Abstract
PURPOSE:
MicroRNAs (miRNAs) have been reported to be involved in tumorigenesis. The aim of this study was to investigate the functional role and prognostic value of miR-652 in gastric cancer (GC).
METHODS:
Quantitative real-time polymerase chain reaction (qRT-PCR) was used to determine the expression levels of miR-652 in human GC tissue samples and GC cell lines. The Kaplan-Meier survival curves and Cox regression analysis were performed to measure the prognostic value of miR-652 in GC. The tumor cell proliferation capacity was estimated by MTT assay, and cell migration and invasion were assessed by Transwell assays. The luciferase reporter assay was performed to confirm the target gene of miR-652.
RESULTS:
MiR-652 was significantly elevated in GC tissues and cell lines (all
CONCLUSION:
MiR-652 functions as an oncogene in GC and promotes tumor progression via targeting RORA. MiR-652 might be a novel predictive marker for the poor prognosis of GC patients.
Introduction
Gastric cancer (GC) is regarded to be one of the most common human cancer, originating from the mucosal epithelium of the stomach [1]. In China, GC affects the quality of patients’ life severely, with high rate of morbidity and mortality [2]. Recently, the number of young patients suffering from GC has a rising tendency [3]. Although the overall incidence of GC declines during the past decades, it remains one of the most common types of cancer and the major causes of cancer-related deaths worldwide [4]. A great progress in the clinical treatment of GC has been made in recent years, but the overall prognosis is always discouraging as a result of postoperative recurrence and metastasis [5]. Thus, researches to investigate the underlying mechanisms of the GC development and metastasis are still of great significance for its clinical treatment [6].
MicroRNAs (miRNAs) is a class of short non-coding RNAs, with the length of approximately 20–24 bases. It can inhibit the translation of the target genes via binding with its 3’-untranslated region (UTR) [7]. An increasing number of miRNAs have been identified to be aberrantly expressed in various types of human cancers, which plays a significant role in tumor formation and progression [8, 9, 10, 11]. In human GC, dysregulation of miRNAs has been reported to be involved in tumor occurrence and development. For instance, Ge et al. found that miR-181a was overexpressed in GC tissues compared to adjacent noncancerous tissues, the upregulation of miR-181a and consequent inhibition of TGF
In the present study, we examined the expression of miR-652 in 135 pairs of GC tissues and matched adjacent non-tumorous tissues. Besides, we further investigated its relationship with clinicopathological features and survival of GC patients. Furthermore, cellular functional experiments were also carried out to calculate the effect that miR-652 had on GC behaviors.
Materials and methods
Patients and sample collection
A total of 135 gastric patients were recruited in this study, who were underwent resection of the primary tumor between June 2008 and March 2011 at Harbin Medical University. These fresh GC tissues and corresponding adjacent normal tissues were immediately snap-frozen in liquid nitrogen and stored at
Cell culture and transfection
The normal gastric epithelium cell line GES-1 and four GC cell lines (SGC-7901, HGC-27, MKN45 and BGC-823) were obtained from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All cells were cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS). All cell cultures were performed in a humidified incubator with 5% CO
To regulate the expression of miR-652, cells were transfected with miR-652 mimic, miR-652 inhibitor or the corresponding negative controls (mimic NC and inhibitor NC), which were synthesized by the company of Gene-Pharma (Shanghai, China). MiR-652 mimic was used to enhance the activity of miR-652, while miR-652 inhibitor was used to inhibit the activity. Mimic NC and inhibitor NC was chemically modified nonsense single-stranded nucleic acids, which have no effect on miR-652 expression after transfected into cells. All the cells were seeded into 24-well plates 24 h before transfection, and the followed transfections were conducted using Lipofectamine 3000 reagent (Invitrogen, USA) according to the manufacturer’s protocols. A total of 48 h after transfection, cells were harvested for the follow-up experiment.
Expression of miR-652 measured by qRT-PCR in GC tissues and cell lines. A. MiR-652 expression was upregulated in the GC tissues compared with the adjacent normal controls (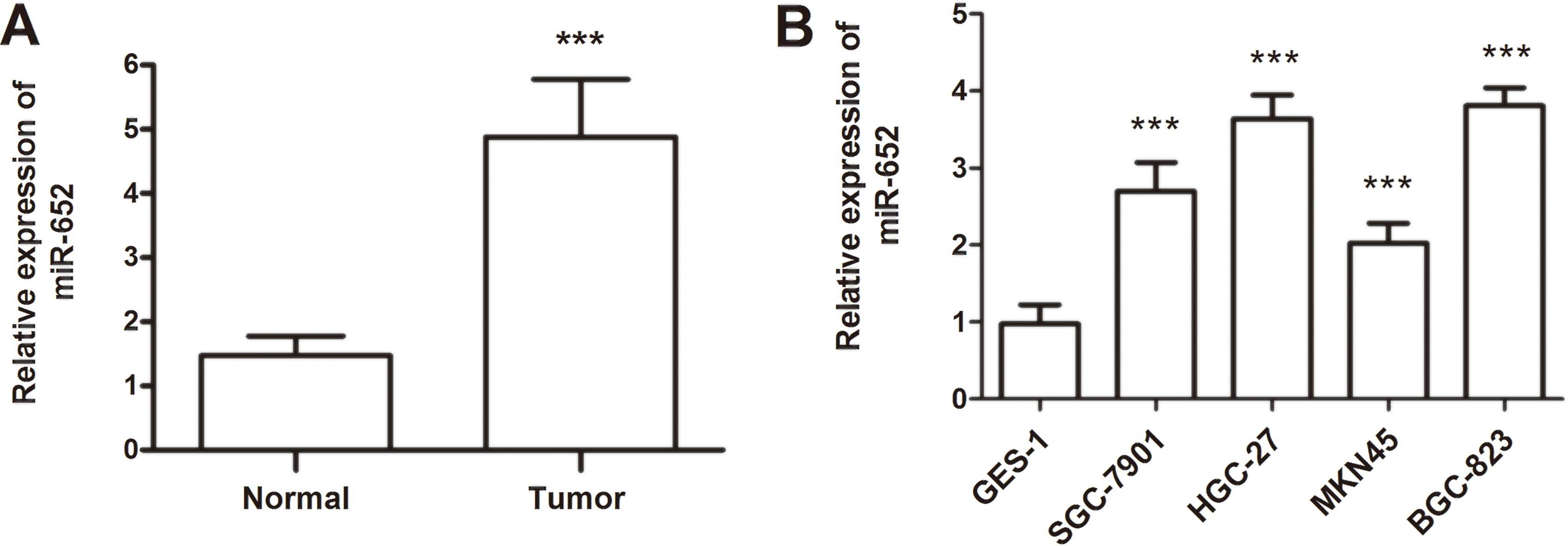
The total RNA was extracted from both GC tissues and cells using Trizol Reagent (Invitrogen, Carlsbad, CA, USA) based on the manufacturer’s protocol. Reverse transcription reactions were performed using the miScript Reverse Transcription Kit (QIAGEN, Germany), and real-time PCR was performed with SYBR green I Master Mix kit (Invitrogen, Carlsbad, CA, USA) using 7300 Real-Time PCR System (Applied Biosystems, USA). The comparative delta CT (2
MTT assay
MTT assay was conducted to measure the cellular proliferation of stably transfected cells. A total of 48 h after transfection, the stably transfected cells were plated in a 96-well plate with the density of 5
Transwell assay
Transwell migration and invasion assays were conducted to evaluate the migration and invasive ability of GC cells. Transwell inserts chambers with 8
Luciferase reporter assay
PmiR-RB-REPORT
Statistical analysis
SPSS version 18.0 software (SPSS Inc., Chicago, IL) and GraphPad Prism 5.0 software (GraphPad Software, Inc., USA) were applied for the data analysis. Differences between two groups were compared by Student’s
Kaplan-Meier survival analysis for the GC patients based on the expression of miR-652. Patients with high miR-652 expression had shorter survival time than those with low miR-652 expression (log-rank 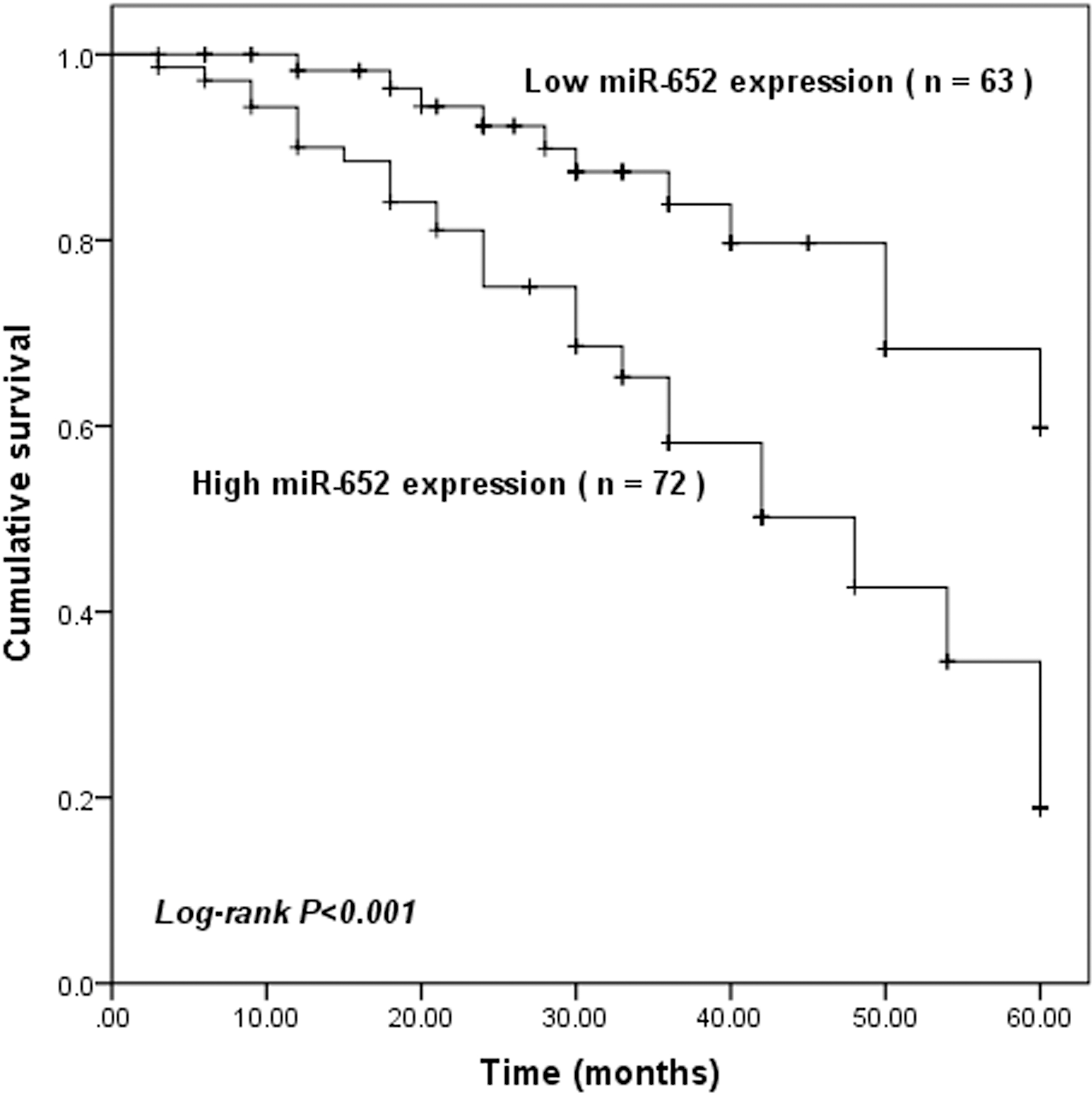
Overexpression of miR-652 in GC tissues and cell lines
The miR-652 expression level was estimated in pairs of tumor tissues and corresponding adjacent normal tissues. The results suggested that miR-652 was mostly up-regulated in the GC tissues compared with matched normal tissues, and the differences reached a significant level (
Effects of miR-652 on cell proliferation in HGC-27 and BGC-823 cells. A. In the two cell lines, expression of miR-652 was significantly increased by miR-652 mimic transfection, but was decreased by miR-652 inhibitor transfection compared with the corresponding negative controls (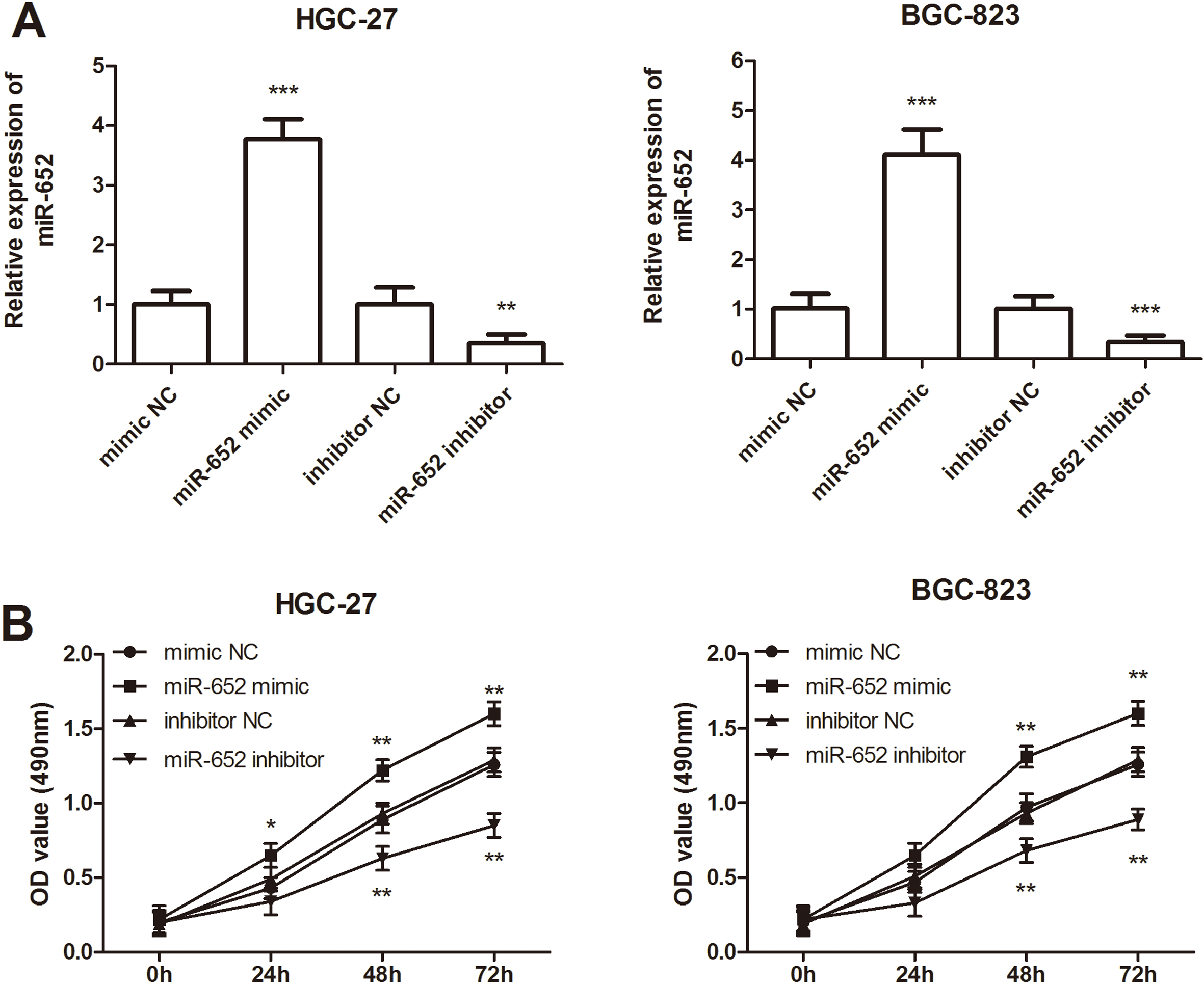
Effects of miR-652 on cell migration and invasion in HGC-27 and BGC-823 cells. A. The results of migration analysis for HGC-27 and BGC-823 cells. B. The results of invasion analysis for HGC-27 and BGC-823 cells. C and D. Overexpression of miR-652 by miR-652 mimic transfection could promote the cell migration and invasion, but the downregulated miR-652 expression could inhibit the cell migration and invasion (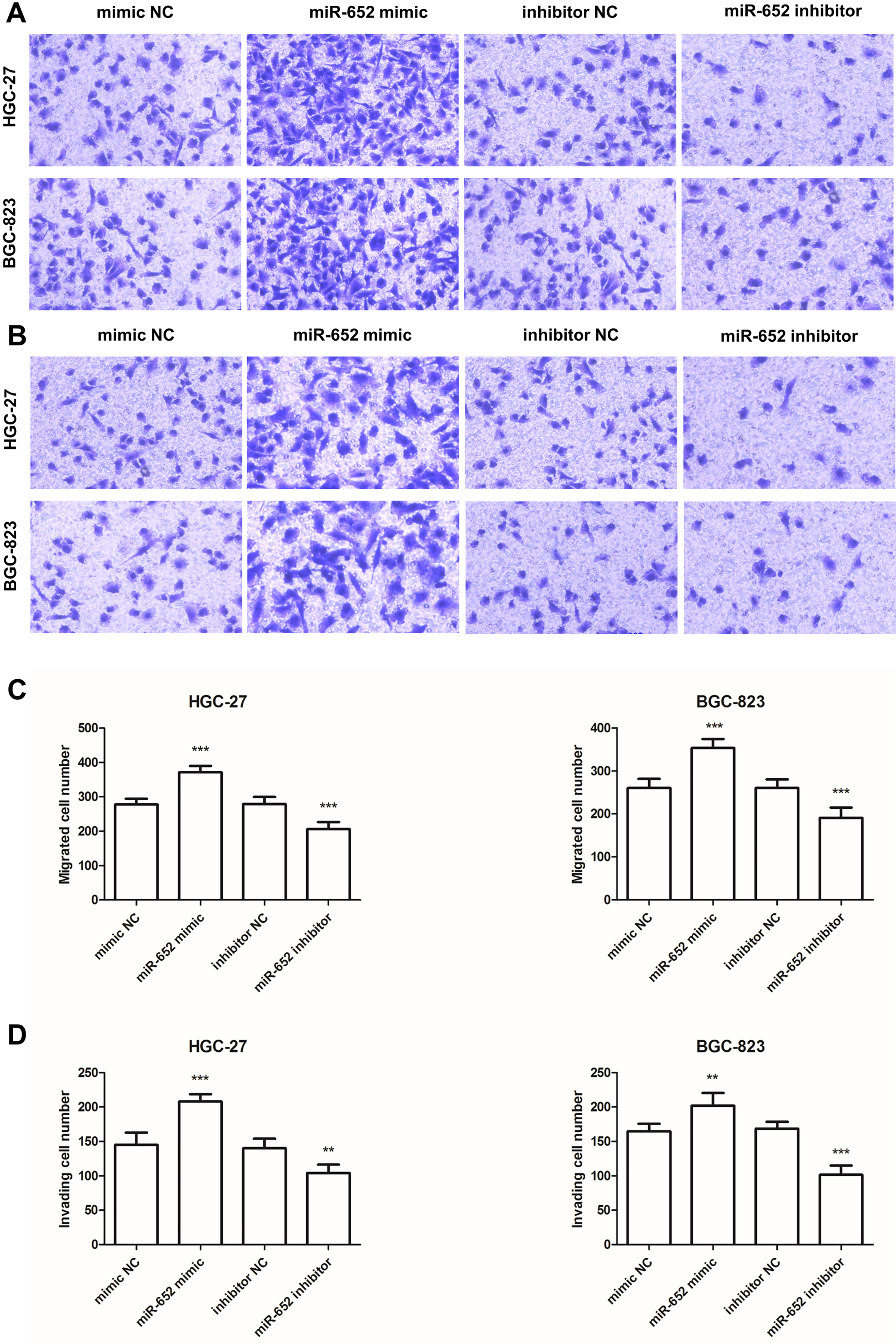
MiR-652 directly targets the conserved RORA 3’-UTR sequence. A. The putative miR-652 binding sequence in the RORA 3’-UTR. B. Analysis of the luciferase activity of the luciferase reporter plasmid containing either wild-type (WT) or mutant (MT) RORA 3’-UTR in BGC-823 cells (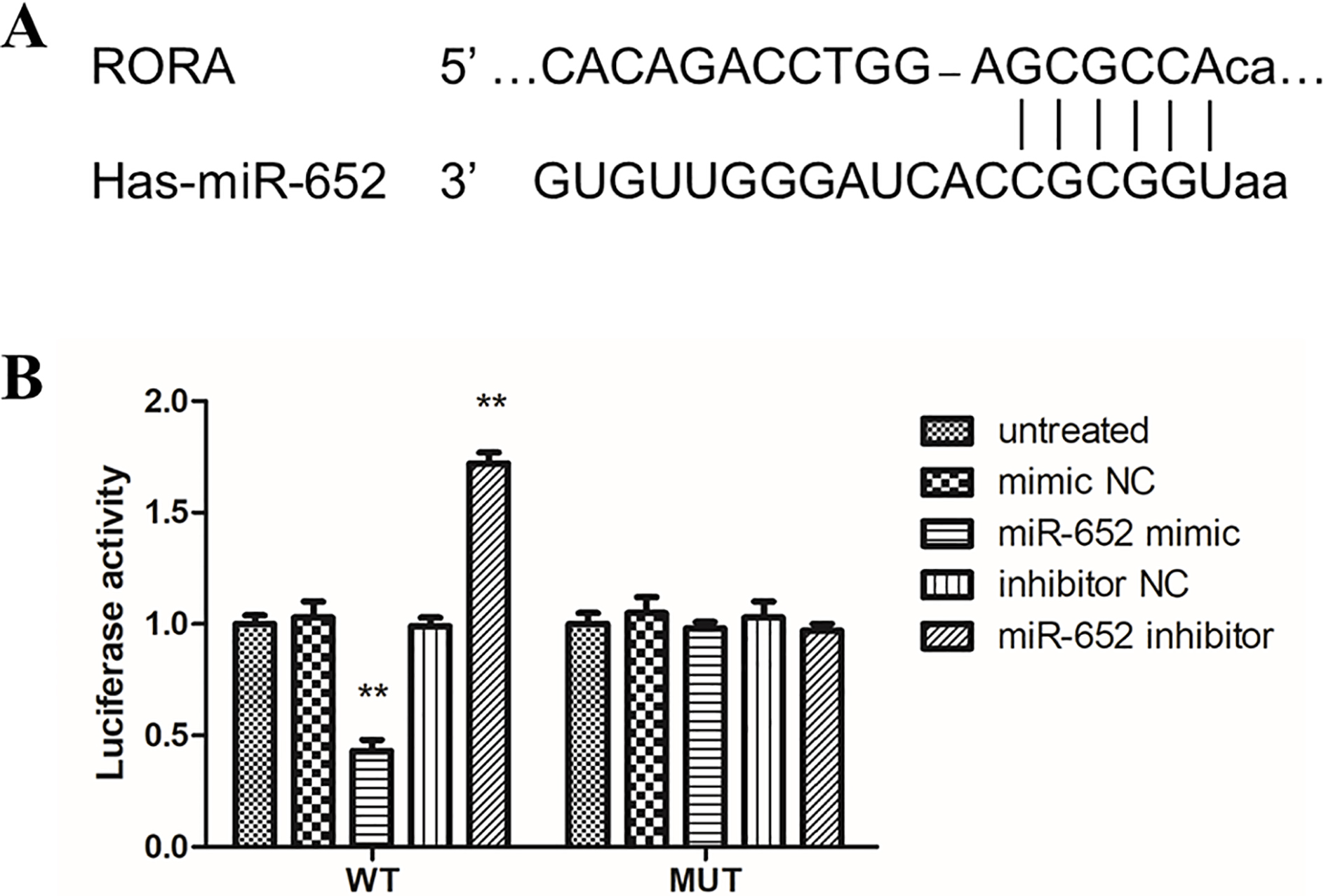
Association of miR-652 with the clinicopathological features of gastric cancer patients
Multivariate Cox regression analysis for miR-652 in gastric cancer patients
The clinical data of the 135 patients were presented in Table 1. The mean expression level of miR-652 in GC tissues was defined as a cutoff to divide the patients into low (
Prognostic value of miR-652 expression for patients with GC
The Kaplan-Meier survival curves and the log-rank test were performed to explore the correlation between miR-652 expression and patients’ survival. We found that patients with high miR-652 expression had significantly lower 5-year overall survival than those with low miR-652 expression (log-rank
Effects of miR-652 on cell proliferation in GC cells
Because the expression level of miR-652 was upregulated to a greater degree in HGC-27 and BGC-823 cell lines, the two cells were used for the subsequent studies. To further evaluate the functional effects of miR-652 on GC cell behaviors, cellular functional experiments were performed by transfecting miR-652 mimics, miR-652 inhibitor or corresponding negative controls into cells. As shown in Fig. 3A, miR-652 expression levels were significantly upregulated in the miR-652 mimic group compared with the mimic NC group, while were downregulated significantly in the miR-652 inhibitor group (all
Effects of miR-652 on cell migration and invasion in GC cells
The transwell assay was also conducted to calculate the involvement of miR-652 in GC cell migration and invasion. The data revealed that, in both HGC-27 and BGC-823 cells, the migrated and invasive cell numbers were increased significantly by miR-652 mimic transfection, while were decreased by miR-652 inhibitor transfection (all
RORA was the target gene of miR-652
According to bioinformatics analysis by online software (TargetScan, miRNAMap and miRDB), RORA was identified to be a potential target of miR-652 (Fig. 5A). Furthermore, the luciferase reporter assay suggested that miR-652 mimic transfection attenuated the luciferase activity of RORA 3’-UTR, but it was not observed when the mutant RORA 3’-UTR expressed. On the contrary, the inhibition of miR-652 significantly promoted RORA 3’-UTR luciferase activity which was not detected under the mutant RORA 3’-UTR expression (
Discussion
Although the incidence of GC shows decrease trend recently as a result of the people’s habits change and the smoking reduction, its survival rate is still modest [17, 18]. Up till now, the etiology and pathogenesis of GC have not been fully understood [19]. GC patients always have no characteristic manifestations in the early stage and can not accept treatment timely, which affects the prognosis of GC patients seriously [20]. Therefore, identifying new molecular markers that can be used as independent prognostic factors for GC is of great significance for the diagnosis and targeted treatment of GC [21]. A number of miRNAs have been reported to express aberrantly in different cancers, and be involved in many processes, such as cell proliferation, apoptosis, and stress response [22, 23, 24, 25]. Recently, a major study has suggested that miR-652 was overexpressed in GC patients than healthy controls [16]. But the function of miR-652 in tumorigenesis and progression of GC are still largely unknown.
In the present study, the expression level of miR-652 was examined in 135 GC patients, and the results revealed that the expression of miR-652 was upregulated significantly in GC tissues, which was further confirmed in GC cell lines. The results were supported by the previous results [16]. Consistently, overexpression of miR-652 has been reported in human prostate cancer cells, which might serve as a biomarker for aggressive prostate cancer [26]. Besides, the expression of miR-652-3p was also found to be significantly upregulated in non-small cell lung cancer (NSCLC) tissues, and showed significant association with lymph node metastasis, TNM stage and poor prognosis [27]. Sun et al. demonstrated that miR-652 was overexpressed in endometrial cancer, which was associated with poor survival rate and early recurrence [28]. All results suggested the carcinogenesis of miR-652 in different human cancers.
The Kaplan-Meier survival curves and log-rank test also demonstrated that patients with high miR-652 expression had significantly lower 5-year overall survival than those with low miR-652 expression. The multivariate Cox analysis further suggested the miR-652 expression and TNM stage to be independent prognostic indicators for overall survival in GC patients. Taken together, these results indicated that miR-652 might be a novel predictive marker for the poor prognosis of GC patients and might play an important role in the tumor progress.
Furthermore, the functional effects of miR-652 on GC cell behaviors were also detected, and overexpression of miR-652 was proved to significantly promote GC cell proliferation, migration and invasion. About the function effects of miR-652 on tumor cell progression, a number of studies have been reported in different human cancers. Nam et al. found that up-regulation of miR-652 enhanced prostate cancer cell growth, migration and invasion by promoting NED through decreased PP2A function [26]. Yang et al. indicated that overexpressing miR-652 resulted in decreased expression of Lgl1 protein, and can promote cell proliferation, migration, invasion and inhibit cell apoptosis [27]. Sun et al. revealed that miR-652 might function as an oncogene in endometrial cancer, and overexpression of miR-652 would promote the tumor progression [28]. All evidence suggested the crucial role of miR-652 in tumor progression.
RORA is a nuclear receptor which belongs to the ROR sub-family [29]. In the present study, RORA was predicted to be a target gene of miR-652 in GC cells according to bioinformatics analysis, which has been widely reported to be involved in cancer progression, including GC [30, 31, 32]. Furthermore, the luciferase reporter assay was performed to confirm RORA to be the target gene of miR-652 in GC. RORA has been reported to be downregulated in human GC tissues, as well as GC cell lines [32]. And the reduction of RORA was reported to be associated with tumor progression and poor prognosis of GC [32]. Combined with the previous evidence, we concluded that miR-652 functions as an oncogene in GC and promotes tumor progression via targeting RORA.
In conclusion, we confirmed that miR-652 expression was increased in GC tissues, as well as GC cell lines. To the best of our knowledge, this is the first study exploring the correlation between altered miR-652 and prognosis in GC patients, and miR-652 was demonstrated to be correlated with clinical outcomes. Moreover, miR-652 was able to promote the tumor progression of GC cells via targeting RORA. Therefore, the present study highlighted that miR-652 might be a useful therapeutic target to improve the survival of GC patients.
Footnotes
Conflict of interest
The authors report no conflicts of interest in this work.
