Abstract
BACKGROUND:
Glioma is the most common primary malignant tumor in the nervous system.
OBJECTIVE:
To investigate the expression of long non-coding RNA Pvt1 oncogene (PVT1) in glioma and its clinical significance.
METHODS:
The expression levels of PVT1 were determined in 59 glioma and 10 normal tissue samples using qRT-PCR. The patients were divided into high and low expression groups and analyzed for their relationship with clinicopathological factors and the survival time using the Kaplan-Meier method.
RESULTS:
The expression levels of PVT1 were significantly higher in glioma tissues than in normal tissues (
CONCLUSIONS:
PVT1 is highly expressed in gliomas and its level is positively related to WHO glioma grade and prognosis of gliomas. Therefore, it may be explored as a new molecular marker for predicting malignancy and prognosis of gliomas.
Background
Glioma is the most common primary malignant tumor in the nervous system, accounting for 40%–60% of all intracranial tumors [1]. Despite rapid advances in surgical, radiotherapy, and chemotherapy techniques for tumor treatments, the median survival time for glioma patients is still only 9 to 12 months [2]. Therefore, identification for new molecular biomarkers of glioma is of great significance in determining the molecular mechanism of the occurrence and development of glioma, designing rational individualized treatments, and judging the prognosis. Long non-coding RNA (LncRNA) is nucleotide of 200nt long. Recent expression profile studies showed that there are significant differences in the expression of LncRNAs between glioma and normal brain tissues [3]. Subsequently, LncRNA is found to be related to the genesis, development and differentiation of glioma [4]. Maher et al. suggested that abnormal expression of LncRNAJi is one of the key factors that lead to increased tumor cell proliferation, invasion and metastasis [5].
The transcript of plasmacytoma variant translocation gene 1 (PVT1), an intergenic LncRNA, was first discovered in mice in 1984 [6]. It has been confirmed that this gene is closely related to lung cancer, breast cancer, liver cancer and colon cancer [7]. However, its relationship with glioma is largely unknown. Therefore, we studied the expression of the gene and analyzed its relationship with tumor grade, clinicopathological factors and prognosis. The findings would help better diagnosis and treatment of the tumor.
Materials and methods
Tissue samples
Ninety-seven tissue samples (fresh tissues, flash-frozen in liquid nitrogen and stored at
Reagents
TRIZOL reagents were purchased from Invitrogen, USA; Prime Script
Real-time quantitative PCR for gene expression
Total RNA was extracted from tissue samples using the TRIZOL reagents and reversely transcripted to cDNA using Prime Script
The data were managed using the Applied Biosys- tems software RQ Manager v1.2.1. Relative expression was calculated by using comparative Ct method and obtaining the fold change value (2-
Statistical analysis
Data were expressed as means
Results
PVT1 level increased with tumor grade
qRT-PCR results showed that PVT1 levels in normal, low and high grade glioma tissues were 4.96
Relative PVT1 levels in normal and gliomas tissues (A) and ROC curve for PVT1 mRNA expression based on the RT-qPCR data (B). The circle shows the point of the highest Youden (Y) index.
We then analyzed the relationship between PVT1 expression and pathological factors. For this purpose, the glioma patients were divided into low and high expression groups based on their PVT1 level. The results showed that the levels were only associated with tumor grade, but not with tumor size, gender, age or Karnofsky performance status (KPS) (Table 1).
Relationship between PVT1 level in glioma tissues and clinicopathological factors
Relationship between PVT1 level in glioma tissues and clinicopathological factors
When the PVT1 level was analyzed for its relationship with patient’s survival time, it was found that patients with high PVT1 level had shorter survival time (
Survival curve of patients with low and high PVT1 level.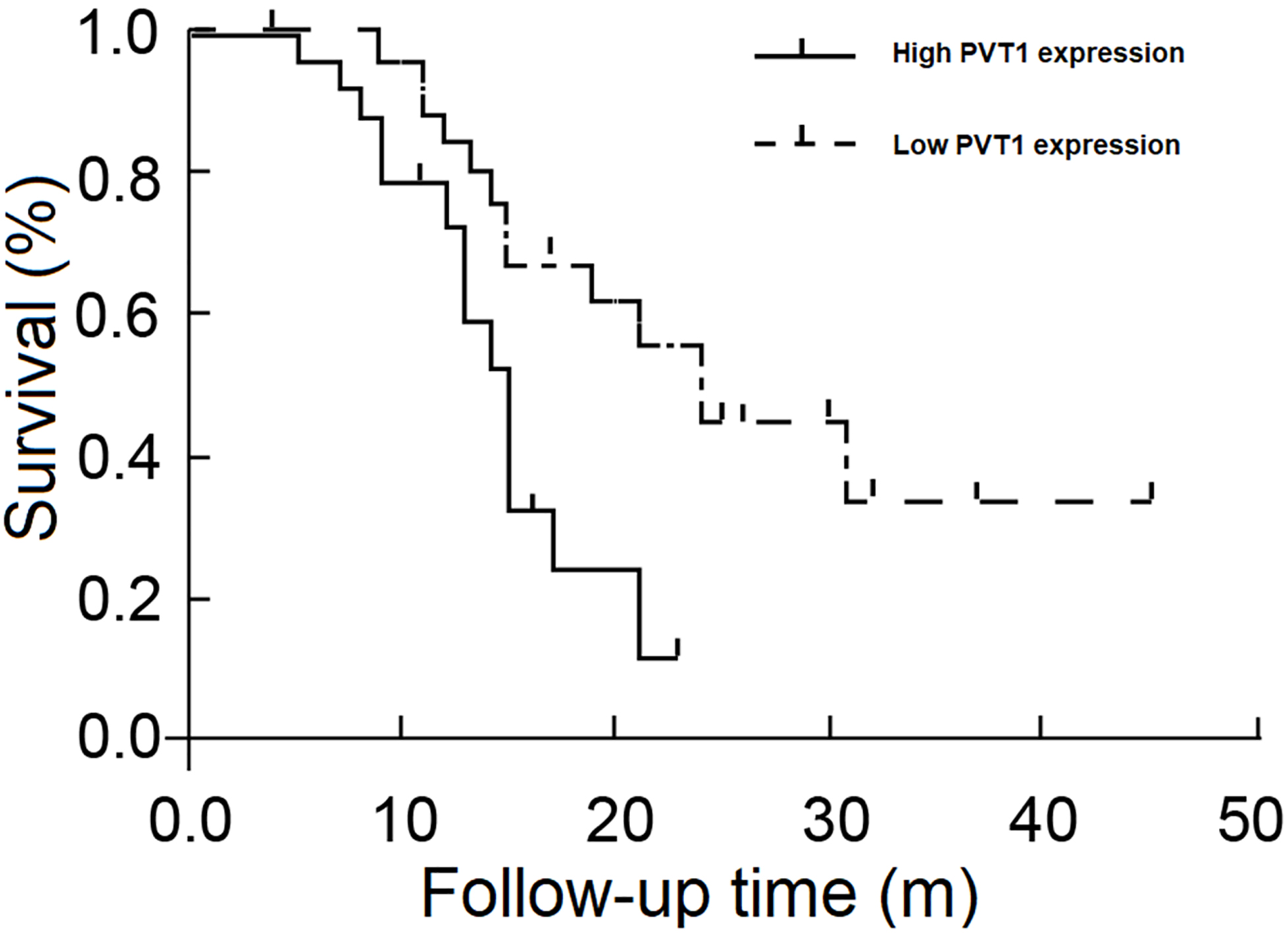
Glioma is the most common malignant tumor of the nervous system. Studies have shown that the type and susceptibility of the tumor are related to specific tumor genes [9, 10]. For example, Wapinski and Chang demonstrated that glioma is mediated by multiple genes and is resulted from disrupted regulatory networks that control the stability, differentiation and proliferation [11], and from the activation of oncogenes and inactivation of suppressor genes [12]. Although LncRNAs were considered non-functional transcription noise [13, 14], recent works have revealed that they play an important role in transcription controls, including DNA methylation, transcriptional activation, transcriptional regulation and transcriptional interference [15].
In glioma, LncRNA Hox antisense RNA (HOTAIR) has been shown to be negatively associated with prognosis and positively with tumor grade [16]. This is likely resulted from the inactivation of HOTAIR target gene PCR2, leading to the promotion of glioma cells [17]. In addition, Qin et al. showed that low LncRNA TSLCA-AS1 expression inhibits the growth of glioma cells probably because of increased methylation of target genes [18].
PVT1 is located in the sense strand of chromosome 8q24 region [19, 20, 21, 22], which is also a region with high DNA amplification in tumors. Abnormal DNA amplification is often an indicator of high cancer risk. For example, in breast cancer and metastatic prostate cancer, the chromosomal 8q24 region often has increased copy numbers of short nucleotide sequences [23]. Furthermore, this region also contains a number of loci that are related to the risk of diseases such as nonsyndromic cleft lip and palate and end-stage renal disease in type 2 diabetes [24]. Because of its special structure and location, the abnormal expression of LncPVT1is often related to tumorigenesis. Recent studies have shown that in many tumors, such as lung cancer, breast cancer, liver cancer, gastric cancer and colon cancer, PVT1 is highly expressed, although the underlying mechanism is still largely unclear [7]. On other hand, downregulation of PVT1has been shown to attenuate the resistance of glioma cells to paclitaxel [25], which may help overcome the drug resistance in cancer therapy.
We found that PVT1 is expressed in glioma and the expression level is higher in the tumors than in normal tissue and is possible related to tumor grade. We also showed that the expression level is related to tumor grade, but not to other factors such as tumor size, age, gender and KPS score. Therefore, we speculated the PVT1 level may be associated with the prognosis of patients with glioma.
Kaplan-Merier analysis showed that PVT1 expression is indeed negatively associated with survival time. A recent studies also showed that it indicates a poor prognosis of glioma via promoting cell proliferation and invasion via target EZH2 [26] and regulates the malignant behaviors of human glioma cells by targeting miR-190a-5p and miR-488-3p [27]. Therefore, LncPVT1 level may be used as a new molecular marker that measures the malignancy and predicts the prognosis of glioma. There are some limitations in the study. The control (healthy) tissues in this study were taken from patients who had brain trauma and it is not unclear if the trauma would have impact on PVT1 expression and the number of cases is relatively small.
Footnotes
Conflict of interest
The authors declare that they have no competing interests.
