Abstract
BACKGROUND:
Recently, the incidence and mortality of retinoblastoma (RB) have gradually increased. Many studies support the pivotal role of microRNAs (miRNAs) in the pathogenesis of RB. Alternation of microRNA-98 (miR-98) expression has been detected in several cancers, excluding RB. This study was designed to assess the regulatory mechanisms of miR-98 in human RB.
METHODS:
RT-qPCR and Western blot analysis were used to detect miR-98 and HMGA2 expression. The effects of miR-98 were explored using the CCK-8 and Transwell assays. Dual-luciferase reporter assay was performed to confirm the relationship between miR-98 and HMGA2.
RESULTS:
In RB, downregulation of miR-98 was identified. Upregulation of miR-98 inhibited proliferation, invasion and migration of RB cells. Further, HMGA2 was confirmed as a direct target gene of miR-98. And knockdown of HMGA2 suppressed the progression of RB. Moreover, upregulation of HMGA2 reversed the suppressive effects in the development of RB. In addition, miR-98 also showed suppressive effect on EMT and Wnt/
CONCLUSION:
MiR-98 targets HMGA2 to act as a tumor suppressor in RB by mediating Wnt/
Introduction
Retinoblastoma (RB) is a malignant tumor derived from photoreceptor precursor cells. RB usually occurs in children under 3 years old and has a family genetic predisposition [13]. RB is the most common intraocular malignancy in infants and children and is rare in adults [18]. Moreover, RB is prone to intracranial and distant metastasis, often endangering children’s lives. For families with a genetic history of RB, genetic testing and genetic counseling can reduce the risk of birth in children with RB [4]. In addition, the prognosis of RB patients is related to many factors, such as tumor size and location, and tumor differentiation [25]. To make matters worse, there are no effective preventive measures for RB. Therefore, early detection, early diagnosis and early treatment are the keys to improving the cure rate and reducing the mortality rate.
Previous studies have demonstrated that microRNAs (miRNAs) are involved in the pathogenesis of human cancers by suppressing the expression of target genes [3]. In RB, different functions of miRNAs as oncogenes or tumor suppressors have been reported. For example, overexpression of miR-497 repressed cell proliferation, invasion and migration in RB by targeting VEGFA [14]. In contrast, miR-125b promoted RB growth through suppressing DRAM2 [2]. Recently, the function of miR-98 in different human cancers has drawn our attention. Yang et al. reported that miR-98 inhibited proliferation and invasion of non-small cell lung cancer (NSCLC) cells through regulating PAK1 [37]. Moreover, low miR-98 expression has been identified as a poor prognostic biomarker for patients with NSCLC [30]. In addition, the inhibitory effect of miR-98 has also been found in hepatocellular carcinoma [15], pancreatic ductal adenocarcinoma [8] and glioma [31]. However, the role of miR-98 is unclear and needs to be investigated in RB.
As a member of the high-mobility group protein family, HMGA2 (high-mobility group A protein 2) is a chromatin remodeling factor that binds to AT-rich regions in DNA [9]. Upregulation of HMGA2 was identified in NSCLC [19] and breast cancer [22]. Moreover, increased expression of HMGA2 was related to a malignant phenotype in pancreatic exocrine tissue [1]. As an oncogene, HMGA2 is a driving factor for tumor metastasis [20]. Moreover, upregulation of HMGA2 promoted metastasis of colorectal cancer and affected survival [33]. More importantly, HMGA2 has been found to be involved in different signaling pathways. The Wnt/
In the present study, we are focusing on exploring whether miR-98/HMGA2 axis can regulate cell proliferation, invasion, migration and epithelial – mesenchymal transition (EMT) in RB through mediating the Wnt/
Materials and methods
Clinical tissues
The RB tissues and normal retina tissues (located
MiR-98 expression was decreased in RB tissues. (A) The expressions of miR-98 in RB tissues. (B) Low miR-98 expression was related to shorter overall survival of RB patients. *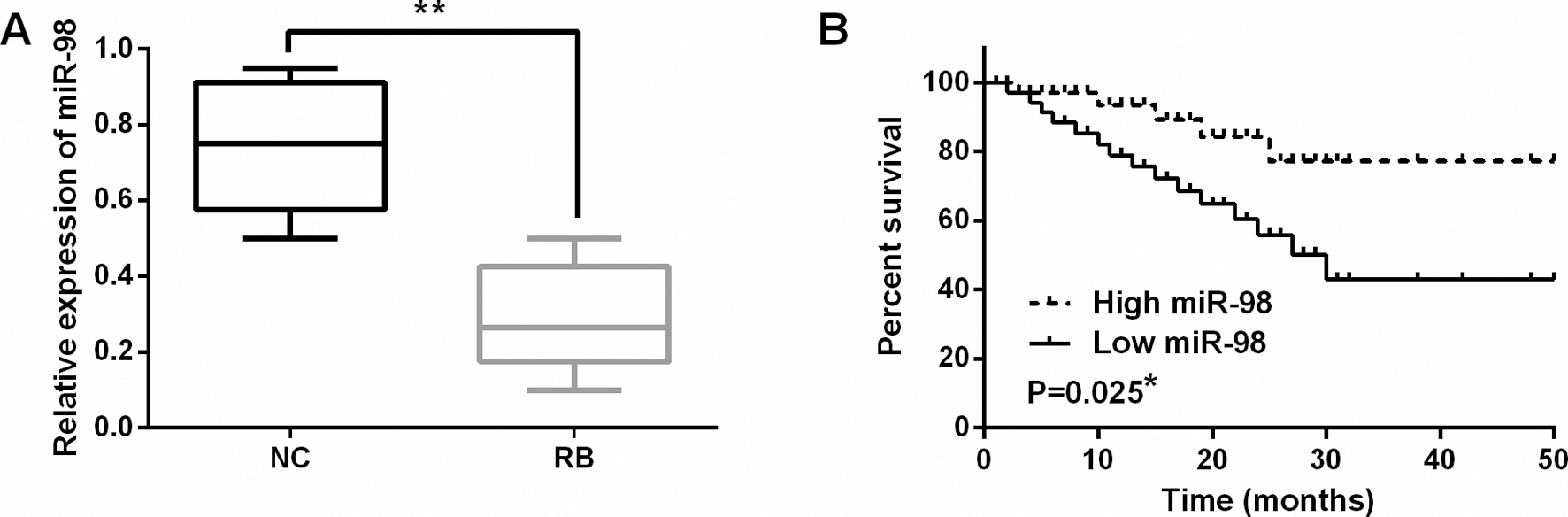
The normal human retinal pigment epithelial cell ARPE-19 and WERI-Rb-1, Y79 cell lines were used for this experiment. These cell lines were obtained from ATCC (Manassas, VA, USA). In RPMI-1640 medium (Gibco, Grand Island, NY, USA), the cells were then seeded with 10% heat-inactivated fetal bovine serum (FBS), 100 U/l penicillin, and 100 U/l streptomycin. The cell lines were incubated in a humidified atmosphere of 95% air and 5% CO
Cell transfection
MiR-98 mimics or inhibitor and negative control (NC) were acquired from Genepharma Co., Ltd (Shanghai, China). HMGA2 siRNA and negative control siRNA were also purchased from Genepharma (Shanghai, China). They were separately transferred to Y79 cells by using Lipofectamine 2000 (Invitrogen, Carlsbad, USA).
Quantitative reverse transcription PCR (qRTPCR)
Total RNA was extracted in RB tissues using TRIzol
Cell counting kit-8 (CCK-8) assay
The CCK-8 assay was used to assess cell viability in RB. First, 3
Transwell assay
Transwell chambers (Millipore) were used to examine cell migration and invasion. First, 5
Dual luciferase reporter assay
The relationship between miR-98 and HMGA2 was verified by dual luciferase reporter assay (Promega Corporation, Madison, WI, USA). The DNA oligonucleotides containing the wild-type 3’-UTR of HMGA2 or the mutant 3’-UTR of HMGA2 were synthesized and cloned into the pmirGLO dual-luciferase miRNA expression vector (Promega Corporation, WI, USA). Next, Y79 cells were co-transfected with the miR-98 mimics and above vectors (Promega Corporation). After 48 h, the firefly and renilla luciferase activities were measured by a dual-luciferase reporter assay system (Promega, Madison, WI, USA) [36].
Western blot analysis
Protein samples were extracted using RIPA lysis buffer (50 mM Tris (pH 7.4), 150 mM NaCl, 1% NP-40, 0.5% sodium deoxycholate). Next, proteins were separated by 10% SDS-PAGE and incubated with 5% skim milk in PVDF membranes at room temperature. Membranes were then incubated with HMGA2, GAPDH, Wnt/
Statistical analysis
Differences between groups were analyzed by SPSS 19.0 using one-way ANOVA with Bonferroni post hoc test. The correlation between miR-98 and clinical features of RB patients was calculated by Chi-squared test. Overall survival rates and survival differences were detected by univariate Kaplan-Meier method with log-rank test. Data were expressed as mean
Relationship between miR-98 expression and their clinic-patholo- gical characteristics of RB patients
Relationship between miR-98 expression and their clinic-patholo- gical characteristics of RB patients
Statistical analyses were performed by the
MiR-98 overexpression inhibited cell proliferation, migration and invasion in RB. (A) The miR-98 expression was detected in WERI-Rb-1, Y79 and ARPE-19 cell lines. (B) MiR-98 expression regulated by miR-98 mimics or inhibitor was examined in Y79 cells. (C, D) MiR-98 mimics or inhibitor regulated the cell proliferation. (E, F) Cell migration and invasion were regulated by miR-98 mimics or inhibitor in RB. *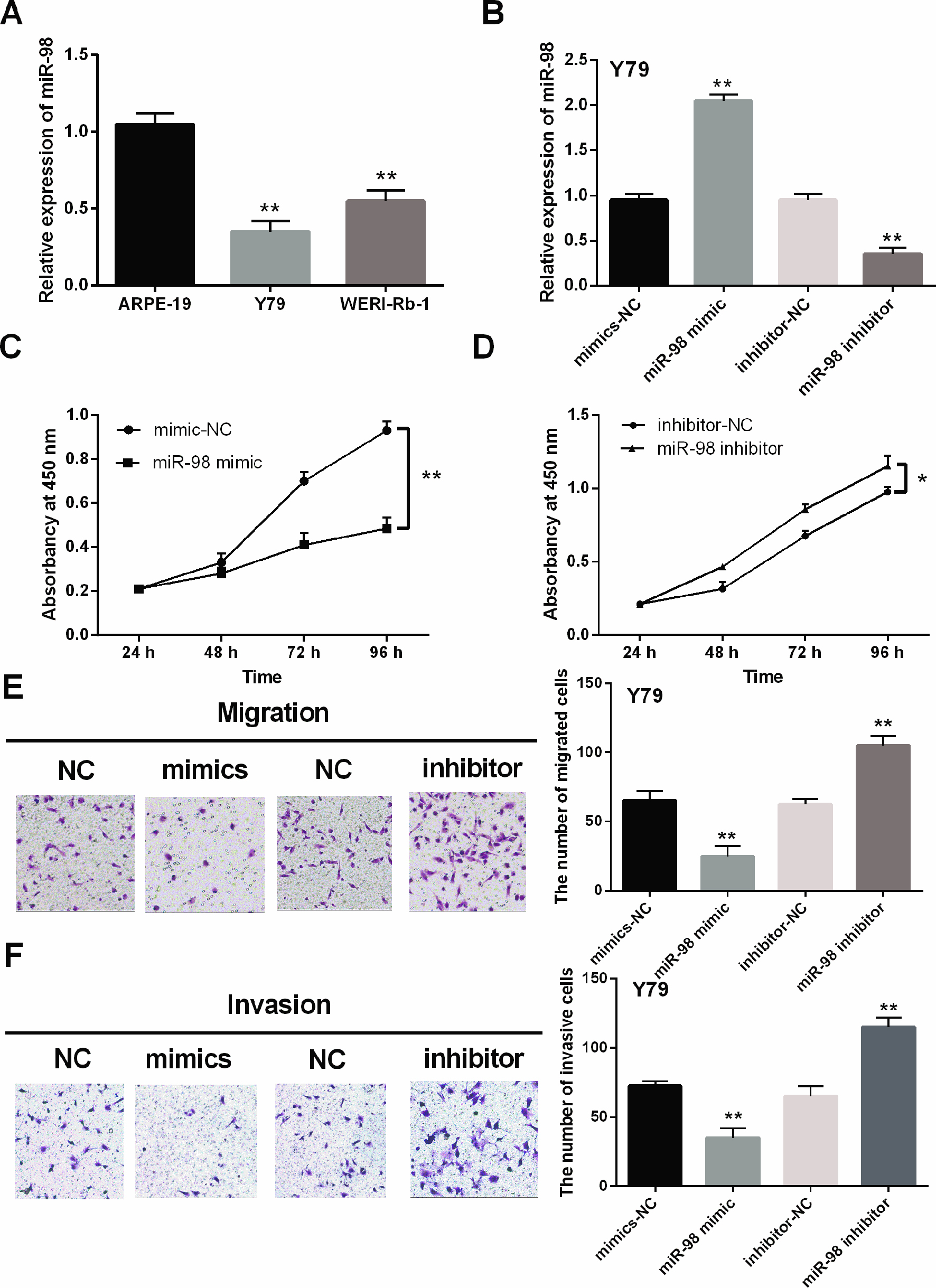
HMGA2 was a direct target gene of miR-98. (A) HMGA2 had binding sites with miR-98. (B) Luciferase reporter assay (C) MiR-98 had negative correlation with HMGA2. (D, E) The expression of HMGA2 were observed in Y79 cells containing miR-98 mimics or inhibitor. **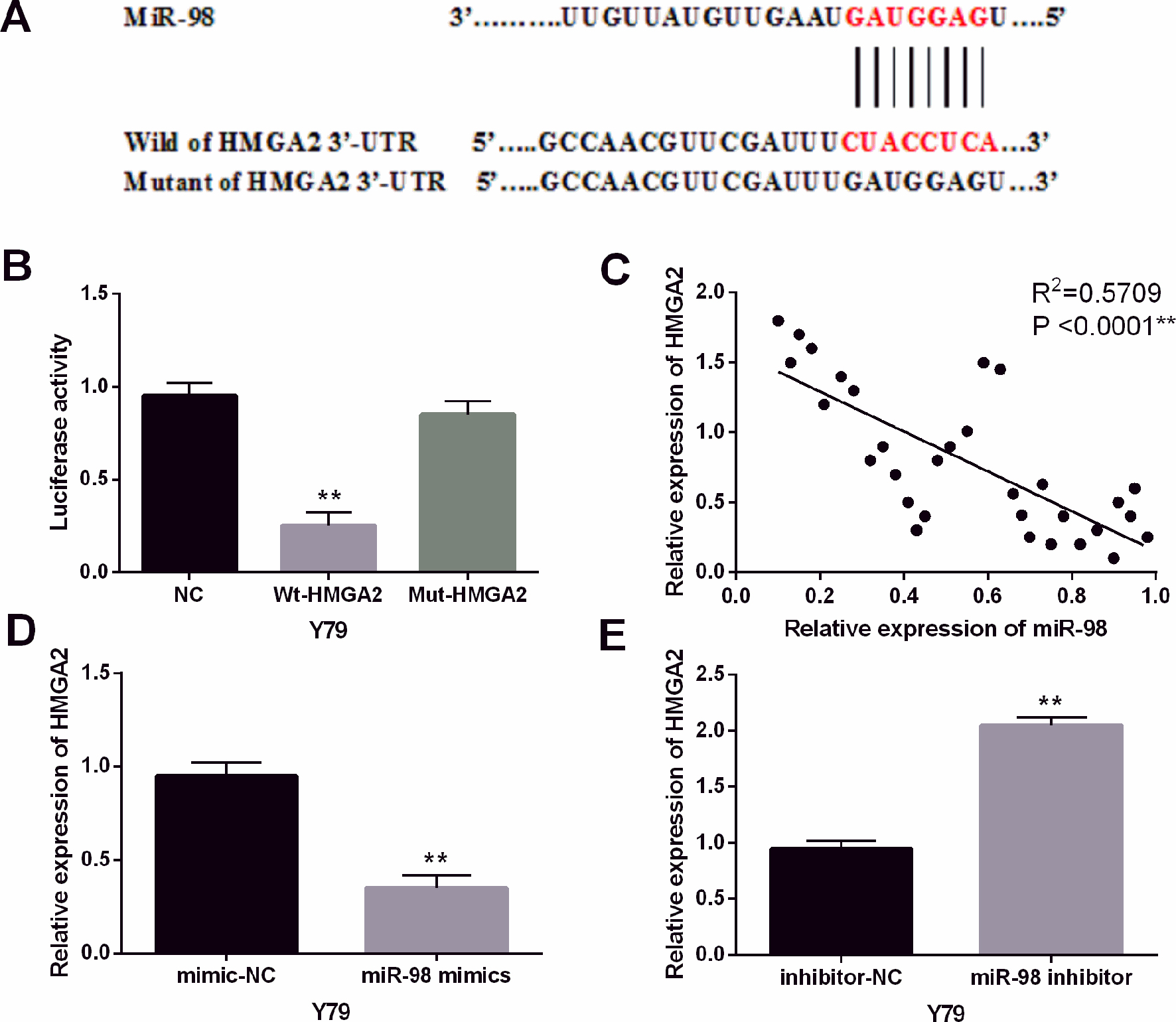
The knockdown of HMGA2 inhibited the development of RB. (A) The expressions of HMGA2 in RB tissues. (B) The HMGA2 expression in WERI-Rb-1, Y79 and ARPE-19 cell lines. (C) The expression of HMGA2 was examined in Y79 cells with HMGA2 siRNA. (D) HMGA2 siRNA regulated the cell proliferation in Y79 cells. (E, F) Cell migration and invasion analysis in cells containing HMGA2 siRNA. **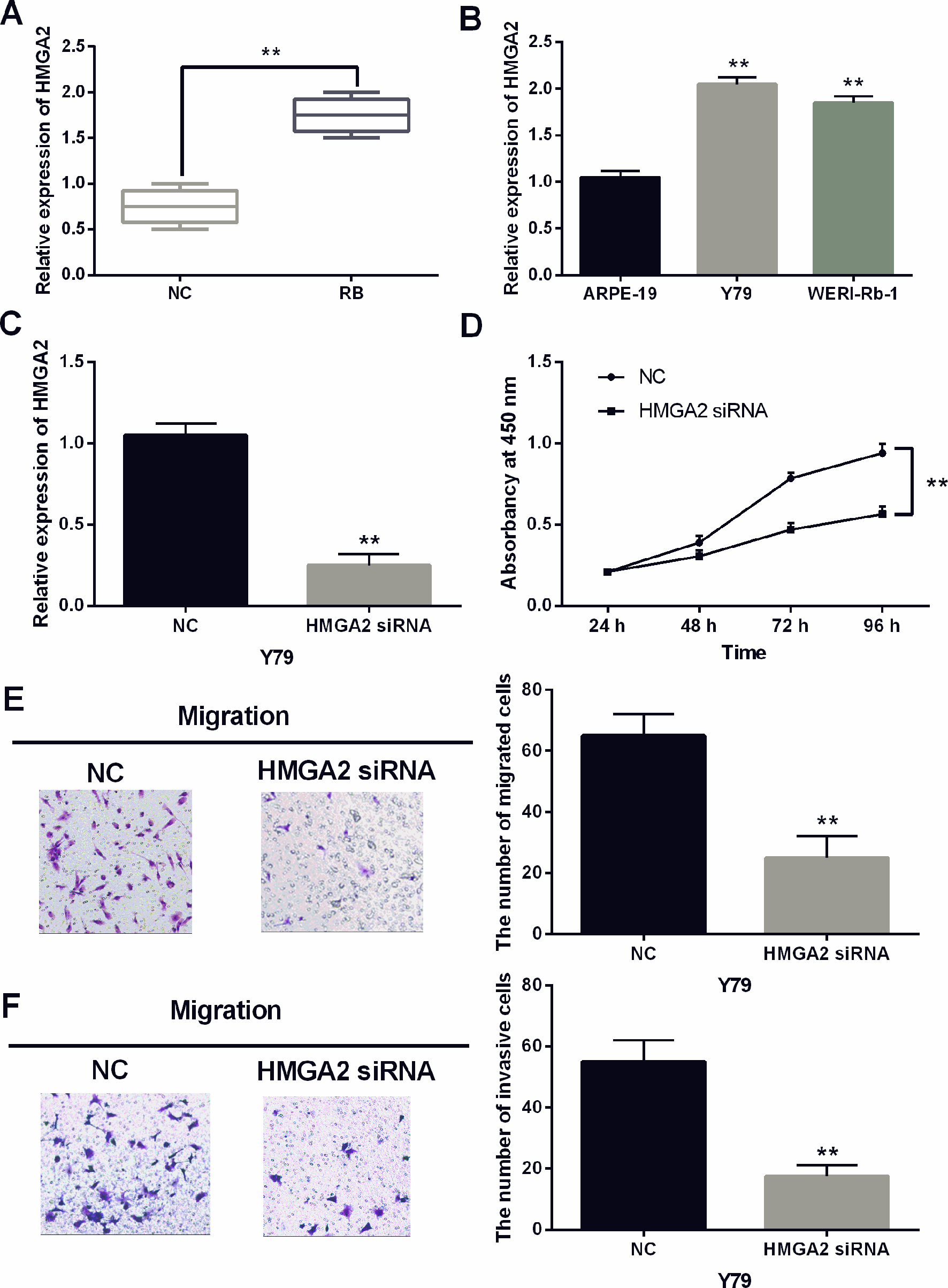
MiR-98 inhibited the progression of RB through targeting HMGA2. (A) The expression of HMGA2 was measured in Y79 cells with HMGA2 vector and miR-98 mimics. (B) The cell proliferation was measured in Y79 cells with HMGA2 vector and miR-98 mimics. (C, D) The cell migration and invasion in Y79 cells with HMGA2 vector and miR-98 mimics. **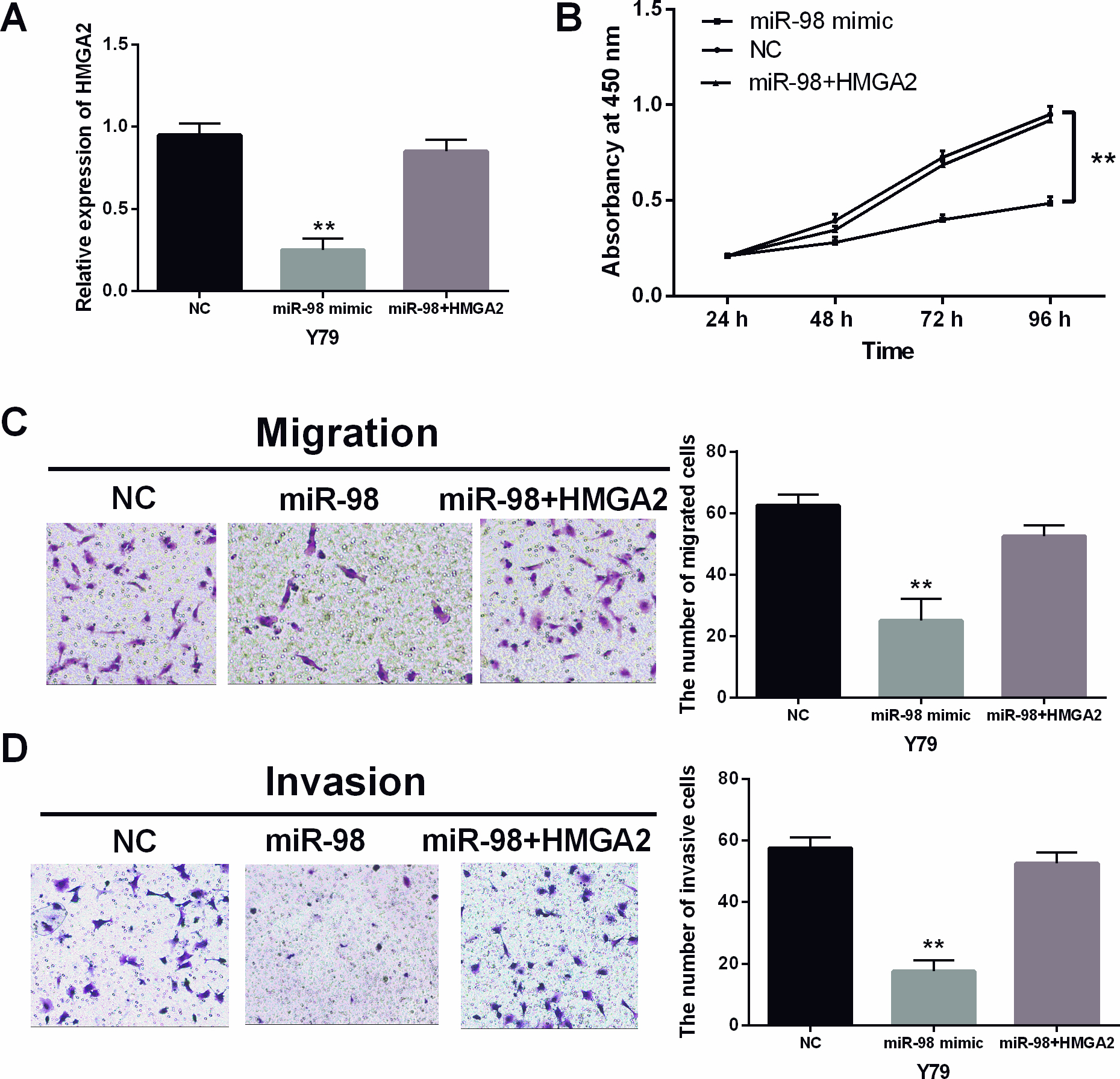
MiR-98 regulated the tumorigenesis of RB through blocking Wnt/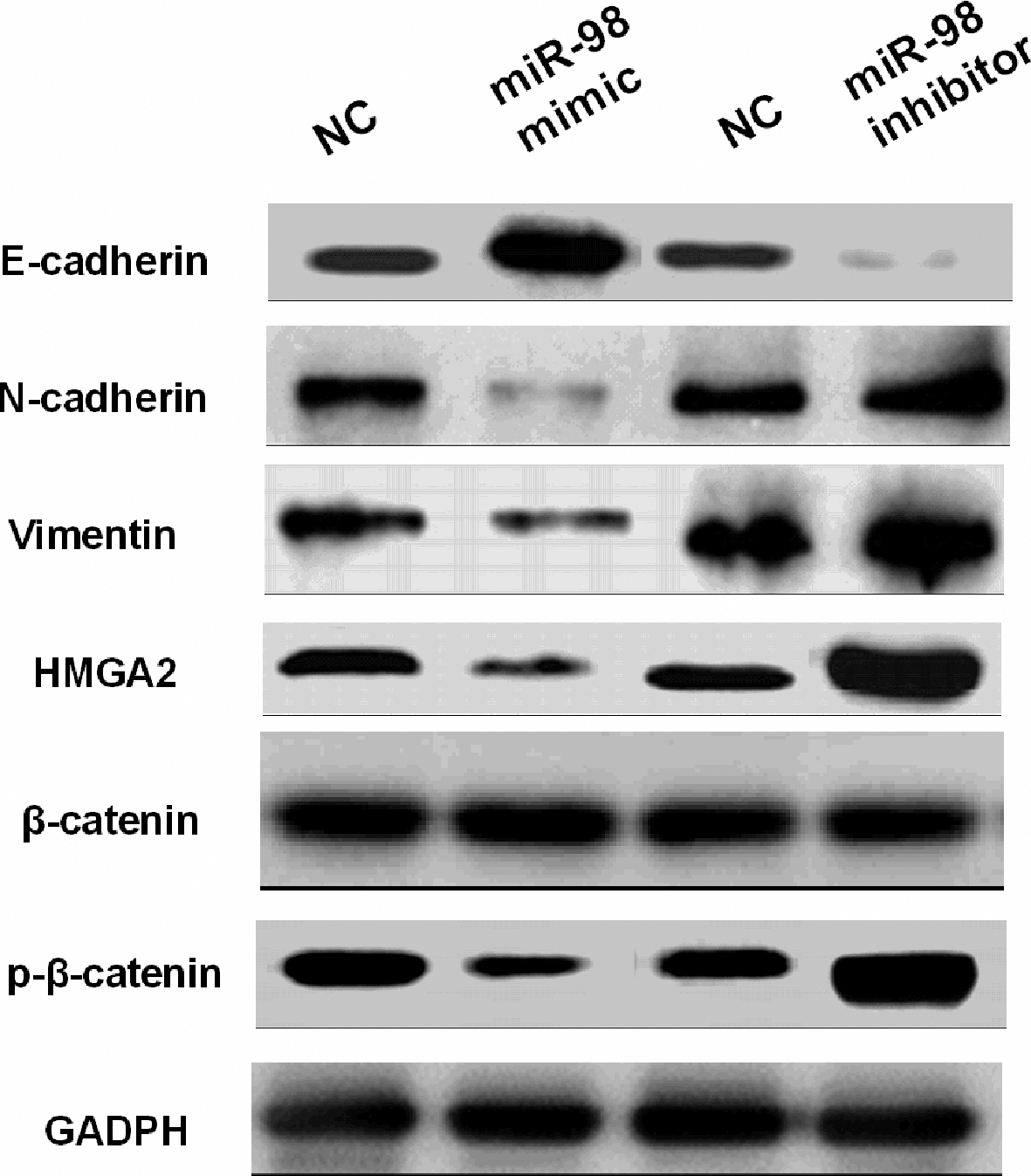
MiR-98 was downregulated in RB tissues
First, the alternation of miR-98 expression was observed in RB tissues by qRT-PCR assay. A lower miR-98 expression was detected in RB tissues than in normal tissues (
Upregulation of miR-98 inhibited cell proliferation, migration and invasion in RB
Next, expression of miR-98 was detected in WERI-Rb-1, Y79 and ARPE-19 cell lines. MiR-98 was also downregulated in WERI-Rb-1 and Y79 cells compared to ARPE-19 cells (
HMGA2 was a direct target gene of miR-98.
Further, theoretical target genes of miR-98 were search from TargetScan (
The knockdown of HMGA2 inhibited the development of RB
Subsequently, HMGA2 expression was observed in RB. Higher expression of HMGA2 was identified in RB tissues than in normal tissues (
MiR-98 inhibited the progression of RB through targeting HMGA2
MiR-98 mimics and HMGA2 vector were then co-transfected into Y79 cells to determine their interaction. The downregulation of HMGA2 induced by miR-98 mimics was recovered by HMGA2 vector in Y79 cells (
MiR-98 regulated the tumorigenesis of RB through blocking Wnt/
-catenin pathway
Finally, the effects of miR-98 on EMT and Wnt/
Discussion
RB is highly malignant, affecting 8,000 children each year, which usually leads to intracranial metastasis [26]. The pathogenesis of RB is a complex process associated with many miRNAs and genes, such as miR-183/LRP6 [29], miR-22/ENO1 [16] and miR-145/ADAM19 [27]. In particular, it had been reported that miR-98 reduced the HMGA2 expression and promoted osteogenic differentiation of mesenchymal stem cells [10]. In head and neck squamous cell carcinoma, HMGA2 had been identified as a target gene of miR-98 [12]. Therefore, the aim of this study was to investigate the role of miR-98 in RB. MiR-98 regulated proliferation, invasion and migration of RB cells by modulating HMGA2 in an attempt to identify new targets for RB therapy.
This study demonstrated the downregulation of miR-98 and upregulation of HMGA2 in RB. HMGA2 was confirmed to be a direct target gene of miR-98. It had been reported that miR-98 negatively regulated HMGA2 expression [35]. A negative correlation between miR-98 and HMGA2 was also identified in RB. Furthermore, low miR-98 expression was related to a shorter overall survival rate in RB patients. Similarly, Wang et al. revealed that low miR-98 expression was associated with poor prognosis in NSCLC patients [30]. In addition, high expression of HMGA2 was identified in RB and miR-98 was negatively correlated with HMGA2 in RB tissues. Similarly, upregulation of HMGA2 was also detected in colorectal cancer, triple-negative breast cancer and osteosarcoma, which was negatively regulated by some miRNAs [7, 11, 28]. Previous studies have further supported our results that miR-98 could directly target HMGA2.
Functionally, overexpression of miR-98 inhibited cell proliferation, invasion and migration in RB by targeting HMGA2. Similarly, miR-98 was reported to inhibit migration, invasion and proliferation of NSCLC cells through targeting ITGB3 [21]. In this study, EMT makers (E-cadherin, N-cadherin and Vimentin) associated with cell metastasis were regulated by miR-98 in RB. We found that miR-98 blocked EMT to inhibit cell metastasis in RB. It was also demonstrated that miR-200c suppressed the migration of RB cells by reversing EMT [23]. Similarly, miR-98 inhibited migration, invasion, proliferation and EMT of HCC cells [39]. In addition, knockdown of HMGA2 inhibited cell proliferation, migration and invasion in RB. Shi et al. proposed that silencing of HMGA2 promoted apoptosis and inhibited cell migration and invasion in prostate cancer, consistent with our study [24]. Further, miR-98 was found to regulate tumorigenesis of RB through blocking the Wnt/
Conclusion
This study preliminarily demonstrated that miR-98 inhibited the development of RB by targeting HMGA2 and mediating the Wnt/
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
