Abstract
BACKGROUND:
Annexin A5 (ANXA5) is a kind of Ca
OBJECTIVE:
To explore what role ANXA5 play in human uterine cervical carcinoma.
MATERIALS AND METHODS:
In this study, a recombined ANXA5 plasmid was constructed and uterine cervical carcinoma cell lines HeLa and SiHa were transfected with it. After ANXA5 overexpression was determined by Western Blot, cell proliferation test was detected by MTT assay and colony formation assay respectively. FACS assay and Hochest33258 staining methods were employed to detect cell apoptosis. To further investigate whether ANXA5 influence cell migration and invasion, wound healing assay and transwell assay were applied. At the same time, the relative mechanism was investigated.
RESULTS:
When ANXA5 expression increased, cell proliferation was inhibited by regulating the expression of bcl-2 and bax while cell metastasis was suppressed by regulating E-cadherin and MMP-9 expression.
CONCLUSION:
ANXA5 overexpression in the uterine cervical carcinoma might play important roles in cell proliferation and metastasis of uterine cervical cancer cells and act as an anti-cancer gene in uterine cervical cancer.
Introduction
Cervical cancer incidence and mortality is the fourth leading cause of death in females worldwide and the second leading cause of mortality among women aged 19–39 years [1], with 500,000 new cases and 250,000 deaths annually worldwide [2]. Although the association between human papilloma virus and tumorigenesis of uterine cervical carcinoma has been clarified, however, whether there are any oncogene activated or suppressor gene inactivated still largely remains elusive. Annexins are a well-known multigene family of Ca
Materials and methods
Cell culture
Human uterine cervical carcinoma cell line HeLa and SiHa were obtained from Shanghai Academy of Medical Sciences (Shanghai, China). The cells were incubated at 37
RT-PCR
Total RNA was isolated from HeLa or SiHa cells with the TRIzol reagent kit according to the manufacturer’s instructions. RNA (2
ANXA5 vector generation and identification
The pcDNA3.1 empty plasmid was obtained from Jikai Biological Engineering Company (Shanghai,China). Both pcDNA3.1 empty plasmid and ANXA5 nucleotide were sliced by BamHI and XhoI incision enzyme so as to obtain sticky ends. The sliced plasmid and the nucleotide was linked by T4 ligase so as to construct the recombined ANXA5 plasmid which was named plasmid pcDNA3.1-ANXA5. Then the recombined plasmid was transformed into DH5
Plasmid transfection of HeLa and SiHa cells
The transfection reagent lipofectamine2000 was purchased from Invitrogen (Invitrogen Co., Carlsbad, CA, USA). At about 50–70% confluence, cells were transfected with the recombined pcDNA3.1-ANXA5 plasmid and the pcDNA3.1 empty plasmid respectively according to the manual. Six hours later, the transfection medium was replaced with complete growth DMEM. Twenty-four hours later, 800
Western blot analysis
The cells were homogenized in a single detergent lysis buffer (50 mM Tris, pH 8.0; 150 mM NaCl; 1% Triton X-100; and 0.5% each of protease and phosphatase inhibitor cocktails) and then centrifuged at 12,000
MTT assay
Cell suspensions (100
Colony formation assay
The cells (6
Fluorescence microscopy
Cells at the logarithmic-growth phase were seeded into 96-well plates (1
Analysis of apoptosis by FACS assay
Cell apoptosis was analyzed by flow cytometry (FACSAria II; BD Biosciences, Franklin Lakes, NJ, USA). Cultured cells were washed twice with phosphate-buffered saline (PBS) and resuspended in binding buffer at 1
Wound healing assay
When the cells reached 80–85% confluence, they were scratched with a micropipette tip in the cell monolayer. After 48 h incubation, recovery of the wound was observed and images were captured by a phase-contrast microscope.
Cell invasion assay
The cell invasion assay was performed using the QCM 24-well cell invasion assay kit (Millipore, Billerica, MA, USA). Each lower chamber contained an additional 600 ml of 0.5% FBS as the chemoattractant. Cells (1
Identification of ANXA5 gene sequence in recombined pcDNA3.1-ANXA5 plasmid by enzyme digestion and sequencing. A. The result of enzyme digestion of pcDNA3.1-ANXA5; B. The sequencing of pcDNA3.1-ANXA5 plasmid in HeLa cells; C. The sequencing of pcDNA3.1-ANXA5 plasmid in SiHa cells.
After dyeing, the lower membrane was decolorized with 33% acetic acid, and the crystal violet was completely eluted. The eluent was measured with a spectrophotometer (Spectramax 190; Molecular Devices, Sunnyvale, CA, USA) at 570 nm. Each assay was replicated three times.
Each experiment was performed at least three times. All the data were expressed as means
Results
The recombined pcDNA3.1-ANXA5 plasmid was successfully constructed
After digested by BamHI and XhoI incision enzyme, the plasmid was put in the agarose gel electrophoresis and at about 1000 bp level, ANXA5 band was observed (Fig. 1A). To assure none of the bases is mistake, the plasmid was sent to the biological company to be sequenced. The result of the sequencing assay confirmed all the bases of ANXA5 gene in the recombined plasmid were correct (Fig. 1B and C).
Screening of ANXA5 overexpression in HeLa and SiHa cells by Western Blot method. ANXA5: the cells transfected with pcDNA3.1-ANXA5 plasmid; Neo: the cells transfected with pcDNA3.1 empty plasmid; Blank: the cells without any treatment. ANXA5 cells group has a significant difference compared with Neo and Blank cells. *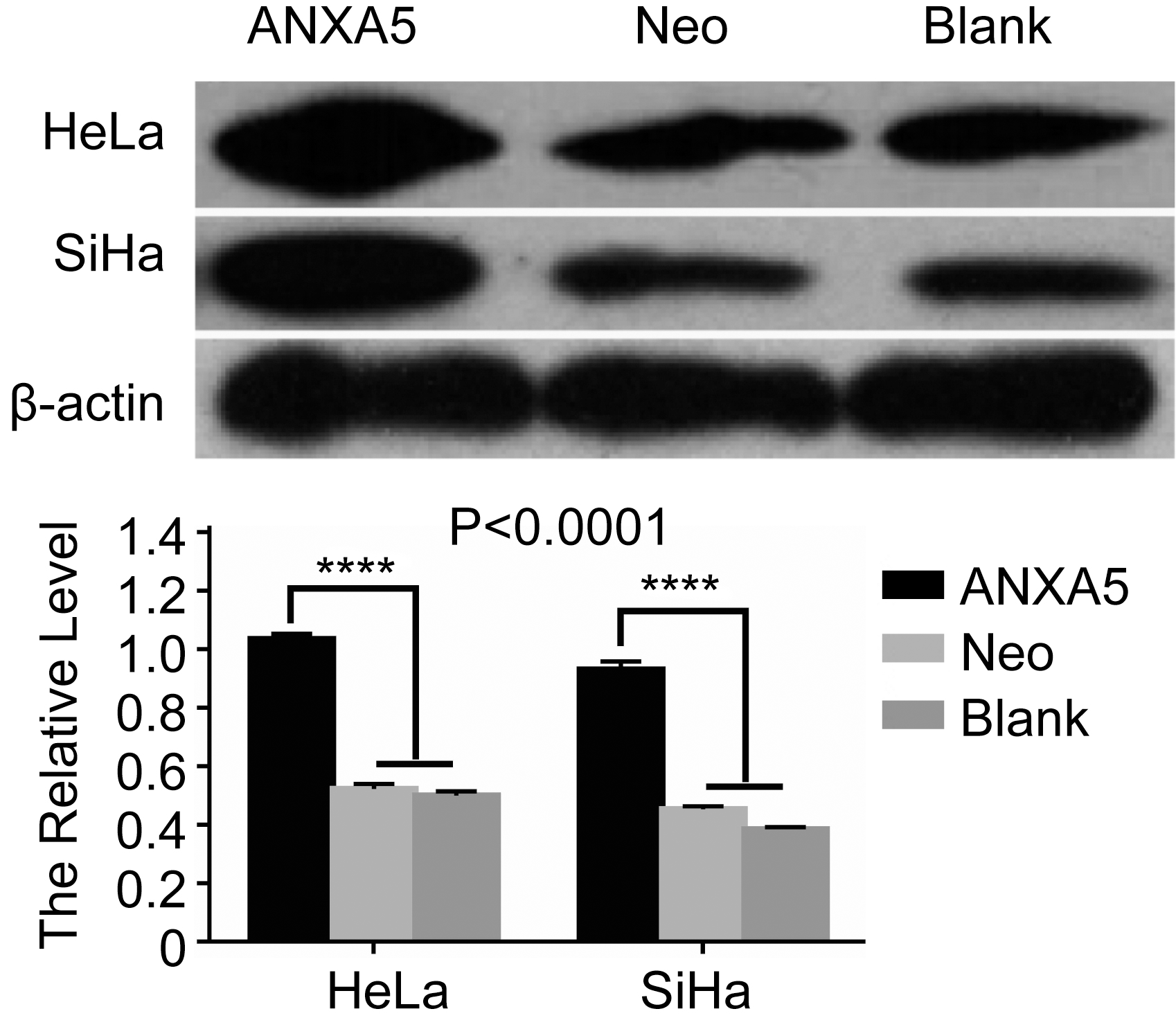
Alteration of ANXA5 overexpression cells in morphology. Original magnification 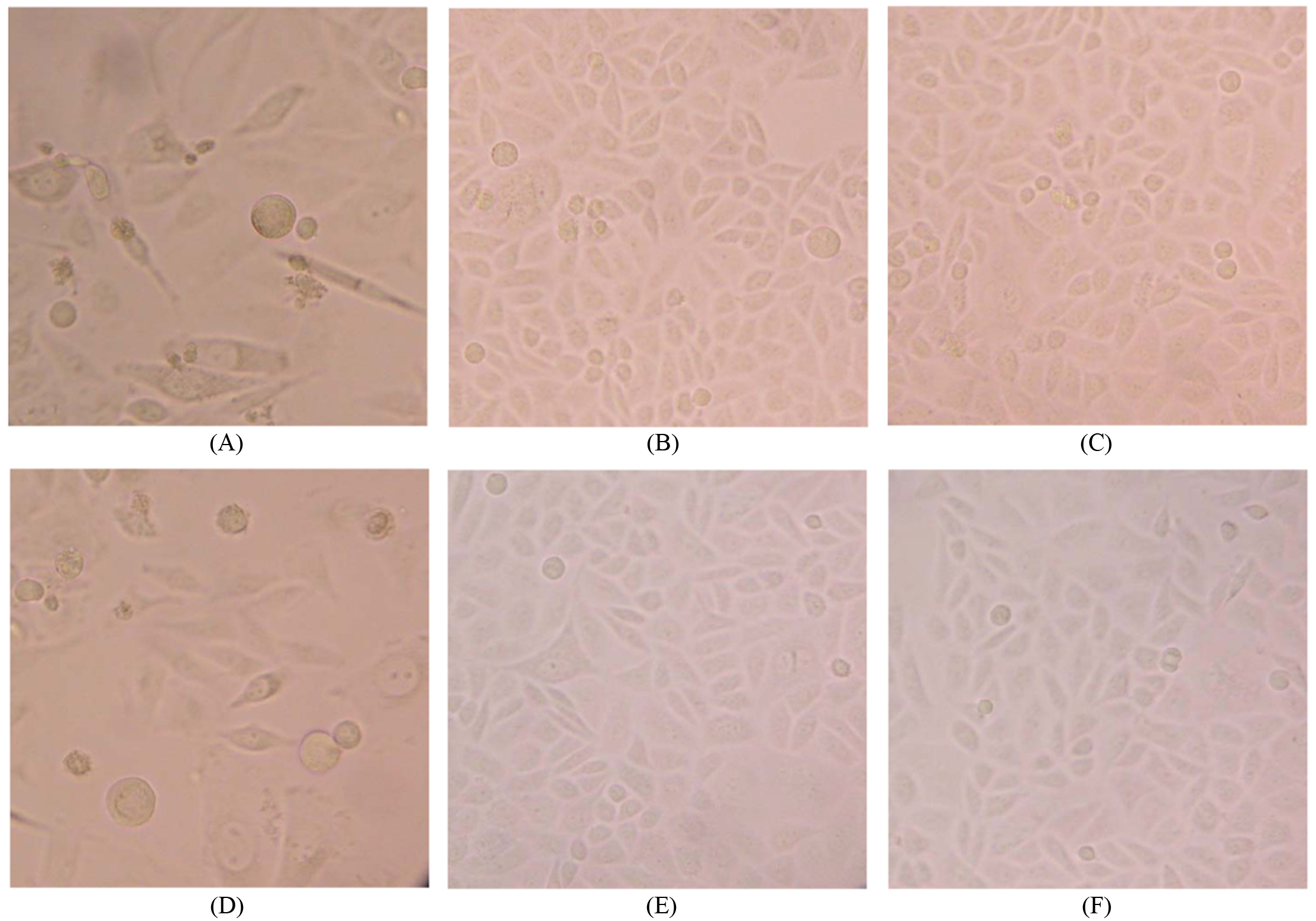
The recombined pcDNA3.1-ANXA5 plasmid was transfected into HeLa cells and SiHa cells respectively with the transfect reagent lipofectimin2000. G418 was used to screen the positive cell clones and two weeks later, the stably ANXA5 transfected cell clones were obtained. To detect whether ANXA5 was overexpressed, Western blot method was applied and ANXA5 overexpression in HeLa(SiHa)/ANXA5 cells groups was confirmed (
Cells morphology under the inverted microscope
The HeLa (SiHa)/ANXA5 cells became looser and irregular, some became thinner and longer like fibroblasts while some became swelled with a prominent nucleolus, rather different from the Neo or Blank cells which were polygonous and grouped together like “paving stones” (Fig. 3).
ANXA5 overexpression suppressed the proliferation of HeLa or SiHa cells
Cell proliferation test was determined by MTT assay and colony formation assay respectively. In MTT assay, subsequent to culture for 12, 24, 36, 48 and 72 h, the
Proliferation of ANXA5 overexpression HeLa or SiHa cells by MTT assay and colony formation assay. A. MTT assay. B. Colony formation assay. ANXA5 cells group has a significant difference compared with Neo and Blank cells. *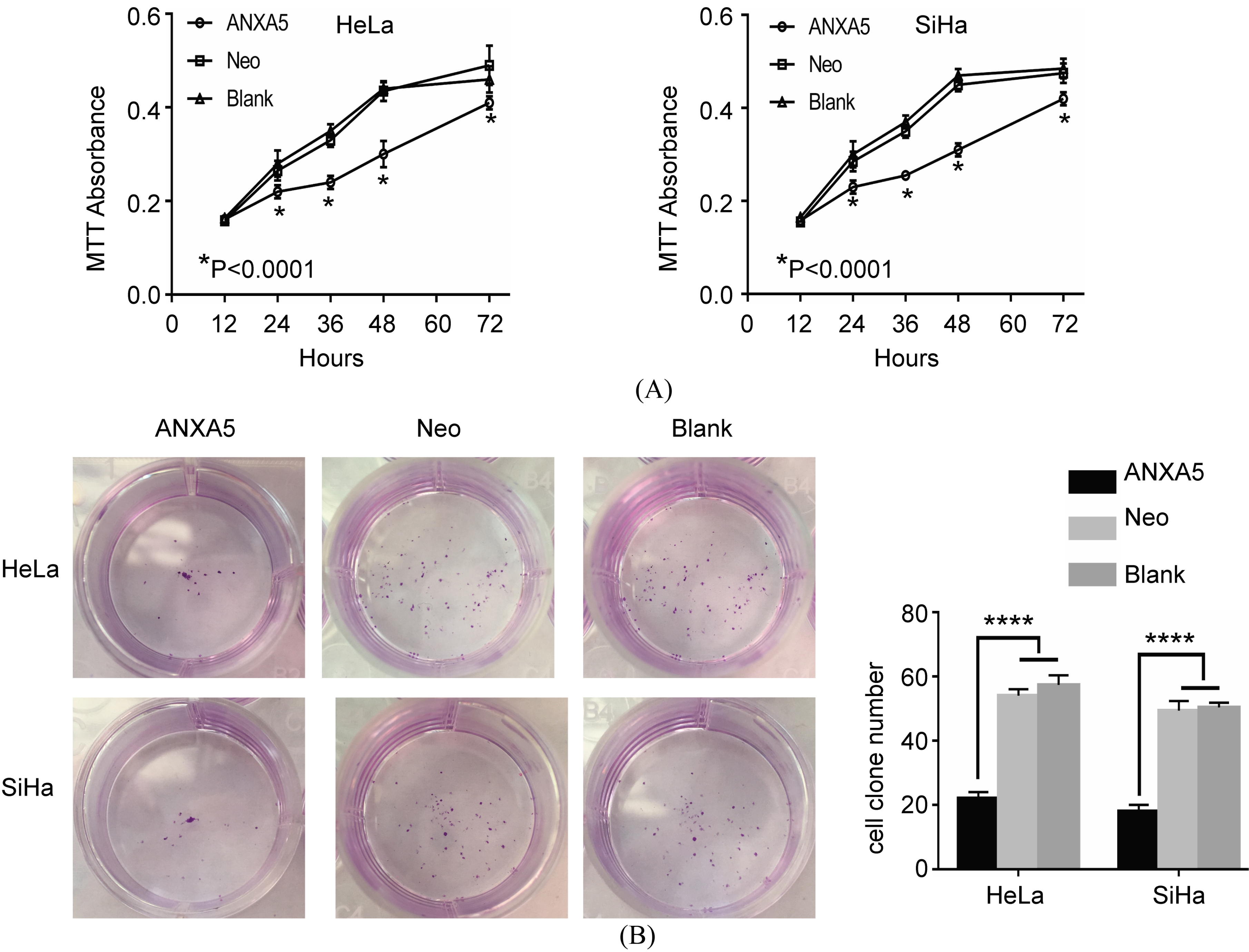
Apoptotic HeLa or SiHa cells stained by Hochest 33258 and FACS assay respectively. A. FACS assay. B. Hochest 33258 staining. Original magnification 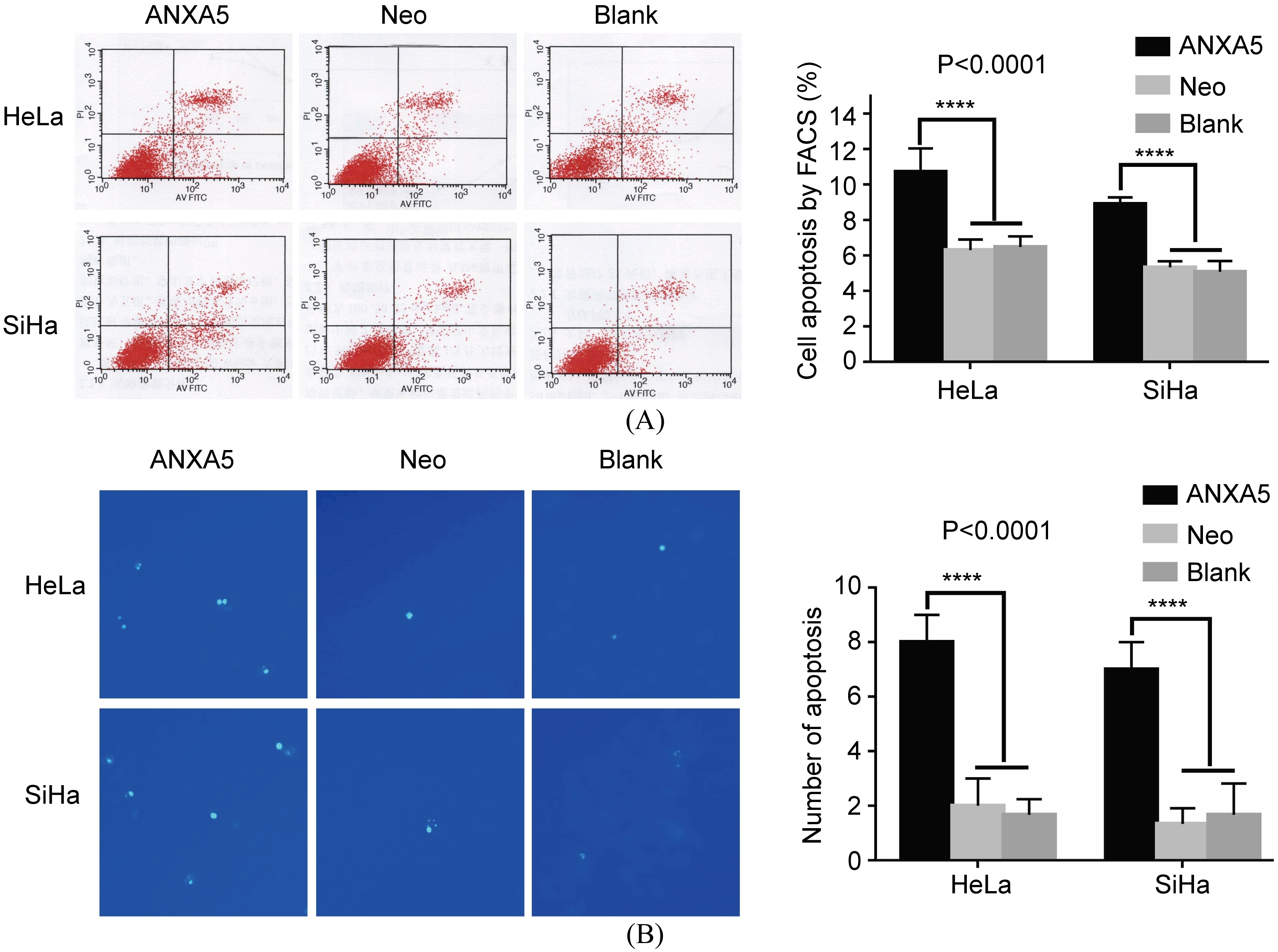
Cell metastasis assay. A. Migration detection by wound healing assay. B. Invasion detection by transwell assay. C. OD570 of the transferred cells. Forty-eight hours after transfection, the ANXA5/HeLa or ANXA5/SiHa cells appeared limited metastasis ability. The difference was significant. *
Bcl-2, Bax, E-cadherin and MMP-9 expression by Western Blot. The expression of bax and E-cadherin increased while that of bcl-2 and MMP-9 deceased. ANXA5 cells group has a significant difference compared with Neo and Blank cells. *
FACS assay and Hochest33258 staining methods were employed. In FACS assay, apoptotic cells in the Hela (SiHa)/ANXA5 group were more than those in both Neo and Blank groups (
ANXA5 overexpression suppressed cell metastasis of uterine cervical cancer cells
To further investigate whether ANXA5 influence cell metastasis, wound healing assay and transwell assay were applied. As shown in Fig. 6A, forty eight hours after the plasmid pcDNA3.1-ANXA5 transfection into the cells, both the ANXA5/Hela and ANXA5/SiHa cells showed limited ability to migrate while the width of the scratch borders in the Neo cells reduced significantly. We then detected the invasion ability of the transfected cells by transwell assay. An obviously decreased number of ANXA5/HeLa or ANXA5/SiHa cells compared to the Neo and Blank cells was detectable which might be attributed to a reduced invasion ability (Fig. 6B). We further collected the eluent of the decolorized lower membrane and detected the absorbance value at 570 nm. The OD570 of the ANXA5 overexpression cell group significantly decreased (Fig. 6C).
Gene expression
To detect the mechanism of ANXA5 on the behavior of the uterine cervical carcinoma cells, the expression of bcl-2, bax, E-cadherin and MMP-9 was further detected. The decreased bcl-2 and the increased bax expression of the ANXA5/HeLa and ANXA5/SiHa cells indicated that ANXA5 might suppress cell proliferation ability by regulating bcl-2 and bax expression. Similarly, the increased E-cadherin expression and decreased MMP-9 expression implied ANXA5 might suppress cell migration and invasion by up-regulating E-cadherin while down-regulating MMP-9 expression (Fig. 7).
Discussion
Previously, we found ANXA5 was up-regulated in uterine cervical cancer [16]. However, the detailed functions ANXA5 might exert had not been discussed. Thus, in this study, we investigated the role of ANXA5 in uterine cervical cancer cells. Our results implied that ANXA5 might act as an anti-cancer protein which could suppress cell proliferation and prompt apoptosis by regulating bcl-2 and bax expression. In addition, ANXA5 could suppress cell migration and invasion by up-regulating E-cadherin and down-regulating MMP-9 expression.
ANXA5 could suppress proliferation and prompt apoptosis in uterine cervical carcinoma cells. Although DT40 cells were found resistant to apoptosis if lacking ANXA5 protein [17] and the interaction of ANXA5 and beta5 integrin can regulate the apoptosis of the growth plate chondrocytes [18], the biological function of endogenous ANXA5 is still unclear. Here, both MTT and colony formation assay were used to detect cell proliferation and the results suggested ANXA5 overexpression inhibited the cell viability which was in line with the report that ANXA5 inhibits neuroblastoma growth
ANXA5 could suppress uterine cervical carcinoma cells migration and invasiveness. To access whether ANXA5 involve in cell metastasis, wound healing assay and transwell assay were employed and the data clearly showed ANXA5 could suppress cell metastasis. These results are in agreement with a study on lung carcinoma cells where ANXA5 inhibited the migration of these cells [34], a study on diffuse large B-cell lymphoma where overexpression of ANXA5 significantly decreased cell invasion, MMP-9 expression and PI3K phosphorylation [35], but in contrast to the study carried out in oral carcinoma cells [11], normal human keratinocytes [36], corneal epithelium [37] and murine hepatocarcinoma cells [38] where a promotion of cell migration by ANXA5 was demonstrated. An explanation for this opposite results might be that ANXA5 exert specific effects on different tumors and different biological species. In addition, we analyzed ANXA5 within its physiological concentration whereas in the study on the corneal epithelium, extracellular ANXA5 protein was applied. To detect the mechanism of ANXA5 involving in cell metastasis, E-cadherin and MMP-9 expression was investigated. E-cadherin is a calcium dependent cell-cell glycoprotein which is thought to contribute to suppression in cancer by decreasing proliferation, invasion and metastasis. MMP-9 is an enzyme that belong to the zinc-metalloproteinases family which involved in the degradation of the extracellular matrix [39]. Both E-cadherin and MMP-9 are regarded as the important members in the process of epithelial-mesenchymal transition (EMT) which is the main mechanism of cell metastasis. We found ANXA5 overexpression could increase E-cadherin but decrease MMP-9 expression. With the result of suppressing cell proliferation, we speculate that ANXA5 might act as an anti-cancer protein during the development of human uterine cervical carcinoma.
In summary, we clarified that ANXA5 up-regulation could suppress cell proliferation and metastasis which implied ANXA5 might act as a tumor suppressor gene on the uterine cervical cancer cells. We have to point out that our results are lack of ANXA5 knockdown in the cells which is the next step of our study. Furthermore, genes in EMT such as snail, slug, vimentin will be detected later and the detailed mechanism of ANXA5 acting on the cancer cells will be investigated in the future.
Footnotes
Acknowledgments
The present study was supported by the Key Subjects in University of Hebei Province (grant No. 2013-4), by the Science and Technology Agency (grant No. 10276142) and by the Education Department of Hebei Province (grant No. Z2014026).
Conflict of interest
The authors declare no conflict of interest.
