Abstract
OBJECTIVES:
In men, human prostate cancer (PCa) has become the second most common cancer. miRNAs are short non-coding RNAs that can inhibit target gene mRNAs. Studies have showed that the alternation of miRNAs expression in cancer is relevant to pathogenesis of tumor. In present study, we aimed to investigate functions of miR-214-5p in PCa.
MATERIALS AND METHODS:
10 paired human prostate tumor tissues and homologous para-tumor tissues were recruited, and the levels of miR-214-5p and CRMP5 were respectively determined by qRT-PCR assay. Luciferase activity analysis was performed to explore the regulation of CRMP5 mRNA 3’UTR by miR-214-5p. Then, cell experiments, including cell proliferation, apoptosis, cell cycle, migration and colony formation ability, were performed after proper plasmids or RNAs transfection.
RESULTS:
In PCa tissues and cell lines, expression of miR-214-5p was decreased compared with para-tumor tissues or normal prostate epithelial cell lines. Luciferase activity assay showed a direct combination of miR-214-5p and CRMP5 mRNA 3’UTR, and indicated that the absence of miR-214-5p in PCa cells may contributes to a high level of CRMP5. Cell experiments showed that miR-214-5p can induce inhibition of tumor cell growth, migration and colony forming efficiency, promotion of apoptosis and G1-phase arrest, on the other hand, co-expression of CRMP5 somewhat counteracted these phenotype induced by miR-214-5p.
CONCLUSION:
Taken together, miR-214-5p shows tumor suppression effects in PCa cells. Loss expression of miR-214-5p in PCa increase levels of CRMP5 through regulating CRMP5 3’UTR, which could be a potential therapy target for PCa.
Introduction
As the fifth major cause of cancer death in men, human prostate cancer (PCa) has an estimated 1,276,106 new cases and 358,989 new deaths in the year 2018 worldwide [1]. The most well-established risk factors for PCa are largely unknown, except older age and genetic susceptibility [2]. Prostate-specific antigen (PSA) testing is commonly used for PCa screening, though it remains controversial. Present studies have been concentrated on searching out approaches that could distinguish prostate malignant cancers from those less mortal, which could be more efficient than routine PSA testing [3].
MicroRNAs (miRNAs) are ubiquitous about 22 nt RNAs that can suppress target mRNA transcription or degrade target mRNA through sufficient or partial complementarity to the 3’ UTR of gene mRNA [4]. The roles of miRNAs mainly involve in tissue development, cell differentiation, cell death and proliferation [5, 6]. Since miRNAs involve in multiple signaling pathways in tumor, Studies showed that the functions of miRNAs in tumor are complicate, it can serve as tumor suppressor factors or oncogenes in various cancer types [7, 8]. Has-miR-214-5p locates on human chromosome1q24.3. In multiple cancer types, including lung cancer [9], prostate cancer [10, 11], cervical cancer [12], ovarian cancer [13], thyroid cancer [14], and hepatocellular cancer [15], miR-214-5p expresses downregulated and performs as tumor suppressor factor. In other cancer types, such as ovarian cancer [16], gastric cancer [17], pancreatic cancer [18] and osteosarcoma [19], miR-214-5p shows a oncogene functions. Therefore, effects of miR-214-5p in cancers are quite complex, hence, study revealing the underlying mechanisms of miR-214-5p in tumor is necessary.
Collapsin response mediator proteins (CRMPs), including CRMP1 to CRMP5, are mainly expressed in nervous system [20]. CRMP5, alias DPYLS5 and DRP5, is the newest identified member of CRMPs, it shows a relative low homology with other CRMPs [21]. CRMP5 is a new member of CRMPs family, and the investigations about the functions of CRMP5 in cancer is few. Studies showed that CRMP5 can interacts with actin and tubulin to regulate neurite outgrowth and polarity [22, 23]. Also, CRMP5 autoantibody was suggested as a marker of lung cancer and thymoma-related autoimmunity disease [24]. Meanwhile, researchers have devoted to reveal the relationship of CRMP5 and tumorigenesis. Moutal et al. reported that CRMP5 can promote glioblastoma cell proliferation and survival through Notch-dependent signaling [25]. Wang et al. showed that CRMP5 has a positive correlation with osteosarcoma growth, high expression of CRMP5 is related to a poor prognosis of osteosarcoma patients [26]. Functions or mechanism of CRMP5 in PCa is rarely reported, here, we aimed to explore the effects and mechanisms of CRMP5 regulated by miR-214-5p in PCa.
Materials and methods
Tissue specimens
Ten paired human prostate cancer tissues and adjacent para-tumor tissue specimens were collected from Department of Urology, Zhujiang Hospital between January 2018 to April 2018. All tissue specimens were maintained in liquid nitrogen until use. This research was approved by the Ethics Committee of Southern Medical University.
Cell culture and cell transfection
Four human PCa cell lines (DU145, C4-2, PC3, LNCaP) and human prostate epithelial cell RWPE-1 were commercially bought from Land Biology Co., Ltd (Guangzhou, China). DU145, C4-2, PC3 and LNCaP cells were cultured with RPMI-1640 medium (Hyclone, USA) containing 10% FBS (Hyclone) and 100
When transfection was performed, cells were seeded in 24-well plates and cultured for 12 h. Then, proper plasmids and RNAs were respectively transfected into cells using Lipofectamine 2000 reagent (Invitrogen, USA) following the instructions. Has-miR-214-5pmimics and has-miR-214-5p inhibitor were synthesized by Ribo Bio Co., Ltd (Guangzhou, China). CRMP5 was amplified from cDNA library and cloned into pCDNA3.1 expression vector. CRMP5 siRNA was synthesized by Ribo Bio Co., Ltd. The normal control (NC) groups were transfected with empty plasmids (pCDNA 3.1
RNA extraction and qRT-PCR assay
Total RNAs of cell and tissues were extracted using TRIzol (Invitrogen) reagent according to the instructions. RNAs concentrations and purity were measured by Eppendorf BioPhotometer plus spectrophotometer (Germany). Total RNAs were reversely transcribed into cDNA using an ImProm-IITM Reverse Transcription System kit (Promega, USA). qRT-PCR assay was performed to determine levels of miRNA or mRNA by 7500 Sequence Detection System (Thermo Fisher Scientific, Applied Biosystems, USA) using SYBR Green PCR Master Mix (TOYOBO, Japan). 2
Western blotting
Cells and tissues were lysed with pre-cooling RIPA lysate buffer supplemented with 100 mM PSMF and Cocktail proteases inhibitor (Sigma, USA). Total protein concentration was measured using a BCA Protein Assay kit (Keygen Biotech, China). Total proteins were mixed with 5
The primary antibodies used are anti-GAPDH (1: 10000 diluted; Kangchen Bio, China), anti-CRMP5 (1:1000 diluted; Abcam, USA). The secondary antibodies used are HRP-conjugated Goat Anti-Rabbit IgG (1:20000 diluted, Southern Biotech, China).
Luciferase activity assay
The fragments including wild type (wt) CRMP5 mRNA 3’UTR and mutant type (Mut) CRMP5 mRNA 3’UTR, which contains a mutant sequence in miR-214-5p predicted binding site, were cloned into luciferase reporter vector psi-CHECK2. Wt-CRMP5-3’UTR-psi-CHECK2 and mut-CRMP5-3’UTR-psi-CHECK2were respectively co-transfected with either miR-214-5p mimics or miR-214-5p inhibitor. 48 h after transfection, cell were collected and luciferase activity analysis were measured by GloMax (Promega) detector using Dual-Luciferase Reporter Assay System (Promega).
Cell proliferation assay
Cell proliferation was measured using MTS analysis 5
Cell apoptosis analysis
Cell apoptosis was measured by using an Annexin V-FITC Cell Apoptosis Detection Kit (KeyGEN Biotech, China). 48 h after transfection, 5
Cell cycle assay
1
miR-214-5p is down-regulated in PCa tissues and cell lines. (A) Relative expression of miR-214-5p in 10 paired human prostate tumor tissues and homologous para-tumor tissues by qRT-PCR. ** indicates 
miR-214-5p direct targets CRMP5 mRNA 3’UTR and suppresses gene expression. (A) TargetScanHuman showed that CRMP5 mRNA 3’UTR (at position 181-187) is a predicted target of miR-214-5p. (B) Wild type (wt) CRMP5 mRNA 3’UTR contains miR-214-5p binding site, and mutant type (Mut) mRNA 3’UTR contains mutant region at miR-214-5p binding site. (C) Wt-CRMP5-3’UTR-pis-CHECK2 and mut-CRMP5-3’UTR-pis-CHECK2 were respectively co-transfected with either miR-214-5p mimics or miR-214-5p inhibitor, then the luciferase activity were measured. (D) Relative expression of CRMP5 in 10 paired human prostate tumor tissues and homologous para-tumor tissues were determined by qRT-PCR analysis. ** indicates 
CRMP5 co-expression recovers cell proliferation altered by miR-214-5p. (A) CRMP protein expression in miR-214-5p mimics, miR-214-5p inhibitor, CRMP5, siCRMP5 and CRMP5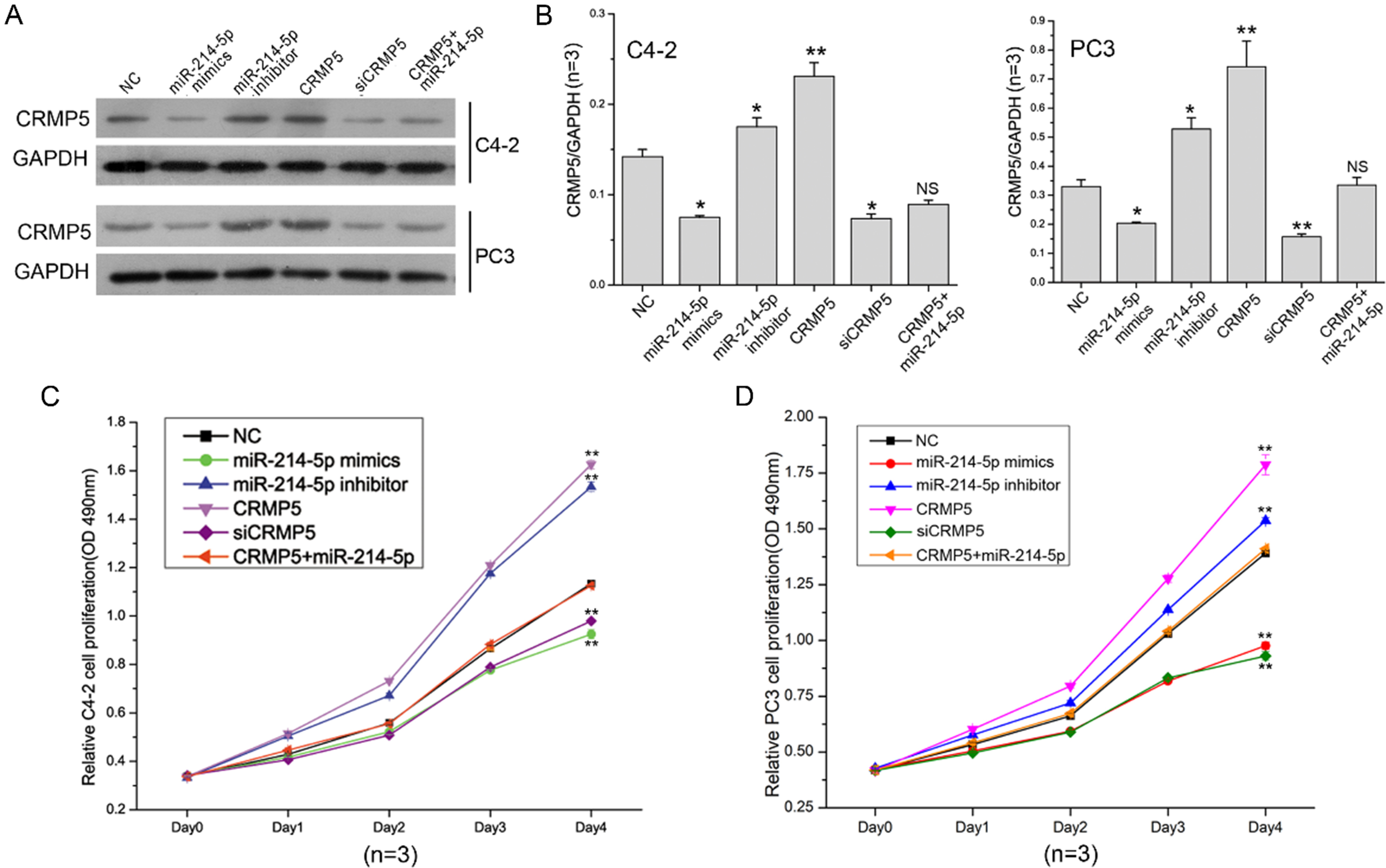
CRMP5 co-expression recovers cell apoptosis and cell cycle arrest altered by miR-214-5p. (A, B) Cell apoptosis were detected in C4-2 and PC3 cells after miR-214-5p mimics, miR-214-5p inhibitor, CRMP5, siCRMP5 and CRMP5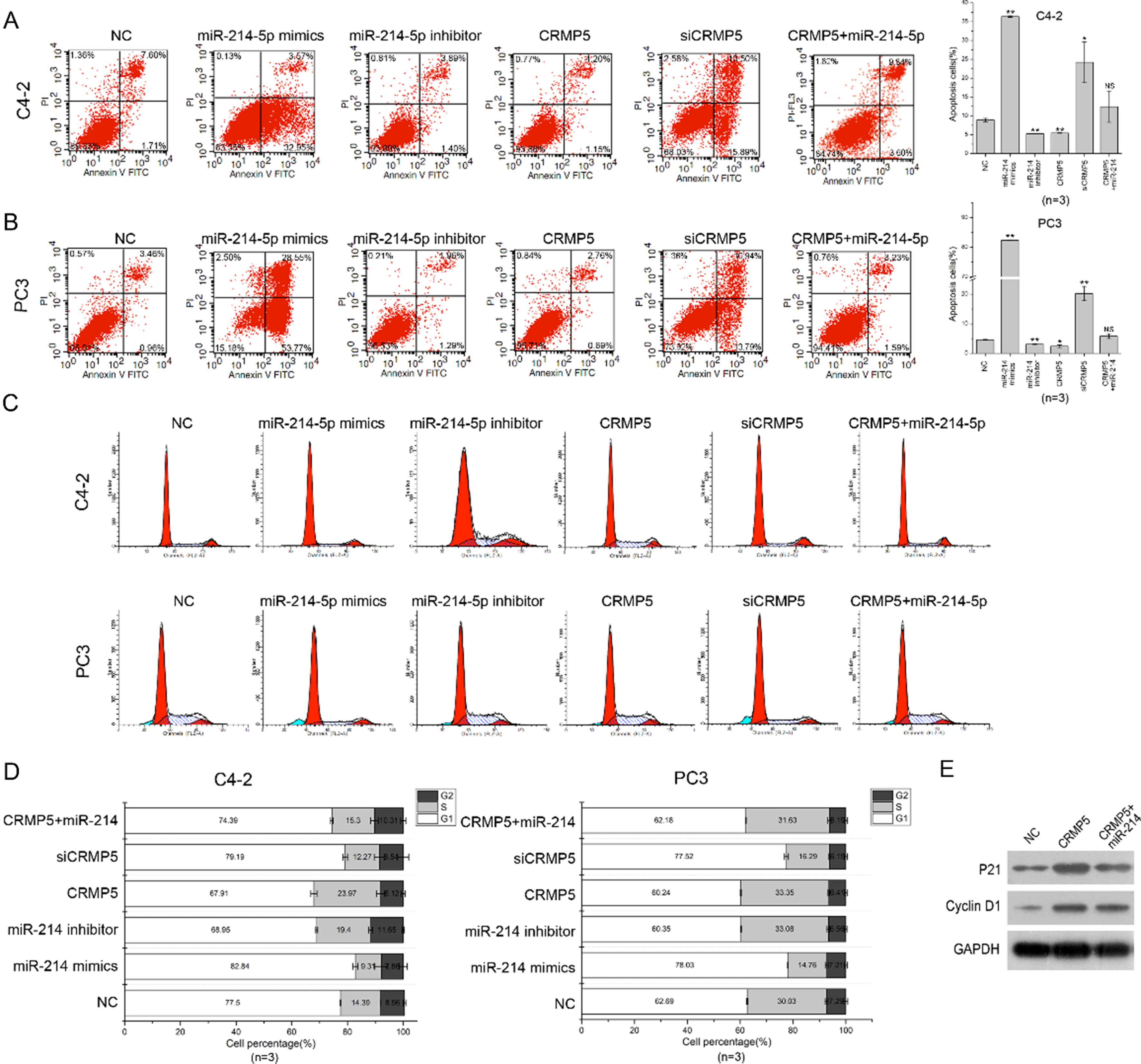
Cell migration was analyzed using 8
Colony forming efficiency analysis
Transfected cells were digested, diluted and counted. 300 cells were seeded in each well of 6-well plates containing 2 ml complete medium. After 7 days, the cells were fixed with 4% paraformaldehyde, washed with PBS, and stained with crystal violet. Images of cell colonies were captured and analyzed by ELISPOT AID iSpot system (Germany).
Statistical analysis
The present data were shown as means
Results
miR-214-5p is down-regulated in human PCa tissues and cell lines
Firstly, we detected miR-214-5p expression in human PCa tissues and cell lines. As shown in Fig. 1A, in 10 paired prostate tissues, levels of miR-214-5p in tumor tissues were much lower than that in homologous para-tumor tissues. Similarly, miR-214-5p expression were significant decreased in PCa cell lines, including DU145, C4-2, PC3 and LNCaP, compared with normal prostate epithelial cell line RWPE-1 (Fig. 1B). All these results suggest that miR-214-5p expression is down-regulated in human PCa tissues and cell lines.
miR-214-5p direct targets CRMP5 mRNA 3’UTR and regulates CRMP5 expression
Online public databases (TargetScan Human 5.1, miRanda and miRDB) were used to determine the binding of CRMP5 mRNA 3’UTR with miR-214-5p. As shown in Fig. 2A, CRMP5 mRNA 3’UTR has a predicted binding site of miR-214-5p at position 181-187. Then, CRMP5 mRNA 3’UTR containing a wild or mutant miR-214-5p binding region (Fig. 2B) were constructed into luciferase reporter vector psi-CHECK2. Wt-CRMP5-3’UTR and mut-CRMP5-3’UTR were respectively co-transfected with miR-214-5p mimics or miR-214-5p inhibitor into C4-2 cells. Luciferase activity analysis indicated that in wt-CRMP5-3’UTR groups, miR-214-5p mimics transfection can significant reduce luciferase activity, miR-214-5p inhibitor transfection can significant increase luciferase activity. However, in mut-CRMP5-3’UTR groups, no significant difference between miR-214-5p mimics transfection and control were detected, neither with miR-214-5p inhibitor transfection and control (Fig. 2C). Taken together, miR-214-5p can direct target CRMP5 mRNA 3’UTR.
miR-214-5p inhibits C4-2 and PC3 cell migration, and CRMP5 co-expression counteracts this effect. (A, B) After miR-214-5p mimics, miR-214-5p inhibitor, CRMP5, siCRMP5 and CRMP5
miR-214-5p inhibits C4-2 and PC3 cell colony forming efficiency, and CRMP5 co-expression counteracts this effect. (A, B) After miR-214-5p mimics, miR-214-5p inhibitor, CRMP5, siCRMP5 and CRMP5
Since miR-214-5p can specifically target CRMP5 3’UTR, we next measured CRMP5 expression in PCa tissues and cell lines. In 10 paired prostate tissues, qRT-PCR assay showed that levels of CRMP5 in tumor specimens were markedly increased compared with corresponding para-tumor specimens (Fig. 2D). Accordingly, CRMP5 mRNA expression in PCa cell lines DU145, C4-2, PC3 and LNCaP were much higher than that in normal epithelial cell line RWPE-1 (Fig. 2E), either with CRMP5 protein expression (Fig. 2F). That is to say, loss of miR-214-5p in PCa cells likely contributes to the promotion of CRMP5 expression.
CRMP5 overexpression counteracts effects of miR-214-5p on cell proliferation, apoptosis and cycle arrest in PCa cells
To further explore functions of miR-214-5p/CRMP5 axis on PCa cell phenotype, cell transfection and cell experiments were performed in C4-2 and PC3 cells. Firstly, miR-214-5p mimics, miR-214-5p inhibitor, CRMP5 and siCRMP5 were respectively transfected into C4-2 and PC3 cells. Western blotting assays showed that miR-214-5p negatively regu lates CRMP5 expression, CRMP5 co-expression with miR-214-5p can somewhat counteracts the repression of miR-214-5p on CRMP5 expression (Fig. 3A and B). In cell proliferation assay, miR-214-5p acts as an inhibitor in C4-2 and PC3 cell growth, CRMP5 overexpression can significantly promote tumor cell growth and recover the growth inhibition of miR-214-5p as well (Fig. 3C and D). Additionally, miR-214-5p can increase the C4-2 and PC3 cell apoptosis, co-transfection with CRMP5 expression plasmids and miR-214-5p mimics can turn the tumor cell apoptosis rate into normal level (Fig. 4A and B). In cell cycle detection, both miR-214-5p mimics and siCRMP5 transfection can arrested C4-2 and PC3 cells at G1 phase of cell cycle, miR-214-inhibitor and CRMP5 plasmids transfection can decrease the cell percentage at G1 phase, CRMP5 plasmids transfection can counteracts the arrestment of G1 phase induced by miR-214-5p to a certain extent (Figs. 4C and 3D). CRMP5 expression promote the expression of P21 and Cyclin D1, and co-expression of miR-214-5p mimics and CRMP5 can counteract this promotion effect to a certain content (Fig. 4E).
CRMP5 overexpression counteracts effects of miR-214-5p on cell migration and colony formation in PCa cells
In further exploration, we found that compared with normal control, both miR-215-5p mimics and CRMP5 siRNA transfection can inhibit the tumor cell migration in trans-well chamber based migration assay, CRMP5 overexpression can increase the number of migration cells, migration cell number in co-expression of CRMP5 and miR-214-5p mimics groups have no significant difference with normal control (Fig. 5A and B). Finally, we measured the effects of miR-214-5p/CRMP5 axis on tumor cell colony forming efficiency. Similarly, the results showed that miR-214-5p and CRMP5 have an opposite effect on the ratio of colony formation. Taken together, miR-214-5p can inhibits PCa progress, CRMP5 co-expression can counteracts suppression effects of miR-214-5p on cell migration and colony forming efficiency (Fig. 6A and B).
Discussion
For the reason of aging, changing in main risk factors and increasing of population, cancer has a growing incident and mortality worldwide. Human prostate cancer (PCa) is the second most frequent cancer and fifth leading cause of cancer death in men, but its etiology is mainly unknown [1]. It could markedly promote the survival if diagnosed at an early PCa stage. Routine diagnostic methods for PCa includes PSA screening and digital rectal examination.
miRNA expression has proved to be altered in cancer, which involves in regulation of multiple signaling pathway and induction of malignant phenotypes of cancer cells. miR-214-5p has been well investigated as oncogene or tumor suppressor in various kinds of human cancers [12, 18]. In human PCa, a miRNA expression profiling showed that miR-214 was down-regulated in tumor tissues compared with corresponding para-tumor tissues, miR-214 and miR-205 together were suggested as potential biomarkers with high precision (89% sensitivity and 80% specificity) for PCa diagnose [10, 27]. At present, the regulation mechanisms of miR-214-5p in human PCa is mostly unknown yet.
CRMPs is intensively expressed in developing and adult nervous system, and plays a role in regulation of nervous system development and neurite outgrowth [21, 28]. It is well investigated that CRMPs can promotes the formation of filopodia and neurite outgrowth by interacting with actin cytoskeleton [22, 29], which is important for tumor cell migration and invasion. L-CRMP1, a novel isoform of CRMP-1, has been proved to promote cancer cell metastasis and invasion [30, 31]. CRMP2 is suggested as prognostic marker and potential new target for cancer therapy in NSCLC [32]. Expression of CRMP4 was revealed to be increased in human colon tumor tissues, and silence of CRMP4 could inhibited tumor cell proliferation [33]. CRMP5 is reported to promote glioblastoma cell proliferation [34]. In osteosarcoma, CRMP5 showed to promote tumor cell proliferation, migration and invasion and tumor growth, CRMP5 expression is associated with poor clinical outcomes in patient with osteosarcoma as well [26]. In our results, we tested the expression of miR-214-5p and CRMP5 in four human PCa cell lines, DU145, C4-2, PC3, LNCaP. Compared with normal prostate cell line, C4-2 and PC3 express much high CRMP5 and low level of miR-214-5p. Hence, we chose these two representative cell types to study the functions of miR-214-5p/CRMP pathway. In addition, the results of functional assays were different, which may due to the differences in AR status of the two cell lines, androgen-sensitive prostate cancer cell line C4-2 cell and androgen-insensitive cell line PC3 [35]. The study about roles of CRMP5 in cancer is increasing, however, the underlying mechanisms need to be uncovered.
In present study, we firstly showed the regulation of CRMP5 by miR-214-5p in human PCa. miR-214-5p can direct target CRMP5 mRNA 3’UTR and inhibit the gene expression. The absence of miR-214-5p in PCa induced a relative high level of CRMPs in tumor cells. Silence of CRMP5 in PCa could inhibit cancer cell proliferation, migration and colony formation ability and promote cell apoptosis. These data indicated that the miR-214-5p/CRMP5 regulation axis should be a potential target pathway for PCa therapy.
