oropharyngeal carcinoma [21]. Increasing the apoptotic rate in tumors leads to killing a large number of tumor cells which improves the efficacy of radiation and decreases the possibility of tumor relapse. According to recent studies, apoptosis is a key pathway for cell death. Remarkably, in our study, we found that up-regulating the expression of miR-125a-5p enhanced the radiosensitivity of cancer cells and the apoptotic rate, while down-regulating the expression of miR-125a-5p reduced the radiation-induced apoptotic rate.
SIRT7 is a member of the sirtuin family of proteins that contains seven sirtuins (SIRT1-7). They are widely expressed in mammals and exert NAD
The present study showed that miR-125a-5p plays an important role in increasing the radiosensitivity of lung tumor cells via SIRT7, which can result in a diversity of cellular sensitivities to radiation. Taken together, our results strongly suggest that miR-125a-5p may become a target for the further development of radiation therapy for lung cancer.
Footnotes
Acknowledgments
This study was supported by Science and Technolo- gy Plan Project of Liaoning Province (No. 201602452, No. 20180540129 No. 20180530095), Personnel Trai- ning Project of Liaoning Cancer Hospital & Insititute (201703), Key Laborotary of Tumor Radiosensitization and Normal Tissue Radioprotection, Liaoning Province, China (2018225102).
