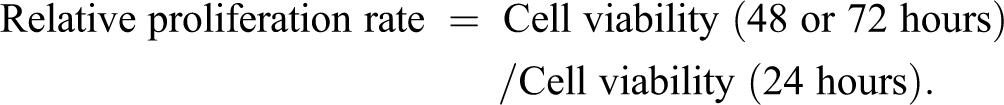Abstract
RNA-binding proteins have been associated with cancer development. The overexpression of a well-known RNA-binding protein, insulin-like growth factor 2 messenger RNA–binding protein 3, has been identified as an indicator of poor prognosis in patients with various types of cancer. Although gastric cancer is a relatively frequent and potentially fatal malignancy, the mechanism by which insulin-like growth factor 2 messenger RNA–binding protein 3 regulates the development of this cancer remains unclear. This study aimed to investigate the role and regulatory mechanism of insulin-like growth factor 2 messenger RNA–binding protein 3 in gastric cancer. An analysis of IGF2BP3 expression patterns reported in 4 public gastric cancer–related microarray data sets from the Gene Expression Omnibus and The Cancer Genome Atlas-Stomach Adenocarcinoma revealed strong expression of this gene in gastric cancer tissues. Insulin-like growth factor 2 messenger RNA–binding protein 3 expression in gastric cancer was further confirmed via quantitative reverse transcription polymerase chain reaction and immunohistochemistry, respectively, in an in-house gastric cancer cohort (n = 30), and the association of insulin-like growth factor 2 messenger RNA–binding protein 3 expression with clinical parameters and prognosis was analyzed. Notably, stronger IGF2BP3 expression significantly correlated with poor prognosis, and significant changes in insulin-like growth factor 2 messenger RNA–binding protein 3 expression were only confirmed in patients with advanced-stage gastric cancer in an independent cohort. The effects of insulin-like growth factor 2 messenger RNA–binding protein 3 on cell proliferation were confirmed through in vitro experiments involving the HGC-27 gastric cancer cell line. MicroR-125a-5p, a candidate microRNA that target on insulin-like growth factor 2 messenger RNA–binding protein 3, decreased in advanced-stage gastric cancer. Upregulation of microR-125a-5p inhibited insulin-like growth factor 2 messenger RNA–binding protein 3, and dual-luciferase report assay indicated that microR-125a-5p inhibited the translation of IGF2BP3 by directly targeting the 3′ untranslated region. These results indicate that the microR-125a-5p/insulin-like growth factor 2 messenger RNA–binding protein 3 axis contributes to the oncogenesis of advanced gastric cancer.
Introduction
Gastric cancer (GC) is among the most prevalent malignancies and causes of cancer-related death worldwide. Although patients with GC have benefited from advances in early diagnostic and therapeutic techniques, there have not been significant improvements in the overall survival of patients with advanced or relapsed disease. 1,2 The etiology of GC involves complex and heterogeneous genetic alterations that affect various oncogenic or tumor-suppressive signaling pathways. A better understanding of the risk factors contributing to each carcinogenic step and the molecular mechanisms underlying GC would facilitate the development of useful cancer prevention or therapeutic strategies. 3 -5
RNA-binding proteins (RBPs) act through versatile RNA-binding domains to play key roles in posttranscriptional events that mediate the localization, stability, alternative splicing, and translation of a large array of target transcripts. 6 Emerging evidence suggests that RBPs are associated with cancer development via intricate posttranscriptional regulatory networks. 7 Notably, previous studies have identified RBP dysregulation in the context of GC. For example, Liang et al demonstrated that polypyrimidine tract–binding protein 3, an essential RBP involved in RNA splicing, contributes to GC metastasis. 8 Wang et al reported that the expression of Lin28B, a RBP that directly binds the 3′ untranslated region (UTR) of NRP-1, was negatively correlated with the survival outcomes of patients with GC. Moreover, Lin28B was significantly overexpressed in GC cells compared with normal gastric epithelial cells. 9 Accordingly, an understanding of the network of interactions between cancer-related RBPs and their transcripts will enable a better understanding of GC biology and may potentially reveal new treatment targets.
Members of the insulin-like growth factor 2 messenger RNA (mRNA)–binding protein (IGF2BP) family, which includes IGF2BP1, IGF2BP2, and IGF2BP3, are the most frequently reported and experimentally validated human RBPs. 10 Photoactivatable ribonucleoside–enhanced crosslinking and immunoprecipitation and RIP studies indicated that IGF2BPs control the localization, translation, or turnover of numerous specific tumor-related mRNAs, such as CTNNB1, KRAS, PTEN, and MYC. 11 Hence, IGF2BPs play significant roles in cell polarization, proliferation, migration, metabolism, and differentiation in the contexts of various cancers. For instance, the upregulation of IGF2BP1 was reported to induce cell proliferation, migration, and invasion and to correlate with reduced survival in patients with hepatocellular carcinoma. 12 Moreover, IGF2BP2 overexpression was found to correlate with poor survival in patients with basal-like breast cancer and esophageal adenocarcinoma. 13 Similarly, variable IGF2BP2 expression may also be of prognostic value in GC. In a first-line combinational therapy study, patients exhibiting disease progression were less likely to harbor the IGF2BP2 polymorphisms rs4402960 and rs6769511, compared with patients with controlled disease. 14
Although the above findings are compelling, IGF2BP3 is the IGF2BP family member most frequently discussed in the context of cancer. Accumulating evidence has identified IGF2BP3 as a promising biomarker or therapeutic target in various cancers. For example, IGF2BP3 has been associated with adverse clinical features and outcomes of colorectal cancer, such as a poor differentiation status, advanced stage, and shorter survival duration. 15 In acute B-lymphoblastic leukemia, IGF2BP3 represents a critical pathogenetic mechanism via targeting oncogenic transcripts. 16 Zhou et al indicated the expression pattern of IGF2BP3 in GC; however, more details should be revealed by further studies. 17
In the present study, we investigated IGF2BP3 expression in GC tissues from different public GC-related cohorts and identified its correlation with patient survival using bioinformatics methods. We further confirmed the expression of IGF2BP3 and its association with clinical parameters in our in-house cohort of patients with GC. The function of IGF2BP3 in GC cells’ proliferation was revealed in vitro. MicroRNAs (miRNAs) have been demonstrated to play multifaceted roles of miRNAs in GC and regarded as novel biomarkers and therapeutic targets. 18 Previous reports showed that miRNAs modulate gastric carcinogenesis and development by regulating GC cell proliferation, cell cycle, apoptosis, invasion, migration, and metastasis through interacting with different molecular signaling pathways. 19,20 Hence, to explain how IGF2BP3 changed in GC, we tried to select candidate miRNAs that could target on IGF2BP3 in GC cells based considering the import role of miRNAs in GC. Finally, miRNA-125a-5p was selected as the interest candidated miRNA and confirmed by a series of in vitro experiments.
Materials and Methods
Bioinformatics Analysis of Public GC-Related Data Sets
Differentially expressed genes (DEGs) in GC tissues included in 4 GC-related Gene Expression Omnibus (GEO) microarray data sets (GSE13861, GSE27342, GSE54129, and GSE63089) were extracted using R software with the “Limma” package. After overlapping the results, a DEG was defined as a gene exhibiting a significant (P < .05) fold change of >2 or <0.5. Subsequently, changes in the expression of IGF2BP3 mRNA between tumor and nontumor tissues in each data set were estimated. IGF2BP3 expression patterns were also explored in The Cancer Genome Atlas-Stomach Adenocarcinoma (TCGA-STAD) database.
Subsequently, the clinical data of 593 and 359 patients (for overall survival and progression-free survival analysis, respectively) with GC included in the Kaplan-Meier plotter (KM plotter) database (http://kmplot.com/analysis/) were subjected to a survival analysis to investigate the role of IGF2BP3 in the prognosis of patients with GC. After dividing patients into high- and low-IGF2BP3 groups using the median of IGF2BP3 expression, the overall and progression-free survival rates were estimated using Kaplan-Meier curves and compared using the log-rank test. The miRwalk, miRcode, and miRanda/mirSVR programs were used to predict miRNAs that might target IGF2BP3, and changes in the miRNA profiles of GC tissues were obtained from TCGA-STAD using R software (version 3.2.5 ) with the “Limma” package.
Patient and Tissue Samples
Thirty GC and matched adjacent nontumor mucosal tissues (lateral distance of >5 cm from the edge of the cancerous region) were collected from patients with clinically and histopathologically confirmed disease who underwent surgical resection at the First Hospital of Lanzhou University. None of the patients had received preoperative antitumor therapies. All tissue samples were harvested and frozen in liquid nitrogen.
Immunohistochemistry
Tumor and nontumor mucosal tissues were fixed in 40 g/L formaldehyde and embedded in paraffin after conventional dehydration. Four-micrometer-thick sections of the tissues were prepared and deparaffinized. Next, immunohistochemical staining to detect IGF2BP3 protein expression in the tissues was performed using a streptavidin–peroxidase method kit (Beijing Solarbio Science and Technology Co, Ltd, Beijing, China) and a mouse monoclonal IGF2BP3-specific primary antibody (1:100 dilution; Santa Cruz Biotechnology, Inc, Dallas, Texas), according to the manufacturer’s protocol.
Real-Time Quantitative Polymerase Chain Reaction
Total RNA was isolated from tissue samples or cultured HGC-27 human GC cells using Trizol reagent (Thermo Fisher Scientific, Carlsbad, California). For detecting the mRNA level, samples of total RNA were then reverse transcribed to complementary DNA (cDNA) using RevertAid H Minus Reverse Transcriptase (Thermo Fisher Scientific) and amplified using the Maxima SYBR Green qPCR Master kit (Thermo Fisher Scientific) with the following primers: IGF2BP3, 5′-AGTTGTTGTCCCTCGTGACC-3′ (forward) and 5′-GTCCACTTTGCAGAGCCTTC-3′ (reverse); Glycerinaldehyd-3-phosphat-Dehydrogenase (GAPDH) (reference gene): 5′-AGGGCTGCTTTTAACTCTGGT-3′ (forward) and 5′-CCCCACTTGATTTTGGAGGGA-3′ (reverse). The following reaction conditions were used: 95°C for 5 minutes; 40 cycles of 95°C for 10 seconds, 60°C for 20 seconds, and 72°C for 20 seconds.
To detect miRNA, samples of total RNA were reverse transcribed into cDNA using the miRNA Reverse Transcription Kit: miScript II RT Kit (QIAGEN, Venlo, the Netherlands) and quantified using the miScript SYBR Green PCR Kit (QIAGEN) with the provided commercial miScript primers specific for miR-125a-5p and SNORD-48 (as reference). The upstream miScript primer sequence of miR-125a-5p was 5′-TCACAGGTTAAAGGGTCTCAGGGA-3′. The upstream miScript primer sequence of SNORD-48 was 5′-GCGACACACTCAGAGTTACCTGG-3′. The downstream primer was miScript Universal Primer. The following reaction conditions were used: 95°C for 15 minutes; 40 cycles of 95°C for 15 seconds, 55°C for 30 seconds, and 70°C for 30 seconds. All polymerase chain reactions were performed according to the manufacturer’s protocols. The 2−ΔΔCT method was used to calculate the relative levels of the target genes with respect to the references and controls.
Cell Culture and Transfection
HGC-27 human GC cells were obtained from the Shanghai Institute of Cell Biology, Chinese Academy of Sciences. The cells were grown in RPMI-1640 culture medium supplemented with 10% fetal bovine serum and penicillin–streptomycin solution in an incubator at 37°C and 5% CO2.
For IGF2BP3 knockdown experiments, 2 different IGF2BP3-specific siRNAs (IGF2BP3-siRNA-#1 and IGF2BP3-siRNA-#2, provided by GenePharma) were transfected into cells using Lipofectamine RNAiMAX Reagent (Thermo Fisher Scientific). The sequence of IGF2BP3-siRNA-#1 and IGF2BP3-siRNA-#2 was 5′-UCACGAUAUCUCCAUUGCAGGAAUU-3′ and 5′-AUACAUUUCAGAGAGCAUAAAGTA-3′, respectively.
For IGF2BP3 overexpression experiments, HGC-27 cells were transfected with pcDNA3.1-IGF2BP3 plasmids via Lipofectamine 2000 Reagent (Thermo Fisher Scientific). For miR-125a-5p overexpression experiments, miR-125a-5p mimics (QIAGEN) were transfected into cells using Lipofectamine RNAiMAX Reagent. The sequence of miR-125-5p was 5′-UCCCUGAGACCCUUUAACCUGUGA-3′. All transfection experiments were conducted according to the manufacturer’s protocols. The Allstar sequence was used as a negative control for the transfection of siRNAs and mimics, while empty vectors were used as controls for the transfection of plasmids.
Cell Proliferation Assay
At 48-hour posttransfection, HGC-27 cells were seeded into 96-well plates at a density of 5000 cells/well. After 24, 48, and 72 hours, cell viability was measured using the CellTiter-Blue kit (Beijing Promega Biotech Co, Ltd, Beijing, China). The relative cell proliferation rate in each group was calculated using the following formula:
Western Blotting
Total proteins were extracted from cells using radioimmunoprecipitation assay buffer supplemented with a protease inhibitor cocktail. A bicinchoninic acid assay was used to determine the concentrations of proteins in the samples. Accordingly, equal amounts of proteins were loaded onto to sodium dodecyl sulfate–polyacrylamide gels for electrophoretic separation, followed by transfer to nitrocellulose membranes. The membranes were blocked in phosphate-buffered saline with Tween-20 buffer containing 5% fat-free milk and incubated in a 1:200 dilution of a mouse monoclonal primary antibody specific for IGF2BP3 (Santa Cruz Biotechnology, Inc) or a 1:5000 dilution of a mouse monoclonal antibody specific for GAPDH (Santa Cruz Biotechnology, Inc) overnight at 4°C. The membranes were then washed and incubated in a 1:2000 dilution of a horseradish peroxidase–conjugated secondary antibody (Santa Cruz Biotechnology, Inc) for 2 hours at room temperature. Finally, the membranes were incubated with an enhanced chemiluminescence reagent (Beijing Applygen Technologies Inc, Beijing, China ) and visualized.
Dual-Luciferase Reporter Assay
Putative miR-125a-5p-binding sites in the wild-type IGF2BP3 3′ UTR sequence and corresponding mutant sequences were subcloned into pMIR-REPORT firefly luciferase reporter vectors (Ambion Inc, Foster City, California) and cotransfected into HGC-27 cells with control vectors containing renilla luciferase. Subsequently, the relative changes in luciferase activity (firefly/renilla) were detected using a dual-luciferase reporter system (Beijing Promega Biotech Co, Ltd).
Statistical Analysis
R software (3.3.2; R Project for Statistical Computing, Vienna, Austria) was used to conduct the statistical analysis of the experimental results in this study. Normally distributed data are presented as means ± standard deviations, and the t test was used to compare values between 2 groups. Non-normally distributed data are presented as median values and were compared using the Mann-Whitney U test. Differences in the clinicopathologic parameters were evaluated using the χ2 test. Survival outcomes were estimated using the Kaplan-Meier method and compared using the log-rank test. A P value of <.05 was considered to indicate statistical significance.
Results
Expression of IGF2BP3 Elevated in GC Tissues and Indicated Poor Prognosis
As few previous reports described the expression of IGF2BP3 in GC, we evaluated the IGF2BP3 expression patterns in 4 public GC-related microarray data sets (GSE13861, GSE27342, GSE54129, and GSE63089) retrieved from the GEO database (sample information was found in Supplementary Table 1). As shown in Figure 1, all 4 GEO cohorts revealed stronger IGF2BP3 expression in GC tissues, with an average fold change in expression of 2.59 in GC tissue relative to nontumor tissue (P < .05). Similarly, an analysis of a TCGA-STAD data set revealed stronger (fold change = 1.81) IGF2BP3 expression in GC tissues, compared with normal tissues (P < .05). Furthermore, we confirmed the association of IGF2BP3 expression with prognosis in a relatively large and mixed cohort from the KM plotter database. Among patients with GC, high IGF2BP3 expression was shown to correlate with lower overall and progression-free survival rates (Figure 2A and B, respectively). Taken together, these findings from database studies suggested that elevated IGF2BP3 expression should be considered a prognostic factor in patients with GC.

Significantly enhanced IGF2BP3 expression was detected in the analyses of 4 GEO gastric cancer–related microarrays and TCGA-STAD data sets. A, Data from GSE13861; (B) Data from GSE27342; (C) Data from GSE54129; (D) Data from GSE63089; (E) Data from TCGA-STAD. *P < .05, compared with normal tissues. GEO indicates Gene Expression Omnibus; IGF2BP3, insulin-like growth factor 2 mRNA-binding protein 3; mRNA, messenger RNA; TCGA-STAD, The Cancer Genome Atlas-Stomach Adenocarcinoma.

IGF2BP3 overexpression correlated with a poor prognosis in 876 patients with primary gastric cancer. A, Overall survival rates and (B) progression-free survival rates according to IGFBP3 expression level. IGF2BP3 indicates insulin-like growth factor 2 mRNA-binding protein 3; mRNA, messenger RNA.
Altered IGF2BP3 Expression Was Only Observed in Advanced GC and Correlated With Clinical Characteristics
We also used quantitative reverse transcription polymerase chain reaction and immunohistochemical staining (Figure 3A and B) to validate the expression patterns of IGF2BP3, respectively, in an independent GC-related cohort of patients in our center. Compared with matched control tissues, we observed only slight, nonsignificant increases in IGF2BP3 expression in malignant tissues from patients with early TNM stage (I + II) GC (P > .05). We found that IGF2BP3 expression significantly elevated in the tumor tissues of patients with advanced TNM stage (III + IV) GC.

Confirmation of elevated IGF2BP3 and IGF2BP3 expressions in an in-house cohort of patients with gastric cancer. A, qRT-PCR revealed a higher expression of IGF2BP3 mRNA expression in patients with advanced TNM stage. B, Representative image of immunohistochemical staining, which confirmed the positive association of IGF2BP3 with advanced TNM stage (original magnification, ×200). *P < .05, compared with normal tissues. IGF2BP3 indicates insulin-like growth factor 2 mRNA-binding protein 3; mRNA, messenger RNA; qRT-PCR, quantitative reverse transcription polymerase chain reaction.
We further grouped 30 patients from our GC cohort into high- and low-IGF2BP3 groups to estimate the associations between IGF2BP3 expression and clinicopathologic characteristics in patients. As summarized in Supplementary Table 2, stronger IGF2BP3 expression was significantly associated with a larger tumor size, increased risk of lymph node involvement, deeper invasion depth, and higher TNM stage.
Insulin-Like Growth Factor 2 mRNA-Binding Protein 3 Promoted GC Cell Proliferation and Was Regulated by MiR-125a-5p In Vitro
In HGC-27 cells, transfection with IGF2BP3-siRNA and pcDNA3.1-IGF2BP3 vectors led to the downregulation and upregulation of IGF2BP3, respectively. As shown in Figure 4, these outcomes respectively inhibited and enhanced the proliferation of HGC-27 cells, as expected.

IGF2BP3 affected the proliferation of HGC-27 human gastric cancer cells. A, IGF2BP3 knockdown suppressed HGC-27 cell proliferation. B, IGF2BP3 overexpression enhanced HGC-27 cell proliferation. *P < .05, compared with IGF2BP3 artificially changed cells. IGF2BP3 indicates insulin-like growth factor 2 mRNA-binding protein 3; mRNA, messenger RNA.
Next, we considered whether the expression of IGF2BP3 may be controlled by miRNAs and used online tools, including miRwalk, miRcode, and miRanda/mirSVR, to predict targeting miRNAs. Subsequently, we identified 6 miRNAs (miR-429, miR-508-3p, miR-4319, miR-761, miR-125a-5p, and miR-338-3p) with potential binding affinity for the 3′ UTR of IGF2BP3 mRNA (Figure 5A). An analysis of the TCGA-STAD data set revealed that only the expression of miR-125a-5p was decreased in GC tissues (Figure 5B and C), and in our cohort, only tissues collected from patients with advanced TNM stage GC exhibited significantly reduced miR-125a-5p expression (Figure 5D).

MicroRNA-125a-5p targeted IGF2BP3 in HGC-27 cells. A, The miRwalk, miRcode, and miRanda/mirSVR databases were used to identify putative IGF2BP3-targeting microRNAs. B and C, miR-338-3p and miR-125a-5p expressions in gastric cancer tissues from the TCGA-STAD data set. *P < .05, compared with normal tissues. D, The microRNA-125a-5p level in GC tissues negatively correlated with TNM stage in our in-house cohort. E, miR-125a-5p overexpression via a mimic inhibited IGF2BP3 mRNA expression (F) and reduced IGF2BP3 protein levels in HGC-27 cells. *P < .05, compared with negative transfection controls. G, miR-125a-5p was directly bound to the 3′ untranslated region of IGF2BP3 mRNA. *P < .05, compared with negative transfection controls. IGF2BP3 indicates insulin-like growth factor 2 mRNA-binding protein 3; mRNA, messenger RNA; TCGA-STAD, The Cancer Genome Atlas-Stomach Adenocarcinoma.
In vitro, we demonstrated that transfection with miR-125a-5p mimics significantly inhibited IGF2BP3 mRNA and protein expression in HGC-27 cells (Figure 5E and F). Moreover, a dual-luciferase reporter assay revealed that miR-125a-5p bound directly to the 3′ UTR of IGF2BP3 mRNA (Figure 5G). Finally, transfection with the miR-125a-5p mimic inhibited the proliferation in HGC-27 cells in vitro (Figure 6).

MicroR-125a-5p (miR-125a-5p) overexpression via mimics inhibited the proliferation of HGC-27 gastric cancer cells. *P < .05, compared with cells transfected with miR-125a-5p mimic.
Discussion
Currently, GC is considered the third leading cause of cancer-related mortality worldwide, 21,22 and the poor survival prognosis of this disease is associated with frequent recurrence, metastasis, and chemoresistance. 23 Although screening programs have been established in most regions, patients are frequently diagnosed with advanced-stage disease. Accordingly, the improvement in prognosis via therapeutically relevant targets is urgently needed.
RBPs have recently emerged as potential cancer therapeutic targets. Particularly, RBPs may affect both the expression and function of oncogenic/tumor-suppressor genes. Previous studies suggest that the expression patterns of RBPs are dysregulated in different types of cancer, which affects the exhibition of various malignant traits. 24 Accordingly, further studies about the role of RBPs in cancers, and particularly GC, are warranted.
As mentioned in the Introduction, IGF2BP3 has been reported to play a key role in tumorigenesis of various carcinomas. 25 Most existing evidence suggests that the overexpression of IGF2BP3 in tumor tissues may serve as a promising early diagnostic and prognostic biomarker. For example, in patients with breast carcinoma, increased IGF2BP3 expression was associated with a poor prognosis. 26 Similarly, Burdelski and colleagues conducted a tissue microarray study of 8877 human cancers and normal tissues and reported that IGF2BP3 overexpression is associated with aggressive tumor features in various types of cancer. 27 Moreover, Wei et al demonstrated that increased IGF2BP3 expression is a reliable marker for the diagnosis of colorectal cancer in endoscopic biopsies, 28 while an elevated level of IGF2BP3 in lung adenocarcinoma enhanced the invasive potential and predicted a poor prognosis. 29 In addition to determining the expression of IGF2BP3 in various cancers, previous studies revealed that this protein might play different roles according to tumor type. For example, IGF2BP3 promoted the growth of glioblastoma by enhancing the production of IGF2. In another study, IGF2BP3 enhanced the proliferation of various types of cancer cells (RD embryonic rhabdomyosarcoma, Hey-A8 human ovarian carcinoma, Cal27 epithelial tongue squamous carcinoma, MCF7 human breast cancer, and HeLa uterine cervical carcinoma cell lines) by interacting synergistically with heterogeneous nuclear ribonucleoprotein M (hnRNPM) in the nucleus to increase the expression of cyclins. 30 In breast cancer, Samanta et al demonstrated that IGF2BP3 stabilized the mRNA encoding an alternative WNT ligand and thus facilitated the activation of TAZ. 31 However, only Zhou et al had previously described the changes in IGF2BP3 expression in GC. 17
To address this gap in the research evidence, we initially investigated the expression patterns of IGF2BP3 in cases of GC included in public GEO databases. To avoid bias, we selected 4 GC-related cohorts comprising a total of 301 tumor tissues and 165 control tissues from different regions and found that significant IGF2BP3 overexpression was observed consistently in tumor tissues (relative to adjacent controls) from all 4 GEO data sets. We also analyzed changes in the mRNA expression profiles of GC tissues included in TCGA-STAD and again observed higher IGF2BP3 mRNA expression relative to normal tissues. These expression patterns suggest a potential role for IGF2BP3 in GC. In the abovementioned data set analysis, IGF2BP3 expression in gastric tumor tissues compared with gastric normal tissue adjacent to the tumor (gNAT). Nevertheless, recently published evidence showed difference between gNAT and real normal gastric mucosa. 32 Gastric normal tissue adjacent to the tumor showed unique gene signature and biological features. Hence, using adjacent normal tissues as healthy control tissue in the characterization of tumor-associated biological processes and pathways could lead to suboptimal results such as false positive and negative. 33
Consistent with this speculation and the findings of Zhou et al, our analysis of patients with GC from the KM plotter database revealed an inverse correlation between IGF2BP3 expression and survival. In a cohort of patients with GC treated at our center, we further identified significant positive associations of IGF2BP3 expression with various characteristics associated with tumor aggressiveness, tumor size, lymph node involvement, invasion depth, and TNM stage. More interestingly, significantly enhanced IGF2BP3 expression was only observed in tissues from patients with advanced-stage GC, suggesting that this RBP plays an oncogenic role in advanced disease. We further conducted in vitro IGF2BP3 expression assays to demonstrate that the artificial overexpression and knockdown of IGF2BP3 could significantly promote and inhibit GC cell proliferation, respectively. Interestingly, however, these changes did not appear to affect other aspects of malignancy, such as cell migration, invasion, and clonality (data not shown).
We additionally explored the mechanism underlying the altered expression of IGF2BP3 in GC cells, particularly the potential regulatory effects of miRNAs. Previous studies demonstrated that miRNA-129-1 inhibited IGF2BP3 and induced cell cycle arrest in glioblastoma, 34 while miRNA-1275 targeted IGF2BP1, 2, and 3 and hindered hepatocellular carcinoma tumor growth. 35 Accordingly, we predicted 6 candidate miRNAs for further investigation. However, only the expression of miR-125a-5p was decreased in GC tissues from the TCGA-STAD data set. In previous reports, miR-125a-5p was shown to inhibit GC cell invasion and metastasis. 36 Moreover, Nishida et al showed that miR-125a-5p expression served as an independent prognostic factor in patients with GC and enhanced the ability of trastuzumab to inhibit cancer cell proliferation. 37 Consistent with these findings, we observed decreased miR-125a-5p expression only in tumor tissues from patients with advanced-stage GC in our cohort. Our in vitro experiments revealed that the overexpression of miR-125a-5p inhibited IGF2BP3 expression in HGC-27 cells. As expected, miR-125a-5p overexpression also inhibited HGC-27 cell proliferation. These findings, together with the results from a dual-luciferase reporter assay, suggest that insufficient miR-125a-5p expression might enhance the overexpression of IGF2BP3 in advanced-stage GC.
Our study was limited by our inability to explain how IGF2BP3 functionally affects the proliferation of GC cells and by the lack of investigation of other potential phenotypic functions of IGF2BP3 in these tumor cells. In conclusion, our evidence supports a role for IGF2BP3 in the tumorigenesis of GC. These mechanistic data may facilitate a better understanding of the pathogenesis of GC and enable the development of potential therapeutic strategies.
Supplemental Material
Supplementary_Table_1_(1) - The Aberrant Expression of MicroRNA-125a-5p/IGF2BP3 Axis in Advanced Gastric Cancer and Its Clinical Relevance
Supplementary_Table_1_(1) for The Aberrant Expression of MicroRNA-125a-5p/IGF2BP3 Axis in Advanced Gastric Cancer and Its Clinical Relevance by Jing Zhang, Fanghui Ding, Dan Jiao, Qiaozhi Li and Hong Ma in Technology in Cancer Research & Treatment
Supplemental Material
Supplementary_Table_2 - The Aberrant Expression of MicroRNA-125a-5p/IGF2BP3 Axis in Advanced Gastric Cancer and Its Clinical Relevance
Supplementary_Table_2 for The Aberrant Expression of MicroRNA-125a-5p/IGF2BP3 Axis in Advanced Gastric Cancer and Its Clinical Relevance by Jing Zhang, Fanghui Ding, Dan Jiao, Qiaozhi Li and Hong Ma in Technology in Cancer Research & Treatment
Footnotes
Authors’ Note
F.D. contributed equally to this work as the first author. This study was approved by the Ethical Committee of First hospital of Lanzhou University (approval no. LDYY2016038). Written informed consent was obtained from the patient or their family.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by internal departmental research funding provided by the Department of Pathology, the First Gospital of Lanzhou University.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.