Abstract
Ovarian cancer (OC) is the third most common gynecological malignancy with the highest mortality worldwide. OC is usually diagnosed at an advanced stage, and the standard treatment is surgery combined with platinum or paclitaxel chemotherapy. However, chemoresistance inevitably appears coupled with the easy recurrence and poor prognosis. Thus, early diagnosis, predicting prognosis, and reducing chemoresistance are of great significance for controlling the progression and improving treatment effects of OC. Recently, much insight has been gained into the non-coding RNA (ncRNA) that is employed for RNAs but does not encode a protein, and many types of ncRNAs have been characterized including long-chain non-coding RNAs, microRNAs, and circular RNAs. Accumulating evidence indicates these ncRNAs play very active roles in OC progression and metastasis. In this review, we briefly discuss the ncRNAs as biomarkers for OC prognosis. We focus on the recent advances of ncRNAs as therapeutic targets in preventing OC metastasis, chemoresistance, immune escape, and metabolism. The novel strategies for ncRNAs-targeted therapy are also exploited for improving the survival of OC patients.
Introduction
Ovarian cancer (OC) ranks fifth in the mortality rate among female malignant tumors, which seriously endangers women’s lives and health. 1 In 2020, the worldwide prevalence of OC in females was 3.4% and the mortality rate was 4.7%. 2 According to the International Federation of Obstetrics and Gynecology, approximately 75% of OC patients are diagnosed in stage III or IV with extensive abdominal metastases. 3 The main obstacle to improving the diagnosis of OC is the lack of effective screening methods for early detection. 4 Although the survival rate of OC patients has improved in the past few decades, the 5-year survival rate for women with stage I epithelial ovarian cancer (EOC) is 92%, while the women diagnosed with the advanced stage OC are still less than 30%.5,6 The median progression-free survival (PFS) for OC ranges from 16 to 21 months, and 75% of patients with the advanced disease undergo recurrence within 18–24 months.7–9
The blood cancer antigen 125 (CA-125) test is the most sensitive and specific early detection marker for OC available.10,11 However, this diagnostic method is still sub-optimal, due to the low sensitivity in the early stage of the disease, and its predictive value for screening is limited. Several new studies have demonstrated the promise of improving OC diagnosis. It was reported that the Risk of Ovarian Cancer Algorithm (ROCA) was used to evaluate continuous CA-125 measurements and showed the improved sensitivity for OC early diagnosis.12,13 In addition, the ROCA followed by transvaginal ultrasound indicated excellent specificity and positive predictive value in the American female population at an average risk of OC. 14 However, useful biomarkers are urgently needed for OC early diagnosis and predicting prognosis.
The basic treatment approach for OC is cytoreductive surgery combined with platinum-based chemotherapy, and the early-stage patients often have better curative effects. 15 With the several lines of medications, OC patients gradually develop chemoresistance, which seriously affects patients’ prognosis. 16 In recurrent OC disease, chemotherapy, antiangiogenic agents, and poly (ADP-ribose) polymerase inhibitors (PARPis) are commonly used in combination. At present, bevacizumab and three PARPis (olaparib, niraparib, rucaparib) have been approved by the Food and Drug Administration as the maintenance treatment after the recurrence of specific patients with platinum-sensitive diseases.17–19 Immunotherapy is a very promising treatment option. 20 However, it needs more preclinical and clinical investigations to further confirm its therapeutic effects.
The recent advances in transcriptomics analysis have demonstrated that molecular non-coding RNAs (ncRNAs) play important roles in various aspects of OC. ncRNAs are functional RNA molecules that are not being translated into proteins but are involved in various physiological and pathological processes. 21 It was found that the dysregulation of certain ncRNAs is related to tumorigenesis, neurological, cardiovascular development, and other diseases. 22 In recent decades, a large number of studies have reported the key role of ncRNAs in cancer progression, metastasis, and drug resistance. 23 And gene expression is regulated under physiological conditions both positively and negatively by various subtypes of ncRNAs. 24 At the same time, some small ncRNA molecules are stable in the bloodstream, which can be used as novel biomarkers for the diagnosis and prognosis of OC in clinic.25,26 In addition, ncRNAs are becoming important therapeutic targets for the treatment of OC by delivering RNA interference (RNAi) or oligonucleotide targeting messenger RNA (mRNA).27–29
In this review, we searched the literature from ‘Pubmed’, ‘web of science’, and other websites closely related with the content of this paper from year 1990 to year 2022. Papers selected are pivotal studies in OC and ncRNA area demonstrating the critical evidence presented in the manuscript. This article briefly introduces the ncRNAs as biomarkers for OC prognosis and focuses on the recent progress of ncRNAs in OC therapeutics, especially for novel targeted therapy development.
Functional characterization of ncRNAs in OC
With the revolution of the high-throughput genome sequencing and array-based technology, approximately 90% of the human genome is transcribed. 30 Although the transcribed genome encodes about 20,000 proteins, this accounts for only 2% of the entire genome. In other words, not all RNAs are translated into functional proteins, which are called ncRNAs, including long-chain non-coding RNAs (lncRNAs), microRNAs (miRNAs), circular RNAs (circRNAs), and other RNAs.30,31 lncRNA molecule consists of more than 200 bases in length, transcribed by RNA polymerase II, capped, and polyadenylated at the 5′ and 3′ ends. 32 miRNA is a small RNA molecule with a sequence of 17–22 nucleotides and has been characterized as ‘molecular rheostat’ or ‘fine-tuner’ of gene expression in different tissues and cell types.33–35 The circRNA varies greatly in length from hundreds to thousands of nucleotides and has been ubiquitously discovered in many species in recent years.36,37 In addition, ncRNAs are usually expressed in a specific manner under a certain cell type, tissue, and developmental stage.38–43
miRNAs are usually combined with a short complementary sequence usually located in the three prime untranslated region (3′-UTR) region of mRNA to regulate the expression of target mRNA, prevent the expression of the corresponding mRNA, or make it degrade. 44 While lncRNAs and circRNAs exhibit gene regulatory mechanisms at the transcriptional and post-transcriptional levels, which regulate gene expression by sponging miRNAs, weakening the interactions between miRNAs and mRNAs through a competitive mechanism.45–49 The corresponding biological functions of these ncRNAs are not only realized by a single RNA regulation but also by interacting with each other.23,45
Research in recent years has demonstrated the important function of ncRNAs in cellular activities associated with OC progression, including cell proliferation, apoptosis, invasion, migration, chemoresistance, angiogenesis, and reprogram energy metabolism.50–53 Studying ncRNAs as prognostic biomarkers could help develop precision medicine for OC patients. Understanding the mechanisms of ncRNAs in the regulation of OC metastasis and chemoresistance holds promise for developing novel therapies to improve the patent’s prognosis.
In addition, OC has high variability in histological subtypes. It contains two categories: EOC which takes 90% of all OC and the rest 10% is non-epithelial OC. 54 EOC is also divided into the most common high-grade serous OC (HGSOC, about 52%), endometrioid ovarian cancer (10%), ovarian clear cell carcinoma (6%), mucinous ovarian cancer (MOC, 6%), and low-grade serous ovarian.54,55 In non-epithelial OC, germ cell tumors and sex cord stromal tumors account for only 3% and 2% of all OC, respectively. 55 Current studies on ncRNAs mainly fall into the EOC. A comprehensive miRNA expression profile can help refine the subtype classification in EOC, opening up new opportunities for identifying clinically applicable markers to improve stratification and diagnosis of OC. 56 It was reported that some ncRNAs showed differential expression levels in subtypes of OC. For example, miR-483-5p was found to be differently expressed in serous EOC and non-serous EOC, with an apparent upregulation in serous EOC. 56 Similarly, the expression of exosomal miR-1290 in HGSOC was reported higher compared to other subtypes of OCs, 57 indicating the different expression of profiles in miRNAs may be associated with different histological types of OC and could have the potential to develop personalized medicine for individual diagnosis and treatment. More deep studies are warranted in the area in future investigation.
NcRNAs as prognostic biomarkers for developing OC personalized treatment
Accurate prediction of the prognosis of OC patients is helpful to guide treatment decisions, which may greatly improve the relapse/relapse-free survival of patients. 58 So far, several methods are used in the prognosis of OC in clinics. First, various molecular markers related to the prognosis of OC can be detected by laboratory experiments and bioinformatics analysis. It was reported that high-level expression of phosphoserine aminotransferase 1 in OC tissue samples was associated with a poor prognosis of patients. 59 The machine learning system also provides the diagnosis and prognosis predictions for EOC patients before the initial intervention. 60 In the past decade, several studies have demonstrated the potential application of liquid biopsy in cancer detection and progression monitoring. Liquid biopsy including circulating tumor cells, circulating tumor DNA, and extracellular vesicles (EVs) holds promising as a new tool for improving OC diagnosis and/or prognosis.61,62 However, the current methods and protein molecules used for OC prognosis still encounter certain limitations. Since most of the findings for biomarker studies are based on The Cancer Genome Atlas (TCGA) database, which lacks valid clinical validation studies for following up and does not have a enough large sample size; thus, they are unable to define the specific molecular mechanisms in OC. 63 Due to the high stability and expression pattern in clinical samples, ncRNAs have shown great potential as prognostic biomarkers for OC.
Based on genome-wide copy number variation, lncRNAs including LOC101927151, LINC00861, and LEMD1-AS1 have been identified as new prognostic markers to predict the survival of OC patients. It was found that the lower the expression of LOC101927151 and the higher the expression of LINC00861 and LEMD1-AS1, the worse the prognosis of the OC patients. 64 In addition, the increase in exosomal lncRNA metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) was found to be highly correlated with the advanced and metastatic phenotype of OC as an independent predictor of the overall survival (OS) of OC patients. 65
Various miRNAs were also reported to be associated with OC prognosis. The miRNA 200 family members provide useful information about prognosis and response to the treatment of HGSOC. Compared with the control, the expression of circulating blood miR-200a, miR-200b, and miR-200c was found to be upregulated in OC patients, which was related to disease stage and reflected tissue expression. 66 It was also reported that using univariate analysis, the higher the concentration of miR-200b in exosomes, the lower the OS in the EOC. 67 Wei et al. 68 showed that OC patients with high expression of miR-199b-3p had longer OS and disease-free survival (DFS). Research by Xie et al. 69 demonstrated that the low miR-1231 expression in tissue indicated a poor prognosis for patients with OC, while upregulation of miR-1231 expression in four human OC cell lines (SKOV3, OVCA433, OV2008, and A2780) inhibited cells growth. A study based on gene expression synthesis (GEO) and bioinformatics analysis found that the prospective pathway signals of miRNA-182 were highly expressed in OC tissues and associated with a poor prognosis. 70 miR-150-5p was reported to be significantly upregulated in recurrent OC tissue specimens compared with primary tissue samples, and its expression was related to the early recurrence and poor survival rate of OC patients. 71 High levels of miR-424 (322) in tumors were found positively to be associated with PFS in patients with OC. 72 A recent clinical study showed that high expression of miR-181a was closely related to the poor prognosis in OC. 73 miR-203 was found to be upregulated and associated with significantly shorter OS and higher risk for OC progression. 74 Furthermore, highly expressed miR-206 was associated with shorter OS in EOC patients who received platinum-based chemotherapy and used to predict chemoresistance to platinum treatment. 75 Some miRNAs are downregulated in OC. For example, the expression level of miR-484 in exosomes was significantly reduced, accompanied with a poor prognosis in OC. 76 Circulating miRNAs were also useful as predictive biomarkers in patients with HGSOC. For example, higher levels of miR-187-5p and miR-6870-5p were associated with both poorer PFS and OS, while miR-1908-5p and miR-6727-5p only acted as prognostic indicators of PFS. 77
Multivariate Cox analysis showed that high expression of circRNA itchy E3 ubiquitin-protein ligase (circ-ITCH) was an independent predictor for the good OS in EOC patients, and high expression of circRNA ABCB10 (circ-ABCB10) was found to be a potential marker for a poor prognosis of OC.78,79 The rates of DFS and OS in patients with low expression of circRNA_LARP4 (circ LARP4) were significantly worse. Therefore, circ LARP4 might be a potential biomarker for the prognosis of OC. 80 The exosomal circular forkhead box protein P1 (circFoxp1) was highly expressed in serum of EOC patients, and through survival analysis, it was found that EOC patients with high exosome circFoxp1 expression had lower OS and DFS, suggesting circFoxp1 was a worse prognostic biomarker. 81
The above-mentioned studies have shown that both tissular and circulating exosome ncRNAs play important roles in the prognosis of OC. Circulating exosome ncRNAs have the potential to be used as biomarkers for predicting the prognosis of OC patients in clinics in a non-invasive manner. With an in-depth understanding of patients’ molecular characteristics using ncRNAs as prognostic markers, the tailored therapies can be developed and applied to those who are expected to benefit the most while limiting ineffective or harmful interventions. 82 And many new therapies that regulate RNAs are being studied extensively, especially miRNAs. 83 Therefore, regulating the expression of certain ncRNAs to change the prognosis of OC might provide the promise for developing the individualized treatment of OC patients and improving their quality of life. The ncRNA biomarkers used for OC prognosis are summarized in Table 1.
The summary of ncRNA biomarkers used for OC prognosis.
circ-ABCB10, circular RNA ABCB10; circFoxp1, circular forkhead box protein P1; circ-ITCH, circRNA itchy E3 ubiquitin-protein ligase; circ-LARP4, circular RNA_LARP4; DFS, disease-free survival; EOC, epithelial ovarian cancer; FIGO, Federation of Obstetrics and Gynecology; GEO, gene expression synthesis; MALAT1, metastasis-associated lung adenocarcinoma transcript 1; OC, ovarian cancer; OS, overall survival; PFS, progression-free survival; TCGA, The Cancer Genome Atlas.
NcRNAs as therapeutic targets for OC treatment
Most patients diagnosed with OC undergo surgery first, followed by platinum-based chemotherapy.84,85 However, the majority of women with advanced EOC, fallopian tube cancer, or primary peritoneal cancer relapse and require additional treatment. 86 According to the American Society of Clinical Oncology guidelines, PARPis are also used in the management of OC. 87 It was reported that ncRNAs are involved in the regulation of many aspects of OC progression,24,50,88 so they can be used as potential therapeutic targets for OC treatment with certain advantages. For example, the specificity of lncRNA has been used to selectively kill tumors without affecting normal tissues. 89 The emerging importance of circRNAs in the initiation and progression of OC also makes them an attractive therapeutic option. 90 For tumor suppressor circRNA, due to its stability and long half-life, it might induce expression in specific cancer cells and produce great anticancer effects. 91 In the following sections, we will focus on the potential of ncRNAs as therapeutic targets for OC treatment from the aspects of metastasis, chemoresistance, immune escape, and metabolic regulation. The ncRNAs as potential therapeutic targets for OC therapy are shown in Figure 1 and Table 2.

ncRNAs as potential therapeutic targets for OC therapy. This diagram illustrates several associated mechanisms by which ncRNAs (lncRNAs, miRNAs, and circRNAs) regulate OC metastasis and chemoresistance, immune escape, and metabolism. Mechanistic studies have shown some ncRNAs that can be used as OC therapeutic targets by inhibiting these potential mechanisms. Treatment of OC by upregulating or downregulating the expression of ncRNAs is a promising way to control OC spread.
ncRNAs as therapeutic targets for OC cell migration, invasion, metastasis, chemoresistance, immune escape, and metabolism reprogramming.

circ-CSPP1, circ-centrosome/spindle pole-associated protein; circ-ITCH, circRNA itchy E3 ubiquitin-protein ligase; circRHOBTB3, circular RNA RHOBTB3; DANCR, differentiation antagonizing non-protein coding RNA; ESRP1, epithelial splicing regulatory protein-1; FEN1, flap structure-specific endonuclease 1; FOXR2, forkhead box 2; GAS5, growth arrest-specific transcript 5; HOTTIP, lncRNA HOXA transcript at the distal tip; IL-6, interleukin 6; MALAT1, metastasis-associated lung adenocarcinoma transcript 1; MICA/B, MHC class I chain-related molecules A/B; MMP, matrix metalloproteinases; ncRNA, non-coding RNA; NRCP, lncRNA ceruloplasmin; PARP1, poly (ADP-ribose) polymerase 1; PD-L1, programmed death-1 ligand 1; PFKFB2, fructose-2,6-biphosphatase 2; PTAR, lncRNA pro-transition associated RNA; SCAI, suppressor of cancer cell invasion; SDC, Syndecan; TP73-AS1, lncRNA P73 antisense RNA 1T; UCA1, urothelial carcinoma associated 1; VEGF, vascular endothelial growth factor; VEGFA, vascular endothelial growth factor A; ZEB1, zinc finger E-box binding homeobox 1.
NcRNAs as therapeutic targets for OC via regulating metastasis
Metastasis accounts for the most lethal reason for the high recurrence and poor prognosis of OC patients.122,123 Therefore, inhibiting tumor metastasis can prevent the recurrence of OC, which is also an important part of OC treatment. Since there is no barrier between the primary tumor and the abdominal cavity, aggregating cells with stem cell characteristics can leave the primary tumor and are implanted in the peritoneum in OC metastasis. 124 It was found that epithelial-to-mesenchymal transition (EMT), cell stemness, angiogenesis, matrix metalloproteinases (MMPs), and changes in cell adhesion molecules promote OC metastasis,122,125–128 which is related to the dysregulation of ncRNAs.
miRNAs are directly regulated by lncRNAs/circRNAs to affect the EMT pathway and related molecules, promoting or inhibiting the metastasis of OC. Liang et al. reported that lncRNA pro-transition-associated RNA (PTAR) was significantly upregulated in the mesenchymal subtype samples, which regulates zinc finger E-box binding homeobox 1 (ZEB1) expression by competitively binding miR-101-3p to promote EMT, leading to increased metastasis of serous OC. Thus, PTAR might be an effective target for OC antimetastatic therapy. 92 It was shown that circ-centrosome/spindle pole-associated protein (circ-CSPP1) sponged miR-1236-3p, to impair its inhibitory effect on ZEB1, and regulated EMT to promote distant metastasis of OC. 114 Overexpression of miR-200b/c was also reported to target ZEB1, which inhibited OC metastatic transmission, and miR-200b/c was related to disease stages.129,130
Several lines of evidence indicate that cancer stem cells (CSCs) are involved in tumor invasion and metastasis.131,132 The lncRNA WDFY3-AS2 sponged miR-139-5p to induce traits in CSCs, which regulates migration and invasion of the chemoresistant OC cell line A2780-DDP. 93 In vitro study showed that inhibition of miR-328 held promise for the development of efficient strategies for eliminating CSCs to prevent OC metastasis and recurrence. 133 In vitro and in vivo experiments had shown that silencing has-circ-0026123 inhibited OC cell proliferation and migration as well as inhibited the expression of CSC differentiation-related markers. 115
It was reported that the formation of a large number of microvessels is the basis for OC growth and metastasis.126,127 The angiogenesis-related ncRNAs regulate the expression of certain molecules that modulate OC angiogenesis, which further promotes OC metastasis. The differentiation antagonizing non-protein coding RNA was reported to regulate the miR-145/vascular endothelial growth factor (VEGF) axis to promote OC angiogenesis, facilitating OC metastasis. 94 miR-205 was a metastasis-associated miRNA in OC and its upregulation was found to positively correlate with microvessel density in OC tissues. 102 circASH2L was highly expressed in OC tissues and cell lines (A2780, TOV112D, OVCAR-3, and SKOV-3) and played a key role in regulating tumorigenesis, angiogenesis, and lymphangiogenesis of OC through the miR-665/VEGFA axis, 116 suggesting it might be a useful target for OC therapy. In addition, circATRNL1 was shown to sponge miR-378 and subsequently activate the Smad4 signaling pathway, which inhibits OC angiogenesis and metastasis. 117
MMPs are a family of secreted or transmembrane enzymes that collectively digest almost all extracellular matrix (ECM) and basement membrane components.134,135 More importantly, MMP-2 and MMP-9 degrade collagen IV, a major ECM component of the basement membrane, and have been implicated as key factors for the invasive and metastatic potential of OC. 124 The study by Wang et al. 95 showed that lncRNA P73 antisense RNA 1T (TP73-AS1) promoted OC cell proliferation and metastasis via the modulation of MMP-2 and MMP-9. miR-134-3p mimic transfection inhibited migration and invasion of SKOV-3 and OVCAR-3 cells and decreased the protein expression levels of cyclooxygenase-2, MMP-2, and MMP-9. 103 In addition, knockdown of circ-CSPP1 also caused a decrease in MMP-2 expression to inhibit OC metastasis, while overexpression of circ-CSPP1 had opposite effects. 114
Inhibition of tumor metastasis by regulating the expression of ncRNAs is an important aspect of OC therapy and might provide a new possible avenue for OC treatment.
NcRNAs as therapeutic targets for OC via decreasing chemoresistance
Nearly 75% of OC patients are highly sensitive to initial anticancer therapy; however, most patients encounter tumor relapse within 2 years, and are unable to respond to available chemotherapeutic compounds due to acquired resistance.136,137 Tumor cells develop several mechanisms to reduce the anticancer effects of cisplatin (CIS)/paclitaxel (PTX) through reducing drug uptake, increasing drug efflux, and inducing drug detoxification by covalently binding to glutathione or metalloprotein. 138 Besides, alterations in DNA damage repair, reactivation of homologous recombination (HR) mechanism, the occurrence of CSCs and EMT, methylation, histone acetylation, and other phenotypic changes, immune cell infiltration, angiogenesis, modification of drug targets, and defect sand hypoxia resistance are all possible reasons for OC chemoresistance.139–145 Extensive studies suggest that ncRNAs are involved in regulating the above-mentioned mechanisms and affecting OC chemoresistance. Therefore, altering the expression of ncRNAs to inhibit OC chemoresistance is also an important strategy for the treatment of OC.
The ATP-binding cassette (ABC) transporter protein family is an energy-dependent transport system for substrate-binding proteins. 146 Multidrug resistance protein 1 (MDR1), multidrug resistance-associated protein 1 (MRP1) and breast cancer resistance protein are the most important ABC transporter protein family involved in drug efflux in tumor chemoresistance mechanisms.147–149 There are many reports on ncRNA-associated regulation of ABC family gene expression, which promotes drug efflux and ultimately leads to the onset of chemoresistance. Wang et al. 96 reported that lncRNA urothelial carcinoma associated 1 was highly expressed in PTX-resistant OC cells, sponging miR-129, regulating the high expression of ABCB1, and accelerating PTX drug efflux. lncRNA MALAT1 was highly expressed in CDDP-OC cells, and induced the expression of MRP1 to accelerate CDDP drug efflux.97,150 miR-495 was reported to inhibit MDR1 expression and reduce drug efflux to reverse MDR in OC. 104
Most chemotherapy drugs, such as platinum-based drugs, directly or indirectly induce DNA damage by activating various signaling pathways, ultimately leading to cell death. Enhanced activity of DNA damage repair was found in CIS-resistant OC cells. 151 PARP is a nuclease that plays a key role in the repair of single-stranded DNA damage. 152 Let-7e is expressed in the low level in OC chemoresistant cells, but PARP1 was highly expressed. The high expression of let-7e was used to target PARP1 and thus inhibited DNA damage repair, which improves the chemoresistance in OC cells. 105 BRCA1 is a key component of the error-free HR double-strand DNA repair pathway. 153 One study reported that miR-9 targeted BRCA1, to improve the chemosensitivity of OC. 106 Another study showed that miR-509-3 in OC enhanced the synthetic lethality of PARPi by regulating HR repair in the HGSOC patient-derived-xenograft model. 107
Accumulating studies show that ncRNAs affects apoptosis as well as the cell cycle that affect the chemotherapy sensitivity.154–157 The lncRNA growth arrest-specific transcript 5 was found to affect the MAPK activity, pro-apoptosis, and cycle arrest, thereby improving chemotherapy sensitivity. 98 It was reported that the high expression of miR-21 decreased PTEN expression, promoted the PI3K/Akt activity, and inhibited OC cell apoptosis. 158 The high expression of miR-21 was also shown to regulate M2 macrophage polarization, inhibit apoptosis, and promote chemoresistance. 108 In OC, the overexpression of circ-Cdr1as was found to enhance DDP-induced apoptosis by regulating the suppressor of cancer cell invasion and miR-1270. 118 circCELSR1 was also highly expressed in PTX-resistant OC cell lines (SKOV-3/PTX and HeyA-8/PTX), and circCELSR1 silencing enhanced PTX-induced OC cytotoxicity by increasing G0/G1 blockade and apoptosis in OC cells. 119
Although advances in understanding ncRNAs in OC chemoresistance have been made in the last decade, the mechanism of action of ncRNA in OC chemoresistance is still not well understood and further studies are urgently needed. This research area is very critical to address the unmet clinical challenge of OC chemoresistance, as well as develop novel treatments to control the recurrence of OC patients. Clarifying these mechanisms of action is also important for laying a solid foundation for the clinical translation of ncRNAs in OC therapy. The putative mechanism of ncRNAs in regulating OC metastasis and chemoresistance is shown in Figure 2.

The putative mechanism of ncRNAs regulates OC metastasis and chemoresistance via target genes and the related pathways. Altered expression of RNAs affects EMT, CSCs, neoangiogenesis, apoptosis pathway, cell cycle, drug efflux, and other biological functions of OC cells, leading to chemoresistance and metastasis.
NcRNAs as therapeutic targets for OC immunotherapy
Cancer immunotherapy has emerged as a promising therapeutic approach in oncology, characterized by the activation of the immune system and induction of tumor immune surveillance or reversal of tumor immune escape. 159 Marth et al. 160 highlighted the therapeutic benefits of immunotherapy and challenges in OC treatment. Figuring out the specific mechanism of immune escape of ncRNAs in OC is an important option for treating OC.
lncRNA HOXA transcript at the distal tip (HOTTIP) was highly expressed in OC tissues, resulting in increasing interleukin 6 expression by binding to c-jun, and promoting programmed death-1 ligand 1 (PD-L1) expression to inhibit T-cell proliferation. Therefore, HOTTIP might be involved in a potential therapeutic strategy by targeting HOTTIP in OC. 99 Another study shown that miR-424(322) inhibited PD-L1 and CD80 expression, promoted the proliferation and survival of CD8+ cytotoxic T lymphocytes and improved the OC immune response. 72 Besides, miR-20a was reported to bind directly to the 3′UTR of MHC class I chain-related molecules A/B (MICA/B) mRNA, leading to its degradation and reducing MICA/B proteins at the plasma membrane. 109 The membrane-bound MICA/B protein was a ligand for the natural killer (NK) group 2 member D receptor and was found on NK cells, γδ+ T cells, and CD8+ T cells, where its reduction enabled tumor cells to evade immune-mediated killing. 109 circ-0005585 was also found to regulate the overexpression of epithelial splicing regulatory protein-1 (ESRP1) by sponging miR-23a/b and miR-15a/15b/16, and the high expression of ESRP1 was related to immunosuppression in OC, 120 suggesting that ESRP1 was a potential therapeutic target for OC immunotherapy. One example of potential effects of ncRNAs as therapeutic targets in OC immunotherapy is shown in Figure 3.
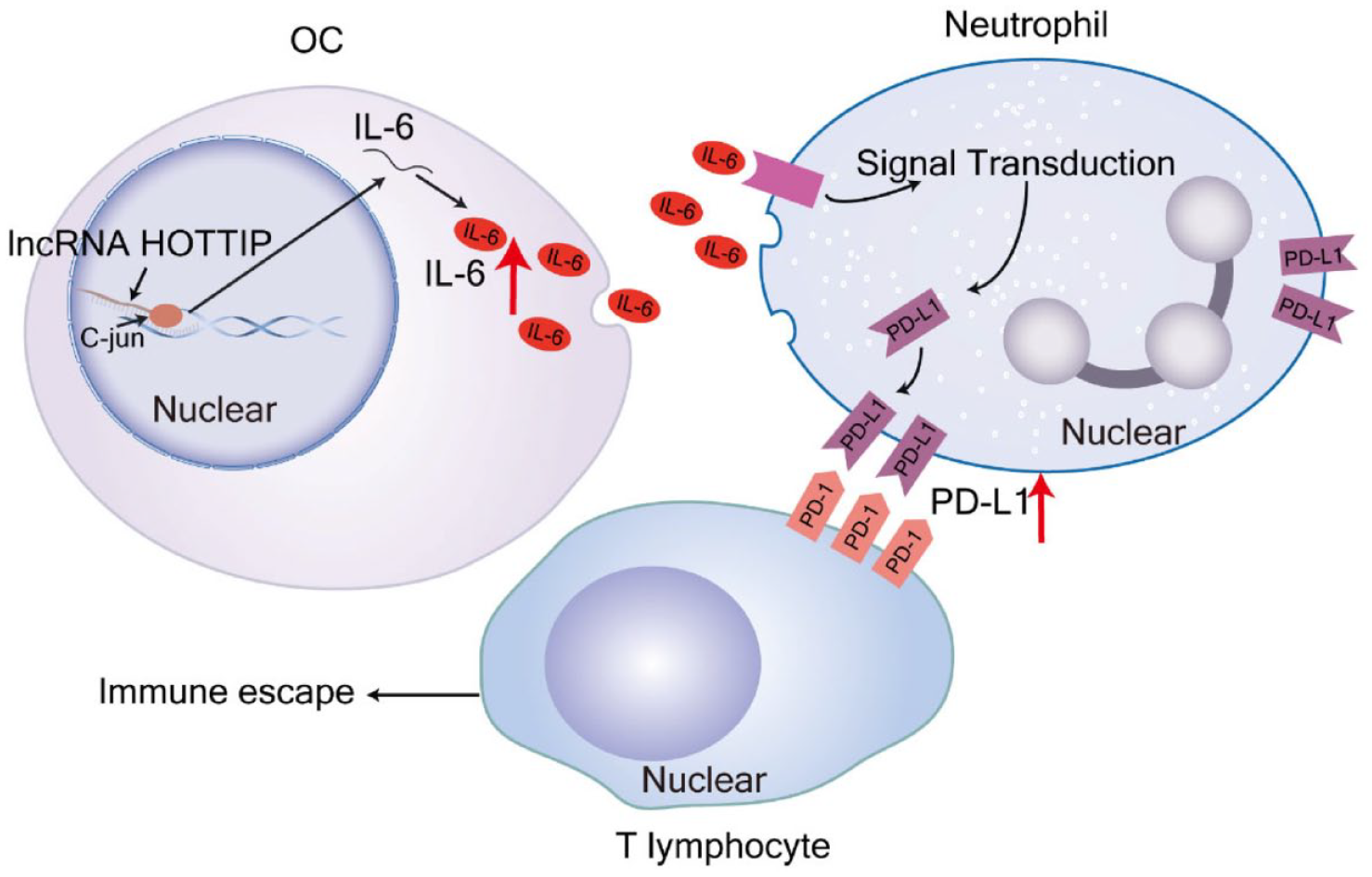
ncRNAs as therapeutic targets for OC immunotherapy. LncRNA HOTTIP is highly expressed in OC, which increases the expression of IL-6 by binding to c-jun and promotes PD-L1 expression in neutrophils to inhibit T-cell proliferation. Therefore, OC can be treated by targeting HOTTIP to increase T-cell proliferation and kill cancer cells.
Immunotherapy is currently a more advanced treatment option for several cancers such as lung cancer, melanoma, and breast cancer. However, very limited reports are present in OC immunotherapy. ncRNAs can regulate immune escape and surveillance, but the specific immune regulatory mechanisms in OC need to be further investigated. ncRNAs’ regulation of immune escape and immune suppression might be used in combination with immunotherapy for better clinical treatment of OC in the future.
NcRNAs as therapeutic targets for OC via regulating metabolism
Reprogramming of energy metabolism is a hallmark of tumors due to genomic instability. 161 Warburg effect in cancer cells is related to aerobic glycolysis in that glucose is metabolized to lactic acid under aerobic conditions. 162
Previous studies demonstrated that lncRNA regulated glycolysis in cancer cells by directly binding to key glycolytic enzymes or by enhancing the transcription of glycolytic enzyme genes activated by lncRNA binding to RNA polymerase II.100,161,163 Mechanistic studies shown that lncRNA LINC00092 binds to a glycolytic enzyme, fructose-2,6-biphosphatase 2, thereby promoting OC metastasis by altering glycolysis and maintaining the local support function of cancer-associated fibroblasts. 101 lncRNA ceruloplasmin was highly expressed in OC tissues, and at the same time, it acted as an intermediate binding partner between STAT1 and RNA polymerase II, leading to increased expression of downstream target genes (such as glucose 6-phosphate isomerase) and regulating glycolysis. 100
miRNAs are also involved in various aspects of tumor metabolism including glucose, lipid, and amino acid metabolism. 164 Overexpression of miR-29b was reported to negatively regulate OC glucose metabolism in vivo. 110 miR-450a was found to reduce amino acid production in OC by regulating targets related to glutamine catabolism. 111 Besides, miR-145 inhibited glutamine metabolism by targeting c-myc. 112 As part of the ncRNA regulatory network, circRNAs were also reported to regulate the metabolism of carbohydrates, lipids, and amino acids by targeting miRNAs or proteins. 165 In OC, high expression of circ-ITCH in A2780 and OVCAR-3 cell lines inhibited glucose consumption and decreased both lactate and ATP content, indicating that circ-ITCH inhibited glycolysis. 113 Furthermore, circRNA RHOBTB3 (circRHOBTB3) overexpression significantly inhibited OC cell glycolysis. 121 So far, research on ncRNAs in metabolic regulation that contributes to OC progression is still very limited. The potential mechanisms of ncRNAs in OC metabolism need to be further explored, and investigating the role of ncRNAs in OC metabolism is a very important and promising area for developing novel OC therapy.
New strategies for ncRNAs-targeted OC therapy
ncRNAs participate in the regulation of multiple molecules in cellular signaling pathways to cause specific reactions. 166 However, many challenges in ncRNA research still exist. In vivo stability, in vivo cell permeability, tissue-specific targeting, and potential off-target effects are the obstacles to the successful conversion of ncRNAs-based compounds from the laboratory to clinic. 167 Given the above reasons, targeted therapy strategies explored in recent years have become hot spots. On the one hand, they can solve the instability of ncRNAs, on the other hand, they hold the potential to aim specific therapeutic targets.
Current research has focused on the development of miRNA nano-formulations to enhance cell uptake, bioavailability, and tumor site accumulation.168,169 The use of nanoparticles (NPs) coupled with antibodies and/or polypeptides can effectively target and sustain the release of miRNAs/anti-miRNAs, which reduces the required therapeutic dose while minimizing systemic and cytotoxicity. 169 Studies have shown that in vivo nanoliposome delivery of miR-15a and miR-16 reduced tumor growth in a preclinical chemoresistant OC orthotopic mouse model to support combination therapy. 170 Another in vivo study using xenograft models has shown that NP-mediated miR-124 reduced OC growth and induced cells sensitive to etoposide. 171 Since the overexpression of miR-21 was related to OC chemoresistance, 169 AS1411 anti-nucleolin aptamer-decorated PEGylated poly(lactic-co-glycolic acid) NPs containing CIS (Ap-CIS-NPs) were used to infect A2780 chemoresistant cells through nucleolar protein-mediated endocytosis and inhibited endogenous miR-21. 172 The targeted delivery of CIS using Ap-CIS-NPs into the miR-21-inhibited cells caused enhanced cell death. 172 The tumor-associated antigen, folate receptor alpha is a GPI membrane protein that is overexpressed in OC. 173 Covalently bound octahedral DNA nanocapsules were functionalized with folate molecules and used as scaffolds to engineer four chelating units with miR-21 complementary sequences to obtain biocompatible Fol-miR-21-NC non-toxic nanostructures, enabling the selective recognition of folate receptor alpha overexpressing cancer cells and sequestration of oncogenic miR-21. 174 Co-administration of doxorubicin and anti-miR-21 exhibited an additive cytotoxicity on tumor cells, laying the foundation for its use as a selective nucleic acid drug. 174 miR-155 was reported to be downregulated in OC-associated dendritic cells (DC) and is essential for DC optimal antigen presentation and T-cell activation. 175 Therefore, the use of polyethylenimine-based nano complexes to deliver miR-155 to tumor-associated DCs increased the expression of miR-155 in vitro and led to enhanced antitumor immunity, thereby increasing the survival rate of mice. 175 A novel nano-targeted co-delivery system modified with hyaluronic acid (HA) was prepared by coating functionalized mesoporous silica NPs (HA-PTX/Let-7a-GNR@MSN) with gold nanorods. 176 This drug delivery system was used in combination with hydrophobic chemotherapeutic drugs PTX and Let-7a and bound to the CD44 receptor, which is highly expressed on the surface of the SKOV3/SKOV3TR cells membrane to overcome MDR in OC. 176 However, as synthesized particles, these NPs might have obvious disadvantages after administration, such as toxicity, loss of targeting ability, and/or rapid clearance from blood circulation.177–179
Exosomes are a subtype of EVs (40–150 nm in diameter), which are considered to be a new generation of nano-scale drug delivery system. Exosomes secreted by different types of cells carry different signal molecules (such as RNAs and proteins), so they have great potential in targeted drug delivery and therapy.180,181 ncRNAs are selectively enriched and stable in exosomes. 182 Exosome ncRNAs play an important role in cell-cell communications, affecting key processes of tumor development such as tumorigenesis, metastasis, angiogenesis, immune regulation, and drug resistance in OC.65,183 Given the important biological functions of exosomal ncRNAs in OC, a strategy specifically targeting exosomes or their cargo might be a promising option for OC treatment. 184 Exosomal miR-21 was demonstrated to transfer from adjacent stromal cells to OC cells, conferring chemoresistance and aggressive phenotype to OC cells, which indicates that preventing exosomal miR-21 transfer from stromal cells was a new way to inhibit the growth of OC. 185 Exosomal miR-146a derived from human umbilical cord mesenchymal stem cells was reported to increase the sensitivity of OC cells to docetaxel and taxane. 186
Although the clinical application of exosomal ncRNAs has a long way to go, new research can help to find cost and time-saving nanotechnology to achieve large-scale production of exosomes. 187 Studies have shown that exosomes engineered by overexpressing miR-92b-3p had the stronger abilities of antiangiogenesis and antitumor than parental OC-derived exosomes, providing a new approach for antiangiogenic therapy of OC. 188 In addition, targeted delivery of miR-484 via RGD-modified exosomes induced normalization of tumor blood vessels in OC, increased tumor sensitivity to chemotherapy, and prolonged survival time after chemotherapy in tumor-bearing mice. 189 The potential of ncRNAs combined with exogenous NPs and endogenous exosomes for OC targeted therapy are shown in Figure 4. The new strategies for ncRNAs-targeted OC therapy are summarized in Table 3.

The potential of ncRNAs combined with exogenous nanomaterials and endogenous exosomes for OC targeted therapy. miR-155 is downregulated in DCs and is essential for DC optimal antigen presentation and T-cell activation. Therefore, the use of PEI-based nano complexes to deliver miR-155 to tumor-associated DCs increases the expression of miR-155 in vitro and leads to enhanced antitumor immunity. Exosomal miR-146a derived from hUCMSCs increases the sensitivity of OC cells to docetaxel and taxane. Both nanomaterials and exosomes containing ncRNAs combined with chemotherapy drugs greatly induce OC cell death.
New strategies for ncRNAs-targeted OC therapy.

Ap-CIS-NPs, AS1411 anti-nucleolin aptamer-decorated PEGylated poly (lactic-co-glycolic acid) nanoparticles containing CIS; CIS, cisplatin; DOPC, 1,2-dioleoyl-sn-glycero-3-phosphatidylcholine; HA-PTX/Let-7a-GNR@MSN, hyaluronic acid (HA) was developed employing GNRs coated with functionalized MSNs (GNR@MSN); hUCMSCs, human umbilical cord mesenchymal stem cells; ncRNA, non-coding RNA; NP, nanoparticle; PEI, polyethylenimine.
Both nanomaterials and exosomes provide a good chance for the application of ncRNAs in OC treatment. As above mentioned, some studies have been conducted on ncRNAs in OC, combined with exosomes/nanomaterials to achieve targeted transport. However, there are still many challenges for overcoming, including the toxicity of NPs, the evasion of the phagocytic system, the inhibition of physiological barriers, and the immune response to the body. 190 In addition, off-target exosomes and nanomaterials that deliver ncRNAs might also exist. However, due to the species specificity of ncRNAs, the results of animal experiments are still facing a huge test before being used in clinical application, and more in-depth exploration is needed.191,192 The current research on ncRNAs targeted therapy mainly focuses on miRNAs, and there are few reports on other types of ncRNAs. More broad research in ncRNAs is an urgent need in the future.
Conclusions and future perspectives
This review mainly focuses on the role of ncRNAs in the prognosis and treatment of OC. We investigate pivotal studies to bring new insights into the clinical application of ncRNAs in OC for future research. The network in which ncRNAs function affects multiple molecular targets that control the biological fate of cells and their responses to oncogenic effects. ncRNAs affect all aspects of OC cells, including proliferation, metastasis, chemoresistance, immune escape, and metabolism, which are in a good position to be used in clinics. 193
It is known that a network effect involves the complex regulatory mechanisms in ncRNAs’ functions. lncRNAs, circRNAs, and miRNAs are competing endogenous RNAs. lncRNAs/cicrRNAs compete with miRNAs to bind to mRNAs and regulate the expression of the corresponding molecules. The interactions among these molecules require further investigations to elucidate the related mechanisms in OC.
And it is important to understand the mechanisms performed by ncRNAs in OC metastasis, chemoresistance, immune escape, and metabolism for developing therapeutic targets. Since the combination of advanced technologies (next-generation sequencing) and new materials (exosome and nanomaterial) has great potential in the application of OC patients. The use of ncRNAs with these advanced technologies enables dual targeting of specific cells and specific genes.
Some nanoscale drug delivery systems, including NPs, liposomes, and nanocapsules, can be used to overcome the shortcomings of traditional drug delivery, preferentially target OC cells and amplify the therapeutic potential. 194 A large number of studies have shown that ncRNAs in exosomes are differentially expressed in serum, plasma, and ascites samples between OC patients and normal controls, and these ncRNAs hold great potential as biomarkers to improve the efficiency of diagnosis, prognosis, and treatment, such as the low expression of miR-484 in OC serum exosomes.76,195 However, there are few studies on targeted therapies combined with ncRNAs in OC treatment, which are worthy of further study.
Transfer RNA-derived fragments (tRFs) are another novel class of small ncRNAs produced through enzymatic cleavage of tRNAs and have been shown to play key regulatory roles similar to miRNAs.196,197 Accumulating evidence has shown that tRFs regulate gene expression at transcriptional and post-transcriptional levels. 196 It was also found that tRFs are expressed in HGSOC and normal ovarian tissues with significant differences, which might provide potential biomarkers for the diagnosis and treatment of HGSOC. 198 Although there have been many studies on tRF in other tumor types, 196 it has rarely been reported in OC research. Plant-derived tRFs-T11 was found to interact with AGO2 to suppress TRPA1 via a RNAi pathway in the A2780 cell, which, in turn, suppresses OC proliferation. 199 Silico analyses and expression profiling were performed using the TCGA-OC database, the GEO dataset, and two institutionally independent cohorts. This analysis highlighted a tRNA GlyGCC-derived internal fragment as a novel molecular predictor of EOC prognosis and supported tRNA may have bright future in the precision medicine decisions in EOC treatment. Like other ncRNAs, tRFs also play important roles in OC progression 200 and need great efforts to deeply investigate their functions and mechanism.
Besides, some small ncRNAs are so stable that they can survive in blood and form the basis for accurate and sensitive screening of major human cancers in a few drops of blood.25,26,201 In addition, the development of diagnostic biomarkers envisaged to be found in urine or blood is ideal to spare patients from the non-invasive procedures usually associated with tissue collection. It is important to find appropriate and effective ncRNAs to improve early diagnosis of OC as well as to serve as prognostic markers, and also as target molecules for therapy.
