Abstract
BACKGROUND:
Radiotherapy is one of main useful therapies in non-small cell lung cancer (NSCLC). Nevertheless, the underlying mechanism between NSCLC cell radiosensitivity and effective treatment remains unclear.
OBJECTIVE:
The aim is to explore the relationship between circular (circ) RNA and NSCLC cell radiosensitivity.
METHODS:
CircRNA plasmacytoma variant translocation 1 (PVT1) and microRNA (miR)-1208 expression in NSCLC cells were assessed using quantitative reverse transcriptase PCR (qRT-PCR). NSCLC cells were transfected with si-PVT1 or miR-1208 inhibitor and then exposed to irradiation. Cellular biology behaviors were detected using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), Terminal deoxynucleotidyl transferase dUTP Nick-End Labeling (TUNEL), colony formation, invasion and western blot. Additionally, binding between circPVT1 and miR-1208 was testified by dual-luciferase reporter and RIP assay.
RESULTS:
CircPVT1 was upregulated in NSCLC cells after irradiation treatment. Silencing circPVT1 induced inhibition of NSCLC cell growth and invasion, accompanied by cell apoptosis and
CONCLUSIONS:
Silencing circPVT1 enhanced radiosensitivity of NSCLC cells by sponging miR-1208.
Introduction
Cancer is one of the most serious diseases with high incidence and mortality rate [1]. Thus, deep understanding mechanisms are of great significance for the treatment of lung cancer. Currently, cancer treatment strategies include radiotherapy, surgery, chemotherapy and immunotherapy [2, 3, 4]. Among them, radiotherapy is one of main means, and about 50% of cancer patients are treated with radiotherapy at their doctor’s recommendation [5]. In addition, patients who respond well to radiotherapy generally show better clinical outcomes in different cancers. Lung cancer is the primary cause of cancer-associated death worldwide [6]. Lung cancer includes two main types, small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), and NSCLC accounts for approximately 85% [7, 8]. Radiotherapy is the firstly considered treatment for inoperable NSCLC patients who have no change of surgery. Unfortunately, clinical resistance of radiotherapy has been identified as a big obstacle during process of NSCLC treatment [9]. So, the improvement of radiosensitivity is extremely significant for effective treatment of NSCLC. A mass of evidence has elucidated that noncoding RNAs (ncRNAs), play significantly appreciated roles during regulating radiation responses, including, microRNAs (miRNAs) and circular RNAs (circRNAs) [10, 11, 12].
CircRNAs are circular ncRNAs that have no 5’-hat and 3’-tail structure [13]. CircRNAs are highly cell-specific and tissue-specific in eukaryotes [14]. A review has detailedly illustrated the relationship between circRNAs and the progression, deterioration, migration as well as recurrence of cancer [15]. CircPVT1 derives from the second exon of PVT1 gene, whose locus is embedded in lncRNA PVT1 and is highly expressed in a variety of cancers [16]. LncRNA PVT1 has been proved to modulate radiosensitivity of NSCLC, and knockdown of lncRNA PVT1 could enhance radiosensitivity [17]. Moreover, circPVT1 has been found to be high-expressed in NSCLC, and circPVT1 dramatically accelerate cell proliferation and invasion [18]. However, it was not well known whether circPVT1 modulated NSCLC radiosensitivity and how it exerted its functions.
MicroRNAs (miRNAs) have the length of 21–23 nucleotides [19] and are another a class of ncRNAs with line structure [20]. MiRNAs also have regulatory effects on cancer cell survival, including NSCLC [21]. For instance, miR-340-5p exhibited a lower expression level in NSCLC tissues and cells, and overexpressing miR-340-5p could limit cell proliferation [22]. Recently, it has been observed that circRNAs can work as miRNA sponges in several types of cancer [23]. For example, in NSCLC cells, circPVT1 could sponge miR-125b to facilitate cell proliferation and invasion [18]. Thus, we hypothesized that circRNA might be involved in radiosensitivity through specific downstream miRNA.
In this study, we for the first time exploited the function of circPVT1 on radiosensitivity of NSCLC cells. In addition, the potential mechanisms of circPVT1 regulating radiosensitivity of NSCLC were preliminarily identified.
Materials and methods
Cell lines and cell culture
Five kinds of human NSCLC cell lines (H226, H1650, A549, H292 and H1299) and normal cell line (bronchial epithelial cell, BEA-2B) were acquired from the American Type Culture Collection (ATCC, Manassas, VA, USA). Cells were cultured in Dulbecco’s modified Eagle’smedium (DMEM) (Thermo Fisher Scientific, Waltham, MA, USA) containing 10% fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 100 U/ml penicillin and 100
Transfection assay
SiRNAs specific targeting circPVT1 (si-PVT1), miR-1208 mimic (miR-1208), miR-1208 inhibitor and their own negative controls were purchased from GenePharma company (Shanghai, China). Cells were cultured until logarithmic phase, and then mixed cell suspension was cultivated in 6-well plates (4
Ionizing radiation treatment
5
3-(4,5-Dimethylthiazol-2-y1)-2,5-diphenyl tetrazolium bromide (MTT) assay
MTT assay was performed to detect cell proliferation as previously reported [26]. When stably transfected cells grew to logarithmic phase, they were planted into a 96-well plate and cultured overnight. Whereafter, 20
TUNEL assay
To assess cell apoptosis, TUNEL staining was carried out [27]. Briefly, transfected cells were fixed with 4% formaldehyde. Then, the rest steps were accomplished, in accordance with specification of TUNEL kit (Roche, Mannheim, Germany). When viewing with a fluorescence microscope (DMI4000B, Leica, Wetzlar, Germany), TUNEL-tagged cells were computed.
Colony formation assay
For analysis of ability of cell survival, colony formation assay was performed as previously reported [28]. After cell transfection and radiation treatment, cells were seeded into 6-well plates (200 cells/well) and cultivated in incubator with 5% CO2 at 37
Invasion assay
To assess the ability of invasion of cell, invasion experiment [29] was conducted with a 24-well transwell chamber plate (BD Biosciences, St Louis, MO, USA). A 24-well transwell chamber plate was divided into two chambers, upper and lower chamber, with an 8
Western blot
For the analysis of expression of related proteins, western blot assay was performed [30]. Proteins were prepared with RIPA (Vazyme, Nanjing, China) were applied to perform the detection of concentration with BCA Kit (Vazyme), equal protein specimens (40
CircPVT1 was highly expressed in NSCLC cells. (A) CircPVT1 levels in human lung cancer cell lines (H226, H1650, A549, H292 and H1299) and normal bronchial epithelial cell line (BEAS-2B) were determined utilizing qRT-PCR. (B) CircPVT1 expression in (0, 2, 4, 6, and 8 Gy) irradiated human lung cancer cell lines H226, H1650, A549, H292 and H1299 cells was tested using qRT-PCR. Compared with BEAS-2B, 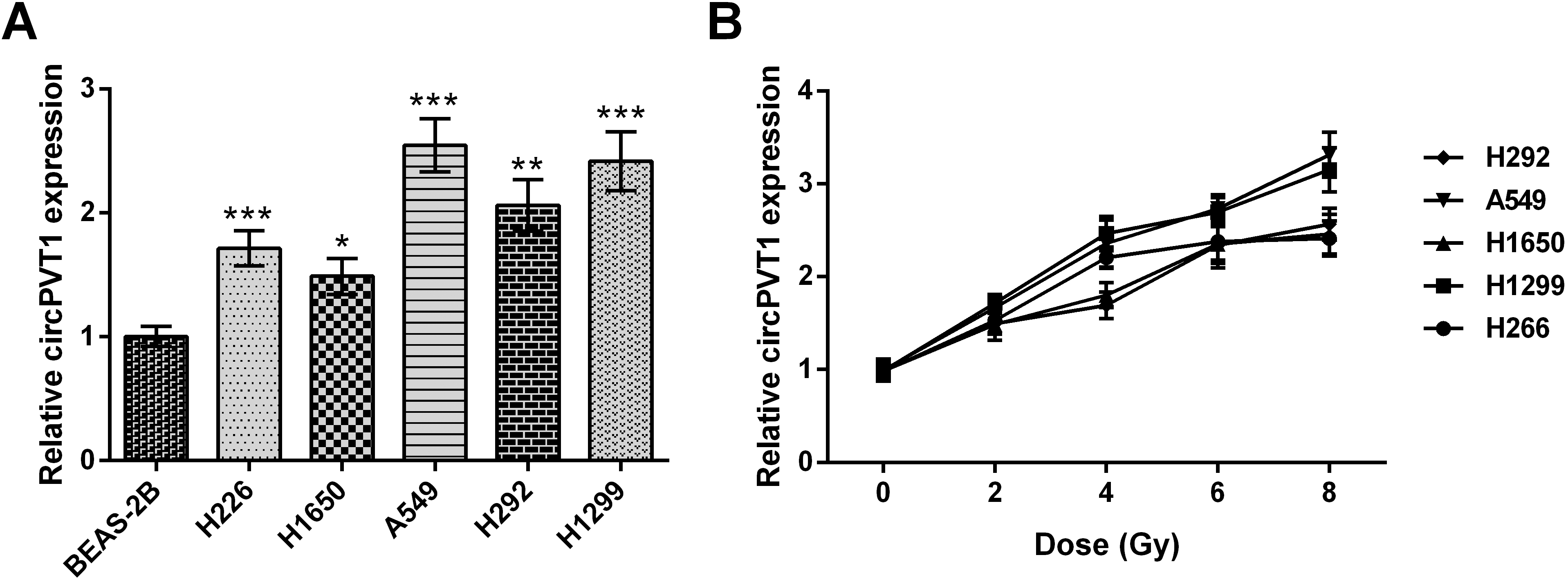
The potential downstream factor of circPVT1 was found out through circular RNA Interactome, namely miR-1208. For verifying targeted association between circPVT1 and miR-1208, the dual-luciferase reporter assay was performed as previously reported [31]. The sequence of circPVT1 untranslated region (3’-UTR) covering the predicted combining sites of miR-1208 or mutated sequences of combining sites were ligated into pGL4 Luciferase Reporter Vector (Promega, Madison, WI, USA), formed circPITX1-WT or circPITX1-MUT, severally. HEK-293T cells were co-transfected with recombinant vectors and miR-1208 (miR-NC). 48 h later, luciferase activity was evaluated with a Dual Luciferase Reporter Assay Kit (Vazyme).
RNA immunoprecipitation (RIP) assay
For investigating the interaction between circPVT1 and miR-1208, RIP was enforced with specific AGO2 antibody [32]. In brief, after HEK-293T cells were subjected to transfection with miR-1208/miR-NC or untreation, cells were lysed with RIRP, and then lysates of cellswere mixed with RIP immunoprecipitation buffer containing magnetic beads conjugated with human anti-Argonaute2 (AGO2) antibody (Abcam, ab32381), or NC mouse IgG (Millipore). After complexes underwent with washing and elution, RNAs were extracted and analyzed with qRT-PCR assay.
Quantitative reverse transcription PCR (qRT-PCR)
All RNAs in cells were collected with Trizol regent (Invitrogen). 1
Silencing circPVT1 repressed cell proliferation in NSCLC cells. (A) CircPVT1 expression in untreated, si-NC-transfected, si-PVT1-transfected NSCLC cell lines (A549 and H1299) was analyzed applying qRT-PCR. (B, C) Viability, (D, E) colony number in un-treated, si-NC-transfected, si-PVT1-transfected, si-NC-transfected plus 8 Gy-irradiated and si-PVT1-transfected plus 8 Gy-irradiated NSCLC cell lines (A549 and H1299) were explored through MTT and colony formation, respectively. 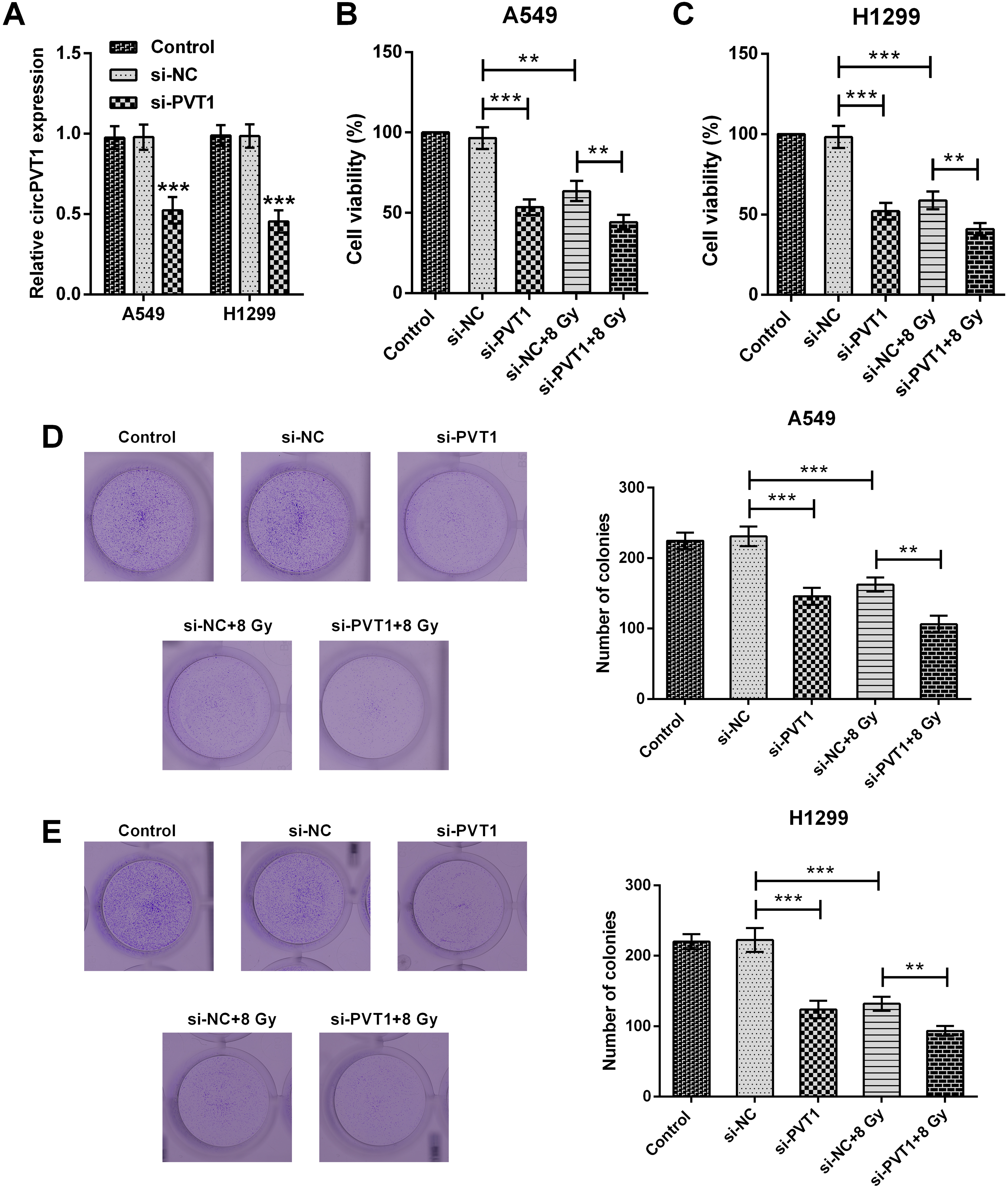
In the study, data was analyzed with Graphpad prism 5 (GraphPad Software, San Diego, CA, USA). The significance of two groups was assessed through one-way ANOVA or Student’s
Silencing circPVT1 accelerated apoptosis in NSCLC cells. (A, B) apoptotic rate and (C) 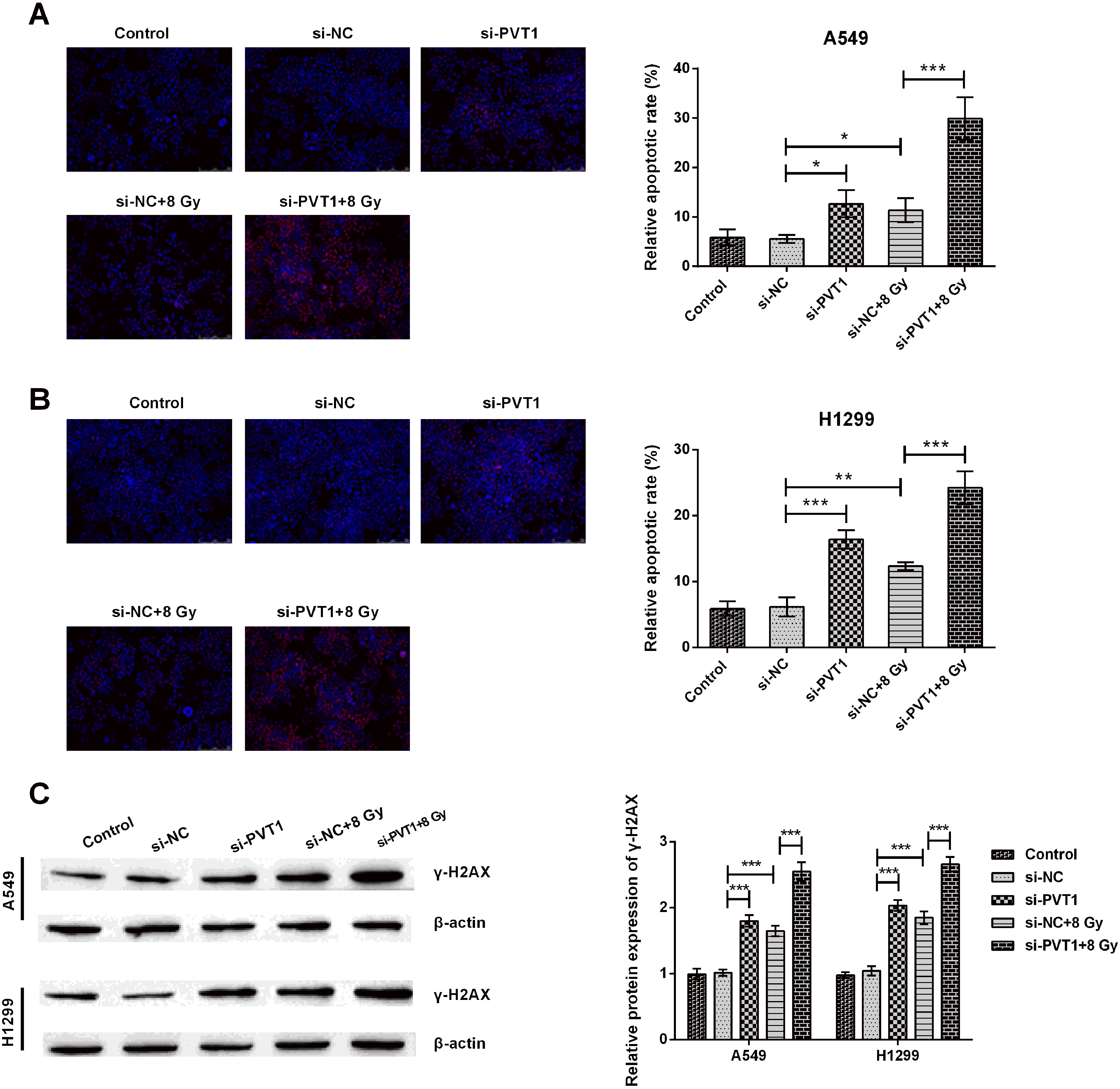
CircPVT1 was highly expressed in NSCLC cells
For exploring expression level of circPVT1 in NSCLC cells, we utilized five kind of NSCLC cell lines (H226, H1650, A549, H292 and H1299) and a kind of normal lung cell line (BEAS-2B). qRT-PCR experiment was adopted to exploit circPVT1 expression level. As displayed in Fig. 1A, circPVT1 expression was significantly higher in all NSCLC cell lines than that in BEAS-2B cells (
Silencing circPVT1 suppressed the invasion of NSCLC cells. (A, B) Relative invasive rate and (C, D) epithelial-mesenchymal transitions (EMT)-related protein (E-cadherin, N-cadherin and Vimentin) expression in un-treated, si-NC-transfected, si-PVT1-transfected, si-NC-transfected plus 8 Gy-irradiated and si-PVT1-transfected plus 8 Gy-irradiated NSCLC cell lines (A549 and H1299) were investigated adopting invasion and western blot assays. The comparison between the two marked groups, 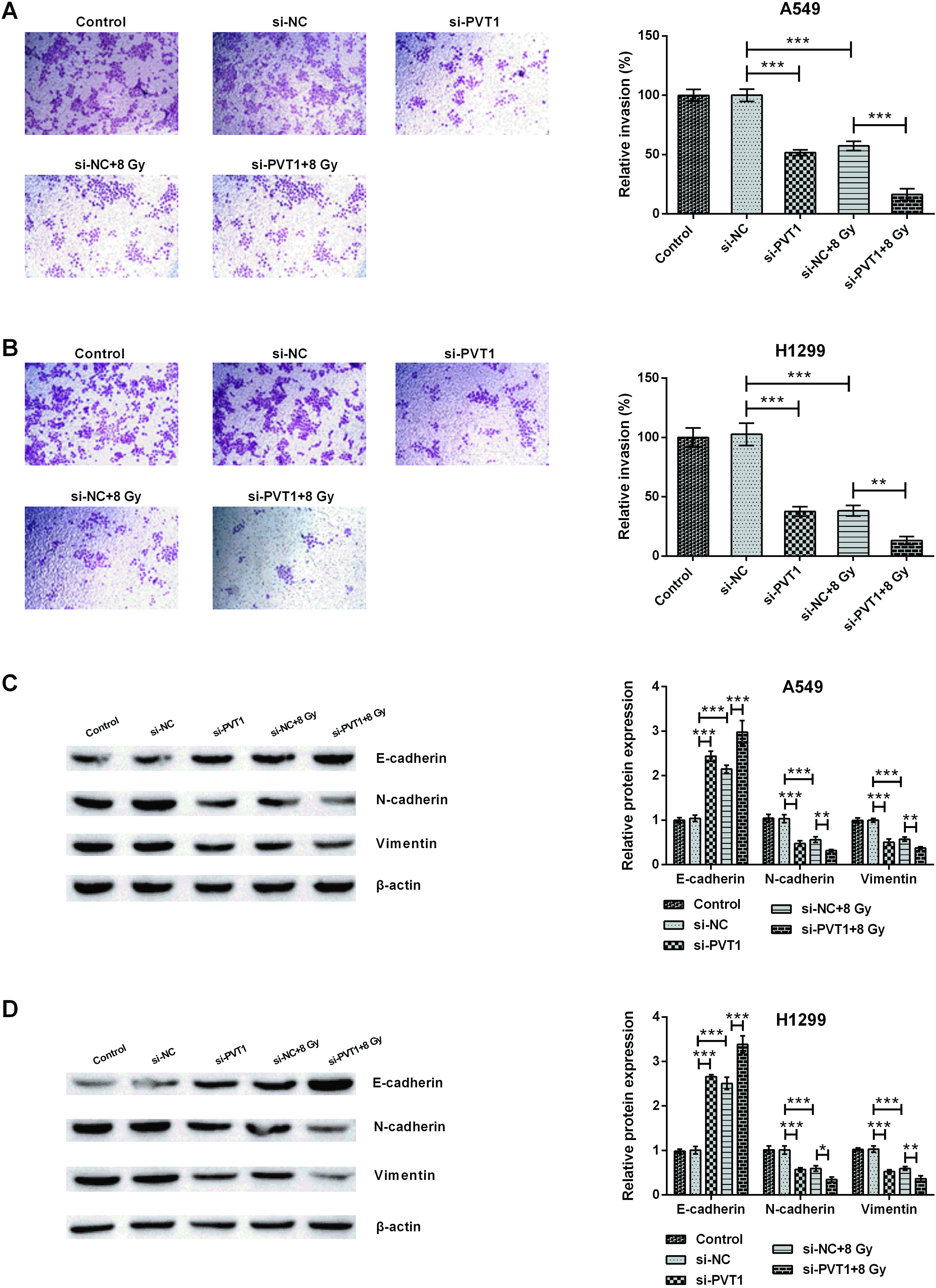
CircPVT1 targeted and oppositely regulated miR-1208. (A) MiR-1208 expression in human lung cancer cell lines (A549 and H1299) and normal bronchial epithelial cell line (BEAS-2B) was determined utilizing qRT-PCR. Compared with BEAS-2B, 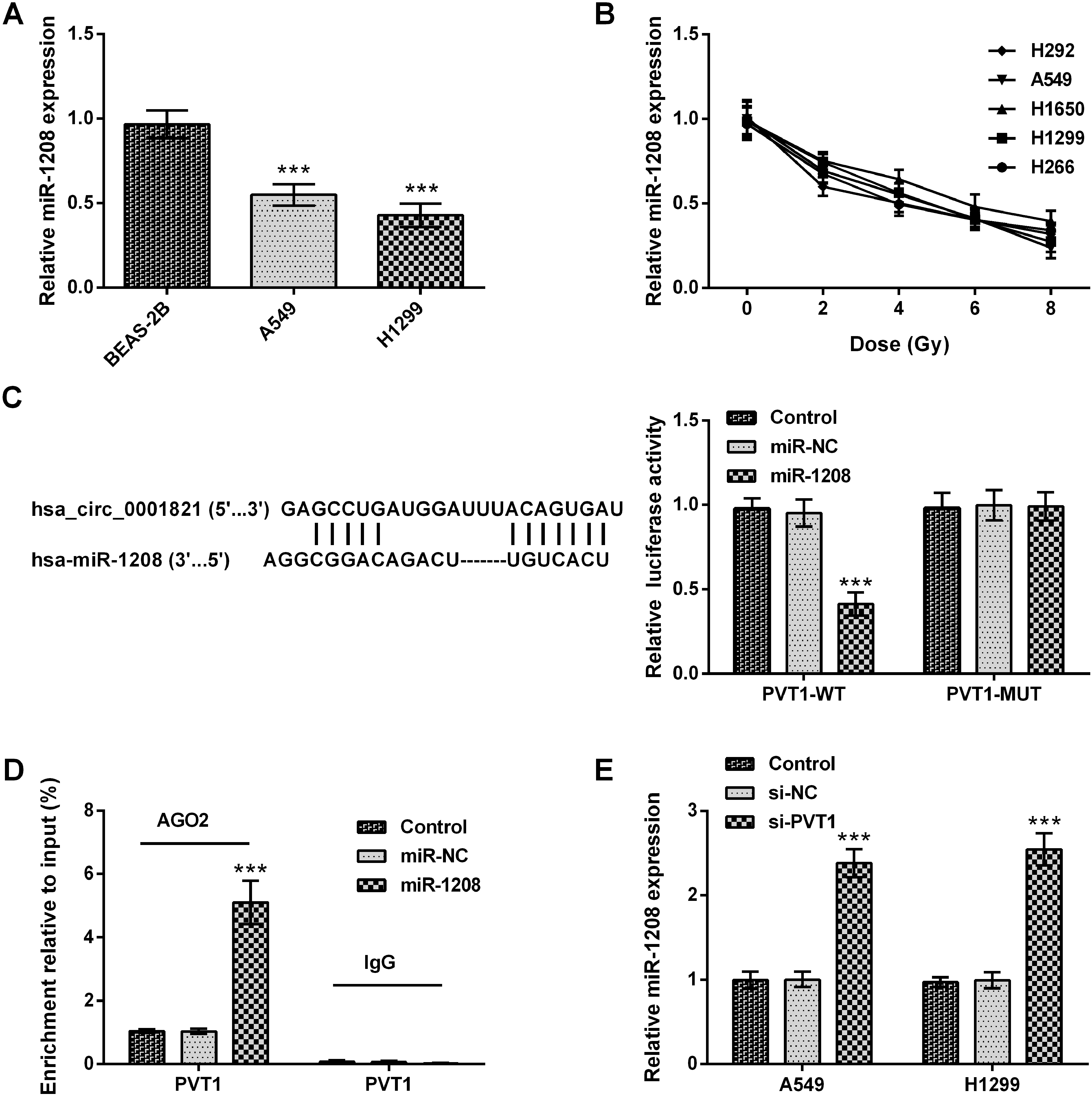
Silencing circPVT1 repressed cell proliferation in NSCLC cells by regulating miR-1208. (A) MiR-1208 expression in untreated, NC inhibitor-transfected and miR-1208 inhibitor-transfected NSCLC cell lines (A549 and H1299) was tested utilizing qRT-PCR. (B, C) Viability and (D, E) colony number, in untreated, si-NC plus NC inhibitor-co-transfected, si-NC plus NC inhibitor-co-transfected plus 8 Gy-irradiated, si-PVT1 plus NC inhibitor-co-transfected plus 8 Gy-irradiated and si-PVT1 plus miR-1208 inhibitor-co-transfected plus 8 Gy-irradiated NSCLC cell lines (A549 and H1299) were explored through MTT and colony formation assay, respectively. The comparison between the two marked groups, 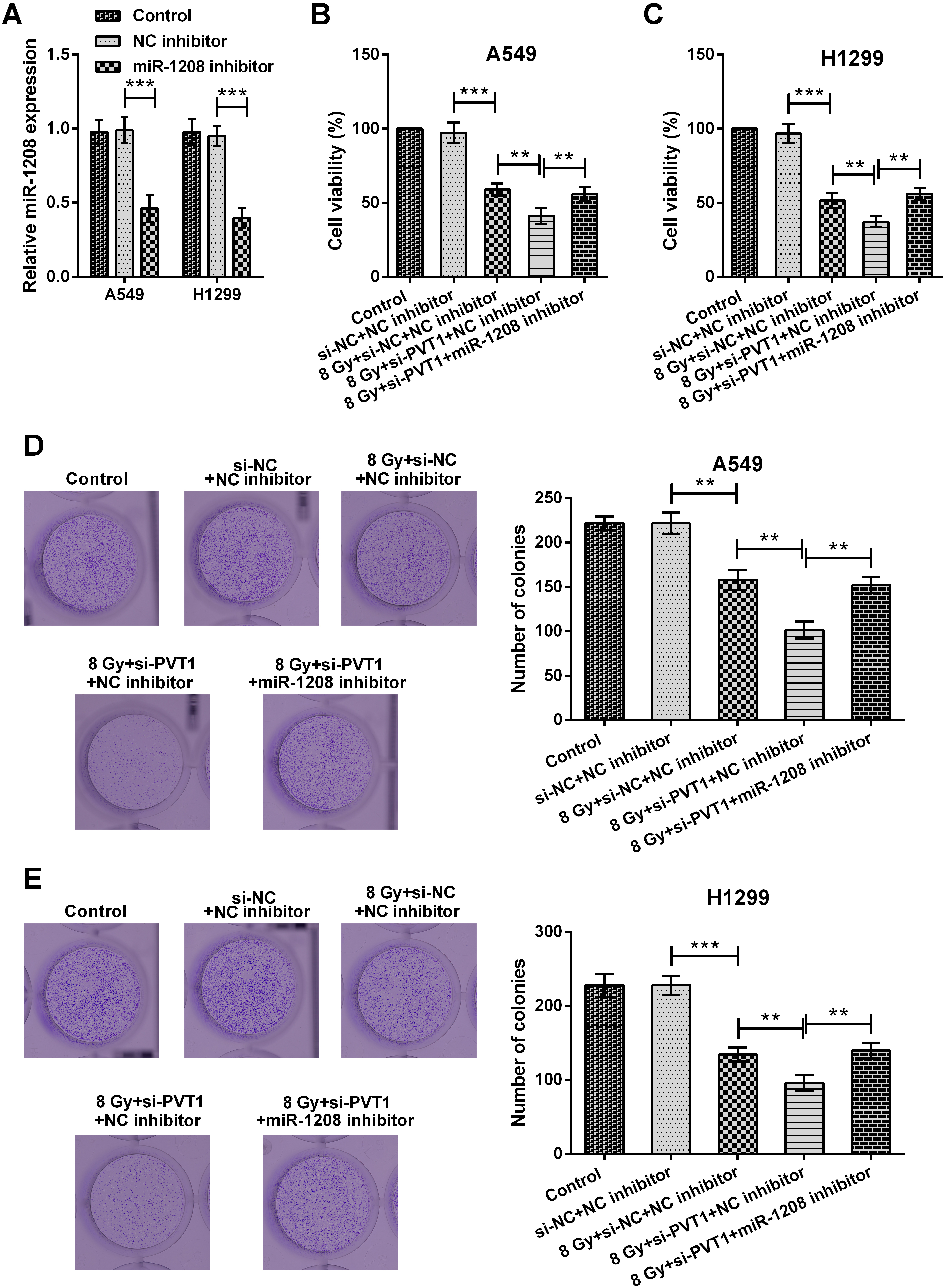
Since circPVT1 upregulation correlates to irradiation treatment, we asked whether circPVT1 knockdown could enhance radiosensitivity in NSCLC cells. To this end, A549 and H1299 cells were transfected with si-PVT1 or si-NC, respectively (both
Silencing circPVT1 inhibited NSCLC cell invasion
Subsequently, the influence of circPVT1 on NSCLC cell invasion was also studied. Figure 4A–B illustrated that circPVT1 knockdown impaired the migration ability of A549 and H1299 cells and 8 Gy radiation treatment further inhibited NSCLC cell invasive ability (
CircPVT1 was a potential sponge of miR-1208
To further explore the molecular mechanism of circPVT1 in regulating NSCLC cell radiosensitivity, the analysis between miRNAs and circPVT1 was conducted. The annlysis from circular RNA Interactome indicated that miR-1208 was a potential downstream miRNA of circPVT1. The results of manifested that miR-1208 was markedly downregulated in A549 and H1299 cells when compared with BEAS-2B cells in which it was identified by qRT-PCR (
Silencing circPVT1 improved radiosensitivity in NSCLC cells by up-regulating miR-1208
To prove the function of miR-1208 on radiosensitivity regulated by circPVT1 in NSCLC cells, miR-1208 inhibitor was transfected to conduct the following experiments. Successful transfection was the key to the subsequent experiments. We found that our transfection assay was successful, as miR-1208 inhibitor transfection lowered miR-1208 expression in A549 and H1299 cells (both
Silencing circPVT1 accelerated apoptosis in NSCLC cells by regulating miR-1208. (A, B) apoptotic rate and (C) 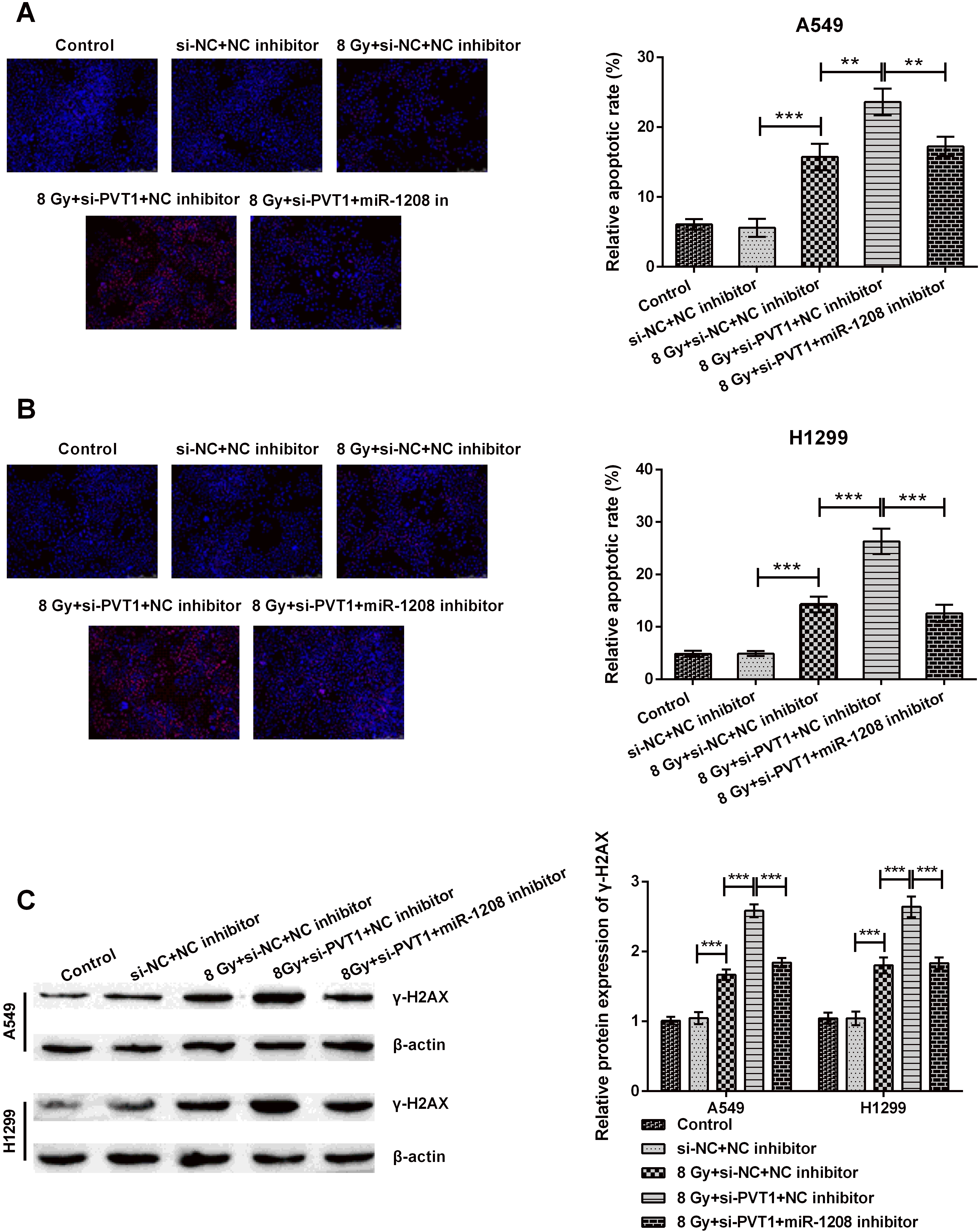
Next, NSCLC cell invasion was examined by transfecting miR-1208 inhibitor. It was seen that miR-1208 inhibitor transfection fortified invasive rate (both
Silencing circPVT1 suppressed the invasion of NSCLC cells by regulating miR-1208. (A, B) Relative invasive rate and (C, D) epithelial-mesenchymal transitions (EMT)-related protein expression (E-cadherin, N-cadherin and Vimentin) in untreated, si-NC plus NC inhibitor-co-transfected, si-NC plus NC inhibitor-co-transfected plus 8 Gy-irradiated, si-PVT1 plus NC inhibitor-co-transfected plus 8 Gy-irradiated and si-PVT1 plus miR-1208 inhibitor-co-transfected plus 8 Gy-irradiated NSCLC cell lines (A549 and H1299) were investigated adopting invasion and western blot assays. The comparison between the two marked groups, 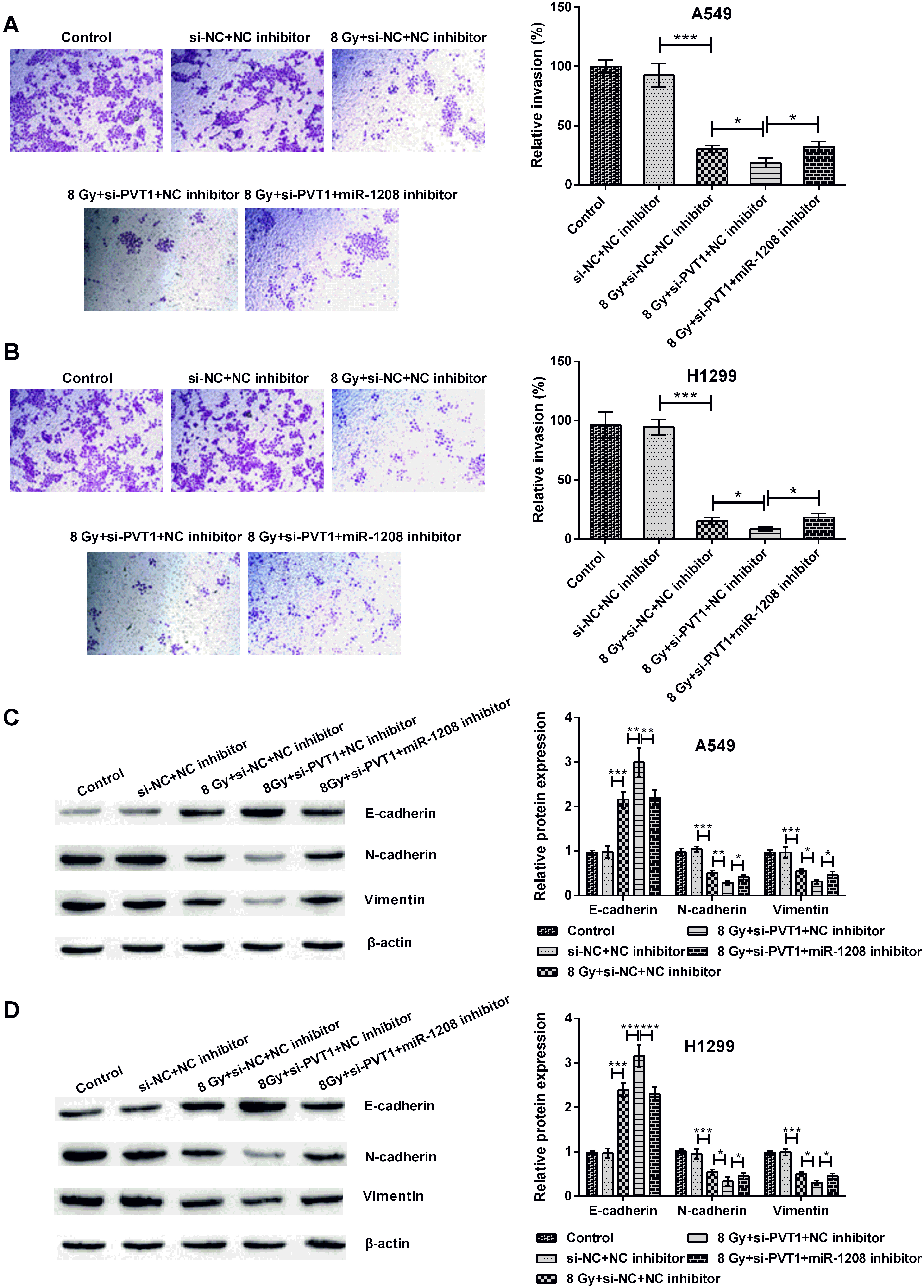
Silencing circPVT1 blocked the PI3K/AKT/mTOR signaling by regulating miR-1208. (A, B) The p-p85 PI3K, p-mTOR and p-AKT expression in untreated, si-NC plus NC inhibitor-co-transfected, si-PVT1 plus NC inhibitor-co-transfected, si-NC plus NC inhibitor-co-transfected plus 8 Gy-irradiated, si-PVT1 plus NC inhibitor-co-transfected plus 8 Gy-irradiated and si-PVT1 plus miR-1208 inhibitor-co-transfected plus 8 Gy-irradiated NSCLC cell lines (A549 and H1299) was detected adopting western blot assay. The comparison between the two marked groups, 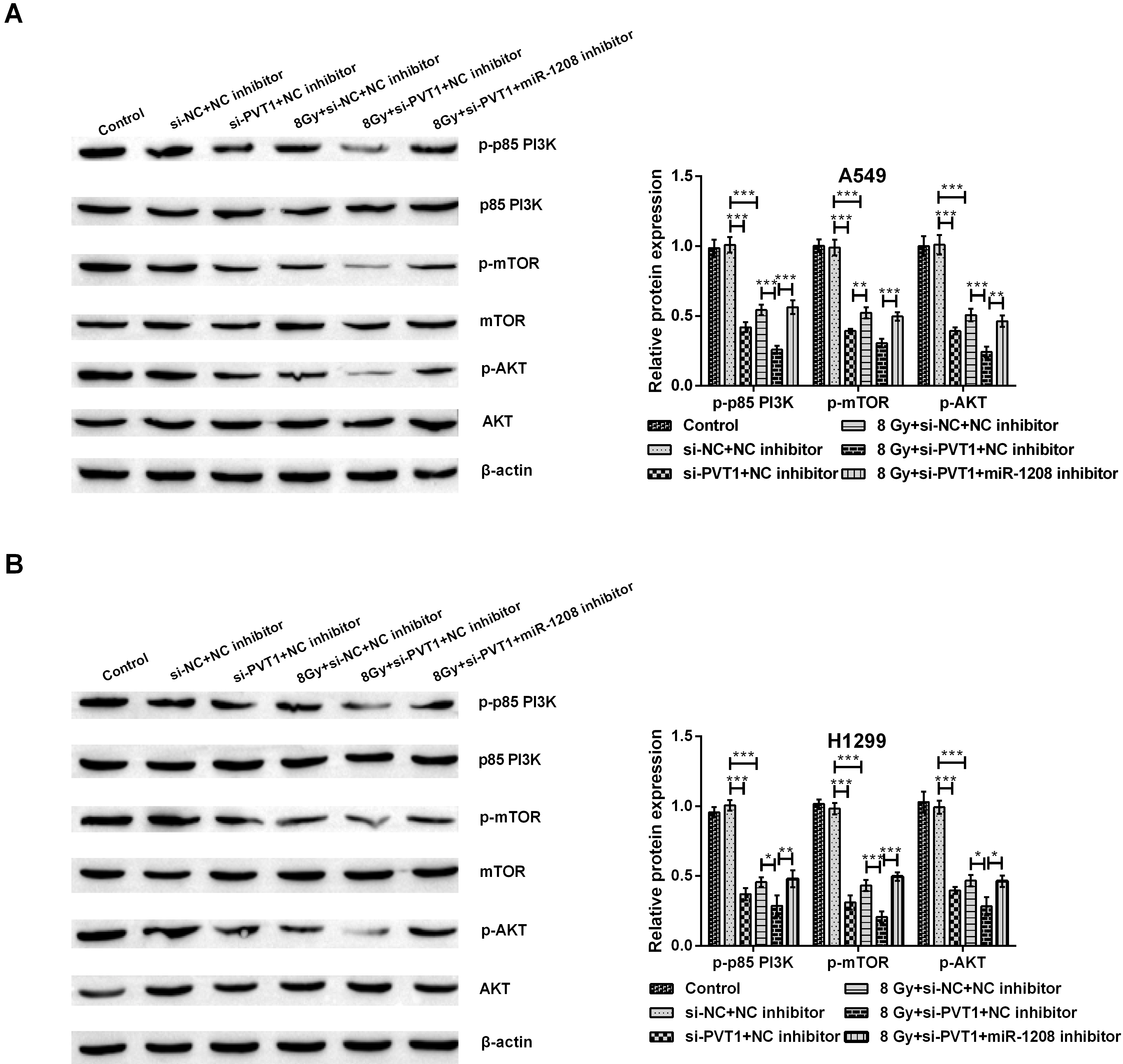
To further exploit the deep mechanisms of circPVT1 and miR-1208 on radiosensitivity of NSCLC cells, we focused on PI3K/AKT/mTOR signaling pathway. The results in Fig. 9A and B indicated that si-PVT1 transfection or 8 Gy radiation decreased the phosphorylation level of PI3K, mTOR and AKT (all
Discussion
Radiotherapy is one of the main methods to treat lung cancer in clinical practice. However, with the resistance of lung cancer cells to radiotherapy, the fact emerged that the survival rate of patients decreased and recurrence rate increased [34, 35]. This provides the impetus for us to look for ways to reduce the resistance of lung cancer to radiotherapy. So, our aim was to explore the possible mechanism of radiotherapy in NSCLC, thereby effectively improving the sensitivity of lung cancer cells to radiotherapy. In our study, we firstly found that circPVT1 was involved in radiosensitivity to NSCLC. Additionally, miR-1208 was observed to be a downstream target of circPVT1. Silencing circPVT1 could impede invasion and strengthen sensitivity of NSCLC to radiotherapy through up-regulating miR-1208. Furthermore, silencing circPVT1 also blocked PI3K/AKT/mTOR pathway through up-regulating miR-1208. This study laid the foundation for better clinical application of radiotherapy.
The improvement of radiosensitivity could availably inhibit cancer cell proliferation and motivate apoptosis, thereby enlarging the outcomes of radiotherapy [36, 37, 38]. CircRNAs are abnormally expressed in cancer cells [39]. It has been reported that circRNAs are involved in radiosensitivity in a mount of cancers. For example, circ_0000285 expression was abnormally increased in nasopharyngeal carcinoma (NPC) tissues with radioresistance, thus circ_0000285 acted as a biomarker for NPC and took part in NPC radiosensitivity [40]. Similarly, circ_0001313 was discovered to be up-regulated in colon cancer cells with low radiosensitivity, and knockdown of circ_0001313 improved radiosensitivity of colon cancer cells [41]. In addition, circMTDH.4 could reduce radioresistance and chemoresistance in NSCLC cells [42]. However, in our study, we discovered that circPVT1 was significantly elevated and silencing circPVT1 could heighten the sensitivity of lung cancer to radiotherapy and suppress invasion in NSCLC. We found a new circRNA associated with radiosensitivity in NSCLC. Our experimental results were similar to those of previous studies. CircPVT1 has been reported to be high-expressed and act as a proliferative factor in numerous cancers, such as NSCLC [24], gastric cancer [43], colorectal cancer (CRC) [44] and esophageal carcinoma [45]. Furthermore, in gastric cancer (GC), circPVT1 played an oncogenic effect, and downregulation of circPVT1 weakened chemotherapeutic resistance of paclitaxel [46]. In lung adenocarcinoma, overexpression of circPVT1 participated in resistance to chemotheraopy through miR-145-5p/ABCC1 axis [47]. Thus, circPVT1 may become a potential target of radiosensitivity enhancement in NSCLC.
In recent years, miRNAs have been identified to be involved in the regulation of radiosentivity in an increasing growing number of cancers, including NSCLC. For instance, miR-200a in NSCLC was low-expressed, and miR-200a could restrain invasion and enhance the sensitivity of NSCLC to radiotherapy through inhibition of HGF/cMet signal pathway [48]. Another article has reported that miR-9 expression levels were evidently downregulated in NSCLC cells, and miR-9 improved the radiosentivity to weaken NSCLC cell proliferative ability [49]. Our result was consistent with previous studied results. In NSCLC cells, miR-1208 was firstly recognized to be down-regulated. Moreover, we learnt about from previous researches that miRNAs acted as the sponge of circRNA and circRNA developed its regulatory effects on radiosentivity through certain miRNA. For example, circ_0073237 knockdown inhibited cancer development and enhanced radiosensitivity through sponging and increasing miR-1183 in glioma [50]. As well as, circMTDH.4 sponged miR-630 to inhibit radiosensitivity of NSCLC [42]. Through using circular RNA Interactome, miR-1208 was found to be a potential downstream target of circPVT1. Silencing circPVT1 enforced radiosensitivity and inhibited invasion in NSCLC.
Cancer process is closely related to a number of signal pathways, including PI3K/AKT/mTOR pathway [51]. Inhibiting PI3K/AKT/mTOR signaling pathway could effectively prevent NSCLC cell proliferation and initiate apoptosis [52, 53]. Furthermore, this signaling also took part in the regulation of radiosensitivity. For instance, in NSCLC, activating PI3K/AKT/mTOR signaling pathway affected radiosensitivity [54]. In addition, a study has shown that targeting PI3K/AKT/mTOR pathway become a new and potential mean of changing radiosensitivity of oral squamous cell carcinoma (OSCC) [55]. In this study, our experimental results revealed that silence of circPVT1 could block PI3K/AKT/mTOR pathway through up-regulating miR-1208. Up to now, although there were no articles reported that circPVT1 and miR-1208 could regulate PI3K/AKT/mTOR pathway, some miRNA was defined to modulate radiosensitivity through the regulation of PI3K/AKT/mTOR signaling pathway. For example, miR-21 could activate PI3K/AKT/mTOR signaling pathway, thereby affecting the sensitivity to radiotherapy in NSCLC cells [54]. Our results hinted that circPVT1 blocked PI3K/AKT/mTOR signaling pathway through up-regulating miR-1208, thereby improving NSCLC cell radiosensitivity.
In conclusion, this study demonstrated that inhibition of circPVT1 could impede NSCLC cell invasion and improve radiosensitivity through up-regulation of miR-1208, and this process was probably regulated by blocking PI3K/AKT/mTOR signaling pathway. These finding provided further insights into the mechanism of radiosensitivity of NSCLC.
Funding
This research was supported by Mechanism of Changes in Immune Status of High-incidence Lung Cancer in the Coal-burning Area of Eastern Yunnan on Comprehensive Treatment, Yunnan Provincial Department of Science and Technology-Kunming Medical University Joint Special General Project (No.2019FE001 (-106)), SPP1 Regulates Female Lung Cancer Cell Stem Resistance and its Mechanism of Drug Resistance in Xuanwei Area (No.2019J1313), Yunnan Provincial Department of Science and Technol-ogy-Kunming Medical University Applied Basic Research Joint Special Fund General Project and Project supported by the Ministry of Science and Technology: A Multi-Center Open Clinical Study of Stereotactic Body Radiotherapy Combined with Immune Checkpoint Inhibitors in the Treatment of Early Non-Small Cell Lung Cancer (No. 2018YFC1313202).
Author contributions
Conception: Meifang Huang, Tianqian Li and Bo Hou.
Interpretation or analysis of data: Qing Wang, Chongxin Li, Huahua Zhou and Bo Hou.
Preparation of the manuscript: Meifang Huang, Shengyi Deng and Zengbo Lv.
Revision for important intellectual content: Yongmei He, Guangying Zhu.
Supervision: Meifang Huang, Bo Hou and Guangying Zhu.
Footnotes
Acknowledgments
This research was supported by Mechanism of Changes in Immune Status of High-incidence Lung Cancer in the Coal-burning Area of Eastern Yunnan on Comprehensive Treatment, Yunnan Provincial Department of Science and Technology-Kunming Medical University Joint Special General Project (No.2019FE001 (-106)), SPP1 Regulates Female Lung Cancer Cell Stem Resistance and its Mechanism of Drug Resistance in Xuanwei Area (No. 2019J1313), Yunnan Provincial Department of Science and Technol-ogy-Kunming Medical University Applied Basic Research Joint Special Fund General Project and Project supported by the Ministry of Science and Technology: A Multi-Center Open Clinical Study of Stereotactic Body Radiotherapy Combined with Immune Checkpoint Inhibitors in the Treatment of Early Non-Small Cell Lung Cancer (No. 2018YFC1313202).
