Abstract
In this study, we investigated the underlying mechanism of the phosphoinositide 3-kinase/Akt– and Wnt/β-catenin-mediated promotion of epithelial-to-mesenchymal transition by epigenetic regulation of histone acetylation in gastric cancer. First, we used immunohistochemistry to detect the expression of phosphorylated Akt, phosphorylated glycogen synthase kinase 3 beta, and β-catenin in gastric cancer tissues and adjacent tissues. In addition, we confirmed that the phosphoinositide 3-kinase/Akt and Wnt/β-catenin signaling pathways were correlated with tumorigenesis, progression, and maintenance of gastric cancer using the phosphoinositide 3-kinase inhibitor LY294002 and an inhibitor of the β-catenin/TCF4 complex, FH535. Epithelial-to-mesenchymal transition–related gene expression was measured by western blotting and quantitative real-time polymerase chain reaction assays. Furthermore, we detected the acetylation of histone H3 lysine 4 and lysine 27 using the FH535 and LY294002 inhibitors at different concentrations for 24 and 48 h. Finally, chromatin immunoprecipitation–quantitative polymerase chain reaction was performed to detect the specific binding of H3K27ac to the promoter of the epithelial-to-mesenchymal transition–related factor, Twist. Taken together, abnormal activation of the phosphoinositide 3-kinase/Akt and Wnt/β-catenin signaling pathway was correlated with the gastric cancer progression and contributed to epithelial-to-mesenchymal transition regulation by controlling histone acetylation.
Introduction
Gastric cancer is one of the most aggressive and deadly tumors in the worldwide. The latest data showed that gastric cancer remains the second most common cause of morbidity in China and is a highly invasive and aggressive malignancy. 1 Pathological diagnoses for gastric cancer include early gastric cancer and advanced gastric cancer. 2 The major treatments for advanced gastric cancer are chemotherapy and molecularly targeted therapy; however, the treatment outcome is not optimistic. 3 Therefore, it is crucial to identify biomarkers of gastric cancer metastasis.
Epithelial-to-mesenchymal transition (EMT) is a process in which epithelial cells are converted to migratory and invasive cells. 4 EMT activation is closely associated with gastric cancer cell motility and invasiveness. 5 A group of EMT-inducing transcription factors (EMT-TFs) are functionally activated during the EMT program. Mesenchymal markers, such as N-cadherin, Snail, Slug, and Twist, activate EMT programs in epithelial cells. However, E-cadherin is an important epithelial factor, and loss of function of this protein may lead to the dissolution of adherens and tight junctions and contribute to cancer progression by increasing proliferation, invasion, and metastasis.6,7 As EMT is critical for the conversion of early-stage tumors into invasive malignancies, it is necessary to explore the molecular mechanism underlying the EMT process.
Abnormal activation of the phosphoinositide 3-kinase (PI3K)/Akt and Wnt/β-catenin pathways has been reported to participate in cancer progression and has been implicated in poor prognosis of gastric cancer, indicating it may be a potential therapeutic target for gastric cancer.8,9 Gastric cancer specimens showed that about 20%–30% gastric cancers presented nuclear β-catenin accumulation. 10 Activation of β-catenin is induced by multiple signaling pathways and associated with malignant transformation. 11 Akt-dependent phosphorylation leads to accumulation of β-catenin in the nucleus by inhibiting glycogen synthase kinase 3 beta (GSK-3β), which is an important component of the canonical Wnt pathway. 12 We previously showed that the PI3K/Akt and Wnt/β-catenin pathways regulated histone methylation using a Wnt inhibitor, FH535 and an AKT inhibitor, LY294002. 13
Histone modification, type of epigenetic modification, is a post-translational modification which includes methylation and acetylation mediated by histone-modifying enzymes.14,15 Histone acetylation is required for histone acetyltransferases (HATs). In addition, histone modification is associated with transcriptional activation or repression. For example, H3K27ac is hallmark of transcriptionally active genes.16,17 Thus, it is important to elucidate the relationship between histone modification and mechanisms of tumorigenesis.
However, the underlying mechanism of the PI3K/Akt and Wnt/β-catenin pathway–mediated promotion of EMT is not been fully eclucidated. Our study provides new insights into the epigenetic regulation of EMT by the PI3K/Akt and Wnt/β-catenin pathways.
Materials and methods
Tissue samples and clinical data collection
Human gastric cancer specimens and corresponding adjacent non-tumorous gastric tissue samples were obtained from 25 Chinese patients at the Tianjin Medical University General Hospital. All the cases were moderately differentiated or well differentiated and were at tumor–node–metastasis (TNM) stage I/II recommended by the American Joint Committee on Cancer (AJCC) in 2010. All patients did not receive chemotherapy or radiotherapy before the experiment. This study was approved by the Research Ethics Committee of Tianjin Medical University General Hospital, China. Written informed consent was obtained from all patients.
Cell culture and drug treatment
The human gastric cancer cell lines SGC-7901 and MGC-803 were purchased from China Academia Sinica (Shanghai, China). All cells were propagated in RPMI-1640 medium supplemented with 10% fetal bovine serum (FBS) and were maintained in a humidified atmosphere with 5% CO2 atmosphere at 37°C. We employed FH535 and LY294002 to separately inhibit the Wnt/β-catenin and PI3K/Akt signaling pathways, respectively. Wnt3a and epidermal growth factor (EGF) were added to the cell lines to activate the signaling pathways.
Western blot analysis
Total protein was extracted using radioimmunoprecipitation assay (RIPA) lysis buffer, and nuclear protein was extracted using a Nuclear and Cytoplasmic Protein Extraction Kit (Beyotime Biotechnology), according to the manufacturer’s protocols. Protein was measured using a standard bicinchoninic acid assay. Equal amounts of protein per lane were separated by 10% and 15% sodium dodecyl sulfate (SDS)–polyacrylamide gel electrophoresis and transferred onto polyvinylidene difluoride (PVDF) membranes. The membrane was blocked at 37°C for 1 h in 5% bovine serum albumin (BSA) or skim milk and then incubated with a specific antibody at 4°C for overnight. The antibodies used in this study were as follows: H3K4ac (1 µg/mL), H3K27ac (1 µg/mL), and β-catenin (1 µg/mL) antibodies were purchased from Abcam; E-cadherin (1 µg/mL), N-cadherin (1 µg/mL), Snail (1 µg/mL), Slug (1 µg/mL), and Twist (2 µg/mL) were from Cell Signaling Technology; glyceraldehyde 3-phosphate dehydrogenase (GAPDH; 0.2 µg/mL) was from Zhongshan Bio Corp.; and H3 (0.2 µg/mL) was from Ray Antibody. Protein expression was visualized using a SuperSignal protein detection kit (Pierce). The membrane was stripped and quantified after normalization with GAPDH and H3.
RNA isolation and quantitative real-time polymerase chain reaction
MGC-803 Cells were grown to ~60% confluence in six-well plates and were incubated with different concentrations of FH535 or LY294002 for 48 h. RNA was extracted from gastric cancer cells using TRIzol (Invitrogen). Equal amounts of RNA were converted into complementary DNA (cDNA) using E-cadherin, N-cadherin, Snail, Slug, Twist, and β-catenin primers. Real-time polymerase chain reaction (RT-PCR) was performed using a Reverse Transcription System (Promega) on a CFX96 Real-Time PCR Detection System (Bio-Rad Laboratories), according to the instructions. Results were normalized to the expression of GAPDH. Optimized PCR was performed as follows: 1 cycle of 95°C for 5 min; 38 cycles of 95°C for 40 s, 57°C for 40 s, and 72°C for 30 s; and a final extension at 72°C for 5 min. Each sample was analyzed in triplicate. The relative expression was calculated via the 2−ΔΔCt method.
Proliferation assay
Cells treated with drugs and dimethyl sulfoxide (DMSO) were plated on 96-well plates with six replicate wells at a cell density of 104 cells/well. Then, 10 µL of Cell Counting Kit-8 (CCK-8; 5 g/L; Sigma) was added into each well. and the cells were incubated for an additional 4 h at 37 °C. The optical density (OD) was measured at 450 nm, and the data are expressed as a percentage of the control and are derived from triplicate samples. For the colony formation assay, 4000 cells treated with drugs and DMSO were seeded onto six-well plates. The medium was replaced every other day for 10 days. The cells were stained with crystal violet prior to capturing the images via camera. The colony formation assay was repeated at least three times.
Cell invasion analysis
A matrigel-coated transwell membrane (Becton, Dickinson and Company) was used to perform transwell invasion assays. The matrigel solidified after 30 min of incubation at 37°C, and then, transfected cells (105) in 100 µL of serum-free RPMI-1640 medium were added to the upper compartment of the chamber. Then, 20% of FBS in RPMI-1640 medium was added to the lower chambers of the wells as a chemoattractant. The cells were incubated for 48 h at 37°C in 5% CO2, and cells on the lower surface were stained with crystal violet. The average number of cells per field was calculated by counting cells in six random fields per well.
Wound-healing assay
Equal numbers of each group of cells (1 × 105) were seeded onto six-well plates. When cell confluence reached approximately 90%, an artificial homogeneous wound was made in the monolayer using a sterile plastic 200 µL micropipette tip. Debris was removed by washing the cells with phosphate-buffered saline (PBS). After 12, 24, and 36 h, the cells that had migrated into the wounded area were photographed at 200× magnification under a light microscope.
Chromatin immunoprecipitation
To test whether H3K27ac bound to the Twist promoter, chromatin immunoprecipitation (ChIP) was performed using the EZ-ChIP kit (Millipore) according to the manufacturer’s protocol. In brief, cross-linked chromatin was sonicated into 200–1000 bp fragments, and Protein G Agarose was added to the antibody/chromatin complexes and incubated overnight at 4°C. The chromatin was immunoprecipitated using anti-H3K27ac (1 µg/mL; Abcam) antibodies. PolyII was used as a positive control. Normal human immunoglobulin G (IgG) was used as a negative control. The Protein G Agarose–antibody/chromatin complex was resuspended in wash buffer and centrifuged to collect the protein/DNA complex. The protein/DNA cross links were reversed to obtain free DNA. Quantitative PCR (qPCR) was conducted according to the method described above to compare the expression of Twist among different groups.
Immunohistochemical staining
For staining, 5-µm sections were cut, dried, and deparaffinized, and immunostaining was performed on the sections using the avidin–biotin complex method. Primary antibodies specific for phosphorylated Akt (p-Akt), p-GSK-3β, and β-catenin (1:100 dilution; Abcam) were diluted in PBS with 0.1% Tween 20 and incubated overnight at 4°C. Finally, the slides were incubated with a secondary antibody (1:100 dilution; Zhongshan Biology) at 37°C for 1 h, followed by incubation in avidin–biotin complex solution for an additional 1 h. Protein expression was detected by incubating with 3,3′-diaminobenzidine (DAB) buffer, and the sections were counterstained with hematoxylin. All images were captured via microscopy (Olympus). The immunohistochemistry score was assessed according to percentage of positive cells: Five high power fields were randomly selected and the percentage of positive cells was used as tagged value of p-Akt/p-GSK-3β/β-catenin, positive cells/cells >10% was marked positive (+); positive cells ≤10% was marked negative (−).
Statistical analysis
The statistical significance of differences between different groups was determined using Student’s t test, while one-way analysis of variance (ANOVA) was used to determine the difference among at least three groups. p values < 0.05 were regarded as significant.
Results
Abnormal activation of the PI3K/Akt and Wnt/β-catenin signaling pathways in gastric cancer
To investigate the activation of the PI3K/Akt and Wnt/β-catenin signaling pathways in gastric cancer progression, we detected the p-Akt, p-GSK-3β, and β-catenin expression levels in 25 paired gastric cancer tissues and corresponding non-tumor tissues by immunohistochemistry. The results showed that the expression of p-Akt, p-GSK-3β, and β-catenin was significantly increased in the gastric cancer tissues compared with the non-tumor tissues (Figure 1(a), Table 1). Next, we examined the transcript levels of p-Akt and p-GSK-3β using quantitative RT-PCR (qRT-PCR), and normalizing to GAPDH. The messenger RNA (mRNA) levels of p-Akt and p-GSK-3β were significantly upregulated in the cancerous tissues compared with non-tumor tissues (p < 0.01, Figure 1(b)). Based on the results above, PI3K/Akt and Wnt/β-catenin signaling pathways were activated in gastric cancer.
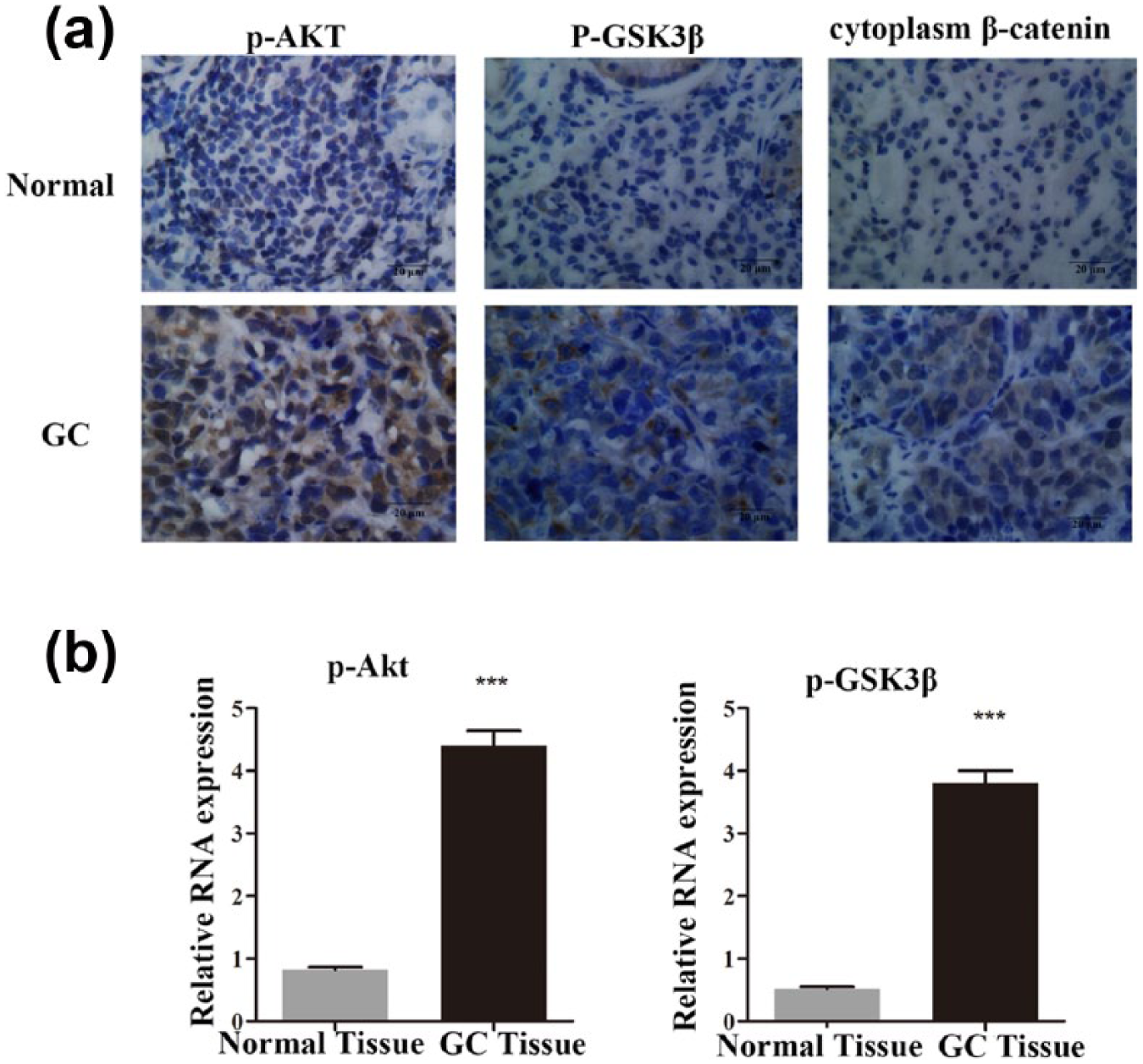
Abnormal activation of the PI3K/Akt and Wnt/β-catenin signaling pathways in gastric cancer. (a) Immunohistochemical analysis of the expression of p-Akt, p-GSK-3β, and β-catenin in 25 paired gastric cancer tissues and corresponding non-tumor tissues. (b) The transcript levels of p-Akt and p-GSK-3β were upregulated in gastric cancer tissues as determines by qRT-PCR.
Immunostaining analysis revealed the expression levels of p-Akt, p-GSK-3β, and β-catenin in normal tissues and gastric adenocarcinoma.

p-GSK-3β: phosphorylated glycogen synthase kinase 3 beta; N: normal; A: adenocarcinoma; IHC: immunohistochemistry.
FH535 and LY294002 inhibit invasion and tumorigenesis in gastric cancer cells by inactivating the PI3K/Akt and Wnt/β-catenin pathways
Based on the results above, we investigated the effect of FH535 18 and LY294002 19 on the invasion, metastasis, and proliferation of gastric cancer cells in vitro. First, we performed transwell assays and wound-healing assays and the results revealed that inhibition of PI3K/Akt and Wnt/β-catenin signaling pathways decreased MGC-803 and SGC-7901 cell invasion and migration, respectively (Figure 2(a) and (b)). In addition, a colony formation assay indicated that treatment with FH535 and LY294002 significantly reduced colonies of the gastric cancer cells (Figure 2(c)). Furthermore, a CCK-8 assay was used to examine the effects of FH535 and LY294002 on the proliferation of SGC-7901 and MGC-803, respectively, cells in vitro. We estimated the proliferation rate of gastric cancer cells at 0, 24, 36, and 48 h, compared with the negative control. FH535 and LY294402 notably repressed the growth of gastric cancer cells (Figure 2(d)). Taken together, these results indicate that the PI3K/Akt and Wnt/β-catenin signaling pathways promote invasion and metastasis, and their upregulation may facilitate the proliferation of gastric cancer cells.

FH535 and LY294002 inhibit the invasion and tumorigenesis of gastric cancer cells by inactivating the PI3K/Akt and Wnt/β-catenin pathways. (a) Transwell assays indicated that FH535 and LY294002 inhibited invasion of SGC-7901 and MGC-803 cells (p < 0.05). (b) Wound0healing assays indicated that FH535 and LY294002 inhibited cell migration in SGC-7901 and MGC-803 cells (p < 0.05). (c) Colony formation assay indicated that FH535 and LY294002 significantly reduced the cell growth of SGC-7901and MGC-803 cells compared with the negative control (p < 0.05). (d) FH535 and LY294002 suppressed the proliferation of SGC-7901 and MGC-803 cells as determined by a CCK-8 assay. (p < 0.05)
PI3K/Akt and Wnt/β-catenin pathways regulated gastric cancer invasion in vitro
Since EMT is characterized by increased cell migration and invasiveness, 20 we evaluated the effect of the PI3K/Akt and Wnt/β-catenin signaling pathways on gastric cancer cell–associated EMT processes. Our research showed that treatment with FH535 and LY294002 was accompanied by a change in EMT. Consequently, we detected the mRNA and protein expression levels of molecular markers of EMT following FH535 and LY294002 treatment. The expression of E-cadherin was increased; in contrast, the mesenchymal markers including N-cadherin, Snail, Slug, Twist, and β-catenin were decreased in the FH535-/LY294002-treated cells (Figure 3(a)–(c)). These results confirmed that the PI3K/Akt and Wnt/β-catenin signaling pathways play a crucial role in gastric cancer cell invasion and migration by regulating EMT.

The PI3K/Akt and Wnt/β-catenin pathways regulated gastric cancer invasion in vitro. (a and b) FH535 and LY294002 treatment induced E-cadherin expression and inhibited N-cadherin, Snail, Slug, Twist and β-catenin expression in gastric cancer cells as determined by western blotting. GAPDH was used as a control. (c) qRT-PCR indicated that treatment with FH535 and LY294002 increased the level of E-cadherin and decreased the levels of N-cadherin, Snail, Slug, Twist, and β-catenin in MGC-803 cells. All experiments were performed in triplicate with three technical replicates.
The PI3K/Akt and Wnt/β-catenin pathways regulate histone acetylation
Our previous study demonstrates that the PI3K/Akt and Wnt/β-catenin signaling pathways regulate histone modification. 13 In this study, we explored the role of the PI3K/Akt and Wnt/β-catenin signaling pathways in regulating histone acetylation. We performed western blotting to examine the levels of H3K4ac and H3K27ac. In MGC-803 and SGC-7901 gastric cancer cells, the H3K4ac and H3K27ac levels were decreased with 20 µM FH535 and 40 µM FH535 when treated for 24 h. Furthermore, the changes were consistent with the prolongation of the FH535 treatment, indicating that there was no significant change in a dose- and time-dependent way (Figure 4(a)). By contrast, the levels of H3K4ac and H3K27ac were increased by activating the Wnt/β-catenin signaling pathways with the treatment of Wnt3a (Figure 4(c)). For the PI3K/Akt signaling pathway, Ly294002 decreased H3K4ac and H3K27ac at concentrations of 10 and 20 µM, respectively. The gastric cancer cell lines MGC-803 and SGC-7901 were treated with LY294002 for 24 and 48 h (Figure 4(b)). However, an upstream activator of the AKT pathway, EGF, increased the levels of H3K4ac and H3K27ac (Figure 4(d)). Thus, both the PI3K/Akt and Wnt/β-catenin signaling pathways increased the levels of H3K4ac and H3K27ac but not in a dose- or time-dependent manner.

The PI3K/Akt and Wnt/β-catenin pathways regulated histone acetylation. (a) The expression levels of H3K4ac and H3K27ac were assessed by western blot analysis following FH535 treatment (20 µM and 40 µM). Histone H3 was used as the nuclear extraction loading control. (b) Treatment with LY294002 (10 µM and 20 µM) decreased the levels of H3K4ac and H3K27ac. (c) Wnt3a (100 ng/mL) activated the Wnt/β-catenin pathway to affect histone acetylation. (d) EGF regulated the PI3K/Akt pathway to increase the levels of histone acetylation.
Epigenetic regulation of Twist expression by the acetylation of H3K27
To determine the underlying mechanism by which the transcription of EMT markers was affected by the PI3K/Akt and Wnt/β-catenin signaling pathway–mediated H3K27 acetylation, we performed ChIP coupled with qPCR analysis (ChIP-qPCR) to detect specific binding to the promoter of EMT markers. First, Genome Browser showed that H3K27ac was enriched in the promoter region of the Twist gene (Figure 5(a)). In addition, ChIP-qPCR results showed that FH535 decreased the binding of H3K27ac to the Twist promoter in gastric cancer cells (Figure 5(b)). Furthermore, H3K27ac level in the promoter of Twist decreased following LY294002 treatment. H3K27ac is an active histone marker, and the results above suggest that Twist was decreased by the suppressive binding of H3K27ac to the Twist promoter, and was induced by inhibition of the PI3K/Akt and Wnt/β-catenin signaling pathways.

Epigenetic regulation of Twist expression by acetylation of H3K27. (a) Genome Browser analysis of H3K27ac enrichment peaks in the Twist promoter region. (b) ChIP demonstrated that the level of H3K27ac in the Twist promoter was decreased following FH535 treatment (p < 0.01). (c) ChIP demonstrated that the level of H3K27ac in the Twist promoter was decreased following LY294002 treatment (p < 0.01). H3K27ac binding to the Twist promoter was assessed by ChIP assays followed by SYBR Green quantitative PCR as described in experimental procedures.
Discussion
Gastric cancer has one of the high morbidity and mortality rate worldwide. Although diverse therapy is available for the treatment of gastric cancer, lots of patients with advanced gastric cancer who undergo surgical resection still develop local or distant metastasis and ultimately die from the cancer. 21 Therefore, it is urgent to explore effective and personalized treatments and to identify biomarkers that will improve the rate of early diagnosis. Our previous study has shown that abnormal activation of the PI3K/Akt and Wnt/β-catenin signaling pathways is related to tumorigenesis of gastric cancer and regulates histone methylation. In this study, we investigated the relationship between histone acetylation and the PI3K/Akt and Wnt/β-catenin signaling pathways in gastric tumorigenesis.
It has been reported that Wnt1 was correlated with the expression of cancer stem cell (CSC) marker CD44, which indicated that Wnt/β-catenin signaling improves the proliferation of gastric CSCs. 11 Moreover, trefoil factor 1 (TFF1) activates β-catenin to enhance cell proliferation through the regulation of the protein phosphatase 2A (PP2A) activity upstream of the AKT and GSK-3β signaling in gastric cancer. 8 The PI3K/Akt and Wnt/β-catenin signaling pathways are involved in the development of gastric cancer.22 –24 The activation of the β-catenin has been reported as an early initiating event in gastric tumorigenesis. 25 We initially observed that p-Akt, p-GSK-3β, and β-catenin were overexpressed in a series of advanced gastric adenocarcinoma specimens and found that the Akt and Wnt pathways were correlated with gastric tumorigenesis. PI3K can interact with AKT molecules inactivating of GSK-3β, which is a critical component of the Wnt pathway and may lead to accumulation of β-catenin in the nucleus, one of the hallmarks for the initiation of the EMT.26 –28 EMT is involved in a minority of cancer cells, including gastric cancer. 29 During the invasion and metastasis of gastric cancer, cells lose epithelial traits, such as E-cadherin, and increase the expression of mesenchymal markers, including N-cadherin, Snail, Slug, Twist, and β-catenin. 30 In this study, we performed western blotting and qRT-PCR analyses and found decreased expression of EMT mesenchymal markers with the Wnt inhibitor FH535 and Akt inhibitor LY294002 in gastric cancer cell lines, indicating that EMT is induced by the Akt and Wnt signaling pathways.
It has been discovered that Twist is overexpressed in gastric cancer and correlated with epigenetic regulation. 31 Previous study demonstrated that DNA methylation, histone modification in complex with Sp1 binding to CpG-rich regions of Twist within the exon 1 region to regulate Twist1 transcription in cells. 32 In our study, we focus on the relationship between Twist and histone modification, which is a type of epigenetic regulation and is independent of alterations in the DNA sequence but causes heritable changes in a cellular phenotype. The first described histone modification, histone acetylation, is associated with actively transcribed genes, indicating a role for histone acetylation in facilitating transcription.33,34 In addition, many reports have demonstrated that histone acetylation is dysregulated in cancer. Thus, we next explored the relationship between the activities of the Akt and Wnt signaling pathways and histone acetylation. The results of western blot analyses, as shown in Figure 4, demonstrated that treatment with FH535 and LY294002 decreased the acetylation of H3K4 and H3K27, which indicates that activation of the Akt and Wnt signaling pathways increased the H3K4 and H3K27 acetylation. Based on the results above, we performed ChIP to analyze the expression of H3K27ac and showed that it was decreased with FH535 and LY294002 treatment at the promoter of Twist, which is a crucial gene for stimulation of EMT.35,36 Therefore, we hypothesized that epigenetic regulation was an approach of the Akt and Wnt signaling pathways inducing EMT by increasing the binding of histone to the promoter of Twist.
In our study, we showed that the PI3K/Akt and Wnt/β-catenin signaling pathways promoted EMT by epigenetically activating the expression of Twist in gastric cancer. Although we validated the effects of the Akt and Wnt signaling pathways on histone acetylation, the exact mechanism requires further investigation. In addition, numerous changes in histone modification are involved in the progression of cancer, 37 and our findings are only the beginning. In conclusion, our study provides new insight into the PI3K/Akt and Wnt/β-catenin signaling pathway–mediated promotion of EMT by epigenetic regulation, indicating that combined therapy of signal inhibitors and epigenetic regulation could be a strategy in the future.
Footnotes
Acknowledgements
Y.S. and Z.-x.L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was supported by funds from National Natural Science Foundation of China (No. 81172356).
