Abstract
Aberrant expression of miR-181d has been noted in multiple human cancers, but its role in gastric cancer (GC) remains unclear. The aim of this study was to investigate the expression, clinical significance and functional role of miR-181d in GC. We applied quantitative real-time polymerase chain reaction (qRT-PCR) to quantify the expression of miR-181d in 131 GC tissues, as well as in GC cell lines. The correlation of miR-181d expression with overall survival of GC patients was analyzed using the Kaplan-Meier survival method. Cox regression analysis was conducted to further determine the prognostic value of miR-181d in GC. Cellular functional experiments were carried out to calculate the effect that miR-181d had on GC behaviors. MiR-181d expression was significantly up-regulated in both GC tissues and cells (all
Introduction
Gastric cancer (GC) is one of the most common type of digestive tract cancers, with high mortality worldwide [1]. Various risk factor have been reported to be associated with GC occurrence, including unhealthy eating habits, smoking, alcohol, as well as virus and bacteria [2]. Up to now, comprehensive treatment in combination with surgery, chemotherapy and radiotherapy remains the most common, as well as effective therapy for GC patients [3]. With the improvements of GC treatment and diagnosis, the overall survival and prognosis remains not so optimistic [4]. The traditional drug therapy for tumors generally means chemotherapy, which always leads to great toxicity to patients and attracts board attention [5]. Recent years, strategies for molecular research becomes a research hotspot, because it has limited and less side effects on normal cells of patients [6]. Thus, it has great significance to explore novel and effective therapeutic targets for GC.
It is well known that microRNAs (miRNAs) are small non-coding RNAs, and serve the function of regulating gene expression at the post-transcription level by binding to the target mRNAs [7, 8]. Previous researches have reported that miRNAs are expressed differentially in various types of cancers [9]. Furthermore, it is also demonstrated that miRNA take participant in various cell procedures such as cell growth, cell cycle, as well as cell development [10]. Recently, the role of miRNAs in GC initiation, development, even progression has aroused more and more attention among scholars. Assumpção et al. conducted a high-throughput sequencing of miRnome of four GC samples, and a number of aberrantly expressed miRNAs have been identified in GC tumor tissues, such as miR-3131, miR-664, miR-150 and so on [11]. Additionally, several miRNAs have been reported to be involved in the tumor progression of GC. For example, Tian et al found that miR-505 was low expressed in GC cells and involved in the development of GC, and HMGB1 was predicted to be a direct target of miR-505 [12]. Guo et al. showed that miR-371-3p was up-regulated in GC tissues which was correlated with aggravation of the GC patients. Besides, the functional assays further proved that miR-371-3p promoted the GC cell proliferation, migration and invasion, suggesting its tumor suppressor role in GC [13]. Notably, miR-181d has been reported to be upregulated in GC tissues compared to normal gastric mucosa [14], but the prognostic value and role of miR-181d in GC remains unknown.
In the present study, the expression patterns and role of miR-181d was validated in GC tissues as well as GC cell lines. Additionally, the clinical and prognostic value of miR-181d in GC were also analyzed.
Materials and methods
Patients and sample collection
Fresh GC tissues and corresponding adjacent normal tissues were obtained form Shengli Oilfield Central Hospital. The adjacent normal tissues were at a distance of more than 5 cm from tumor margin were collected, with the size of about 1.0 cm
Cell culture and transfection
Human GC cell lines (NCI-N87, AGS, and MGC-803) and the normal gastric mucosa cell GES-1 were acquired from the Cell Bank of the Chinese Academy of Sciences (Shanghai, China). All cell lines were routinely grown in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS) and incubated at 37
The miR-181d mimic, miR-181d inhibitor and the corresponding negative controls of mimic and inhibitor (mimic NC and inhibitor NC) were synthesized and purified by Gene-Pharma (Shanghai, China). Then the GC cell lines were seeded into 6-well plates, and divided into four groups, including miR-181d mimic NC group (mimic NC group), miR-181d mimic group, miR-181d inhibitor NC group (inhibitor NC group), and miR-181d inhibitor group, which were following transfected with miR-181d mimic NC, miR-181d mimic, miR-181d inhibitor NC or miR-181d inhibitor respectively by using Lipofectamine 3000 reagent (Invitrogen, USA) following the manufacturer’s protocols. A total of 48 h after transfection, samples were collected for further experiments.
RNA extraction and quantitative real-time polymerase chain reaction (qRT-PCR)
Trizol Reagent (Invitrogen, Carlsbad, CA, USA) was applied for the total RNA extraction from GC tissues and the cultured GC cell lines according to the manufacture’s protocol. RNA was reverse transcribed into cDNA using the miScript Reverse Transcription Kit (QIAGEN, Germany). qRT-PCR was performed using SYBR green I Master Mix kit (Invitrogen, Carlsbad, CA, USA) and 7300 Real-Time PCR System (Applied Biosystems, USA) to estimate the expression of miR-181d. Then the relative expressions of miR-181d were normalized to U6. The expression fold change was determined using the comparative delta CT (2
MTT assay
Cellular proliferation of the stably transfected cells was calculated using an MTT assay. Transfected cells were collected at 48 h post-transfection and seeded into a 96-well plate with the density of 5
Transwell assay
Cell migration was measured using 6.5 mm Transwell inserts with 8
Statistical analysis
All statistical analysis were performed using SPSS version 18.0 software (SPSS Inc., Chicago, IL, USA) and GraphPad Prism 5.0 software (GraphPad Software, Inc., USA). Student’s
Results
Overexpression of miR-181d in GC tissues and cell lines
The expression of miR-181d was examined in a set of 131 paired samples using qRT-PCR. The results suggested that miR-181d was significantly up-regulated in GC tissues when compared to the adjacent normal tissues (
Association of miR-181d with the clinicopathological features of gastric cancer patients
Association of miR-181d with the clinicopathological features of gastric cancer patients
Expression of miR-181d measured by qRT-PCR in GC tissues and cell lines. A. MiR-181d expression was upregulated in the GC tissues compared with the adjacent normal controls (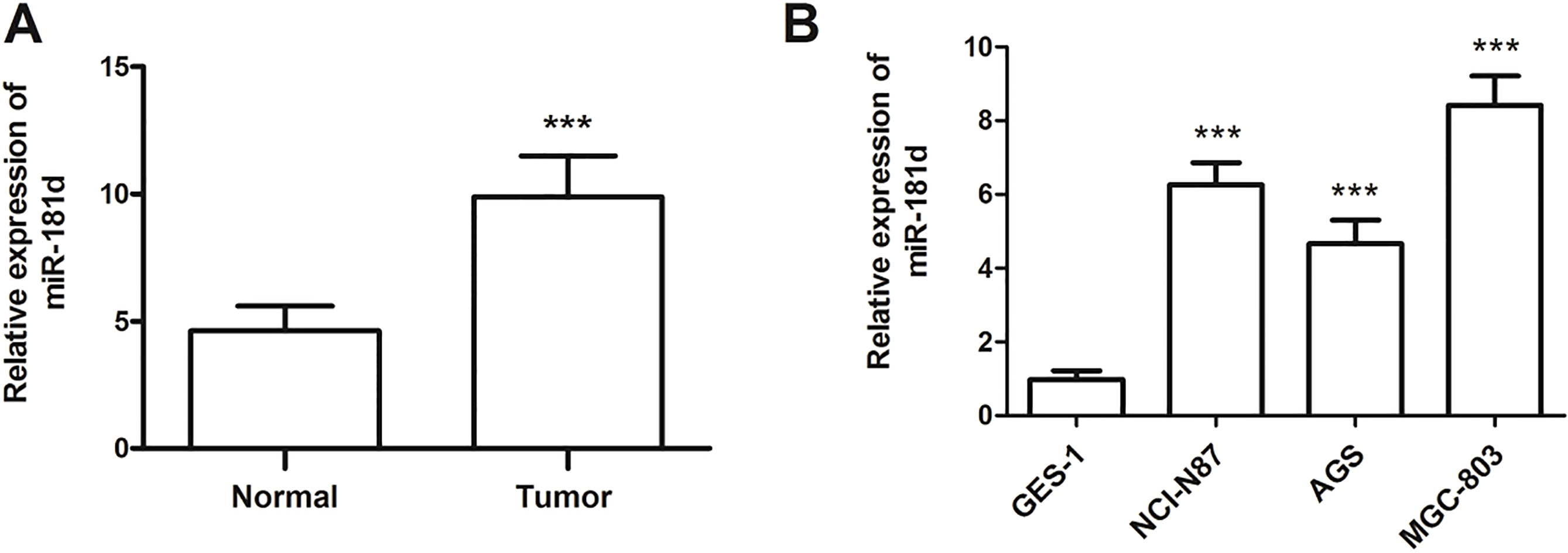
The correlation of miR-181d with the clinicopathological characteristics was further investigated. As shown in Table 1, all GC patients were divided into low (
Prognostic value of miR-181d expression for patients with GC
The Kaplan-Meier survival curves was applied to calculate the prognostic value of miR-181d. The results revealed that the GC patients with high miR-181d expression had a shorter survival time, when compared with those presenting a low miR-181d expression (log-rank
Multivariate Cox regression analysis for miR-181d in gastric cancer patients
Multivariate Cox regression analysis for miR-181d in gastric cancer patients
Kaplan-Meier survival analysis for the GC patients based on the expression of miR-181d. Patients with high miR-181d expression had shorter survival time than those with low miR-181d expression (log-rank 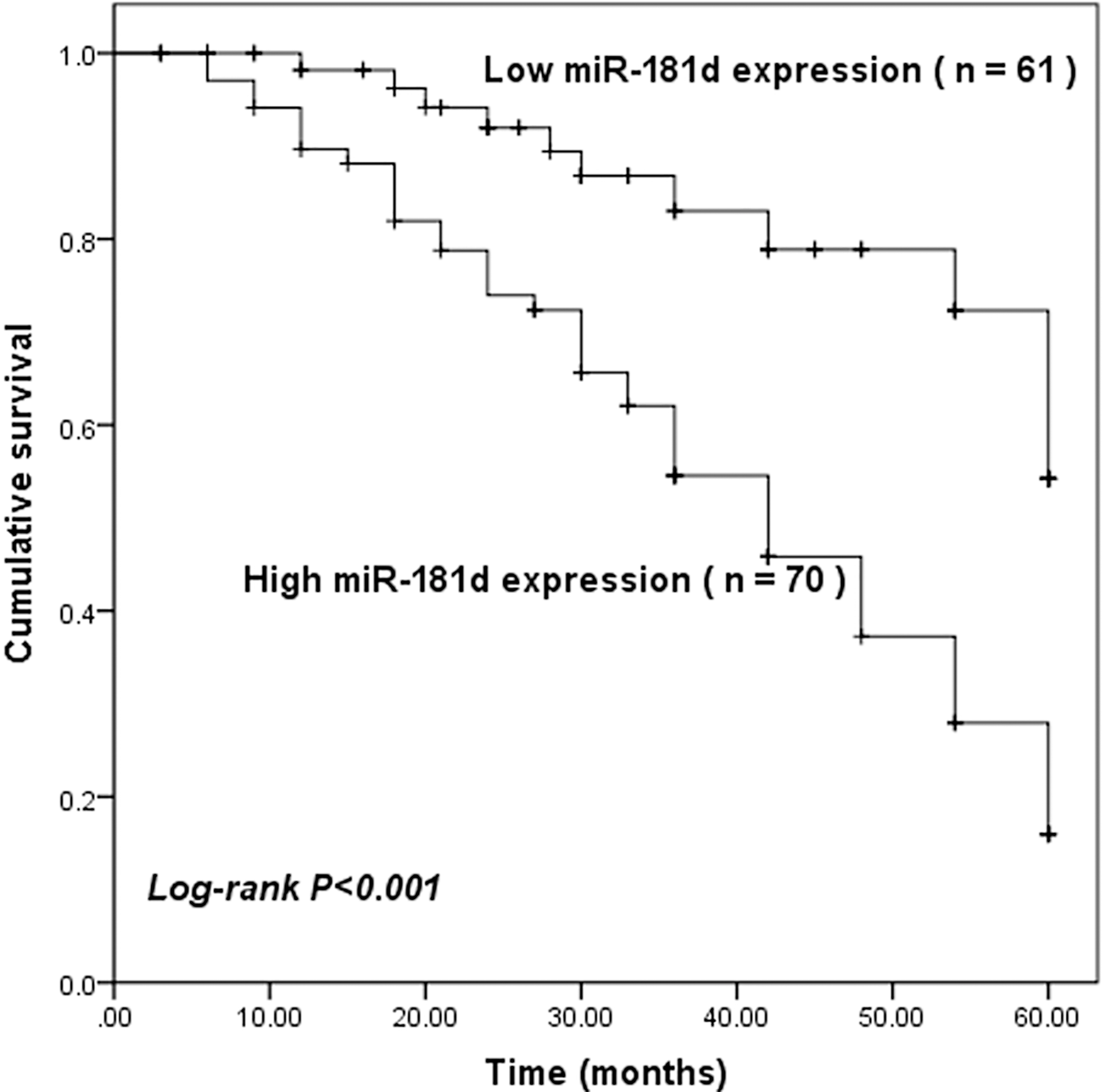
Since miR-181d expression levels were upregulated to a greater extent in NCI-N87 and MGC-803 cells, the two cell lines were used in subsequent experiments. NCI-N87 and MGC-803 cells were then transfected with miR-181d mimic, inhibitor or the corresponding negative controls. As shown in Fig. 3A, in both NCI-N87 and MGC-803 cells, the miR-181d expression levels were significantly higher in the miR-181d mimic group compared with the mimic NC group, while were lower in the miR-181d inhibitor group (all
Effects of miR-181d on cell proliferation, migration and invasion in NCI-N87 and MGC-803 cells. A. In the two cell lines, expression of miR-181d was significantly increased by miR-181d mimic transfection, but was decreased by miR-181d inhibitor transfection compared with the corresponding negative controls (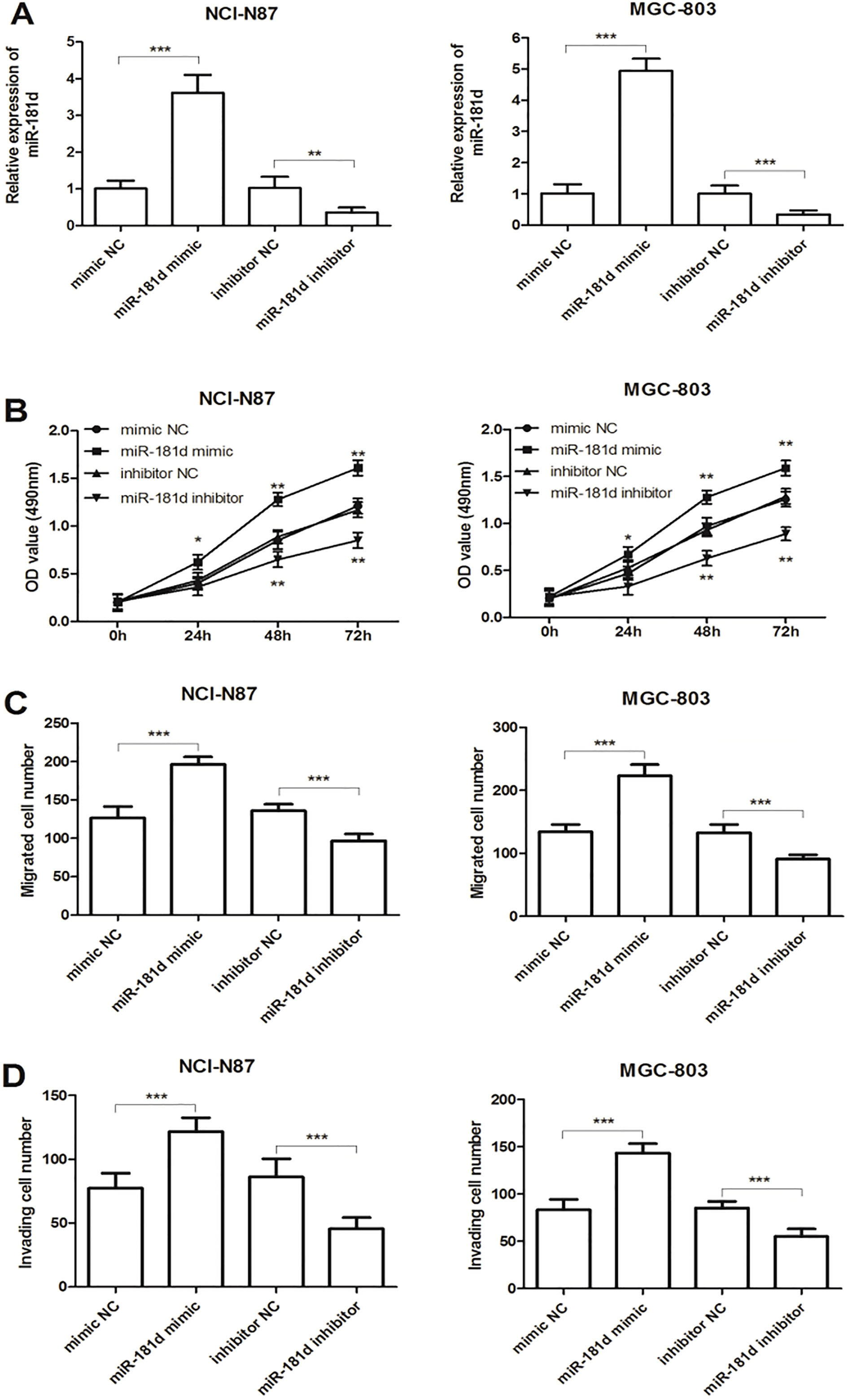
GC is regarded to be a major cause of cancer related mortality worldwide, as a result of the untimely diagnosis [15]. It is reported that 98% of patients with early GC are alive at five years after the clinical treatment, but patients with advanced GC may have relatively poor prognosis as a result of distant metastasis [16]. Tumor recurrence and metastasis are considered to be the main causes of poor prognosis for GC patients [17]. As previous studies reported, the proliferation and metastasis of GC cells can be affected by various genes and multiple factors [18, 19]. MiRNA has huge abilities of post transcriptional regulation, in addition to possessing broad application prospect in tumor therapy. Aberrant expression of miRNAs has been reported in many kinds of human cancers, and the dysregulation of miRNAs plays a crucial role in the progression of different cancers [20, 21, 22]. Assumpção et al. have identified a number of aberrantly expressed miRNAs in GC through a high-throughput sequencing of miRnome of four GC samples, such as miR-3131, miR-664, miR-150 and so on [11]. Previous evidences have demonstrated that miR-181d was upregulated in GC [23, 24], but its clinical and function role have not been studied.
MiR-181d is a member of miR-181 family, which contains miR-181a, miR-181b, miR181c and miR-181d. MiR-181d has been reported to be abnormally expressed in many kinds of human cancers. For example, Li et al. found that miR-181d was downregulated in both Esophageal squamous cell carcinoma (ESCC) cell lines and clinical samples, and DERL1 might be the target gene of miR-181d in ESCC [25]. Huang et al. demonstrated that miR-181d expression level was lower in colorectal cancer (CRC) tissues, and the down-regulation of miR-181d was significantly associated with poor prognosis for CRC patients [26]. Wang et al. revealed that low expression of miR-181d was detected in both glioma tissues and cell lines, indicating miR-181d to be a glioma suppressor [27]. In contrast, up-regulation of miR-181d was detected in human pancreatic carcinoma and cell lines, which can promote the cancer cell proliferation, migration and fluorouracil resistance, and NKAIN2 was considered as the direct target [28]. In the present study, the expression level of miR-181d was proved to be up-regulated in both GC tissues and cell lines. As reported in the previous evidences, miR-181d was up-regulated in GC [29]. Besides, Ueda et al. performed miRNA microarray in 353 gastric samples and proved that miR-181d was up-regulated in GC, which was also reported by An et al. [24, 29]. All evidences supported our present results. Additionally, our study documented that the expression level of miR-181d showed significant association with TNM stage and lymph node metastasis. The effect that miR-181d had on GC behaviors was further investigated, and cellular functional experiments indicated that up-regulated miR-181d expression promoted cell proliferation, migration and invasion in GC. We concluded that miR-181d might be an oncogene in GC, and play a vital role in the cancer progression.
We further investigated the clinical value of miR-181d in GC prognosis, and Kaplan-Meier survival analysis suggested that GC patients in the higher miR-181d expression group had shorter survival time than those in the low miR-181d expression group. Multivariate Cox regression analysis demonstrated that miR-181d expression and TNM stage were two independent prognostic marker for GC. Accordingly, miR-181d might be a novel predictive marker for the poor prognosis of GC patients and involved in the development of GC.
About the oncogenic function of miR-181d, several studies have been reported in different kind of cancer, which supported the present results. Consistently, Zhang et al. found that miR-181d had higher expression level in human pancreatic carcinoma, and down-regulated miR-181d can inhibit pancreatic cancer proliferation, migration in vitro and suppress the growth of cancer explant in vivo [28]. Strotbek et al. reported the oncogenic function of miR-181family in luminal breast cancer including miR-181d, and PH domain and leucine-rich repeat protein phosphatase 2 (PHLPP2) was proved to be the target gene of miR-181d, which may be involved in PI3K-Akt pathway [30]. PHLPP2 is an isoform of the PHLPP, which has been proved to induce cell apoptosis and inhibit tumor metastasis of several kind of human cancers [31, 32]. Notably, Ding et al. recently reported that PHLPP2 mRNA expression was lower in GC tissues compared to adjacent normal tissues, and high malignant-degree tumor tissues have lower PHLPP2 expression [33]. Furthermore, previous researches have suggested that PHLPP2 can regulate the phosphatase activity of Akt, and further be involved in regulating Akt signaling pathway [34, 35]. Interestingly, Akt signaling pathway has been widely reported to show closely correlation with GC progression [36, 37]. The present results revealed that miR-181d expression was up-regulated in GC, we inferred that miR-181d may promote tumor progression in GC via targeting PHLPP2 and activating Akt signaling pathway. But further studies are needed to verify our hypothesis.
In summary, the present study suggested that miR-181d had a high expression level in both GC tissues and cell lines. MiR-181d may serve a role as an oncogene in GC by promoting cell proliferation, migration and invasion. And miR-181d might be a novel predictive marker for the prognosis of GC patients, miR-181d may act as a potential novel therapeutic target in GC. Further researches involved in target genes are still in demand to further explore the mechanism of miR-181d in GC.
Footnotes
Conflict of interest
The authors have declared no conflict of interest.
